Abstracts
Annelid worms represent a significant part of freshwater benthic communities worldwide and Oligochaeta is a particularly species-rich group. Dero (A) bimagnasetus (Naididae) previously found and described from a small marsh in Surinam in 1974, has now been found for the first time in Barra Lake, MG, Brazil. Due to the scarce biological data and absence of ecological information in the literature regarding this species we are presenting morphological information on the specimens obtained and the physical and chemical characteristics of the habitat they were found. This species occurred only in the littoral zone of Barra Lake, in muddy, low oxygen, low conductivity and low organic matter sediment. The four individuals collected ranged 3.17-4.15 mm total length; 0.25 - 0.26 mm body width and 0.16-0.21 mm3 total volume. Considering the present anthropic pressures on freshwater biota and fast biodiversity losses worldwide it is now recognized that attention must be paid to low abundance species and the urgency for preservation of their habitats.
Oligochaeta; taxonomy; new record; freshwater biota; benthos
Vermes anelídeos representam uma parte significativa das comunidades de água doce em todo o mundo e Oligochaeta é um grupo particularmente rico em espécies. Dero (A) bimagnasetus (Naididae) havia sido encontrado e descrito em um pequeno pântano no Suriname em 1974, e agora também coletado na lagoa Barra, MG, Brasil. Devido ao baixo número de espécimes coletados e à ausência de informações ecológicas sobre esta espécie, estamos apresentando informações sobre a morfometria dos espécimes obtidos e as características físicas e químicas de seu habitat. Esta espécie ocorreu apenas na região litorânea da lagoa da Barra, em sedimento lamacento e com pouca matéria orgânica, baixa concentração de oxigênio e baixa condutividade elétrica na água. Os quatro indivíduos encontrados tiveram 3,17-4,15 mm de comprimento total; 0,25-0,26 mm de largura e 0,16-0,21 mm3 de biovolume. Considerando as atuais pressões antrópicas sobre a biota de água doce e as rápidas perdas de biodiversidade em todo o mundo, é agora reconhecido que muita atenção deve ser dada às espécies pouco abundantes e que é urgentemente necessário preservar seus habitats.
Oligochaeta; taxonomia; novo recorde; biota de água de doce; bentos
1. Introduction
Aquatic Oligochaeta form an important and particularly rich-species component of benthic and pleustonic invertebrate aquatic environments around the world. There has been a considerable advance in taxonomical research for this group, but this has not so far been accompanied by ecological studies. The understanding of biological diversity in relation to its origin and maintenance requires a great deal of ecological information and there is nowadays a consensus that, in order to conserve biodiversity, both taxonomical and ecological studies must be pursued at a much faster pace.
Dero (Aulophorus) Schmarda,
1861, is well represented among the Oligochaeta occurring in Brazil, with 8 species
recorded. Only 1 species, D. (A.)
furcatus Müller, has a cosmopolitan distribution and in
Brazil it has been recorded in seven states: 1) Paraná state, in Patos
Lake, in the municipality of Taquarussu by Montanholi-Martins and Takeda (2001)MONTANHOLI-MARTINS, MC. and TAKEDA, AM., 2001. Spatial and temporal
variations of Oligochaetes of the Ivinhema River and Patos Lake in the Upper
Paraná River Basin, Brazil. Hydrobiologia, vol. 463, no. 1-3, p. 197-205.
http://dx.doi.org/10.1023/A:1013163927814.
http://dx.doi.org/10.1023/A:101316392781...
; in Ivinhema River,
between the municipalities of Taquarussu and Jateí, by Montanholi-Martins and Takeda (2001)MONTANHOLI-MARTINS, MC. and TAKEDA, AM., 2001. Spatial and temporal
variations of Oligochaetes of the Ivinhema River and Patos Lake in the Upper
Paraná River Basin, Brazil. Hydrobiologia, vol. 463, no. 1-3, p. 197-205.
http://dx.doi.org/10.1023/A:1013163927814.
http://dx.doi.org/10.1023/A:101316392781...
and Behrend et al. (2009)BEHREND, RDL., FERNANDES, SEP., FUJITA, DS. and TAKEDA, AM., 2009.
Eight years of monitoring aquatic Oligochaeta from the Baía and Ivinhema
Rivers. Brazilian Journal of Biology, vol. 69, no. 2, supl. 0, p. 559-571.
http://dx.doi.org/10.1590/S1519-69842009000300011.
PMid:19738963
http://dx.doi.org/10.1590/S1519-69842009...
; in Baía
River, in the municipality of Batayporã by Behrend et al. (2009)BEHREND, RDL., FERNANDES, SEP., FUJITA, DS. and TAKEDA, AM., 2009.
Eight years of monitoring aquatic Oligochaeta from the Baía and Ivinhema
Rivers. Brazilian Journal of Biology, vol. 69, no. 2, supl. 0, p. 559-571.
http://dx.doi.org/10.1590/S1519-69842009000300011.
PMid:19738963
http://dx.doi.org/10.1590/S1519-69842009...
