Abstract
The effect of the intracellular microenvironment in the presence of an oxygen vector during expression of a fusion protein in Escherichia coli was studied. Three organic solutions at different concentration were chosen as oxygen vectors for fumarase expression. The addition of n-dodecane did not induce a significant change in the expression of fumarase, while the activity of fumarase increased significantly to 124% at 2.5% n-dodecane added after 9 h induction. The concentration of ATP increased sharply during the first 6 h of induction, to a value 7600% higher than that in the absence of an oxygen-vector. NAD/NADH and NADP/NADPH ratios were positively correlated with fumarase activity. n-Dodecane can be used to increase the concentration of ATP and change the energy metabolic pathway, providing sufficient energy for fumarase folding.
Keywords:
Oxygen vector; Energy; Fusion protein; Expression; Redox metabolism
Introduction
Molecular oxygen is an environmental and developmental signal that regulates cellular bioenergetics.11 Haddad JJ. Oxygen sensing and oxidant/redox-related pathways. Biochem Biophys Res Commun. 2004;316:969-977.,22 Zhang DL, Guan D, Liang JB. Reducing lactate secretion by ldhA Deletion in l-glutamate-producing strain Corynebacterium glutamicum GDK-9. Braz J Microbiol. 2014;45(4):1477-1483. During aerobic fermentation, oxygen plays an important role as the terminal electron acceptor in the respiratory chain that drives the energy metabolism.33 Schlepütz T, Gerhards JP, Büchs J. Ensuring constant oxygen supply during inoculation is essential to obtain reproducible results with obligatory aerobic acetic acid bacteria in vinegar production. Process Biochem. 2013;48(3):398-405. However, oxygen requirements in aqueous growth medium systems have been estimated to be 14% higher than for conventional aerobic processes. In the case of submerged fermentations, the supply of oxygen to the aqueous phase is one of the limiting factors. Oxygen-vectors are hydrophobic liquids in which oxygen has a higher solubility than in water.44 Giridhar R, Srivastava AK. Productivity enhancement in l-sorbose fermentation using oxygen vector. Enzyme Microbial Technol. 2000;27:537-541. Wang55 Jianlong W. Enhancement of citric acid production by Aspergillus niger using n-dodecane as an oxygen-vector. Process Biochem. 2000;35:1079-1083. reported the oxygen solubility in n-dodecane was 54.9 mg/L at 35 °C under atmospheric conditions, compared with 34.9 mg/L in water.
Indeed a number of researchers have used oxygen vectors to enhance oxygen solubility successfully,66 Da Silva TL, Reis A, Roseiro JC, Hewitt CJ. Physiological effects of the addition of n-dodecane as an oxygen vector during steady-state Bacillus licheniformis thermophillic fermentations perturbed by a starvation period or a glucose pulse. Biochem Eng J. 2008;42:208-216. and as a consequence, oxygen vectors have been applied in several different culture systems.55 Jianlong W. Enhancement of citric acid production by Aspergillus niger using n-dodecane as an oxygen-vector. Process Biochem. 2000;35:1079-1083.
6 Da Silva TL, Reis A, Roseiro JC, Hewitt CJ. Physiological effects of the addition of n-dodecane as an oxygen vector during steady-state Bacillus licheniformis thermophillic fermentations perturbed by a starvation period or a glucose pulse. Biochem Eng J. 2008;42:208-216.-77 Narta U, Roy S, Kanwar SS, Azmi W. Improved production of l-asparaginase by Bacillus brevis cultivated in the presence of oxygen-vectors. Bioresour Technol. 2011;102:2083-2085. Examples of common hydrocarbon oxygen vectors include n-dodecane, n-hexadecane, perfluorocarbons and numerous vegetable oils.88 Liu YS, Wu JY. Use of n-hexadecane as an oxygen vector to improve Phaffia rhodozyma growth and carotenoid production in shake-flask cultures. J Appl Microbiol. 2006;101(5):1033-1038.
