Abstracts
The development of cercariae from Schistosoma mansoni strains BH and SJ in Biomphalaria glabrata and Biomphalaria tenagophila treated with saccharose was studied. The molluscs were maintained in dechlorinated tap water containing 0.01% saccharose. After one week of treatment with saccharose, B. glabrata and B. tenagophila were exposed to ten S. mansoni miracidia, from BH and SJ strains respectively. Control snails of both species were maintained in dechlorinated tap water without saccharose and exposed to the same number of miracidia. There was no significant difference between the infection rates of snails treated or not with saccharose. However, the two groups of B. glabrata had significantly greater infection rates than the corresponding B. tenagophila groups. Molluscs treated with saccharose had a lower survival rate, with the greatest mortality occurring immediately before and at the beginning of cercariae release. Treatment with saccharose did not result in the release of more cercariae, but larvae from molluscs so treated showed a greater capacity to penetrate mouse skin, which was attributed to the greater energy supply during larval development in the mollusc.
Planorbidae; Biomphalaria glabrata; Biomphalaria tenagophila; Schistosoma mansoni; saccharose
Foi estudado o desenvolvimento de cercárias de Schistosoma mansoni das linhagens BH e SJ em Biomphalaria glabrata e Biomphalaria tenagophila, respectivamente, tratadas com sacarose. Os moluscos foram mantidos em água declorada contendo 0,01% de sacarose. Após uma semana de tratamento com sacarose, B. glabrata e B. tenagophila foram expostas a dez miracídios de S. mansoni das linhagens BH e SJ, respectivamente. Moluscos controles de ambas as espécies mantidos em água declorada sem sacarose foram expostos ao mesmo número de miracídios. Não houve diferença significativa entre as taxas de infecção dos moluscos tratados ou não com sacarose. Entretanto, os dois grupos de B. glabrata tiveram taxas de infecção significativamente maiores que as correspondentes aos grupos de B. tenagophila. Moluscos tratados com sacarose apresentaram menor taxa de sobrevivência, com a maior mortalidade ocorrendo imediatamente depois e no início da liberação de cercárias. O tratamento com sacarose não resultou na maior liberação de cercárias, mas larvas de moluscos tratados com esse açúcar mostraram maior capacidade de penetrar na pele de camundongo, o que foi atribuído ao maior suprimento de energia durante o desenvolvimento larval no molusco.
Planorbidae; Biomphalaria glabrata; Biomphalaria tenagophila; Schistosoma mansoni; sacarose
Influence of saccharose on the development of cercariae from Schistosoma mansoni strains BH and SJ
Influência da sacarose no desenvolvimento de cercárias das linhagens BH e SJ de Schistosoma mansoni
Bruno, T. I. B; Zanotti-Magalhães, E. M; Magalhães, L. A.; Carvalho, J. F.
Departamento de Parasitologia, Instituto de Biologia, Universidade Estadual de Campinas, UNICAMP, C.P. 6109, CEP 13083-970, Campinas, SP, Brazil
Correspondence to Correspondence to Eliana M. Zanotti-Magalhães Departamento de Parasitologia Instituto de Biologia, Universidade Estadual de Campinas, UNICAMP C.P. 6109, CEP 13083-970, Campinas, SP, Brazil E-mail: emzm@unicamp.br
ABSTRACT
The development of cercariae from Schistosoma mansoni strains BH and SJ in Biomphalaria glabrata and Biomphalaria tenagophila treated with saccharose was studied. The molluscs were maintained in dechlorinated tap water containing 0.01% saccharose. After one week of treatment with saccharose, B. glabrata and B. tenagophila were exposed to ten S. mansoni miracidia, from BH and SJ strains respectively. Control snails of both species were maintained in dechlorinated tap water without saccharose and exposed to the same number of miracidia. There was no significant difference between the infection rates of snails treated or not with saccharose. However, the two groups of B. glabrata had significantly greater infection rates than the corresponding B. tenagophila groups. Molluscs treated with saccharose had a lower survival rate, with the greatest mortality occurring immediately before and at the beginning of cercariae release. Treatment with saccharose did not result in the release of more cercariae, but larvae from molluscs so treated showed a greater capacity to penetrate mouse skin, which was attributed to the greater energy supply during larval development in the mollusc.
