Abstracts
Studies on the anatomy of Piper leaves demonstrate the presence of a subepidermal tissue distinct from the adjacent epidermis, which cells show thin walls and hyaline contents. Some authors consider such cells a hypodermal tissue, while others refer to them as components of a multiple epidermis. In this study, the nature of this subepidermal tissue was investigated through the analysis of leaf ontogeny in three Piper species. The analysis showed that the referred tissue originates from the ground meristem and, thus, should be considered a hypodermis. The studied species suggests that the role of the hypodermis would be to protect the photosynthetic apparatus from excess light, regulating the intensity of light reaching the chlorophyll parenchyma.
hypodermis; multisseriate epidermis; Piperaceae
Os estudos de anatomia foliar de espécies de Piper revelam a presença de um tecido subepidérmico distinto da epiderme e cujas células apresentam paredes finas e conteúdo hialino. Estas células são referenciadas por alguns autores como um tecido hipodérmico e por outros, como sendo constituintes de uma epiderme múltipla. Nesse estudo verificou-se a natureza deste tecido subepidérmico a partir da análise da ontogênese foliar de três espécies de Piper. A análise revelou que o referido tecido tem origem do meristema fundamental e, portanto, deve ser denominado de hipoderme. Para as espécies avaliadas, sugere-se que a hipoderme teria função de, proteger o aparato fotossintético do excesso de luminosidade, regulando a intensidade luminosa que atinge o parênquima clorofiliano.
hipoderme; epiderme multisseriada; Piperaceae
1 Introduction
Piperaceae species present a multisseriate epidermis, according to Fahn (1974)Fahn, A., 1974. Plant anatomy. Oxford: Pergamon Press. 611
p.. Yet, for the five existing genera
in this family (Stevens, 2013STEVENS, PF., 2013. Angiosperm Phylogeny Website. Version 12.
Missouri Botanical Garden. Available from:
<http://www.mobot.org/MOBOT/research/APweb/>. Access in: 20 Jun.
2013.
http://www.mobot.org/MOBOT/research/APwe...
), the
multisseriate epidermis is reported only in Peperomia species
(Takemori et al., 2003TAKEMORI, NK., BONA, C. and ALQUINI, Y., 2003. Anatomia comparada
das folhas de espécies de (Piperaceae) - I. Ontogênese do tecido aqüífero e dos
estômatos. PeperomiaActa Botanica Brasilica, vol. 17, no. 3, p.
387-394.; Evert, 2006Evert, RF., 2006. Esau’s plant anatomy: meristems, cells, and
tissues of the plant body: their structure, function, and development. 3rd ed.
New Jersey: John Wiley & Sons, Inc. 601 p.), which show succulent leaves.
Subepidermal layers formed by conspicuous, hyaline cells were observed on the adaxial surface of another Piperaceae species, Ottonia martiana Miq.; in this species, the subepidermal tissue was also seen on the abaxial surface, but only next to the central rib of the leaf (Souza et al., 2004SOUZA, LA., MOSCHETA IS. and OLIVEIRA JHG., 2004. Comparative morphology and anatomy of the leaf and stem of Peperomia dahlstedtii C.DC., Miq. and Kunth (Piperaceae). Ottonia martianaPiper diospyrifoliumGayana Botanica, vol. 61, no. 1, p. 6-17.). In this case, the subepidermal tissue was not considered a part of the epidermis, neither a hypodermis, being emphasized that further studies on the leaf ontogeny were needed.
The dermal system of Piper leaves is usually described as formed by
a unisseriate epidermis and the adjacent cell layers, called hypodermis (Yuncker, 1973YUNCKER, TG., 1973. The Piperaceae of Brazil - II. Hoehnea, vol. 3,
p. 29-284.; Vianna and Akisue, 1997VIANNA, WO. and AKISUE, G., 1997. Caracterização morfológica de L.
Piper aduncumLecta, vol. 15, no. 1-2, p.
11-62.; Nascimento and Vilhena-Potiguara, 1999NASCIMENTO, ME. and VILHENA-POTIGUARA, VCR., 1999. Aspectos
anatômicos dos órgãos vegetativos de C. DC. (Piperaceae) e suas estruturas
secretoras. Piper hispidinerviumBoletim do Museu Paraense
Emílio Goeldi, vol. 15, no. 1, p. 39-104.; Cutter, 2002CUTTER, EG., 2002. Anatomia vegetal: experimentos e interpretação.
