Abstracts
This paper presents a floristic survey of the wood component of cerrado s.s. (eastern face) and gallery forest (western face) areas carried out in the southern part of the Serra de Maracaju. The dispersal syndromes and floristic relations of this portion of the Serra were compared to those of other formations from different regions of Brazil. Between October 2007 and September 2008, monthly collections identified 144 species. As for dispersal syndromes, zoochory prevails, followed by autochory and anemochory. The gallery forest is a heterogeneous formation that shelters floristic elements shared with the Atlantic rain forest, the Amazonian forest, semi-deciduous seasonal forests and dry forests of the Pantanal. Low similarity between the two areas analyzed makes this region unique and suggests that the Serra de Maracaju is greatly influenced by the semi-deciduous seasonal forests of the southeastern region.
cerrado; floristic survey; gallery forest; floristic similarity
Ao sul da Serra de Maracaju, foi efetuada a florística do componente lenhoso de áreas de cerrado s.s. (Face Leste) e Mata de Galeria (Face Oeste), com investigações das síndromes de dispersão e das relações florísticas desta porção da Serra com outras formações em diferentes regiões do Brasil. Os dados obtidos resultaram em 144 espécies, em coletas mensais, no período de outubro de 2007 a setembro de 2008. Para as síndromes de dispersão, verificou-se predominância da zoocoria, seguida por autocoria e anemocoria. A Mata de Galeria da Serra de Maracaju é uma formação heterogênea, que detém elementos florísticos compartilhados com a Floresta Pluvial Atlântica, a Floresta Amazônica, a Floresta Estacional Semidecidual e as Florestas Secas do Pantanal. A baixa similaridade entre as áreas analisadas confere um caráter único à região e sugere que a Serra de Maracaju recebe maior influência das Florestas Estacionais Semideciduais da Região Sudeste.
cerrado; levantamento florístico; mata de galeria; similaridade florística
ECOLOGY
Floristic analysis and dispersal syndromes of woody species of the Serra de Maracaju, Mato Grosso do Sul, Brazil
Análise florística e síndromes de dispersão de espécies lenhosas da Serra de Maracaju, Mato Grosso do Sul, Brasil
Ramos, WM.I; Sartori, ALB.II,* * e-mail: albsartori@gmail.com
IPrograma de Pós-graduação em Biologia Vegetal, Universidade Federal de Mato Grosso do Sul UFMS, Campo Grande, MS, Brazil
IILaboratório de Botânica, Centro de Ciências Biológicas e da Saúde, Universidade Federal de Mato Grosso do Sul UFMS, CEP 79070-900, Campo Grande, MS, Brazil
ABSTRACT
This paper presents a floristic survey of the wood component of cerrado s.s. (eastern face) and gallery forest (western face) areas carried out in the southern part of the Serra de Maracaju. The dispersal syndromes and floristic relations of this portion of the Serra were compared to those of other formations from different regions of Brazil. Between October 2007 and September 2008, monthly collections identified 144 species. As for dispersal syndromes, zoochory prevails, followed by autochory and anemochory. The gallery forest is a heterogeneous formation that shelters floristic elements shared with the Atlantic rain forest, the Amazonian forest, semi-deciduous seasonal forests and dry forests of the Pantanal. Low similarity between the two areas analyzed makes this region unique and suggests that the Serra de Maracaju is greatly influenced by the semi-deciduous seasonal forests of the southeastern region.
Keywords: cerrado, floristic survey, gallery forest, floristic similarity.
RESUMO
Ao sul da Serra de Maracaju, foi efetuada a florística do componente lenhoso de áreas de cerrado s.s. (Face Leste) e Mata de Galeria (Face Oeste), com investigações das síndromes de dispersão e das relações florísticas desta porção da Serra com outras formações em diferentes regiões do Brasil. Os dados obtidos resultaram em 144 espécies, em coletas mensais, no período de outubro de 2007 a setembro de 2008. Para as síndromes de dispersão, verificou-se predominância da zoocoria, seguida por autocoria e anemocoria. A Mata de Galeria da Serra de Maracaju é uma formação heterogênea, que detém elementos florísticos compartilhados com a Floresta Pluvial Atlântica, a Floresta Amazônica, a Floresta Estacional Semidecidual e as Florestas Secas do Pantanal. A baixa similaridade entre as áreas analisadas confere um caráter único à região e sugere que a Serra de Maracaju recebe maior influência das Florestas Estacionais Semideciduais da Região Sudeste.