9 Cascaval D, Galaction AI, Folescu E, et al. Comparative study on the effects of n-dodecane addition on oxygen transfer in stirred bioreactors for simulated, bacterial and yeasts broths. Biochem Eng J. 2006;31(1):56-66.-1010 Menge M, Mukherjee J, Scheper T. Application of oxygen vectors to Claviceps purpurea, cultivation. Appl Microbiol Biotechnol. 2001;55(4):411-416. However, it is, not uncommon that overexpressed recombinant proteins in Escherichia coli (E. coli) show a discrepancy between high specific activity and increased expression level.1111 Baneyx F, Mujacic M. Recombinant protein folding and misfolding in Escherichia coli. Nat Biotech. 2004;22:1399-1408. The expression of protein is a complicated process which is regulated by multiple-signaling pathways, including ones affected by the availability of nutrients, growth factors, and the intracellular microenvironment (ATP, NADH and NADPH).1212 Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell. 2003;115:577-590.,1313 Fu J, Wang Z, Chen T, et al. NADH plays the vital role for chiral pure d-(-)-2, 3-butanediol production in Bacillus subtilis under limited oxygen conditions. Biotechnol Bioeng. 2014;111:2126-2131.
Most studies have concerned the application of oxygen to enhance the yield of target product; however the effects of the intracellular microenvironment (ATP, NADH and NADPH) in the presence of oxygen vector during expression of a fusion protein in E. coli have been less extensively studied. Therefore, in the present study, the intracellular microenvironment response to n-dodecane in E. coli was investigated, especially the effects on the expression of fumarase.
Materials and methods
Microorganisms, plasmids and growth conditions
Growth conditions and the recombinant E. coli BL21-pET22b-fumR, which produces Rhizopus oryzae fumarase, have been previously described.1414 Song P, Li S, Ding Y, Xu Q, Huang H. Expression and characterization of fumarase (FUMR) from Rhizopus oryzae. Fungal Biol. 2011;115:49-53. The expression of fumarase was induced by adding IPTG (0 h, final concentration 1 mmol/L). Cells were grown for additional 6 h at 30 °C and were harvested by centrifugation.
Oxygen vector
When fumarase expression was induced by adding IPTG, oxygen vector (n-hexane, n-dodecane and n-hexadecane) was immediately added to the sterile fermentation broth at different volumetric fractions as appropriate.
Preparation of cell-free extracts and enzyme assays
Fumarase was purified according to our previously research,1414 Song P, Li S, Ding Y, Xu Q, Huang H. Expression and characterization of fumarase (FUMR) from Rhizopus oryzae. Fungal Biol. 2011;115:49-53. and assayed according to the method described by Kanarek and Hill.1515 Kanarek L, Hill RL. The preparation and characterization of fumarase from swine heart muscle. J Biol Chem. 1964;239:4202-4206. Protein concentration was determined using the Bradford assay.1616 Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248-254. One unit of fumarase activity was defined as the production of 1 µmol of fumarate or l-malate per minute under standard reaction conditions.
Gel electrophoresis
To determine the amount of soluble fumarase, the supernatant and the pellet fraction after cell disruption were analyzed by SDS-PAGE on a 12.5% separation gel.1717 Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680-685. A Mini-PROTEAN Tear Cell (Bio-RAD, Hercules, CA, USA) was used for electrophoresis.
Molecular weight markers were purchased from TaKaRa Bio Group. After electrophoresis, the protein bands were visualized with Coomassie Brilliant blue staining.1414 Song P, Li S, Ding Y, Xu Q, Huang H. Expression and characterization of fumarase (FUMR) from Rhizopus oryzae. Fungal Biol. 2011;115:49-53.
Intracellular ATP
The intracellular ATP concentration was analyzed by an HPLC system (Dionex P680 pump, Chromeleon controller, and Dionex UVD 170U Detector; Dionex Corporation, CA, USA).1818 Liu Y, Lv C, Xu Q, Li S, Huang H, Ouyang P. Enhanced acid tolerance of Rhizopus oryzae during fumaric acid production. Bioprocess Biosyst Eng. 2015;38:323-328. One milliliter cell suspension was harvested by centrifugation at 15,000 × g for 1 min at -8 °C and the harvested cells were mixed with 1 mL methanol at -40 °C. A reagent containing 50% perchloric acid was used to extract ATP. The solution was filtered through a 0.22 µm membrane, if necessary, and 20 µL was injected into the HPLCsystem. The mobile phase comprised phosphate buffer solution/acetonitrile/water (86:4:10) at a flow rate of 1.0 mL/min. The phosphate buffer solution was composed of NaH2PO4 10.93 g/L, Na2HPO4 3.04 g/L, and tetrabutylammonium bromide 3.22 g/L (pH 6.5). A Sepax HP-C18 column (250 mm × 4.6 mm) was used at 35 °C. The column effluent was monitored at 254 nm.