; 2) São Paulo state, in
Pinheiros and Jardim Europa districts, in the municipality of São Paulo by
Marcus (1943)MARCUS, E., 1943. Sobre Naididae do Brasil. Boletim de Zoologia,
vol. 32, no. 7, p. 3-181.; in ornamental
fish breeding ponds, associated with the gastropod Pomacea
bridgesii Reeve, in the municipality of Araraquara by Gorni and Alves (2006)GORNI, GR. and ALVES, RD., 2006. Naididae (Annelida,
Oligochaeta) associated with Pomacea bridgesii (Reeve)
(Gastropoda, Ampullaridae). Revista Brasileira de Zoologia, vol. 23,
no. 4, p. 1059-1061.
http://dx.doi.org/10.1590/S0101-81752006000400011.
http://dx.doi.org/10.1590/S0101-81752006...
; in Tietê
River, in the municipality of Barra Bonita by Suriani et al. (2007)SURIANI, AL., FRANÇA, RS., PAMPLIN, PA., MARCHESE, M., LUCCA,
JV. and ROCHA, O., 2007. Species richness and distribution of oligochaetes in
six reservoirs on Middle and low Tietê River (SP, Brazil). Acta
Limnologica Brasiliensia, vol. 19, no. 4, p. 415-426.; in Tietê River, in the
municipality of Bariri by Pamplin et al.
(2005)PAMPLIN, PAZ., ROCHA, O. and MARCHESE, M., 2005. Riqueza de
espécies de Oligochaeta (Anellida, Clitellata) em duas represas
do rio Tietê (São Paulo). Biota Neotropica, vol. 5, no. 1,
p. 63-70.
http://dx.doi.org/10.1590/S1676-06032005000100007.
http://dx.doi.org/10.1590/S1676-06032005...
; 3) Mato Grosso state, in Rio Negro River
(Pantanal biome) by Takeda et al.
(2000)TAKEDA, AM., PEREIRA, MCF. and BARBOSA, FAR., 2000. Zoobenthos
survey of the Pantanal, Mato Grosso do Sul, Brasil. Bulletin of Biological
Assessment and Engineering, vol. 18, no. 3, p. 49-55.; 4) Pernambuco state, in the municipality of Cabo
by Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.;
in the municipality of Bom Jardim by Marcus
(1944)MARCUS, E., 1944. Sobre Oligochaeta límnicos do Brasil. Boletim
de Zoologia, vol. 43, no. 8, p. 5-101. and Du Bois-Reymond
Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in the municipality of Mercês by Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in the
municipality of Salgadinho by Du Bois-Reymond Marcus
(1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; 5) Pará state, in the municipality of
Fordlândia by Du Bois-Reymond Marcus
(1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949aDU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and
tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural
de Montevideo, vol. 3, no. 51, p. 1-15., b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; in Cururú River, an indigenous
land of Munduruku (a tributary of Tapajós River), in the municipality
of Jacareacanga by Du Bois-Reymond Marcus
(1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in Acará River, in the municipality of Óbidos
by Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.,1949b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; in the south of Belém city by
Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.;
in the municipality of Itaituba by Du Bois-Reymond
Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; and in the municipality of Humaitá by Marcus (1943)MARCUS, E., 1943. Sobre Naididae do Brasil. Boletim de Zoologia,
vol. 32, no. 7, p. 3-181.; Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949aDU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and
tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural
de Montevideo, vol. 3, no. 51, p. 1-15., b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; 6)
Minas Gerais state, in São Pedro stream, in the municipality of Juiz de Fora by
Stephan (2007)STEPHAN, NNC., 2007. Distribuição Espacial e Temporal dos
insetos e Oligochaeta aquáticos da Sub-Bacia do córrego São
Pedro. Juiz de Fora: Universidade Federal de Juiz de Fora. 61 p.
Dissertação de Mestrado em Ciências Biológicas,
Comportamento e Biologia Animal.; in Piumhi
River, in the municipality of Piumhi by Suriani-Affonso et al. (2011)SURIANI-AFFONSO, AL., FRANÇA, RS., MARCHESE, M. and ROCHA, O.,
2011. Environmental factors and benthic Oligochaeta (Annelida,
Clitellata) assemblages in a stretch of the Upper São Francisco River
(Minas Gerais state, Brazil). Brazilian Journal of Biology, vol. 71,
no. 2, p. 437-446. PMid:21755161.; Rio Grande do Sul state, in a
rice field channels, in the municipality of Mostarda by Stenert (2009)STENERT, C., 2009. Estrutura da comunidade de macroinvertebrados
aquáticos em arrozais do Rio Grande do Sul. São Carlos: Universidade
Federal de São Carlos. 205 p. Tese de Doutorado em Ecologia e Recursos
Naturais..
Another 3 species, D. (A.)
lodeni Brinkhurst, D.
(A.) vagus Leidy and
D. (A.) hymanae
Naidu, can be considered widely distributed. In Brazil, D.