Palavras-chave: cerrado, levantamento florístico, mata de galeria, similaridade florística.
1. Introduction
Characterized as savanna-like vegetation, Cerrado, which covers 22% of the Brazilian territory (Ratter and Dargie, 1992), prevails on the Central Plateau and is the second largest plant formation (Rodrigues, 2005).
The floristic unit of cerrado seems to be especially determined by the woody flora, which is more homogeneous than the herbaceous one along the physiognomic gradient (Batalha et al., 2001), and its flora is considered the richest among the world savannas, with high levels of endemism (Ribeiro and Walter, 1998). The Cerrado is influenced by adjacent biomes with which it shares certain species (Ribeiro and Walter, 1998; Oliveira Filho and Ratter, 1995). Since it occupies a region bordered by the Chaco, the caatinga and the Amazonian and Atlantic forests, it serves as a corridor connecting them (Oliveira Filho and Ratter, 1995).
The flora of the state of Mato Grosso do Sul is considered a vegetational mosaic because of the influences of the Amazonian forest to the North, of the Atlantic forest to the East, of the Chaco to the West, of the seasonal forests of the river Paraná basin to the South and of the Central Plateau Cerrado in its center. Cerrado constitutes about 65% (Costa et al., 2003) of the natural formations of this state. It is represented by several of its different phytophysiognomies found in plains and on hill tops.
High mountain environments are little explored scientifically. In addition, high rates of deforestation contribute to the loss of valuable information on both their flora and their ecological and biogeographical aspects. Furthermore, between the hills are valleys and rock outcrops with different phytophysiognomies and more fertile soils, which possibly contribute to increasing species diversity (van den Berg and Oliveira-Filho, 2000).
Gallery forests occupy only 5% of the area covered with Cerrado (Silva Júnior, 2004). Yet, they present high species diversity, are fairly heterogeneous in terms of sheltering many elements common to the Atlantic rain forest and the Amazonian forest (Oliveira Filho and Ratter, 1995). Due to their connection to other grassland and savanna formations typical of Central Brazil (Ribeiro and Walter, 1998), gallery forests can act as corridors between forest fragments and help maintain the genetic flow.
Seed dispersal represents an important phase of the plant reproductive cycle. It is also critical to regenerate the natural populations and communities (Janzen, 1970). Dispersal mechanisms are essential to the natural distribution of species and to the movements and exchanges of genetic material within and without populations. In tropical forests, zoochory (Howe and Smallwood, 1982) prevails over abiotic syndromes while in less humid habitats, anemochory predominates (Vieira et al., 2002).
The aim of this study is to observe the dispersal syndrome of two areas, one of cerrado s.s. and one of gallery forests, and to explore the possible floristic relations in the southern part of the Serra de Maracaju. To do so, we sought to answer the following questions: 1) Which syndrome prevails in each area? 2) What are the floristic relations between the Serra de Maracaju and other Brazilian plant formations?
2. Material and Methods
The study area (Figure 1) is located in the southern part of the Serra de Maracaju, state of Mato Grosso do Sul, Brazil, where it separates the Central Plateau from the Pantanal plain (Brasil, 1997). It is covered with cerrado sensu stricto on its eastern face (20° 27' 17" S and 55° 29' 24" W) and with gallery forest on its western face (20° 26' 88" S and 55° 29' 77" W).
The study area comprises low to medium (between 2 and 15 m high) trees. The gallery forest shelters 3-30 meter high trees and it is characterized by high moisture and rich herbaceous vegetation, essentially cryptogams and epiphytes. It also presents an area with closed vegetation difficult to access, with rock outcrops and stone walls. A narrow (approximately 2.0 m wide) creek runs through the whole valley bottom before it flows into the Aquidauana River.