NAD(P)-NAD(P)H pool isolation and cycling assay
Samples were removed from aerobic cultures, and the dinucleotides were extracted by the method described by Vemuri.1919 Vemuri GN, Eiteman MA, McEwen JE, Olsson L, Nielsen J. Increasing NADH oxidation reduces overflow metabolism in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 2007;104:2402-2407. Recycling assays of the extracts containing specific dinucleotide species were performed in triplicate.2020 Gibon Y, Larher F. Cycling assay for nicotinamide adenine dinucleotides: NaCl precipitation and ethanol solubilization of the reduced tetrazolium. Anal Biochem. 1997;251:153-157.
Results and discussion
Effects of oxygen vectors on fumarase activity
Three oxygen vectors (n-hexane, n-dodecane and n-hexadecane) at a final concentration of 1.5% (v/v) were immediately added to the fermentation broth. The oxygen vectors added in this study were all organic solvents, which have toxicity effects on cells at high concentrations.2121 Sikkema J, Poolman B, Konings WN, de Bont JA. Effects of the membrane action of tetralin on the functional and structural properties of artificial and bacterial membranes. J Bacteriol. 1992;174:2986-2992.
22 Uribe S, Rangel P, Espinola G, Aguirre G. Effects of cyclohexane, an industrial solvent, on the yeast Saccharomyces cerevisiae and on isolated yeast mitochondria. Appl Environ Microbiol. 1990;56:2114-2119.-2323 Vermue M, Sikkema J, Verheul A, Bakker R, Tramper J. Toxicity of homologous series of organic solvents for the gram-positive bacteria Arthrobacter and Nocardia Sp. and the gram-negative bacteria Acinetobacter and Pseudomonas Sp.. Biotechnol Bioeng. 1993;42:747-758. In this study, 1.5% n-hexane was found to be toxic, and was observed to inhibit growth of the cell and cause cell lysis after 3 h. As a consequence, the concentration of n-hexane was decreased to 0.6%. The effects of oxygen vectors on cell growth and fumarase activity were investigated after 6 h of induction (Table 1). The biomass increased after adding 1.5% n-dodecane and n-hexadecane, while it was still inhibited by the 0.6% n-hexane.
Enhancement of fumarase activity was detected. With 1.5% n-dodecane, fumarase activity reached the maximum value of 12.47 U/mg, which increased by 94% compared with control experiments. Therefore, this vector was selected for the subsequent investigations.
The concentrations (0-3.5%, v/v) of the n-dodecane were added to investigate their influences on cell growth and fumarase activity. Table 2 shows that the addition of n-dodecane as an oxygen vector at low concentrations increased biomass production and fumarase activity. At concentration of 2.5% n-dodecane, the activity was higher than that observed in the concentrations of 1, 1.5 and 2, despite lower biomass, which may indicate a better folding of the enzyme. However, at volumetric fractions of n-dodecane higher than%, cell lysis and decreased fumarase activity were observed, which might have been because higher concentrations of n-dodecane are toxic to cells.
Effects of concentrations of 2.5% (v/v) n-dodecane on the time-course profiles of fumarase activity
The effect of the presence of 2.5% n-dodecane is shown in Fig. 1 illustrates that expression of the fumR gene was induced by adding IPTG; meanwhile the biomass was inhibited by IPTG. The OD600 and fumarase activity during the induction process were depicted that the n-dodecane keep the activity and biomass at higher level compared with n-dodecane absence. After 9 h of induction, the biomass and the fumarase activity both reached the maximum value. The 2.5% n-dodecane addition increased fumarase activity to 14.21 U/mg, which is 124% enhancement compared with the absence of n-dodecane.
Effect of 2.5%(v/v) n-dodecane on the time-course profiles of fumarase activity. ■, Fumarase activity without IPTG or n-dodecane; ●, fumarase activity with IPTG but without n-dodecane; ▲, fumarase activity with IPTG and n-dodecane; □, OD600 without IPTG or n-dodecane; ○, OD600 with IPTG but without n-dodecane; △, OD600 with IPTG and n-dodecane.