(A.) lodeni was recorded in 3
states: 1) São Paulo state, in Tietê River, in the municipality of
Bariri by Pamplin et al. (2005)PAMPLIN, PAZ., ROCHA, O. and MARCHESE, M., 2005. Riqueza de
espécies de Oligochaeta (Anellida, Clitellata) em duas represas
do rio Tietê (São Paulo). Biota Neotropica, vol. 5, no. 1,
p. 63-70.
http://dx.doi.org/10.1590/S1676-06032005000100007.
http://dx.doi.org/10.1590/S1676-06032005...
and
Suriani et al. (2007)SURIANI, AL., FRANÇA, RS., PAMPLIN, PA., MARCHESE, M., LUCCA,
JV. and ROCHA, O., 2007. Species richness and distribution of oligochaetes in
six reservoirs on Middle and low Tietê River (SP, Brazil). Acta
Limnologica Brasiliensia, vol. 19, no. 4, p. 415-426.; in
Tietê River, in the municipality of Barra Bonita by Suriani et al. (2007)SURIANI, AL., FRANÇA, RS., PAMPLIN, PA., MARCHESE, M., LUCCA,
JV. and ROCHA, O., 2007. Species richness and distribution of oligochaetes in
six reservoirs on Middle and low Tietê River (SP, Brazil). Acta
Limnologica Brasiliensia, vol. 19, no. 4, p. 415-426.; in Infernão Lake
(Ecological Station of Jataí), in the municipality of Luís
Antônio by Correia and Trivinho-Strixino
(1998)CORREIA, LCS. and TRIVINHO-STRIXINO, S., 1998. Macroinvertebrados da
rizosfera de Scirpus cubensis na Lagoa do Infernão (Estação
Ecológica de jataí-SP): Estrutura e função. Acta
Limnologica Brasiliensia, vol. 10, no. 1, p. 37-47. and Trivinho-Strixino et
al. (2000)TRIVINHO-STRIXINO, S., CORREIA, LCS. and SONODA, K., 2000.
Phytophilous chironomidae (Diptera) and other macroinvertebrates in
the ox-bow Infernão Lake (Jatai Ecological Station, Luiz Antonio, SP,
Brazil). Revista Brasileira de Biologia, vol. 60, no. 3, p. 527-535.
http://dx.doi.org/10.1590/S0034-71082000000300018.
PMid:11188879
http://dx.doi.org/10.1590/S0034-71082000...
; 2) Pará state, in the municipality of
Fordlândia by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253.;
Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949a)DU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and
tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural
de Montevideo, vol. 3, no. 51, p. 1-15.; in Cupari River, in the
municipality of Óbidos by Marcus
(1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253.; Du Bois-Reymond Marcus
(1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949aDU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and
tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural
de Montevideo, vol. 3, no. 51, p. 1-15., b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; 3) Paraná state, in
Baía River, in the municipality of Baytaporã by Behrend et al. (2009)BEHREND, RDL., FERNANDES, SEP., FUJITA, DS. and TAKEDA, AM., 2009.
Eight years of monitoring aquatic Oligochaeta from the Baía and Ivinhema
Rivers. Brazilian Journal of Biology, vol. 69, no. 2, supl. 0, p. 559-571.
http://dx.doi.org/10.1590/S1519-69842009000300011.
PMid:19738963
http://dx.doi.org/10.1590/S1519-69842009...
.
D. (A.) vagus was recorded in three states, only: 1) Minas Gerais state, in Timboré Lake (at the natural lakes system of Middle Rio Doce Valley), in the municipality of Dionísio by Campitelli-Ramos (2008); 2) Rio Grande do Sul state, in a rice field channel, in the municipality of Mostarda by Stenert (2009)STENERT, C., 2009. Estrutura da comunidade de macroinvertebrados aquáticos em arrozais do Rio Grande do Sul. São Carlos: Universidade Federal de São Carlos. 205 p. Tese de Doutorado em Ecologia e Recursos Naturais.; 3) Mato Grosso do Sul state, in Rio Negro River (Pantanal biome) by Takeda et al. (2000)TAKEDA, AM., PEREIRA, MCF. and BARBOSA, FAR., 2000. Zoobenthos survey of the Pantanal, Mato Grosso do Sul, Brasil. Bulletin of Biological Assessment and Engineering, vol. 18, no. 3, p. 49-55.. D. (A.) hymanae was recorded only in São Paulo state in Tietê River, in the municipality of Promissão by Suriani et al. (2007)SURIANI, AL., FRANÇA, RS., PAMPLIN, PA., MARCHESE, M., LUCCA, JV. and ROCHA, O., 2007. Species richness and distribution of oligochaetes in six reservoirs on Middle and low Tietê River (SP, Brazil). Acta Limnologica Brasiliensia, vol. 19, no. 4, p. 415-426..
The remaining 4 species are endemic to the Neotropical region: D. (A.) bimagnasetus, D. (A.) costatus Du Bois-Reymond Marcus and D. (A.) carteri Stephenson, or to the American continents: D. (A.) borellii Michaelsen. According to Harman (1974)HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36., D. (A.) is one of the oldest taxa among Neotropical Oligochaeta and this region may be its centre of dispersal, and also a centre of origin. In Brazil, D. (A.) bimagnasetus was only recorded in Minas Gerais state, in Barra Lake (at the natural lake system of Middle Rio Doce Valley), in the municipality of Dionísio by Campitelli-Ramos (2008).
Dero (A.) hymanae and D. (A.) costatus were recorded only in São Paulo state: in Pinheiros River, in the municipality of São Paulo by Marcus (1942MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de Zoologia, vol. 25, no. 6, p. 153-253., 1943)MARCUS, E., 1943. Sobre Naididae do Brasil. Boletim de Zoologia, vol. 32, no. 7, p. 3-181. and Du Bois-Reymond Marcus (1944)DU BOIS-REYMOND MARCUS, E., 1944. Notes on fresh-water Oligochaeta from Brazil. Comununicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 1, no. 20, p. 1-8.; in Lagoa Dourada Reservoir associated with the sponge Metania spinata Carter and with the submerged macrophytes Mayaca fluviatilis (Mayacaceae) and Najas sp. (Najadaceae), in the municipality of Itirapina, by Gorni and Alves (2008)GORNI, GR. and ALVES, RG., 2008. Naididae species (Annelida: Oligochaeta) associated with the sponge Metania spinata (Carter, 1881) (Porifera: Metaniidae) from a southeastern Brazilian reservoir. Acta Limnologica Brasiliensia, vol. 20, no. 3, p. 261-263..