Key words: Planorbidae, Biomphalaria glabrata, Biomphalaria tenagophila, Schistosoma mansoni, saccharose.
RESUMO
Foi estudado o desenvolvimento de cercárias de Schistosoma mansoni das linhagens BH e SJ em Biomphalaria glabrata e Biomphalaria tenagophila, respectivamente, tratadas com sacarose. Os moluscos foram mantidos em água declorada contendo 0,01% de sacarose. Após uma semana de tratamento com sacarose, B. glabrata e B. tenagophila foram expostas a dez miracídios de S. mansoni das linhagens BH e SJ, respectivamente. Moluscos controles de ambas as espécies mantidos em água declorada sem sacarose foram expostos ao mesmo número de miracídios. Não houve diferença significativa entre as taxas de infecção dos moluscos tratados ou não com sacarose. Entretanto, os dois grupos de B. glabrata tiveram taxas de infecção significativamente maiores que as correspondentes aos grupos de B. tenagophila. Moluscos tratados com sacarose apresentaram menor taxa de sobrevivência, com a maior mortalidade ocorrendo imediatamente depois e no início da liberação de cercárias. O tratamento com sacarose não resultou na maior liberação de cercárias, mas larvas de moluscos tratados com esse açúcar mostraram maior capacidade de penetrar na pele de camundongo, o que foi atribuído ao maior suprimento de energia durante o desenvolvimento larval no molusco.
Palavras-chave: Planorbidae, Biomphalaria glabrata, Biomphalaria tenagophila, Schistosoma mansoni, sacarose.
INTRODUCTION
Adult schistosomes accumulate a considerable amount of glycogen, which is continuously produced by the worms in alternating cycles of synthesis and degradation. The rate of glucose used by schistosomes is extremely high since within a 1 h period they can take up from the surrounding medium an amount of glucose equivalent to 15%-26% of their dry body weight. Males contain more than twice as much glycogen as females (Bueding, 1950). High glycogen consumption is also observed in miracidia and cercariae.
Cheng & Snyder (1962) showed that glycogen is important for the development of trematode larvae in the snail host since stored glycogen is the main energy source, especially for motility of the cercariae. Christie et al. (1974a, b) reported that S. mansoni larvae caused nutritional drainage, including a decrease in glycogen levels, in the intermediate host Biomphalaria glabrata. Release of cercariae by the snails decreased the host's glucose requirements by 7.5%, which corresponded to the amount of glucose used by the cercariae for the synthesis of proteins, nucleic acids, and lipids. Pinheiro & Amato (1994) observed that in Bradybaena similaris infected with Eurytrema coelomaticum, the hemolymph glucose levels were lower and there was a reduction in the glycogen content of the digestive gland and cephalopodal mass.
In this work, we examined the influence of saccharose exposure on the susceptibility of B. glabrata and B. tenagophila to infection by two strains (BH and SJ) of S. mansoni. These planorbid species are important vectors of S. mansoni. B. glabrata has a wide geographical distribution in Brazil and a high susceptibility to S. mansoni infection. In contrast, B. tenagophila, which is restricted to southeastern and southern Brazil, generally shows a low susceptibility to S. mansoni infection, but is responsible for the spread of Manson's schistosomiasis in southern Brazilian states. Paraense & Corrêa (1963) described the S. mansoni strains BH and SJ based on the observation that B. glabrata from Belo Horizonte (MG, Brazil) was susceptible to the local strain of the trematode, but was resistant to infection by S. mansoni from São José dos Campos (SP, Brazil). Likewise, B. tenagophila from São José dos Campos was susceptible to the sympatric S. mansoni strain (SJ) but was resistant to infection by the trematode strain (BH) from Belo Horizonte.