The addition of n-dodecane did not induce a significant change in the expression of fumR gene (Fig. 2), while the fumarase activity was increased significantly. It is possible that the accurate folding caused the higher activity. Protein folding and expression are complicated processes that are regulated by multiple-signaling pathways including one affected by the availability of nutrients, growth factors, intracellular ATP levels, internal redox state (NADH/NAD),2424 De Graef MR, Alexeeva S, Snoep JL, Teixeira de Mattos MJ. The Steady-State Internal Redox State (NADH/NAD) reflects the external redox state and is correlated with catabolic adaptation in Escherichia coli. J Bacteriol. 1999;181:2351-2357. and environmental stress.1212 Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell. 2003;115:577-590. The addition of an oxygen-vector possibly influenced oxygen transport through the respiratory chain, subsequently changing the intracellular microenvironment.
Effect of n-dodecane on the expression of fumR gene. SDS PAGE analysis of the expression levels of the fumR at 37□. The expression of the fumarase was induced by adding IPTG (final concentration 1 mM). Cells were grown for additional 6 h at 180 rpm and 37□ and were harvested by centrifugation. The cells were normalized to their end OD600. M: M: protein marker; 1: pET-22b-fumR,-IPTG, - n-dodecane; 2:pET-22b-fumR,+IPTG, - n-dodecane:3:pET-22b-fumR,+ - IPTG,+ n-dodecane.
Changes of intracellular ATP
The universal molecular carrier for biological energy is ATP, and both protein biosynthesis and folding require ATP hydrolysis. The effect of n-dodecane and IPTG on ATP concentration are shown in Fig. 3. With the absence of IPTG, the concentration of ATP was continuously rising. While IPTG and n-dodecane were both used, the concentration of ATP reached the maximum value at 6 h. And during the first 6 h of induction, the concentration of ATP increased sharply to a value 7700% higher than that obtained without an oxygen-vector, and then it dropped significantly. However, fumarase activity (Fig. 1) was increased continuously while the expression level almost kept the same after 6 h induction (Fig. 2). It has been suggested that molecular chaperones actively facilitate protein folding by using the energy of ATP hydrolysis to alter the accessible conformations for a non-native protein.2525 Lin Z, Madan D, Rye HS. GroEL stimulates protein folding through forced unfolding. Nat Struct Mol Biol. 2008;15:303-311.,2626 Kim YE, Hipp MS, Bracher A, et al. Molecular chaperone functions in protein folding and proteostasis. Annu Rev Biochem. 2013;82:323-355.
The effect of different oxygen vectors on ATP concentration. □,-IPTG -oxygen vector; ■,-IPTG-oxygen vector; △,+ n-dodecane; ▲, +n-dodecane +IPTG.
Chaperones are essential for maintaining protein-folding homeostasis and play important roles in the cellular stress response. By binding to aggregation-sensitive folding intermediates, chaperones inhibit aberrant interactions between proteins. The most intensively studied folding chaperones, such as the GroEL-ES and DnaK systems, facilitate substrate protein folding through ATP- and cofactor-driven conformational changes.2525 Lin Z, Madan D, Rye HS. GroEL stimulates protein folding through forced unfolding. Nat Struct Mol Biol. 2008;15:303-311.
26 Kim YE, Hipp MS, Bracher A, et al. Molecular chaperone functions in protein folding and proteostasis. Annu Rev Biochem. 2013;82:323-355.-2727 Saibil H. Chaperone machines for protein folding, unfolding and disaggregation. Nat Rev Mol Cell Biol. 2013;14(10):630-642. Although a number of ATP-independent chaperones have been reported, most chaperones use cycles of ATP binding and hydrolysis to assist protein folding were well studied.2727 Saibil H. Chaperone machines for protein folding, unfolding and disaggregation. Nat Rev Mol Cell Biol. 2013;14(10):630-642.,2828 Stull F, Koldewey P, Humes JR, et al. Protein folding occurs while bound to the ATP-independent chaperone Spy. Nat Struct Mol Biol. 2016;23(1):53-58. The mechanism of nucleotide sensing in group II chaperonins in eukaryotes has been revealed. Nucleotide-sensing loop (NSL) was studied as an ATP binding site. Functional analysis using NSL mutants shows a significant decrease in ATPase activity, suggesting that the NSL is involved in timing of the protein folding cycle. The binding of ATP and subsequent hydrolysis promote the closure of the multi-subunit rings where protein folding occurs.2929 Pereira JH, Ralston CY, Douglas NR, et al. Mechanism of nucleotide sensing in group II chaperonins. EMBO J. 2012;31(3):731-740.