São Paulo: Roca. 336 p.; Albiero et al.,
2005aALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2005a.
Morfoanatomia dos órgãos vegetativos de Piper crassinervium HB
& K. (Piperaceae). Acta Botanica Brasilica, vol. 19, no. 2, p.
305-312., bALBIERO, ALM., SOUZA, LA., MOURÃO, KSM., ALMEIDA, OJG. and LOPES,
WAL., 2005b. Morfo-anatomia do caule e da folha de Piper
gaudichaudianum Kuntze (Piperaceae). Acta Farmaceutica Bonaerense,
vol. 24, no. 4, p. 550-554., 2006ALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2006.
Morfoanatomia dos órgãos vegetativos de Sw. (Piperaceae). Piper
hispidumRevista Brasileira de Farmacognosia, vol. 16, no. 3, p.
379-391.). In turn, some authors such as Gogosz et al. (2012)GOGOSZ, AM., BOEGER, MRT., NEGRELLE, RRB. and BERGO, C., 2012.
Anatomia foliar comparativa de nove espécies do gênero . Piper
(Piperaceae)Rodriguesia, vol. 63, no. 2, p. 405-417.
http://dx.doi.org/10.1590/S2175-78602012000200013.
http://dx.doi.org/10.1590/S2175-78602012...
describe a multiple epidermis in the
leaves of nine Piper species. At maturity, it is difficult to
distinguish the multiple epidermis from the hypodermis and this controversy usually
leads to the conclusion that studies on leaf ontogeny are needed.
An attempt to elucidate the discussion on the origin of subepidermal tissues was the observation by Albiero et al. (2005a)ALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2005a. Morfoanatomia dos órgãos vegetativos de Piper crassinervium HB & K. (Piperaceae). Acta Botanica Brasilica, vol. 19, no. 2, p. 305-312. of rare divisions in the protodermis of Piper crassinervium H.B. & K. leaves, suggesting the presence of a multisseriate epidermis. Although the authors could observe these divisions, they were not able to confirm the nature of the subepidermal tissue, since studies on leaf development were not conducted.
Under this scenario, this study aimed on clarifying the origin of the subepidermal layers of Piper L. leaves, through anatomical and ontogenetic analyses of Piper aduncum L., P. aff. hispidinervum and P. hispidinervum C.DC. leaves.
2 Material and Methods
Stem apexes and leaf primordia of Piper aduncum, Piper aff. hispidinervum e Piper hispidinervum were collected at Departamento de Biologia, at Universidade Federal de Lavras, DBI-UFLA, where plants are cultivated in a greenhouse. These individuals are originated from EMBRAPA Acre (Rio Branco, Acre).
Botanical materials were fixed in Karnovsky’s solution (Karnovsky, 1965KARNOVSKY, MJ., 1965. A formaldehyde-glutaraldehyde fixative of high
osmolality for use in electron microscopy. The Journal of Cell Biology, vol. 27,
p. 137-138.) and stored in 70% Ethyl Alcohol. Permanent
slides were produced from the dehydration of materials in an ethanol series, and
infiltration within hydroxyethyl methacrylate (Feder and O’Brien, 1968Feder, N. and O’BRIEN, TP., 1968. Plant microtechnique: some
principles and new methods. American Journal of Botany, vol. 55, no. 1, p.
123-142. http://dx.doi.org/10.2307/2440500.
http://dx.doi.org/10.2307/2440500...
), following the manufacturer’s instructions.
Transverse 5 μm sections were obtained with a rotation microtome, stained with 0.05%
Toluidine Blue O (O’Brien et al., 1965O’BRIEN, TP., FEDER, N. and McCULLY, ME., 1965. Polychromatic
staining of plant cell walls by toluidine blue O. Protoplasma, vol. 59, p.
368-373.), and
mounted on Permount™.
Images were produced under a Carl Zeiss™ trinocular microscope, with an image capture device. Scales were projected under the same optical conditions.