MATERIAL AND METHODS
Melanic specimens of B. glabrata and B. tenagophila 6-8 mm in diameter, obtained from populations in Belo Horizonte (MG, Brazil) and São José dos Campos (SP, Brazil), respectively, were maintained in the laboratory and used in these experiments. The S. mansoni strains BH and SJ (Paraense & Corrêa, 1963) were obtained from Swiss albino mice infected with cercariae derived from trematode populations sympatric with B. glabrata and B. tenagophila, respectively.
Two experimental groups were set up for each mollusc species. Group A consisted of molluscs treated with saccharose whereas the snails in group B were not. Both groups were exposed to S. mansoni. The concentration of saccharose (União, Limeira, SP, Brazil) used was 0.01% since preliminary experiments showed that higher glucose concentrations caused high snail mortality. The appropriateness of this concentration was confirmed by the elevated glucose levels in the hemolymph of the molluscs after one week of treatment. The water of both mollusc groups was changed twice a week throughout the experiments and the snails were fed with lettuce ad libitum. Sixty specimens of B. glabrata were used in each group whereas 160 specimens of B. tenagophila were used since this species is more resistant to infection.
The parameters monitored included the prepatent period, infection and mortality rates, production of cercariae, the ability of cercariae to penetrate mice, and that of miracidia to penetrate the snails.
After one week of treatment with saccharose, the snails were exposed to 10 miracidia for 2 h in individual glass dishes containing 2 ml of dechlorinated tap water at 28ºC. The extent of miracidial penetration after this period was assessed by using a stereomicroscope to count the miracidia reamining in the water.
Twenty-one days after infection, the snails were examined weekly for three months to assess the elimination of cercariae and to determine the final snail infection rate. To observe the release of cercariae, the molluscs were placed individually in glass dishes with 2 ml of dechlorinated tap water at 28ºC for 2 h. The number of cercariae released was determined using a 2% ninhydrin solution according to Paraense & Corrêa (1989). The prepatent period was defined as the period between the date of snail infection and the beginning of cercariae release. Snail mortality was determined weekly from the beginning of the experiment. The ability of cercariae to penetrate mice was tested using larvae obtained after exposure of the molluscs to light and heat for 2 h. A suspension of 100 cercariae was placed in a test tube and the tail of the mouse was immersed in this liquid for 2 h. At the end of this period, the cercariae present in the test tubes were counted to establish the rate of cercarial penetration (Magalhães, 1969a).
Statistical analysis
The data were analyzed using version 6.08 of Statistical Analysis System (SAS®) software. For mortality rate, the Lifetest procedure was used, with the parameters being time (week), mollusc species (B. glabrata- Bg or B. tenagophila - Bt), and treatment (with saccharose (A) or without saccharose (B)). The log rank statistic was applied to the four survival curves obtained to test the hypothesis of equality in survival.
The infection rate was analysed by the Catmod (categorical modeling) procedure, which included the species, the treatment, and the interaction between them. Cercariae production was analysed using the General Linear Models (GLM) procedure, by considering the mollusc species (Bg or Bt), the treatment (A or B), and the interaction between them (Mollusc* treatment). Multiple comparisons were done using the method of Ryan-Gabriel-Welsch (SAS Institute Inc., 1989).
RESULTS
After exposure of the snails to miracidia for 2 h, no larvae were observed in the glass dishes containing B. glabrata or B. tenagophila. Tables 1 and 2 show the results of the prepatent period, the number of snails releasing cercariae, and the number of surviving snails. Cercariae release began in the fifth week after infection in both groups of B. glabrata (treated or not with saccharose) and in B. tenagophila treated with saccharose. B. tenagophila treated only with dechlorinated tap water released cercariae after the sixth week of infection.
Of 60 specimens of B. glabrata treated with saccharose, 38 (63.3%) released cercariae. Similarly, 31 (51.7%) of the 60 specimens of B. glabrata treated only with dechlorinated tap water also released cercariae. For B. tenagophila, the number of molluscs infected with S. mansoni was low (1.9%) regardless of the treatment. This difference between species was significant (p = 0.0000). Treatment with saccharose had no effect on the infection rate in either species (p = 0.5957).