According to Ribeiro and Walter (1998), the concept of cerrado adopted here includes such physiognomies as forest formations (riparian forests, gallery forests, dry forests and cerradão), savannas (cerrado sensu stricto, parque de cerrado, palm groves and veredas (wet savannas) and grasslands (campos sujos open shrubby savannas), campos rupestres (rocky grasslands) and campos limpos (open grasslands).
Fertile angiosperm individuals of trees, shrubs and palms tree were collected monthly along trails, between October 2007 and September 2008, in a total area sampled of 10 ha. We classified the species in families, according to APG III (2009), and growth habit, with reference to Whittaker (1975). Collected samples were deposited at the herbarium CGMS of the Federal University of Mato Grosso do Sul, at Campo Grande. Diaspore analysis was based on the materials collected in the field and the classification of dispersal syndromes is according to van der Pijl (1982).
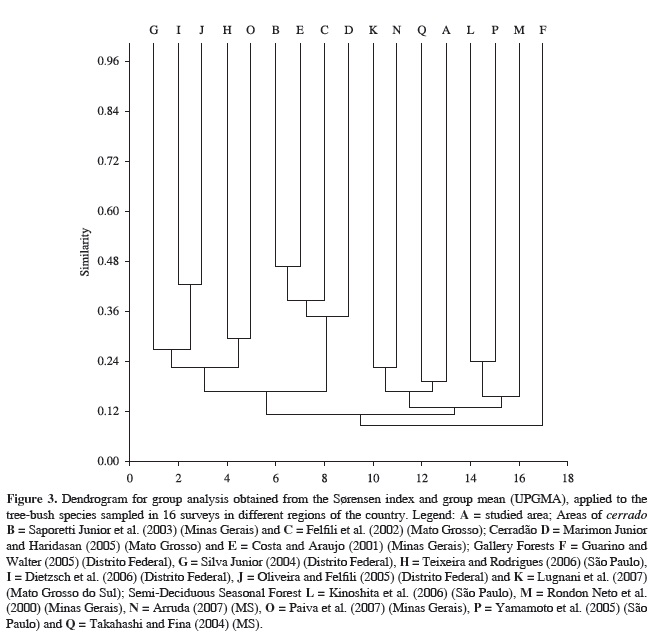
The floristic similarity between the studied gallery forest and 16 other surveys carried out in different regions (Table 1) was assessed using the Sørensen similarity index (Müller-Dombois and Ellenberg, 1974). Similarity relations were generated by the Unweighted Pair-Group Method using Arithmetic Averages (UPGMA) method with the software PAST (Hammer et al., 2001). The results were converted into a dendrogram (Sneath and Sokal, 1973).
3. Results
Floristic composition 144 species distributed into 104 genera and 45 families (Table 2) were found. Among them were 53 trees and 45 shrubs in the gallery forest and 66 trees and 43 shrubs species in the cerrado s.s.
Among the 98 species found in the gallery forest, 34 are not shared with the area of cerrado s.s. The most representative families are Fabaceae (15), Rubiaceae (6), Piperaceae (6), Malvaceae (5), Euphorbiaceae (4) and Salicaceae (4) to which 41% of the total of species belongs. The most representative genera were Piper, Casearia, Trichilia, Miconia and Bauhinia.
Forty-eight of the 109 species (Table 2) collected in the cerrado s.s. are exclusive to it, while the others also grow in the area of gallery forest. Fifty-four percent of the total species belong to the following families: Fabaceae (24), Malvaceae (9), Bignoniaceae (6), Euphorbiaceae (5), Annonaceae (5), Malphighiaceae (5) and Melastomataceae (5). The most representative genera were Miconia, Casearia, Piper, Luehea, Bauhinia, Tabebuia and Annona.
