Abstract
The quality of forage production is a prerequisite to raising livestock. Therefore, income losses in this activity, primarily cattle raising, can result in the impossibility of economic activity. Through the qualitative and quantitative anatomical study of Urochloa hybrida cv. Mulato II and U. brizantha cv. Marandu, we searched for descriptions and compared changes in the individual vegetative body from populations with death syndrome pastures (DPS). Specimens were collected at different physiological stages from farms in northern Mato Grosso. After collection, the individuals were fixed in FAA50 and stored in 70% alcohol. Histological slides were prepared from the middle third of the sections of roots, rhizomes, and leaves, and the proportions and characteristics of tissues were evaluated in healthy, intermediate, and advanced stages of DPS. Changes were compared between cultivars. With the advancement of the syndrome, the following changes were observed: a more marked decrease in the length of roots in U. hybrida; disorganization of the cortical region of the roots and rhizome cultivars; fungal hyphae in roots and aerenchyma formation in U. hybrida; a decrease in sclerenchyma fiber proportions in roots and leaves; sclerification of the epidermis of U. brizantha rhizomes; and an increase in pericyclic fibers in U. hybrida. Furthermore, there was a decrease in the volume of epidermal cells of the abaxial face of the leaves of both cultivars, with a greater reduction in U. hybrida; a gradual decrease in thickness in the midrib of leaves similar to leaf mesophyll; conduction system obstructions; partial or total cell lysis in roots and rhizomes affected by the syndrome. Obstructions in sieve tube element and companion cells, and sometimes obstruction in xylem vessel elements. The evolution of DPS in cultivars was similar, but there were variations, arising probably from the physiological response to stress, such as aerenchyma formation in the root and increased pericycle in the rhizome of U. hybrida.
Keywords:
forage; plant anatomy; fungal hyphae
Resumo
A produção de forragem de qualidade é um pressuposto indispensável à atividade pecuária moderna. Dessa forma, perdas de rendimento nesta atividade preliminar à criação bovina, podem inviabilizar esta atividade econômica. Através de estudos anatômicos qualitativos e quantitativos em Urochloa hybrida cv. Mulato II e U. brizantha cv. Marandu buscou-se descrever e comparar as alterações do corpo vegetativo de indivíduos de populações que apresentam síndrome da morte das pastagens (SMP). Foram coletados indivíduos em estádios morfológicos distintos em propriedades rurais no norte de Mato Grosso, Amazônia Meridional. Após a coleta, os indivíduos foram fixados em FAA50 e armazenados em álcool 70%. Lâminas histológicas foram confeccionadas a partir de secções do terço médio de raízes, rizomas e folhas, onde foram avaliadas as características e proporções dos tecidos em indivíduos saudáveis, indivíduos em estádio intermediário e também em estádio mais avançado da SMP, bem como comparadas as alterações entre as duas cultivares. Com o avanço da síndrome verificou-se diminuição mais acentuada da extensão de raízes em U. hybrida. A região cortical das raízes e do rizoma das cultivares exibiu desorganização, sendo observada nas raízes a presença de hifas fúngicas e a formação de aerênquima em U. hybrida. As proporções de fibras esclerenquimáticas diminuíram nas raízes e folhas. No rizoma, a epiderme de U. brizantha apresentou esclerificação e U. hybrida exibiu aumento da quantidade de fibras pericíclicas. Houve diminuição do volume das células epidérmicas da face abaxial das folhas de ambas as cultivares, com maior diminuição em U. hybrida. A região da nervura central das folhas apresentou gradativa diminuição com espessura similar ao mesofilo foliar. O sistema de condução apresentou obstruções, onde elementos de tubo crivado e células companheiras do floema das raízes e rizomas afetados pela síndrome exibiram parcial ou total lise celular e os elementos de vaso do xilema por vezes se mostraram obstruídos. A evolução da SMP nas cultivares estudadas é semelhante, contudo houve variações decorrentes provavelmente da resposta fisiológica ao estresse, como a formação de aerênquima na raiz, alteração e aumento da espessura do periciclo do rizoma de U. hybrida.