D. (A.) carteri was recorded in 4 states in Brazil: 1) São Paulo state, in the Emas Reservoir, in the municipality of Pirassununga by Marcus (1943)MARCUS, E., 1943. Sobre Naididae do Brasil. Boletim de Zoologia, vol. 32, no. 7, p. 3-181.; 2) Piauí state, in the Caicós Reservoir by Marcus (1944)MARCUS, E., 1944. Sobre Oligochaeta límnicos do Brasil. Boletim de Zoologia, vol. 43, no. 8, p. 5-101. and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20.; 3) Pernambuco state, in the municipality of Recife, in Capoeira and São Bento district by Marcus (1944)MARCUS, E., 1944. Sobre Oligochaeta límnicos do Brasil. Boletim de Zoologia, vol. 43, no. 8, p. 5-101.; in the municipality of Goiana by Marcus (1944)MARCUS, E., 1944. Sobre Oligochaeta límnicos do Brasil. Boletim de Zoologia, vol. 43, no. 8, p. 5-101.; 4) Pará state, in the municipality of Fordlândia by Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20., 1949aDU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 3, no. 51, p. 1-15., b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de Montevideo, vol. 3, no. 56, p. 1-6.; in Cururú River, in the municipality of Jacareacanga by Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20.; in São Manuel River, in the municipality of Óbidos by Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20., in Cuminá River by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de Zoologia, vol. 25, no. 6, p. 153-253.; Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20., in Salgado Lake by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de Zoologia, vol. 25, no. 6, p. 153-253. and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20.; in Alemquer river by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de Zoologia, vol. 25, no. 6, p. 153-253. and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20..
D. (A.)
borellii was recorded in 6 states in Brazil: 1) São
Paulo state, in Santa Clara stream, in the municipality of Araraquara by Alves and De Lucca (2000)ALVES, RG. and DE LUCCA, JV., 2000. Oligochaeta (Annelida:
Clitellata) como indicador de poluiçao orgânica em dois
córregos pertencentes a Bacia do Ribeirao do Ouro Araraquara (Sao
Paulo-Brasil). Brazilian Journal of Ecology, vol. 4, no. 1-2, p.
112-117.; in
Infernão Lake, in the municipality of Luís Antônio by Trivinho-Strixino et al. (2000)TRIVINHO-STRIXINO, S., CORREIA, LCS. and SONODA, K., 2000.
Phytophilous chironomidae (Diptera) and other macroinvertebrates in
the ox-bow Infernão Lake (Jatai Ecological Station, Luiz Antonio, SP,
Brazil). Revista Brasileira de Biologia, vol. 60, no. 3, p. 527-535.
http://dx.doi.org/10.1590/S0034-71082000000300018.
PMid:11188879
http://dx.doi.org/10.1590/S0034-71082000...
;
2) Mato Grosso do Sul state, in Negro River (Pantanal Biome) by Takeda et al. (2000)TAKEDA, AM., PEREIRA, MCF. and BARBOSA, FAR., 2000. Zoobenthos
survey of the Pantanal, Mato Grosso do Sul, Brasil. Bulletin of Biological
Assessment and Engineering, vol. 18, no. 3, p. 49-55.; 3) Mato
Grosso state by Michaelsen (1900)MICHAELSEN, W., 1900. Oligochaeta. Berlin: R. Friedländer und
Sohn. Das Tierreich, no. 10.;
4) Pará state, in Tapajós river by Du
Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in the municipality of
Fordlândia by Du Bois-Reymond Marcus
(1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949aDU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and
tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural
de Montevideo, vol. 3, no. 51, p. 1-15., b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; in Cururú river, in the
municipality of Jacareacanga by Du Bois-Reymond
Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in the municipality of Santarém by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253. and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.; in
Trombetas River, in the municipality of Óbidos by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253. and Du
Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., in Acará river by Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6., in Cuminá River by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253. and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., in
Salgado Lake by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253.;
Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.,
in Alemquer River by Marcus (1942)MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de
Zoologia, vol. 25, no. 6, p. 153-253.
and Du Bois-Reymond Marcus (1947)DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20.;
5) Amazon state, in the municipality of Macaés by Du Bois-Reymond Marcus (1947DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from
Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo,
vol. 2, no. 44, p. 1-20., 1949b)DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of
Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de
Montevideo, vol. 3, no. 56, p. 1-6.; 6) Minas Gerais state, in São Pedro
Stream, in the municipality of Juiz de Fora by Stephan (2007)STEPHAN, NNC., 2007. Distribuição Espacial e Temporal dos
insetos e Oligochaeta aquáticos da Sub-Bacia do córrego São
Pedro. Juiz de Fora: Universidade Federal de Juiz de Fora. 61 p.
Dissertação de Mestrado em Ciências Biológicas,
Comportamento e Biologia Animal..