Dispersal syndromes Among the species sampled, zoochory (57.3%) prevails, followed by the autochory (22%) and anemochory (20.7%) in both habits. Some species were found to have more than one dispersal syndrome.
Among the 53 tree species collected in the gallery forest, 57.6% are zoochoric, 20.3% autochoric and 22.1% anemochoric. As for the 45 bush species, zoochory (64.5%) and autochory (25%) present similar values, while anemochory shows much lower values (10.5%) (Figure 2).
In the cerrado s.s., out of the 66 tree species, 52.4% are zoochoric, 22%, autochoric and 25.6% anemochoric. The values for the 43 bush species do not differ much: zoochory (52%), autochory (27%) and anemochory (21%) (Figure 2).
Floristic comparison The floristic list of the gallery forest studied here and that of 16 surveys of different remnants were fed into a presence/absence matrix, totaling 780 binomials. The floristic relations of the gallery forest in the southern part of the Serra de Maracaju to these other remnants were converted into a dendrogram that reveals two main groups (Figure 3). The first one comprised remnants of gallery forests and semi-deciduous seasonal forests in Mato Grosso do Sul, Minas Gerais, São Paulo and Distrito Federal, which showed low similarity (S = 0,15) among themselves and with all the other forests studied. The second one was constituted by remnants of gallery forest, semi-deciduous seasonal forest, cerrado s.s. and cerradão in the Distrito Federal, São Paulo, Minas Gerais and Mato Grosso. All the cerrado remnants were grouped and the highest similarity values were recorded for those of Minas Gerais (S = 0,48). Two gallery forests of the Distrito Federal presented the second highest similarity value (S = 0,40). There was also a grouping between remnants of gallery forest and semi-deciduous seasonal forests of the southeastern region, but it showed lower similarity values (S = 0,30).
4. Discussion
Floristic composition The species richness found in the present study (144 species) is similar to that by Mendonça et al. (1998) who surveyed woody species of Cerrado. Among the species common to the gallery forest of Central Brazil are Xylopia aromatica, Unonopsis lindmanii, Diospyros hispida, Emmotum nitens of which the two latter only occur in that region (Oliveira Filho and Ratter, 1995). Thirty-four species of this gallery forest (western face) were not found in the area of cerrado s.s. (eastern face), but have already been reported in different Brazilian plant formations: Sloanea guianensis, Myrciaria floribunda, Salacia elliptica, Andira fraxinifolia were mentioned in areas of Atlantic rain forest (Guilherme et al., 2004; Durigan et al., 2008); Diospyros obovata, Trichillia elegans, Esenbeckia grandiflora, Chrysophyllum marginatum, Aloysia virgata were registered in areas of semi-deciduous seasonal forests (Jurinitz and Jarenkow, 2003); Abuta grandifolia, Mouriri guianensis were considered as Amazonian elements that can be found in gallery forests (Prance and Schaller, 1982); Calycophylum multiflorum was cited in dry forests in the Pantanal (Prance and Schaller, 1982). Thus, elements of the gallery forest can be common to the Atlantic rain forest, the Amazonian forest (Oliveira Filho and Ratter, 1995), semi-deciduous seasonal forests (van den Berg and Oliveira-Filho, 2000) and dry forests in the Pantanal (Prance and Schaller, 1982). The occurrence of the above mentioned species suggests that the gallery forest of the Serra de Maracaju shelters floristic elements from different plant formations. In addition to these floristic elements exclusive to the western face, 64 species are shared with the adjacent cerrado (eastern face).
The area of cerrado s.s. (eastern face) shares species not only with the western face, but also with the vegetation along the Aquidauana River, which explains the high number of species sampled (109). A significant number of woody species were also reported in other Brazilian regions with areas of cerrado s.s. (Ratter et al., 1997). In cerrado s.s., the occurrence of given species as Annona crassiflora, Caryocar brasiliensis, Curatella americana, Dimorphandra mollis, Machaerium acutifolium, Qualea grandiflora, Qualea multiflora and Tabebuia aurea helps characterize this plant formation. According to Ribeiro and Walter (1998), the above mentioned species are considered as typical of cerrado s.s..