3 Results
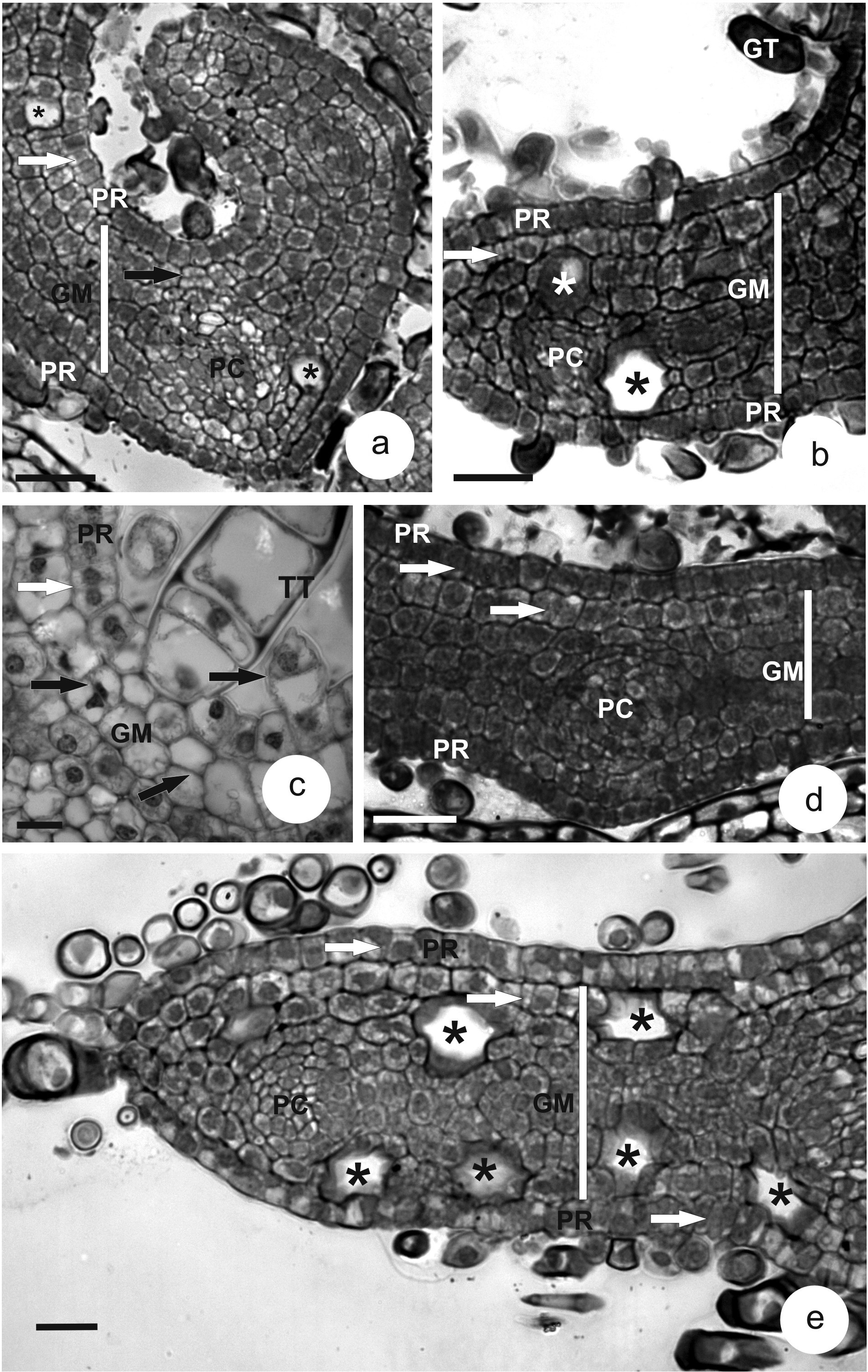
In the undifferentiated tissues of leaf primordia, the protodermis is unisseriate on both surfaces. Protodermal cells are cuboid to slightly elongated, radially (Figure 1a-e). Anticlinal and periclinal divisions are seen in this tissue, although they differentiate only into glandular (Figure 1b) and tector (Figure 1c) trichomes.
Transverse sections of leaf primordia in three Piper species. a-c. P. aduncum. d. P. aff. hispidinervum. e. P. hispidinervum. (BLACK ARROW = periclinal division; WHITE ARROW = anticlinal division; * = oil idioblast; GM = ground meristem; GT = glandular trichome; HY = hypodermis; PC = procambium; PR = protodermis; TT = tector trichome). Bars = 25 µm (a, b, d); 20 µm (e); 10 µm (c).
The ground meristem, located between the two protodermal surfaces, is formed of cells of varied shapes with high meristematic activity. On the adaxial surface, the ground meristem next to the protodermis shows periclinal divisions, specifically close to the leaf ribs (Figure 1a, c), although anticlinal divisions are more frequent (Figure 1b, d, e). At this developmental stage, some differentiating oil idioblasts are seen (Figure 1a, b, e).