In this paper we report the first occurrence of Dero (A.) bimagnasetus in Brazil, describing some of its morphological characteristics and the environmental conditions of the habitat in which this species was found. Before the present record, this species was only recorded within the Neotropical region in a small marsh located in the Surinam (Harman, 1974HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36.), its type locality.
2. Material and Methods
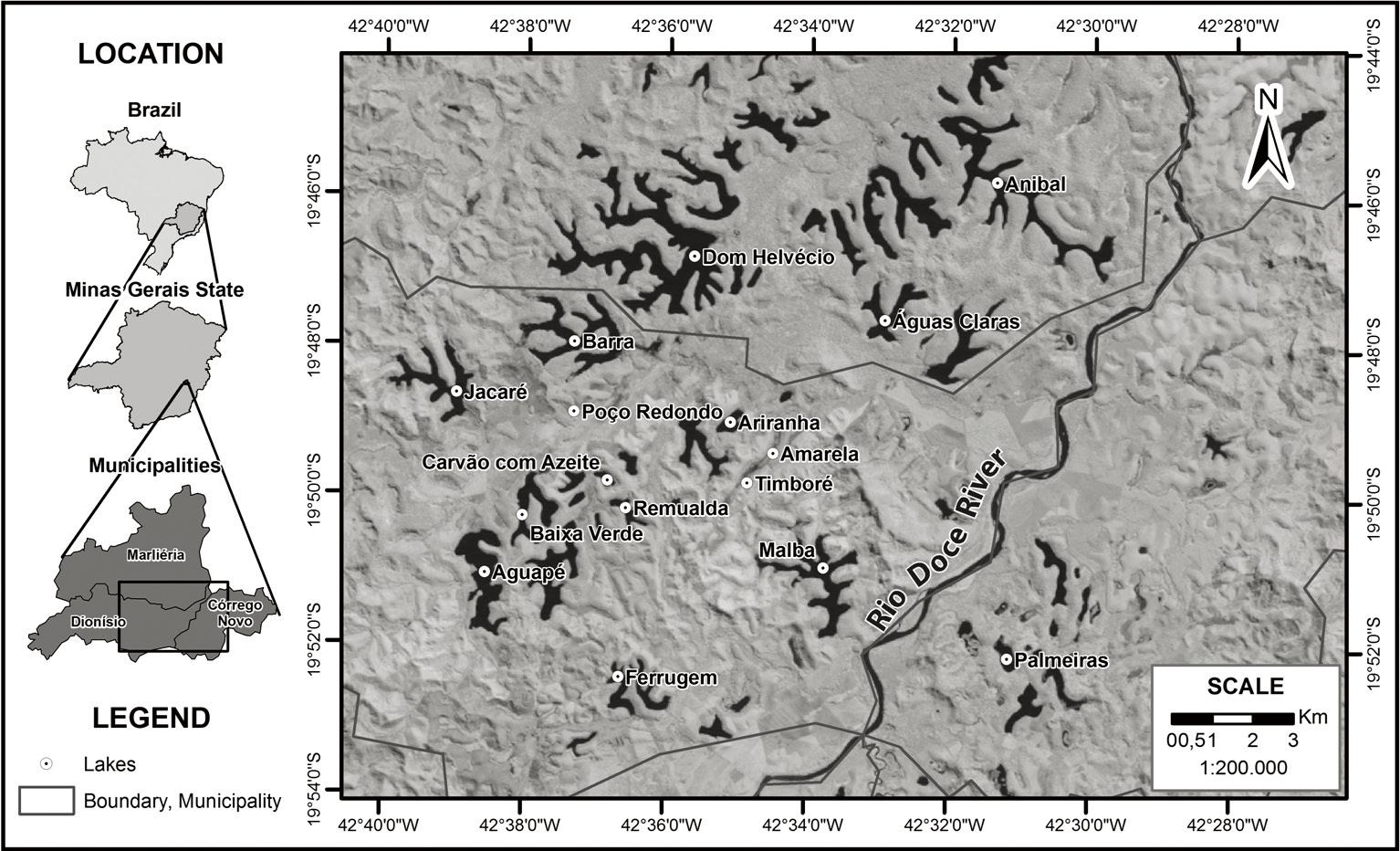
Barra Lake (see in Figure 1) is a relatively large lake in the Middle Rio Doce Valley, Minas Gerais state, Brazil (19° 48′ 00.2″ S; 42° 37′ 17.3″ W). The lake is surrounded by Eucalyptus forest from reforestation, mixed with native Atlantic forest regrowth.
Schematic drawing and aerial view showing Barra Lake location in Minas Gerais state, Brazil. Source: LANDSAT 5 TM, National Institute for Space Research (INPE). http://www.inpe.br/
Sampling was carried out at 5 sites on the lake, 1 in the limnetic and 4 in the littoral zone, on one sampling date, March 03, 2004, in the morning (08:00 h - 11:00 h). Geographical coordinates were recorded with a GPS Garmin 130.
Water pH, electrical conductivity and dissolved oxygen parameters were measured at 3 depths in the water column and on the bottom with the multi-probe sensor Horiba U-10. Water samples for the analysis of total phosphorus and nitrogen concentrations were collected at the surface layer with a Van Dorn bottle, stored in polyethylene bottles and frozen shortly thereafter. The total nitrogen and total phosphorus concentrations were determined in the laboratory, according to the procedures described in Mackereth et al. (1978)MACKERETH, FJH., HERON, J. and TALLING, FJ., 1978. Water analysis: some revised methods for limnologists. Freshwater Biological Association Scientific Publication. Kendall: Titus Wilson e Sons. Freshwater Biological Association Scientific Publication, no. 36..