Dispersal syndromes Zoochory prevails in the woody components, which was expected since the characteristic species of all the forest layers yield zoochoric fruits (Mikich and Silva, 2001). According to Howe and Smallwood (1982), in tropical forests, between 50% and 90% of the trees and bush produce zoochoric fruits, confirming the importance of biotic agents in the genetic flow of forest habitats. The anemochoric and autochoric syndromes did not differ in percentage (in both areas) which must be a local pattern, because this region is surrounded by hills under the influence of waterways and strong winds, which favor abiotic syndromes. The results obtained for autochory differ from those found by Vieira et al. (2002), who suggested that this strategy is rarely found in cerrado s.s.. Autochory is a dispersal mechanism related only to mother plants involving the release of ripe seeds. Thus, autochoric species probably depends on a secondary seed-disperser, since many have no efficient dispersal mechanisms (van der Pijl, 1982). Some species were classified as both autochoric and zoochoric based on the initial dehiscence of their fruits that later exhibited some seed ornamentation, as arils, observed in Copaifera martii, which suggests that a secondary agent participates in its dispersal. Since they have mucilaginous or fleshy arils, Guazuma ulmifolia and Guarea Guidonia are probably dispersed by birds, as mentioned by Howe and Smallwood (1982).
Floristic comparison the low similarity rates reported for the gallery forest (western face) of the Serra de Maracaju reveal a heterogeneous area, forming groupings close to those of semi-deciduous seasonal forests and gallery forest in Mato Grosso do Sul. Based on the analysis of different tree phytophysiognomies found in Central Brazil, Felfili et al. (2002) asserted that gallery forests are the richest, and the most diverse and heterogeneous ones. They present the lowest similarity rates between themselves. Therefore, even physically closest gallery forests can present floristic compositions that differ from one to the other (Silva Júnior, 2004), indicating a vegetation mosaic (Sampaio et al., 2000). Floristic similarity analysis showed that the gallery forest studied here is more similar to the semi-deciduous seasonal forests of southeastern Brazil. This aspect is corroborated by Oliveira Filho and Ratter (1995) and van den Berg and Oliveira Filho (2000), who found strong floristic relations between the gallery forest of the central part of the Central-Western Region and the semi-deciduous seasonal forests of the Paraná River basin.
The grouping formed by the set of remnants of gallery forest, cerrado and cerradão possibly shares a species with a distribution pattern concentrated in the central region of Brazil, as suggested by Oliveira Filho and Ratter (1995). Interspersed in this grouping was a subgroup formed by gallery forests (São Paulo) and semi-deciduous seasonal forests (Minas Gerais) probably gathered because they share species whose distribution extends from southeastern Brazil to the Central Plateau.
5. Conclusions
The woody component in the southern part of the Serra de Maracaju is very dependent on the fauna for their dispersion in the gallery forest and cerrado s.s., which shows the need of conserving these communities to ensure the action of dispersers since many avoid clean areas because of the high risk of predation. The abiotic syndromes make up an important percentage and have a fundamental role in the Serra de Maracaju formed by a set of steep hills which act as physical barriers and require agents like wind, water and action of gravity so that winged fruits or no winged can be dispersed.
The low similarity among the areas analyzed gives a unique floristic characteristic to this region. This was shown to be the case with gallery forests, in other places, which were characterized as very heterogeneous forests in floristic terms, but with low similarity when compared to other areas. The similarity between the areas analyzed suggests that the Serra de Maracaju is greatly influenced by the semi-deciduous seasonal forests of the southeastern region.
Received November 28, 2011
Accepted March 20, 2012
Distributed February 28, 2013
- Angiosperm Phylogeny Group – APG III., 2009. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG III. Botanical Journal of the Linnean Society, vol. 161, p. 105-121. http://dx.doi.org/10.1111/j.1095-8339.2009.00996.x
- ARRUDA, LDO., 2007. Florística e diversidade em um fragmento de Floresta Estacional Semidecidual Aluvial em Dourados-MS. Floresta, vol. 37, no. 2, p. 189-199.