Procambial rings, with more rectangular and diminute cells, are observed immersed in the ground meristem (Figure 1a, b, d, e).
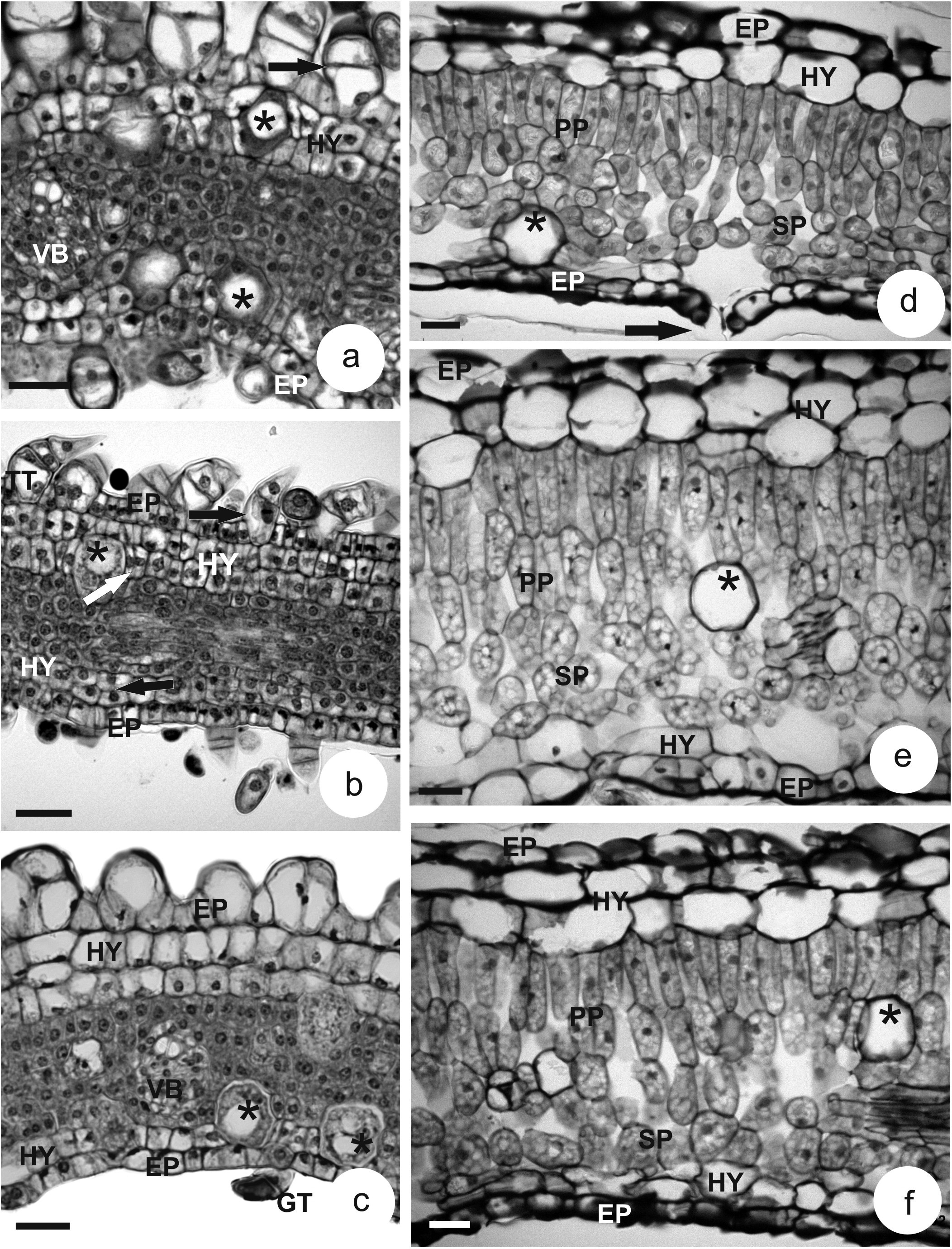
At a later stage, young leaf, the epidermis become more radially elongated (Figure 2a-c) and it shows tector (Figure 2a-c) and glandular trichomes (Figure 2c).