Sediment samples were taken with a Van Veen grab (337 cm2) for both benthic macroinvertebrates and sediment particle size analysis. The organic matter content of the sediment was determined as in Buchman and Brady (1979)BUCHMAN, HO. and BRADY, NC., 1979. Natureza e propriedades do solo. 5th ed. Rio de Janeiro: Freitas Bastos. and sand, silt and clay fractions were determined as described by Suguio (1973)SUGUIO, K., 1973. Introdução à Sedimentologia. São Paulo: Editora Edgar Blucher Ltda., and this information was used to classify sediment texture (Camargo et al., 1987CAMARGO, MN., KLAMT, E. and KAUFFMAN, JH., 1987. Classificação de solos usada em levantamentos pedológicos no Brasil. Sociedade Brasileira de Ciência do Solo, Campinas, vol. 12, p. 11-33.).
Two sediment samples were taken in each sampling site. Samples were sieved through a net of 0.21 mm mesh. The retained material was preserved in 8% formaldehyde solution. In the laboratory, organisms were sorted out and identified under a Zeiss Stemi SV 6 stereo-microscope and Zeiss Axioskop 2 Plus microscope, using specialised literature, mainly the taxonomical keys of Righi (1984)RIGHI, G., 1984. Oligochaeta. Manual de Identificação de Invertebrados Límnicos do Brasil, Brasília, CNPq, 48 p. and Brinkhurst and Marchese (1989)BRINKHURST, RO. and MARCHESE, MR., 1989. Guia para la identificación de oligoquetos acuáticos continentales de Sud y Centroamerica. 2th ed. Santo Tomé: Associación de Ciencias Naturales del Litoral..
Specimens were photographed and measured in a Zeiss Axiocam HCr
microscope with an image acquisition system. Individual biovolumes were calculated
from linear dimensions using the cylinder equation (Sun and Liu, 2003SUN, J. and LIU, D., 2003. Geometric models for calculating cell
biovolume and surface area for phytoplankton. Journal of Plankton Research, vol.
25, no. 11, p. 1331-1346.
http://dx.doi.org/10.1093/plankt/fbg096.
http://dx.doi.org/10.1093/plankt/fbg096...
).
3. Results
Dero (A.) bimagnasetus (Figure 2) specimens were found among the macroinvertebrates collected at site 4, one of the four littoral sites sampled in Barra Lake. Since this is the first record of this species in Brazilian freshwater environments, a general characterisation of the physical and chemical conditions in site 4 is presented (as shown in Table 1).
General view of Dero (Aulophorus) bimagnasetus under optical microscope at 50 times magnification.
Values of pH, electrical conductivity (Cond), temperature (Temp) and concentrations of dissolved oxygen (DO), total nitrogen (N) and total phosphorus (P) in the water column at site 4 (S: 19° 48′ 002″, W: 42° 37′ 173″) in the littoral zone of Barra Lake, Middle Rio Doce Valley, Minas Gerais State, Brazil. X: Average; SD: standard deviation.
The water pH was nearly neutral, and the conductivity was low. The dissolved oxygen concentration at point 4 was significantly lower than at the other sites. Also, there was a dense stand of macrophytes (Eleocharis sp., Nymphaea sp. and Utricularia sp.) at this site, not seen at any other sampling site in the littoral zone of the lake (Campitelli-Ramos, 2008). In the previous record of this species in Surinam, D. (A.) bimagnasetus was first recorded among rotting plant debris (Harman, 1974HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36.). However, in Barra Lake, D. (A.) bimagnasetus was collected from muddy sediment with little organic matter, as shown in Table 2.
Sediment fractions (sand, silt and clay), percent organic matter and textural classification of sediment at site 4, Barra Lake, Middle Rio Doce Valley, Minas Gerais State, Brazil, where individuals of Dero (Aulophorus) bimagnasetus were found.
Only 4 individuals of this species were found in the littoral zone of Barra lake, and linear dimensions and biovolumes of the specimens are presented (as shown in Table 3). The total length varied from 3.17 to 4.15 mm; width from 0.25 to 0.32 mm and biovolume from 0.16 to 0.35 mm3.
Length, width and biovolume of four individuals of Dero (Aulophorus) bimagnasetus found in the sediment collected at site 4, littoral region of Barra Lake, Middle Rio Doce Valley, Minas Gerais State, Brazil. SD: standard deviation.
4. Discussion
D. (A.) bimagnasetus was among the less abundant species sampled in the Barra Lake, which could be the result of a small number of samples taken, instead of an actual species rarity. They were also among the smallest sized organisms in the benthic assemblages when compared with other macroinvertebrates collected in the sediment at site 4.
The distinctive morphological feature of D. (A.) bimagnasetus is the occurrence in the ventral bundles of III of a single giant chaetae and 1 or 2 giant chaetae in the ventral bundle of IV (see in Figure 3 and Figure 4). Among the Naidinae, this character also appears in other species, e.g., Pristina aequiseta Bourne. However, in the genus Dero no other species possess such giant chaetae (Brinkhurst and Wetzel, 1984BRINKHURST, RO. and WETZEL, MJ., 1984. Aquatic Oligochaeta of the world: Supplement. A catalogue of new freshwater species, descriptions and revisions. Canada: Fisheries and Oceans. p. 44-101. Canadian Technical Report of Hydrography and Ocean Sciences, no. 44.).