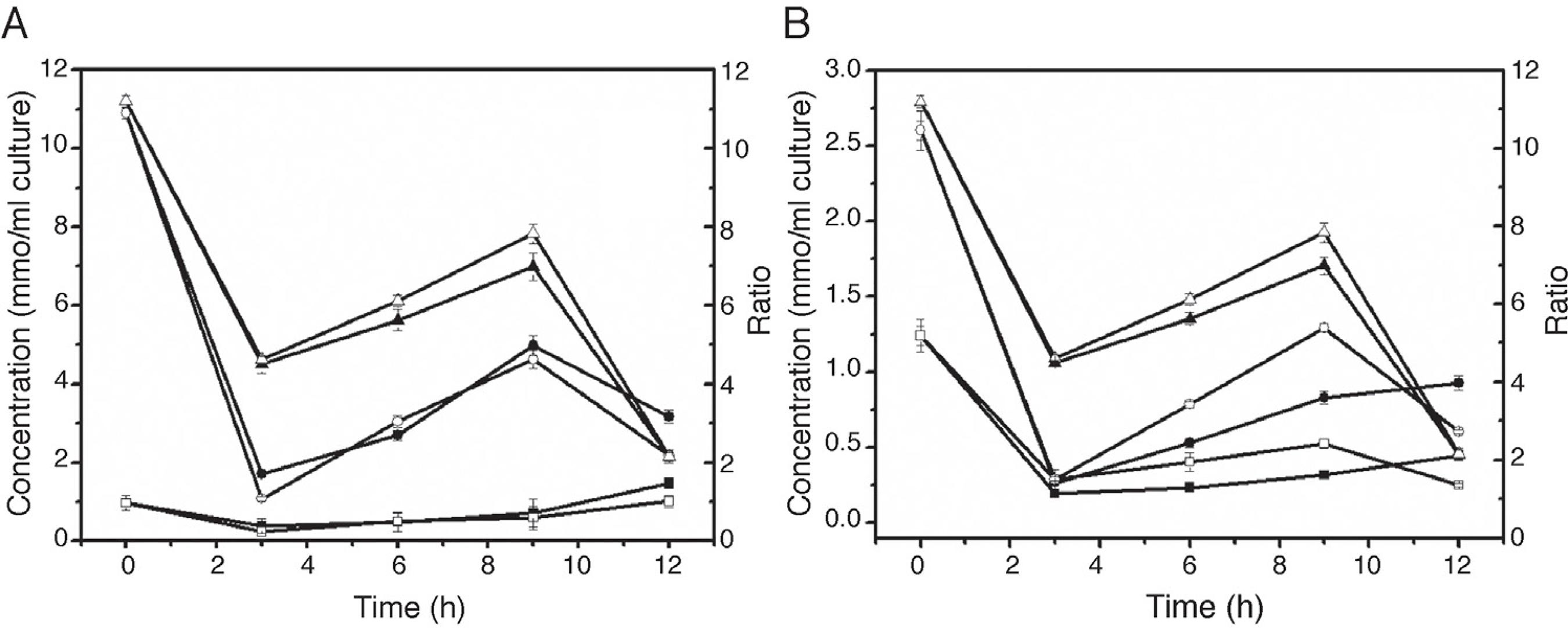
Changes of intracellular redox state
NAD, NADH, NADP and NADPH are important cofactors involved in biological redox reactions. These cofactors play an important role in preserving and regulating the intercellular redox state. In this study, the concentration changes of those cofactors were investigated after adding IPTG and n-dodecane. After adding IPTG, the concentration of NAD and NADH declined (Fig. 4A). During the induction, the concentration of NAD and NAD/NADH ratio gradually increased, reaching the maximum values at 9 h, and subsequently decreased, while the concentration of NADH increased slowly. The addition of oxygen vectors enable the intracellular NAD/NADH ratio increase more significantly. In addition, the same trend occurred in the changes in concentration of NADP, NADPH, and NADP/NADPH ratio (Fig. 4B). NAD/NADH and NADP/NADPH ratios were positively correlated with fumarase activity.