Transverse sections of Piper leaves. a-c. young leaves. d-f. mature leaves. a, d. P. aduncum; ARROW = stomata. b, e. P. aff. hispidinervum. c, f. P. hispidinervum. (* = oil idioblast; EP = epidermis; GT = glandular trichome; HY = hypodermis, PP = palisade parenchyma; SP = spongy parenchyma; VB = vascular bundle; BLACK ARROW: periclinal division; WHITE ARROW: anticlinal division). Bars = 25 µm (a-c); 50 µm (d-f).
In the mesophyll region, two cell layers differentiate next to the epidermis of the adaxial and abaxial surface, forming a tissue with hyaline, cuboid to radially elongated cells (Figure 2a-c).
The differentiation of the two cell layers on the upper surface, sometimes three layers (Figure 2c), and one a two cell layers on the lower surface are distinguished from the remaining layers of the ground meristem, especially seen through changes in cell shape and their hyaline contents. The cells from the meristematic layers, which will form the photosynthetic tissue, are isodiametric and show dense contents (Figure 2a-c). Collateral vascular bundles differentiate in this region (Figure 2a, c)
Mature leaves in the studied species are hypostomatic (Figure 2d) and present a unisseriate epidermis, with few tector and secretory trichomes. The subepidermal tissue, the hypodermis, of both surfaces presents typical parenchyma cells, with hyaline contents and thin walls. On the adaxial surface, the hypodermis varies from one to three layers of cells of varied shapes, from rectangular to isodiametric. On the abaxial surface, one to two layers are formed (Figure 2d-f).
The mesophyll is bilateral, showing palisade and spongy parenchyma; oil idioblasts (Figure 2d-f) and collateral vascular bundles are dispersed in this region.
4 Discussion
The hypodermis is characterized by the differentiation of cells originated from the ground meristem, while the multisseriate epidermis derives from periclinal divisions of the protodermis (Fahn, 1974Fahn, A., 1974. Plant anatomy. Oxford: Pergamon Press. 611 p.). In the studied Piper species, periclinal divisions were observed in the protodermis, but these originated the epidermal annexes, not the subepidermal tissue; thus, the latter should be referred to as a hypodermis. The rare periclinal divisions observed in the protodermal cells of Piper crassinervium H.B. & K. leaves (Albiero et al., 2005aALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2005a. Morfoanatomia dos órgãos vegetativos de Piper crassinervium HB & K. (Piperaceae). Acta Botanica Brasilica, vol. 19, no. 2, p. 305-312.) demonstrate that the intensity of divisions is enough to form trichomes, but not a subepidermal tissue, confirming that the subepidermal tissue originates from the ground meristem, not from the protodermis.
The hypodermal tissue frequently occurs in more resistant leaves, with scleromorphic
features, being usually absent in thin, flexible leaves, in addition to being found
mainly next to the central rib of leaves (Mauseth,
1988MAUSETH, JD., 1988. Plant anatomy. California: Cummings Publishing
Company. 560 p.). It is evident in this study that, although the analyzed
Piper species show thin and flexible leaves, the hypodermis is
present on both surfaces in a varied number of layers; furthermore, it can also
differentiate into a collenchyma next to the central rib on adaxial leaf surfaces.
The number of hypodermal layers is used by several authors as a taxonomic character
to identify species in the genus (Albiero et al.,
2005aALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2005a.
Morfoanatomia dos órgãos vegetativos de Piper crassinervium HB
& K. (Piperaceae). Acta Botanica Brasilica, vol. 19, no. 2, p.
305-312., bALBIERO, ALM., SOUZA, LA., MOURÃO, KSM., ALMEIDA, OJG. and LOPES,
WAL., 2005b. Morfo-anatomia do caule e da folha de Piper
gaudichaudianum Kuntze (Piperaceae). Acta Farmaceutica Bonaerense,
vol. 24, no. 4, p. 550-554., 2006ALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2006.
Morfoanatomia dos órgãos vegetativos de Sw. (Piperaceae). Piper
hispidumRevista Brasileira de Farmacognosia, vol. 16, no. 3, p.
379-391.; Gogosz et al.,
2012GOGOSZ, AM., BOEGER, MRT., NEGRELLE, RRB. and BERGO, C., 2012.
Anatomia foliar comparativa de nove espécies do gênero . Piper
(Piperaceae)Rodriguesia, vol. 63, no. 2, p. 405-417.
http://dx.doi.org/10.1590/S2175-78602012000200013.
http://dx.doi.org/10.1590/S2175-78602012...