Dero (Aulophorus) bimagnisetus setae. A: normal ventral seta of II; B: giant ventral seta of III; C: giant ventral seta of IV; D: normal posterior ventral seta; E: needle seta. Modified from Harman (1974)HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36..
Detailed view of Dero (Aulophorus) bimagnasetus giant setae located at segments III (A) and VI (B) (pair), under optical microscope.
D. (A.) bimagnasetus shares with its congeners D. (A.) furcatus, D. (A.) borellii, D. (A.) lodeni, D. (A.) hymanae and D. (A.) indicus Naidu, the presence of a bifid or pectinate needle chaetae, beginning at segment V. However, according to Harman (1974)HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36., who described this species, the presence of giant chaetae in the ventral bundles indicates a possible sexual reproductive mode, not previously found in this genus. The name of this species is therefore related to the very large ventral chaetae on segments III and IV.
The fact that up to now D. (A.) bimagnasetus had been found only in Surinam and now in Southeastern Brazil, suggests that this might be a low abundance species. Considering the increasing anthropic pressure on freshwater habitats and consequent biodiversity losses by habitat destruction, a great attention should be paid to this species and its habitat, in order to preserve it, as well the all other components of benthos community in Barra Lake. We also expect that reporting its occurrence in Brazil and its distinctive features may contribute to finding it in other localities, and to get a deeper knowledge of its ecological requirements.
Acknowledgements – The authors would like to thank the Brazilian Federal Research and Education Council (CNPq) for the financial support for the research (PROBIO/MMA, Covenant 952/02) and scholarship (process number: 134419/2006-5) and FAPESP for the scholarship (process number: 06/54264-5). We also thank Ricardo Milanetti Degani for his help with map elaboration and image edition.
References
- ALVES, RG. and DE LUCCA, JV., 2000. Oligochaeta (Annelida: Clitellata) como indicador de poluiçao orgânica em dois córregos pertencentes a Bacia do Ribeirao do Ouro Araraquara (Sao Paulo-Brasil). Brazilian Journal of Ecology, vol. 4, no. 1-2, p. 112-117.
- BEHREND, RDL., FERNANDES, SEP., FUJITA, DS. and TAKEDA, AM., 2009. Eight years of monitoring aquatic Oligochaeta from the Baía and Ivinhema Rivers. Brazilian Journal of Biology, vol. 69, no. 2, supl. 0, p. 559-571. http://dx.doi.org/10.1590/S1519-69842009000300011. PMid:19738963
» http://dx.doi.org/10.1590/S1519-69842009000300011 - BRINKHURST, RO. and WETZEL, MJ., 1984. Aquatic Oligochaeta of the world: Supplement. A catalogue of new freshwater species, descriptions and revisions. Canada: Fisheries and Oceans. p. 44-101. Canadian Technical Report of Hydrography and Ocean Sciences, no. 44.
- BRINKHURST, RO. and MARCHESE, MR., 1989. Guia para la identificación de oligoquetos acuáticos continentales de Sud y Centroamerica. 2th ed. Santo Tomé: Associación de Ciencias Naturales del Litoral.
- BUCHMAN, HO. and BRADY, NC., 1979. Natureza e propriedades do solo. 5th ed. Rio de Janeiro: Freitas Bastos.
- CAMARGO, MN., KLAMT, E. and KAUFFMAN, JH., 1987. Classificação de solos usada em levantamentos pedológicos no Brasil. Sociedade Brasileira de Ciência do Solo, Campinas, vol. 12, p. 11-33.
- CAMPITELLI-RAMOS, R.,2008. Estudo da composição taxonômica e da densidade de macroinvertebrados bentônicos no sistema de lagoas naturais do vale do médio Rio Doce (MG), com ênfase na espécie de molusco exótica Melanoides tuberculata (Müller, 1774). São Carlos: Universidade de São Paulo. 146 p. Dissertação de mestrado em Ciências da Engenharia Ambiental.
- CORREIA, LCS. and TRIVINHO-STRIXINO, S., 1998. Macroinvertebrados da rizosfera de Scirpus cubensis na Lagoa do Infernão (Estação Ecológica de jataí-SP): Estrutura e função. Acta Limnologica Brasiliensia, vol. 10, no. 1, p. 37-47.
- DU BOIS-REYMOND MARCUS, E., 1944. Notes on fresh-water Oligochaeta from Brazil. Comununicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 1, no. 20, p. 1-8.
- DU BOIS-REYMOND MARCUS, E., 1947. Naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 2, no. 44, p. 1-20.
- DU BOIS-REYMOND MARCUS, E., 1949a. Further notes on naidids and tubificids from Brazil. Comunicaciones Zoologicas del Museo de Historia Natural de Montevideo, vol. 3, no. 51, p. 1-15.
- DU BOIS-REYMOND MARCUS, E., 1949b. A new tubificid from the Bay of Montevideo. Comunicaciones Zoologicas del Museu de Historia Natural de Montevideo, vol. 3, no. 56, p. 1-6.