Effect of n-dodecane on intracellular redox state. A: Effect of n-dodecane on the NAD/NADH ratio. ■, the concentration of intracellular NADH without adding n-dodecane; ●, the concentration of intracellular NAD without adding n-dodecane; ▲,NAD/NADH ratio without adding n-dodecane; □, the concentration of intracellular NADH adding with n-dodecane; ○, the concentration of intracellular NAD adding with n-dodecane; △,NAD/NADH ratio adding with n-dodecane. B: Effect of n-dodecane on the NADP/NADPH ratio. ■, the concentration of intracellular NADPH without adding n-dodecane; ●, the concentration of intracellular NADP without adding n-dodecane; ▲,NADP/NADPH ratio without adding n-dodecane; □, the concentration of intracellular NADPH adding with n-dodecane; ○, the concentration of intracellular NADP adding with n-dodecane; △,NADP/NADPH ratio adding with n-dodecane.
The proper folding and stability of many proteins depend on the formation of disulfide bonds.3030 Gilbert HF. Protein disulfide isomerase and assisted protein folding. J Biol Chem. 1997;272:29399-29402. The higher NAD/NADH and NADP/NADPH ratios are more suitable for the formation of disulfide bonds, resulting in proper protein folding. Protein Data Bank (PDB) contains several fumarase crystal structures, although none of them has disulfide bonds. R. oryzae fumarase presents 6 cysteines residues, being possible the formation of disulfide bonds, differently from the others fumarase. Hence, one possibility is that the additional NADH oxidation occurs through electron transport chain (ETC) and more ATP is produced to provide enough energy for the formation of disulfide bonds, and for protein folding. NADPH can also enter the mitochondrial ETC through complex reactions. With the presence of n-dodecane, the over-express fumarase from R. oryzae by recombinant E. coli causes cells to produce more ATP for the protein expressing and folding, requiring more energy from NADH oxidation through the respiratory chain.
Conclusions
The effect of oxygen-vectors disturbance on the intracellular microenvironment of the submerge fermentation of fumarase overexpression in E. coli was investigated. In this study, the addition of n-dodecane increased fumarase activity and the concentration of ATP. Redox ratios significantly increased, while a significant change in the expression of the fumR gene was not induced (Fig. 4). Thus, on the basis of the above phenomenon, it may be inferred that the addition of an oxygen-vector changed the intracellular energy metabolism. A more oxidative environment can facilitate the correct folding of a protein that present disulfide in the surface. This fumarase possess 6 Cys that could be involved in structural disulfide formation.
In conclusion, application of n-dodecane as an oxygen vector can greatly enhance the expression of fumarase in recombinant E. coli, and the biological energy molecule ATP may play a key role in protein expression for both high expression levels and specific activities, which suggests a new strategy in the heterologous expression of protein.
Acknowledgements
This work was supported by the Natural Science Foundation of Jiangsu Province (BK 20161048), the National science fund for colleges and universities in Jiangsu Province (17KJB530006), and the Key Project of Jiangsu Collaborative Innovation Center of Chinese Medicinal Resources Industrialization.
References
-
1Haddad JJ. Oxygen sensing and oxidant/redox-related pathways. Biochem Biophys Res Commun 2004;316:969-977.
-
2Zhang DL, Guan D, Liang JB. Reducing lactate secretion by ldhA Deletion in l-glutamate-producing strain Corynebacterium glutamicum GDK-9. Braz J Microbiol 2014;45(4):1477-1483.
-
3Schlepütz T, Gerhards JP, Büchs J. Ensuring constant oxygen supply during inoculation is essential to obtain reproducible results with obligatory aerobic acetic acid bacteria in vinegar production. Process Biochem 2013;48(3):398-405.
-
4Giridhar R, Srivastava AK. Productivity enhancement in l-sorbose fermentation using oxygen vector. Enzyme Microbial Technol 2000;27:537-541.
-
5Jianlong W. Enhancement of citric acid production by Aspergillus niger using n-dodecane as an oxygen-vector. Process Biochem 2000;35:1079-1083.
-
6Da Silva TL, Reis A, Roseiro JC, Hewitt CJ. Physiological effects of the addition of n-dodecane as an oxygen vector during steady-state Bacillus licheniformis thermophillic fermentations perturbed by a starvation period or a glucose pulse. Biochem Eng J 2008;42:208-216.
-
7Narta U, Roy S, Kanwar SS, Azmi W. Improved production of l-asparaginase by Bacillus brevis cultivated in the presence of oxygen-vectors. Bioresour Technol 2011;102:2083-2085.