; Raman et al., 2012Raman, V., GALAL, AM. and KHAN, IA., 2012. An investigation of the
vegetative anatomy of Piper sarmentosum, and a comparison with
the anatomy of Piper betle (Piperaceae). American Journal of
Plant Sciences, vol. 3, no. 8, p. 1135-1144.
http://dx.doi.org/10.4236/ajps.2012.38137.
http://dx.doi.org/10.4236/ajps.2012.3813...
).
Although the subepidermal tissue in Piper is originated from the
ground meristem, for another genus in the Piperaceae, Peperomia,
the analysis of leaf ontogeny demonstrated the presence of a multisseriate epidermis
(Takemori et al., 2003TAKEMORI, NK., BONA, C. and ALQUINI, Y., 2003. Anatomia comparada
das folhas de espécies de (Piperaceae) - I. Ontogênese do tecido aqüífero e dos
estômatos. PeperomiaActa Botanica Brasilica, vol. 17, no. 3, p.
387-394.). The difference
in the origin of the subepidermal tissue in species of the same family may be
related, in the case of Peperomia, to an evolutionary convergence
to the epiphytic habit, as defended by Reginato et
al. (2009)Reginato, M., BOEGER, MRT. and GOLDENBERG, R., 2009. Comparative
anatomy of the vegetative organs in A. Gray (Melastomataceae), with emphasis on
adaptations to epiphytism. PleiochitonFlora, vol. 204, no. 10,
p. 782-790. http://dx.doi.org/10.1016/j.flora.2008.11.006.
http://dx.doi.org/10.1016/j.flora.2008.1...
. These authors report that species from different families and
orders, with the same epiphytic habit, were subjected to a selective pressure to
adapt to the environment, as exemplified by the presence of leaves with water
reserves.
Reginato et al. (2009)Reginato, M., BOEGER, MRT. and GOLDENBERG, R., 2009. Comparative
anatomy of the vegetative organs in A. Gray (Melastomataceae), with emphasis on
adaptations to epiphytism. PleiochitonFlora, vol. 204, no. 10,
p. 782-790. http://dx.doi.org/10.1016/j.flora.2008.11.006.
http://dx.doi.org/10.1016/j.flora.2008.1...
also reported that the
subepidermal tissues (hypodermis or multisseriate epidermis) in the succulent leaves
of Peperomia, Pleiochton (Melastomataceae) and representatives of
the Gesneriaceae, were related to water storage, occupying a great proportion of the
leaf volume (between one third and half of it). The proportion of photosynthetic and
water tissues, in addition to features such as homogeneous mesophylls with big,
vacuolated cells, may also evidence a CAM photosynthetic metabolism (Smith and
Winter, 1996 apud Reginato et al. 2009Reginato, M., BOEGER, MRT. and GOLDENBERG, R., 2009. Comparative
anatomy of the vegetative organs in A. Gray (Melastomataceae), with emphasis on
adaptations to epiphytism. PleiochitonFlora, vol. 204, no. 10,
p. 782-790. http://dx.doi.org/10.1016/j.flora.2008.11.006.
http://dx.doi.org/10.1016/j.flora.2008.1...
).
Such an anatomical pattern is not seen in Piper species, although
studies on their photosynthetic metabolism are still needed.
For Rudall (2007)RUDALL, PJ., 2007. Anatomy of flowering plants - an introduction to
structure and development. 3nd ed. Cambridge: Cambridge University Press. 145
p., the presence of a
hypodermis may be a xeromorphic feature, as well as the presence of thick cuticles
and epidermis, which in combination would diminish the light intensity reaching the
photosynthetic tissue. Among the xeromorphic features reported by Rudall (2007)RUDALL, PJ., 2007. Anatomy of flowering plants - an introduction to
structure and development. 3nd ed. Cambridge: Cambridge University Press. 145
p., the presence of a hypodermis in
Piper is always cited, although the other characteristics are
most probably absent due to the environmental conditions to which
Piper species are adapted: tropical forest environment.
Although the species grow in the woods, their seeds germinate preferably in areas
where the incidence of light is higher (Bergo et
al., 2010Bergo, CL., SILVA, RC., OHLSON, OC., BIASI, LA. and PANOBIANCO, M.,
2010. Luz e temperatura na germinação de sementes de pimenta longa () e
pimenta-de-macaco (. Piper hispidinervumPiper
aduncum)Revista Brasileira de Sementes, vol. 32, no. 3, p. 170-176.
http://dx.doi.org/10.1590/S0101-31222010000300019.
http://dx.doi.org/10.1590/S0101-31222010...