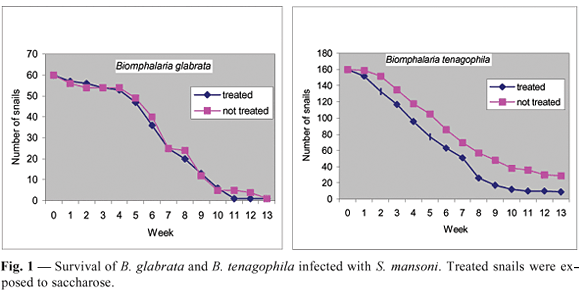
Molluscs treated with saccharose showed lower survival compared to those exposed only to chlorinated water. In this treatment, mortality was higher in the weeks that preceded the start of cercariae release (Fig. 1). The equality test applied to the survival curves showed a significant difference between them (p = 0.001).
Treatment with saccharose did not result in a greater release of cercariae by either species (p = 0.0843, Table 3). However, B. glabarata released a significantly larger number of cercariae than did B. tenagophila (p = 0.0055).
The ability of the cercariae to penetrate was tested in 20 Swiss albino mice, each of which was exposed to 100 cercariae. The cercariae used were from B. glabrata treated or not with saccharose. In the mice (n = 10) exposed to cercariae from B. glabrata treated with saccharose, only 20 of the 1000 cercariae used did not penetrate the skin, whereas in mice (n = 10) exposed to cercariae from B. glabrata not treated with saccharose, 68 cercariae out of 1000 did not penetrate. In the mice (n = 5) exposed to 100 cercariae of strain SJ from B. tenagophila treated with saccharose, a total of 23 cercariae did not penetrate, whereas in the 5 mice exposed to 100 cercariae from B. tenagophila treated with dechlorinated water, 46 cercariae did not penetrate mouse skin. Thus, the treatment with saccharose enhanced the ability of cercariae to penetrate mice (p = 0.05).
DISCUSSION
A large variety of free sugars, such as glucose, fructose, sucrose, and others released by aquatic plants may benefit organisms such as snails. Glucose is taken up mainly across the integument while maltose may be taken up by drinking, or as glucose after hydrolysis by exogenous maltase (Thomas et al., 1990). Glucose and trehalose are the major sugars in the hemolymph and digestive gland-gonad complex of B. glabrata. The levels of these sugars decreased significantly after infection with Echinostoma caproni (Perez et al., 1994).
As shown here, mollusc mortality was significantly greater in groups treated with saccharose. The death of infected snails was greater in the period preceding the release of cercariae. A high mortality of snails treated with saccharose occurred between the fifth and eighth week after infection, in the same period that cercariae were being formed and released. Since the susceptibility of the snails to S. mansoni was only verified by observing the release of cercariae, and because this observation was made only once a week, it was not possible to determine whether some snails died before releasing cercariae. This could partly explain the similar rates of snail infection in the two groups of each species. In groups treated with saccharose, there may have been a larger consumption of energy by sporocysts, with a consequent increase in larval vitality, which would have produced greater damage to the snails, thus elevating the mortality rate.
Variations in glucose concentrations occur in healthy and infected snails. However, in infected molluscs, this variation is more evident after the third week of infection, when the molluscs show a significant reduction in hemolymph glucose levels (Cheng & Lee, 1971). This depletion results from the use of glucose by the parasites and coincides with the time of formation of secondary sporocysts and cercariae (Cheng & Lee, 1971). Statistical analysis revealed no significant difference in the number of cercariae produced by the groups treated or not with saccharose. However, 38 specimens of B. glabrata treated with saccharose released 66,060 larvae compared to 37,604 cercariae produced by 31 specimens of B. glabrata not treated with saccharose.
According to Bruce et al. (1969), in their free-living phase cercariae metabolize the glycogen acquired during intramolluscan development. This observation could explain the greater ability of cercariae from molluscs treated with saccharose to penetrate mouse skin as compared to cercariae from non-sugar treated molluscs.
Our results on the susceptibility of B. glabrata and B. tenagophila exposed to the corresponding sympatric strains of S. mansoni confirmed the observations by Magalhães (1969b), who reported a larger infectivity of B. glabrata.