-
8Liu YS, Wu JY. Use of n-hexadecane as an oxygen vector to improve Phaffia rhodozyma growth and carotenoid production in shake-flask cultures. J Appl Microbiol 2006;101(5):1033-1038.
-
9Cascaval D, Galaction AI, Folescu E, et al. Comparative study on the effects of n-dodecane addition on oxygen transfer in stirred bioreactors for simulated, bacterial and yeasts broths. Biochem Eng J 2006;31(1):56-66.
-
10Menge M, Mukherjee J, Scheper T. Application of oxygen vectors to Claviceps purpurea, cultivation. Appl Microbiol Biotechnol 2001;55(4):411-416.
-
11Baneyx F, Mujacic M. Recombinant protein folding and misfolding in Escherichia coli. Nat Biotech 2004;22:1399-1408.
-
12Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell 2003;115:577-590.
-
13Fu J, Wang Z, Chen T, et al. NADH plays the vital role for chiral pure d-(-)-2, 3-butanediol production in Bacillus subtilis under limited oxygen conditions. Biotechnol Bioeng 2014;111:2126-2131.
-
14Song P, Li S, Ding Y, Xu Q, Huang H. Expression and characterization of fumarase (FUMR) from Rhizopus oryzae. Fungal Biol 2011;115:49-53.
-
15Kanarek L, Hill RL. The preparation and characterization of fumarase from swine heart muscle. J Biol Chem. 1964;239:4202-4206.
-
16Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248-254.
-
17Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970;227:680-685.
-
18Liu Y, Lv C, Xu Q, Li S, Huang H, Ouyang P. Enhanced acid tolerance of Rhizopus oryzae during fumaric acid production. Bioprocess Biosyst Eng 2015;38:323-328.
-
19Vemuri GN, Eiteman MA, McEwen JE, Olsson L, Nielsen J. Increasing NADH oxidation reduces overflow metabolism in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 2007;104:2402-2407.
-
20Gibon Y, Larher F. Cycling assay for nicotinamide adenine dinucleotides: NaCl precipitation and ethanol solubilization of the reduced tetrazolium. Anal Biochem 1997;251:153-157.
-
21Sikkema J, Poolman B, Konings WN, de Bont JA. Effects of the membrane action of tetralin on the functional and structural properties of artificial and bacterial membranes. J Bacteriol 1992;174:2986-2992.
-
22Uribe S, Rangel P, Espinola G, Aguirre G. Effects of cyclohexane, an industrial solvent, on the yeast Saccharomyces cerevisiae and on isolated yeast mitochondria. Appl Environ Microbiol 1990;56:2114-2119.
-
23Vermue M, Sikkema J, Verheul A, Bakker R, Tramper J. Toxicity of homologous series of organic solvents for the gram-positive bacteria Arthrobacter and Nocardia Sp. and the gram-negative bacteria Acinetobacter and Pseudomonas Sp.. Biotechnol Bioeng 1993;42:747-758.
-
24De Graef MR, Alexeeva S, Snoep JL, Teixeira de Mattos MJ. The Steady-State Internal Redox State (NADH/NAD) reflects the external redox state and is correlated with catabolic adaptation in Escherichia coli. J Bacteriol 1999;181:2351-2357.
-
25Lin Z, Madan D, Rye HS. GroEL stimulates protein folding through forced unfolding. Nat Struct Mol Biol 2008;15:303-311.
-
26Kim YE, Hipp MS, Bracher A, et al. Molecular chaperone functions in protein folding and proteostasis. Annu Rev Biochem 2013;82:323-355.
-
27Saibil H. Chaperone machines for protein folding, unfolding and disaggregation. Nat Rev Mol Cell Biol 2013;14(10):630-642.
-
28Stull F, Koldewey P, Humes JR, et al. Protein folding occurs while bound to the ATP-independent chaperone Spy. Nat Struct Mol Biol 2016;23(1):53-58.
-
29Pereira JH, Ralston CY, Douglas NR, et al. Mechanism of nucleotide sensing in group II chaperonins. EMBO J 2012;31(3):731-740.
-
30Gilbert HF. Protein disulfide isomerase and assisted protein folding. J Biol Chem. 1997;272:29399-29402.
Edited by
Publication Dates
-
Publication in this collection
Jul-Sep 2018
History
-
Received
2 June 2016 -
Accepted
28 Sept 2017