; Pacheco Junior et al.,
2013PACHECO JUNIOR, F., SILVA, JB., NEGREIROS, JRS., SILVA, MRG. and
FARIAS, SB., 2013. Germination and vigor of long-pepper seeds (Piper
hispidinervum) as a function of temperature and light. Revista
Ciência Agronômica, vol. 44, no. 2, p. 325-333.
http://dx.doi.org/10.1590/S1806-66902013000200015.
http://dx.doi.org/10.1590/S1806-66902013...
) such as edges and clearings. Because the leaves of
Piper species are exposed to this increased luminosity, they
are subject to the stress caused by excess of light. Additionally, environmental
variations are larger in clearings and edges (Tabarelli and Mantovani, 1999Tabarelli, M. and Mantovani, W., 1999. Clareiras naturais e a
riqueza de espécies pioneiras em uma floresta atlântica montana. Brazilian
Journal of Biology, vol. 59, no. 2, p. 251-261.), particularly with respect to light
incidence, temperature and humidity, factors which directly influence the morphology
of the leaves.
The studied Piper species present flexible, thin leaves, with hypodermis on both leaf surfaces (but more prominent on the adaxial surface), which occupy up to one fourth of the leaf. It is probable that the hyaline hypodermis of Piper aduncum, P. aff. hispidinervum and P. hispidinervum is related to their photosynthetic efficiency, as it would protect the photosynthetic apparatus from excess light, regulating light intensity as seen in the Bromeliaceae (Brighigna et al., 1984Brighigna, L., FIORDI, AC. and PALANDRI, MR., 1984. Structural characteristics of mesophyll in some species. TillandsiaPhytomorphology, vol. 34, p. 191-200.) and in Peperomia (Takemori et al., 2003TAKEMORI, NK., BONA, C. and ALQUINI, Y., 2003. Anatomia comparada das folhas de espécies de (Piperaceae) - I. Ontogênese do tecido aqüífero e dos estômatos. PeperomiaActa Botanica Brasilica, vol. 17, no. 3, p. 387-394.). Although several authors report that the hypodermis may function as a protective tissue against the excessive solar radiation, this hypothesis should be tested physiologically.
5 Conclusion
The distinct subepidermal layers seen in the mature leaves of the studied Piper species are originated from the ground meristem. Therefore these layers should be referred as hypodermis.
Acknowledgements
To EMBRAPA Acre, for the botanical materials used in the anatomical analyses. To the undergraduate student Beatriz Pereira Ezequiel, for the production of slides.
References
- ALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2005a. Morfoanatomia dos órgãos vegetativos de Piper crassinervium HB & K. (Piperaceae). Acta Botanica Brasilica, vol. 19, no. 2, p. 305-312.
- ALBIERO, ALM., SOUZA, LA., MOURÃO, KSM., ALMEIDA, OJG. and LOPES, WAL., 2005b. Morfo-anatomia do caule e da folha de Piper gaudichaudianum Kuntze (Piperaceae). Acta Farmaceutica Bonaerense, vol. 24, no. 4, p. 550-554.
- ALBIERO, ALM., PAOLI, AAS., SOUZA, LA. and MOURÃO, KSM., 2006. Morfoanatomia dos órgãos vegetativos de Sw. (Piperaceae). Piper hispidumRevista Brasileira de Farmacognosia, vol. 16, no. 3, p. 379-391.
- Bergo, CL., SILVA, RC., OHLSON, OC., BIASI, LA. and PANOBIANCO, M., 2010. Luz e temperatura na germinação de sementes de pimenta longa () e pimenta-de-macaco (. Piper hispidinervumPiper aduncum)Revista Brasileira de Sementes, vol. 32, no. 3, p. 170-176. http://dx.doi.org/10.1590/S0101-31222010000300019.
» http://dx.doi.org/10.1590/S0101-31222010000300019 - Brighigna, L., FIORDI, AC. and PALANDRI, MR., 1984. Structural characteristics of mesophyll in some species. TillandsiaPhytomorphology, vol. 34, p. 191-200.
- CUTTER, EG., 2002. Anatomia vegetal: experimentos e interpretação. São Paulo: Roca. 336 p.
- Evert, RF., 2006. Esau’s plant anatomy: meristems, cells, and tissues of the plant body: their structure, function, and development. 3rd ed. New Jersey: John Wiley & Sons, Inc. 601 p.
- Fahn, A., 1974. Plant anatomy. Oxford: Pergamon Press. 611 p.