In summary, the exposure of B. glabrata and B. tenagophila to saccharose resulted in a larger energy reserve that negatively affected snail survival, especially during the period preceding the release of cercariae, and also significantly increased the ability of cercariae to penetrate mouse skin.
Received February 7, 2003 - Accepted December 15, 2003 - Distributed February 28, 2005
- BRUCE, J. I., WEISS, E., STIREWALT, M. A. & LINCICOME, D. R., 1969, Schistosoma mansoni: glycogen content and utilization of glucose, pyruvate, glutamate and citric acid cycle intermediates by cercariae and schistosomules. Exp. Parasitol., 26: 29-40.
- BUEDING, E., 1950, Carbohydrate metabolic of Schistosoma mansoni. J. Gen. Physiol., 33: 475-495.
- CHENG, T. C. & SNYDER, R. W., Jr., 1962, Studies on host-parasite relationships between larval trematodes and their hosts. I. A review. II. The utilization of the host's glycogen by the intramolluscan larvae of Glypthelmins pennsylvaniensis Cheng, and associated phenomena. Trans. Amer. Microsc. Soc., 81: 209-228.
- CHENG, T. C. & LEE, F. O., 1971, Glucose levels in the mollusc Biomphalaria glabrata infected with Schistosoma mansoni. J. Invert. Pathol., 18: 395-399.
- CHRISTIE, J. D., FOSTER, W. B. & STAUBER, L. A., 1974a, The effect of parasitism and starvation on carbohydrate reserves of Biomphalaria glabrata. J. Invert. Pathol., 23: 55-72.
- CHRISTIE, J. D., FOSTER, W. B. & STAUBER, L. A., 1974b, 14C up take by Schistosoma mansoni from Biomphalaria glabrata exposed to 14C-glucose. J. Invert. Pathol., 23: 297-302.
- MAGALHÃES, L. A., 1969a, Técnica para avaliação da viabilidade de penetração de cercárias de Schistosoma mansoni em Mus musculus. O Hospital, 75: 1663-1666.
- MAGALHÃES, L. A., 1969b, Estudos dos dados obtidos de uma população de Biomphalaria glabrata de Belo Horizonte infectadas por Schistosoma mansoni da mesma cidade, e de uma população de Biomphalaria tenagophila de Campinas infectada por Schistosoma mansoni de São José dos Campos. Rev. Soc. Bras. Med. Trop., 3: 195-196.
- PARAENSE, W. L. & CORRÊA, L. R., 1963, Susceptibility of Australorbis tenagophilus to infection with Schistosoma mansoni. Rev. Inst. Med. Trop. S. Paulo, 5: 23-29.
- PARAENSE, W. L. & CORRÊA, L. R., 1989, A potential vector of Schistosoma mansoni in Uruguay. Mem. Inst. Oswaldo Cruz, 84: 281-288.
- PEREZ, M. K., FRIED, B. & SHERMA, J., 1994, 1994, High performance thin-layer chromatographic analysis of sugars in Biomphalaria glabrata (Gastropoda) infected with Echinostoma caproni (Trematoda). J. Parasitol., 80: 336-338.
- PINHEIRO, J. & AMATO, S. B., 1994, Eurytrema coelomaticum (Digenea, Dicrocoleliidae): the effect of infection on carbohydrate contents of its intermediate snail host, Bradybaena similaris (Gastropoda, Xanthonychidae). Mem. Inst. Oswaldo Cruz, 89: 407-410.
- SAS INSTITUTE INC., 1989, SAS/STATÔ User's Guide, Version 6.4, Vol 1. Cary, NC, USA, 943p.
- THOMAS, J. D., KOWALCZYK, C. & SOMASUNDARAM, B., 1990, The biochemical ecology of Biomphalaria glabrata, a freshwater pulmonate mollusc: the uptake and assimilation of exogenous glucose and maltose. Comp. Biochem. Physiol., 95: 511-528.
Publication Dates
-
Publication in this collection
16 Nov 2005 -
Date of issue
Feb 2005
History
-
Accepted
15 Dec 2003 -
Received
07 Feb 2003