- GORNI, GR. and ALVES, RD., 2006. Naididae (Annelida, Oligochaeta) associated with Pomacea bridgesii (Reeve) (Gastropoda, Ampullaridae). Revista Brasileira de Zoologia, vol. 23, no. 4, p. 1059-1061. http://dx.doi.org/10.1590/S0101-81752006000400011.
» http://dx.doi.org/10.1590/S0101-81752006000400011 - GORNI, GR. and ALVES, RG., 2008. Naididae species (Annelida: Oligochaeta) associated with the sponge Metania spinata (Carter, 1881) (Porifera: Metaniidae) from a southeastern Brazilian reservoir. Acta Limnologica Brasiliensia, vol. 20, no. 3, p. 261-263.
- HARMAN, WJ., 1974. The Naididae (Oligochaeta) of Surinam. Zoologische Verhandelingen., vol. 133, no. 1, p. 1-36.
- MACKERETH, FJH., HERON, J. and TALLING, FJ., 1978. Water analysis: some revised methods for limnologists. Freshwater Biological Association Scientific Publication. Kendall: Titus Wilson e Sons. Freshwater Biological Association Scientific Publication, no. 36.
- MARCUS, E., 1942. Sobre algumas Tubificidae do Brasil. Boletim de Zoologia, vol. 25, no. 6, p. 153-253.
- MARCUS, E., 1943. Sobre Naididae do Brasil. Boletim de Zoologia, vol. 32, no. 7, p. 3-181.
- MARCUS, E., 1944. Sobre Oligochaeta límnicos do Brasil. Boletim de Zoologia, vol. 43, no. 8, p. 5-101.
- MICHAELSEN, W., 1900. Oligochaeta. Berlin: R. Friedländer und Sohn. Das Tierreich, no. 10.
- MONTANHOLI-MARTINS, MC. and TAKEDA, AM., 2001. Spatial and temporal variations of Oligochaetes of the Ivinhema River and Patos Lake in the Upper Paraná River Basin, Brazil. Hydrobiologia, vol. 463, no. 1-3, p. 197-205. http://dx.doi.org/10.1023/A:1013163927814.
» http://dx.doi.org/10.1023/A:1013163927814 - PAMPLIN, PAZ., ROCHA, O. and MARCHESE, M., 2005. Riqueza de espécies de Oligochaeta (Anellida, Clitellata) em duas represas do rio Tietê (São Paulo). Biota Neotropica, vol. 5, no. 1, p. 63-70. http://dx.doi.org/10.1590/S1676-06032005000100007.
» http://dx.doi.org/10.1590/S1676-06032005000100007 - RIGHI, G., 1984. Oligochaeta. Manual de Identificação de Invertebrados Límnicos do Brasil, Brasília, CNPq, 48 p.
- SUGUIO, K., 1973. Introdução à Sedimentologia. São Paulo: Editora Edgar Blucher Ltda.
- SUN, J. and LIU, D., 2003. Geometric models for calculating cell biovolume and surface area for phytoplankton. Journal of Plankton Research, vol. 25, no. 11, p. 1331-1346. http://dx.doi.org/10.1093/plankt/fbg096.
» http://dx.doi.org/10.1093/plankt/fbg096 - SURIANI, AL., FRANÇA, RS., PAMPLIN, PA., MARCHESE, M., LUCCA, JV. and ROCHA, O., 2007. Species richness and distribution of oligochaetes in six reservoirs on Middle and low Tietê River (SP, Brazil). Acta Limnologica Brasiliensia, vol. 19, no. 4, p. 415-426.
- SURIANI-AFFONSO, AL., FRANÇA, RS., MARCHESE, M. and ROCHA, O., 2011. Environmental factors and benthic Oligochaeta (Annelida, Clitellata) assemblages in a stretch of the Upper São Francisco River (Minas Gerais state, Brazil). Brazilian Journal of Biology, vol. 71, no. 2, p. 437-446. PMid:21755161.
- STEPHAN, NNC., 2007. Distribuição Espacial e Temporal dos insetos e Oligochaeta aquáticos da Sub-Bacia do córrego São Pedro. Juiz de Fora: Universidade Federal de Juiz de Fora. 61 p. Dissertação de Mestrado em Ciências Biológicas, Comportamento e Biologia Animal.
- STENERT, C., 2009. Estrutura da comunidade de macroinvertebrados aquáticos em arrozais do Rio Grande do Sul. São Carlos: Universidade Federal de São Carlos. 205 p. Tese de Doutorado em Ecologia e Recursos Naturais.
- TAKEDA, AM., PEREIRA, MCF. and BARBOSA, FAR., 2000. Zoobenthos survey of the Pantanal, Mato Grosso do Sul, Brasil. Bulletin of Biological Assessment and Engineering, vol. 18, no. 3, p. 49-55.
- TRIVINHO-STRIXINO, S., CORREIA, LCS. and SONODA, K., 2000. Phytophilous chironomidae (Diptera) and other macroinvertebrates in the ox-bow Infernão Lake (Jatai Ecological Station, Luiz Antonio, SP, Brazil). Revista Brasileira de Biologia, vol. 60, no. 3, p. 527-535. http://dx.doi.org/10.1590/S0034-71082000000300018. PMid:11188879
» http://dx.doi.org/10.1590/S0034-71082000000300018
Publication Dates
-
Publication in this collection
May 2014
History
-
Received
31 July 2012 -
Accepted
22 Feb 2013