- Feder, N. and O’BRIEN, TP., 1968. Plant microtechnique: some principles and new methods. American Journal of Botany, vol. 55, no. 1, p. 123-142. http://dx.doi.org/10.2307/2440500.
» http://dx.doi.org/10.2307/2440500 - GOGOSZ, AM., BOEGER, MRT., NEGRELLE, RRB. and BERGO, C., 2012. Anatomia foliar comparativa de nove espécies do gênero . Piper (Piperaceae)Rodriguesia, vol. 63, no. 2, p. 405-417. http://dx.doi.org/10.1590/S2175-78602012000200013.
» http://dx.doi.org/10.1590/S2175-78602012000200013 - KARNOVSKY, MJ., 1965. A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. The Journal of Cell Biology, vol. 27, p. 137-138.
- MAUSETH, JD., 1988. Plant anatomy. California: Cummings Publishing Company. 560 p.
- NASCIMENTO, ME. and VILHENA-POTIGUARA, VCR., 1999. Aspectos anatômicos dos órgãos vegetativos de C. DC. (Piperaceae) e suas estruturas secretoras. Piper hispidinerviumBoletim do Museu Paraense Emílio Goeldi, vol. 15, no. 1, p. 39-104.
- O’BRIEN, TP., FEDER, N. and McCULLY, ME., 1965. Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma, vol. 59, p. 368-373.
- PACHECO JUNIOR, F., SILVA, JB., NEGREIROS, JRS., SILVA, MRG. and FARIAS, SB., 2013. Germination and vigor of long-pepper seeds (Piper hispidinervum) as a function of temperature and light. Revista Ciência Agronômica, vol. 44, no. 2, p. 325-333. http://dx.doi.org/10.1590/S1806-66902013000200015.
» http://dx.doi.org/10.1590/S1806-66902013000200015 - Raman, V., GALAL, AM. and KHAN, IA., 2012. An investigation of the vegetative anatomy of Piper sarmentosum, and a comparison with the anatomy of Piper betle (Piperaceae). American Journal of Plant Sciences, vol. 3, no. 8, p. 1135-1144. http://dx.doi.org/10.4236/ajps.2012.38137.
» http://dx.doi.org/10.4236/ajps.2012.38137 - Reginato, M., BOEGER, MRT. and GOLDENBERG, R., 2009. Comparative anatomy of the vegetative organs in A. Gray (Melastomataceae), with emphasis on adaptations to epiphytism. PleiochitonFlora, vol. 204, no. 10, p. 782-790. http://dx.doi.org/10.1016/j.flora.2008.11.006.
» http://dx.doi.org/10.1016/j.flora.2008.11.006 - RUDALL, PJ., 2007. Anatomy of flowering plants - an introduction to structure and development. 3nd ed. Cambridge: Cambridge University Press. 145 p.
- SOUZA, LA., MOSCHETA IS. and OLIVEIRA JHG., 2004. Comparative morphology and anatomy of the leaf and stem of Peperomia dahlstedtii C.DC., Miq. and Kunth (Piperaceae). Ottonia martianaPiper diospyrifoliumGayana Botanica, vol. 61, no. 1, p. 6-17.
- STEVENS, PF., 2013. Angiosperm Phylogeny Website. Version 12. Missouri Botanical Garden. Available from: <http://www.mobot.org/MOBOT/research/APweb/>. Access in: 20 Jun. 2013.
» http://www.mobot.org/MOBOT/research/APweb/ - Tabarelli, M. and Mantovani, W., 1999. Clareiras naturais e a riqueza de espécies pioneiras em uma floresta atlântica montana. Brazilian Journal of Biology, vol. 59, no. 2, p. 251-261.
- TAKEMORI, NK., BONA, C. and ALQUINI, Y., 2003. Anatomia comparada das folhas de espécies de (Piperaceae) - I. Ontogênese do tecido aqüífero e dos estômatos. PeperomiaActa Botanica Brasilica, vol. 17, no. 3, p. 387-394.
- VIANNA, WO. and AKISUE, G., 1997. Caracterização morfológica de L. Piper aduncumLecta, vol. 15, no. 1-2, p. 11-62.
- YUNCKER, TG., 1973. The Piperaceae of Brazil - II. Hoehnea, vol. 3, p. 29-284.
-
(With 2 figures)
Publication Dates
-
Publication in this collection
May 2015
History
-
Received
25 July 2013 -
Accepted
06 Jan 2014