- BATALHA, MA., MANTOVANI, W. and MESQUITA JUNIOR, HN., 2001. Vegetation Structure in Cerrado Physiognomes in South-Eastern Brazil. Brazilian Journal of Biology, vol. 61, no. 3, p. 475-483. PMid:11706576. http://dx.doi.org/10.1590/S1519-69842001000300018
- Brasil. Ministério do Meio Ambiente, 1997. Plano de Conservação da Bacia do Alto Paraguai PCBAP/Projeto Pantanal. Diagnóstico dos meios físico e biótico – meio biótico Brasília: PNMA. vol. 2, tomo 3.
- COSTA, AA. and ARAÚJO, GM., 2001. Comparação da Vegetação Arbórea de Cerradão e de Cerrado na Reserva do Panga, Uberlândia, Minas Gerais. Acta Botanica Brasilica, vol. 15, no. 1, p. 63-72.
- COSTA, RB., SALLES, AT. and MOURA, HHS., 2003. Degradação de reservas florestais particulares e desenvolvimento sustentável em Mato Grosso do Sul. Interações. Revista Internacional de Desenvolvimento Local, vol. 4, no. 7, p. 41-46.
- DIETZSCH, L., REZENDE, AV., PINTO, JRR. and PEREIRA, BAS., 2006. Caracterização da Flora arbórea de dois fragmentos de Mata de galeria do Parque Canjerana, DF. Cerne, vol. 12, no. 3, p. 201-210.
- DURIGAN, G., BERNACCI, LC., FRANCO, GADC., ARBOCZ, GF., METZER, JP. and CATHARINO, ELM., 2008. Estádio sucessional e fatores geográficos como determinantes da similaridade florística entre comunidades florestais no Planalto Atlântico, Estado de São Paulo, Brasil. Acta Botanica Brasilica, vol. 22, no. 1, p. 51-62. http://dx.doi.org/10.1590/S0102-33062008000100007
- FELFILI, JM., NOGUEIRA, PE., SILVA JÚNIOR, MC., MARIMON, BS. and DELITTI, WBC., 2002. Composição Floristica e Fitossociologia do Cerrado Sentido Restrito no Municipio de Água Boa MT. Acta Botanica Brasilica, vol. 16, no. 1, p. 103-112.
- GUARINO, ESG. and WALTER, BMT., 2005. Fitossociologia de dois trechos inundáveis de Matas de Galeria no Distrito Federal, Brasil. Acta Botanica Brasilica, vol. 19, no. 3, p. 431-442. http://dx.doi.org/10.1590/S0102-33062005000300003
- GUILHERME, FAG. and MORELLATO, LPC., ASSIS, MA., 2004. Horizontal and vertical tree community structure in a lowland Atlantic Rain Forest, Southeastern Brazil. Revista Brasileira de Botânica, vol. 27, no. 4, p. 725-737.
- HAMMER, O., HARPER, DAT. and RYAN, PD., 2001. PAST: Palaeontological Statistics software package for education and data analysis. Palaeontologia Eletronica, vol. 4, no. 1, p. 9. Available from: <http://folk.uio.no/ohammer/past>
- HOWE, HF. and SMALLWOOD, J., 1982. Ecology of seed dispersal. Annual Review of Ecology and Systematics, vol. 13, p. 201-228. http://dx.doi.org/10.1146/annurev.es.13.110182.001221
- JANZEN, DH., 1970. Herbivores and the number of tree species in tropical forests. American Naturalist, vol. 104, p. 501-528. http://dx.doi.org/10.1086/282687
- JURINITZ, CF. and JARENKOW, JA., 2003. Estrutura do componente arbóreo de uma floresta estacional na Serra do Sudeste, Rio Grande do Sul, Brasil. Revista Brasileira de Botanica, vol. 26, no. 4, p. 475-487. http://dx.doi.org/10.1590/S0100-84042003000400006
- KINOSHITA, LS., TORRES, RB., FORNI-MARTINS, ER., SPINELLI, T., AHN, YJ. and CONSTÂNCIO, SS., 2006. Composição florística e síndromes de polinização e de dispersão da mata do Sítio São Francisco, Campinas, SP, Brasil. Acta Botanica Brasilica, vol. 20, no. 2, p. 313-327.
- LUGNANI, JS., RESENDE, UM. and BUENO, ML., 2007. Comparação Entre Duas Formações Vegetacionais Arbóreas do Parque Estadual do Prosa-PEP, Campo Grande, MS. Revista Brasileira de Biociências, vol. 5, no. 2, p. 453-455.
- MARIMON JUNIOR, BHM. and HARIDASAN, M., 2005. Comparação da vegetação arbórea e características edáficas de um cerradão e um cerrado sensu stricto em áreas adjacentes sobre solo distrófico no leste de Mato Grosso, Brasil. Acta Botanica Brasilica, vol. 19, no. 4, p. 913-926. http://dx.doi.org/10.1590/S0102-33062005000400026
- MENDONÇA, RC., FELFILI, JM., WALTER, BMT., SILVA JÚNIOR, MC., REZENDE, AV., FILGUEIRAS, TS. and NOGUEIRA, PE., 1998. Flora vascular do cerrado. In SANO, SM. and ALMEIDA, SDP. (Eds.). Cerrado: ambiente e flora. Planaltina: Embrapa-CPAC.
- MIKICH, SB. and SILVA, SM., 2001 Composição Florística e Fenologia das Espécies Zoocóricas de Remanescentes de Floresta Estacional Semidecidual no Centro-Oeste do Paraná, Brasil. Acta Botanica Brasilica, vol. 15, no. 1, p. 89-113.
- MÜLLER-DOMBOIS, D. and ELLENBERG, H., 1974. Aims and methods in vegetation ecology New York. John Wiley and Sons.
- OLIVEIRA, ECL. and FELFILI, JM., 2005. Estrutura e dinâmica da regeneração natural de uma mata de galeria no Distrito Federal, Brasil. Acta Botanica Brasilica, vol. 19, no. 4, p. 801-811. http://dx.doi.org/10.1590/S0102-33062005000400016
- OLIVEIRA FILHO, AT. and RATTER, JA., 1995. A study of the original of central Brazilian forest by analysis of plant species distribution patterns. Edinburgh Journal of Botany, vol. 52, p. 141-194. http://dx.doi.org/10.1017/S0960428600000949
- PAIVA, LV., ARAÚJO, GM. and PEDRONI, F., 2007. Structure and dynamics of a woody plant community of a tropical semi-deciduous seasonal forest in the "Estação Ecológica do Panga", municipality of Uberlândia, Minas Gerais, Brazil. Revista Brasileira de Botânica, vol. 30, no. 3, p. 365-373.
- PRANCE, GT. and SCHALLER, GB., 1982. Preliminary study of some vegetation types of the Pantanal, Mato Grosso, Brazil. Brittonia, vol. 34, p. 228-251. http://dx.doi.org/10.2307/2806383
- RATTER, JA. and DARGIE, TCD., 1992. An analysis of the floristic composition of 26 cerrado areas in Brazil. Edinburgh Journal of Botany, vol. 49, p. 235-250. http://dx.doi.org/10.1017/S0960428600001608
- RATTER, JA., RIBEIRO, JF. and BRIDGEWATER, S., 1997. The Brazilian cerrado vegetation and threats to its biodiversity. Annals of Botany, vol. 80, no. 3, p. 223-230. http://dx.doi.org/10.1006/anbo.1997.0469
- RIBEIRO, JF. and WALTER, BMT., 1998. Fitofisionomia do Bioma Cerrado. In SANO, SM. and ALMEIDA, SDP. (Eds.). Cerrado: ambiente e flora. Planaltina: Embrapa-CPAC. p. 89-152.
- RODRIGUES, MT., 2005. A biodiversidade dos cerrados: conhecimento atual e perspectivas, com uma hipótese sobre o papel das matas de galerias na troca faunística durante ciclos climáticos. In SCARIOT, A., SOUSA-SILVA, J. C. and FELFILI, JM. (Eds.). Cerrado: Ecologia, Biodiversidade e Conservação. Brasília: MMA. p. 235-246.
- RONDON NETO, RMR., BOTELHO, SA., FONTES, MAL., DAVIDE, AC. and FARIA, JMR., 2000. Estrutura e Composição Florística da Comunidade Arbustivo-Arbórea de uma Clareira de Origem Antrópica, em uma Floresta Estacional Semidecídua Montana, Lavras-MG, Brasil. Cerne, vol. 6, no. 2, p. 79-94.
- SAMPAIO, AB., WALTER, BMT. and FELFILI, JM., 2000. Diversidade e Distribuição de Espécies Arbóreas em Duas Matas Galeria na Micro-Bacia do Riacho Fundo, Distrito Federal. Acta Botanica Brasilica, vol. 14, no. 2, p. 197-214.
- SAPORETTI JUNIOR, AW., MEIRA NETO, JAA. and ALMADO, RP., 2003. Fitossociologia de Cerrado Sensu Stricto no Municipio de Abaté-MG. Árvore, vol. 27, no. 3, p. 413-419.
- SILVA JÚNIOR, MC., 2004, Fitossociologia e estrutura diamétrica da mata de galeria do taquara, na reserva ecológica do IBGE, DF. Árvore, vol. 28, no. 3, p. 419-428.
- SNEATH, PH. and SOKAL, RR., 1973. Numerical Taxonomy San Francisco: W. H. Freeman and Company. p. 573.
- TAKAHASHI, A. and FINA, BG., 2004. Síndromes de dispersão de sementes de uma área do morro do Paxixi, Aquidauana, MS, Brasil. In Anais do IV Simpósio sobre recursos naturais e sócio-economicos do Pantanal, 2004. Corumbá
- TEIXEIRA, AP. and RODRIGUES, RR., 2006. Análise florística e estrutural do componente arbustivo-arbóreo de uma floresta de galeria no Município de Cristais Paulista, SP, Brasil. Acta Botanica Brasilica, vol. 20, no. 4, p. 803-813. http://dx.doi.org/10.1590/S0102-33062006000400006
- VAN DEN BERG, E. and OLIVEIRA FILHO, AT., 2000. Composição florística e estrutura fitossociológica de uma floresta ripária em Itutinga, MG, e comparação com outras áreas. Revista Brasileira de Botânica, vol. 23, no. 3, p. 231-253. http://dx.doi.org/10.1590/S0100-84042000000300002
- VAN DER PIJL, L., 1982. Principles of dispersal in higher plants Berlin: Springer-Verlag. http://dx.doi.org/10.1007/978-3-642-87925-8
- VIEIRA, DLM., AQUINO, FG., BRITO, MA., FERNANDES-BULHÃO, C. and HENRIQUES, RPB., 2002. Síndromes de dispersão de espécies arbustivo-arbóreas em cerrado s. s. do Brasil Central e savanas amazônicas. Revista Brasileira de Botânica, vol. 25, p. 215-220. http://dx.doi.org/10.1590/S0100-84042002000200009
- WHITTAKER, RH., 1975. Communities and ecosystems 2nd ed. New York: Collier-Macmillan Canada, Ltd.
- YAMAMOTO, LF., KINOSHITA, LS. and MARTINS, FR., 2005. Florística dos componentes arbóreo e arbustivo de um trecho da Floresta Estacional Semidecídua Montana, município de Pedreira, SP. Revista Brasileira de Botânica, vol. 28, no. 1, p. 191-202. http://dx.doi.org/10.1590/S0100-84042005000100016
Publication Dates
-
Publication in this collection
18 Apr 2013 -
Date of issue
Feb 2013
History
-
Received
28 Nov 2011 -
Accepted
20 Mar 2012