Palavras-chave:
forrageira; anatomia vegetal; hifas fúngicas
1 Introduction
Use of pastures is the most common and cheaper to create cattle in Brazil, either beef or dairy cattle. The maintenance of the forage productivity, combined with the climate and soil conditions needed for a sustainable system over time, constitutes one of the major livestock problems because the degradation of these ecosystems occurs on most of the farms. The search for the solution of this problem not only involves the identification of forage materials suited to different conditions, but also to the optimum levels of fertilizer to ensure the longevity and production of such forage (Sanchês et al., 2013SANCHÊS, S.S.C., GALVÃO C.M.L., RODRIGUES, R.C., SIQUEIRA, J.C., JESUS A.P.R., ARAÚJO, J.S., SOUSA, T. V. R. and SILVA-JUNIOR, A. L., 2013. Produção de forragem e características morfofisiológicas do capim-mulato cultivado em latossolo do cerrado em função de doses de nitrogênio e potássio. Revista Brasileira de Agropecuária Sustentável, vol. 3, no. 1, pp. 81-89.).
The forages of the genre Urochloa P. Beauv. (Poaceae) arouse great interest among farmers because of their high dry matter production, as well as adaptability, persistence, ease of establishment, and a good nutritional value; these forages also have reduced disposition to disease and present good growth during the entire year, not only during the rainy season (Costa et al., 2006COSTA, K.A.P., ROSA, B. and OLIVEIRA, I.P., 2006. Efeito da estacionalidade na produção de matéria seca e composição bromatológica da cv. Marandu. Brachiaria brizanthaCiência Animal Brasileira, vol. 6, no. 3, pp. 187-193.). Especially U. ruziziensis, U. brizantha (Hochst. ex A. Rich.) R.D. Webster and U. decumbens (Stapf) R.D. Webster have been used for many years in tropical pastures in Brazil (Pereira et al., 2012PEREIRA, R.C., DAVIDE, L.C., TECHIO, V.H. and TIMBÓ, A.L.O., 2012. Duplicação cromossômica de gramíneas forrageiras: uma alternativa para programas de melhoramento genético. Ciência Rural, vol. 42, no. 7, pp. 1278-1285. http://dx.doi.org/10.1590/S0103-84782012000700023.
http://dx.doi.org/10.1590/S0103-84782012...
).
These species have advantages as well as limitations to be used as forages, and it is desirable that their features are combined into new cultivars with more limited and realistic edaphoclimatic parameters. Strategies to explore the variability in individuals include carrying out artificial crosses, with induction to a hybridization, and manipulation of the sexuality and apomixis (Valls and Peñaloza, 2004VALLS, J.F.M. and PEÑALOZA, A.D.P., 2004. Recursos genéticos de gramíneas forrageiras para a pecuária. In: 41ª Reunião Anual da Sociedade Brasileira de Zootecnia, 2004. Campo Grande. Campo Grande: Sociedade Brasileira de Zootecnia. CD-ROM.).
In this direction, the Centro Internacional de Agricultura Tropical (CIAT) has made available a new option of forage, the Urochloa hybrida cv. Mulato II (CIAT 36087), the result of crossing and selection between Urochloa ruziziensis and Urochloa decumbens cv. Basilisk, with accesses of U. brizantha (Teodoro et al., 2012TEODORO, M.S.R., PINHO COSTA, K.A., DIAS, F.J.S., SIMON, G.A., SAENZ, E.A.C., SEVERIANO, E.C. and CRUVINEL, W.S., 2012. Composição bromatológica dos capins Marandu e Mulato II submetidos a diferentes alturas de resíduo. Global Science and technology, vol. 5, no. 3, p. 37-146.).
Urochloa hybrida cv. Mixed II is a perennial grass, with a height close to 1 meter at peak production, with linear leaves, lanceolate of an intense green color, with an average length of 35 to 40 cm, width of 2.5 to 3.0 cm, and an abundant pubescence (Argel et al., 2007ARGEL, P.J., MILES, J.W., GUIOT, J.D., CUADRADO, H. and LASCANO, C.E., 2007. Cultivar Mulato II (Brachiaria hybrida CIAT 36087): Gramínea de alta qualidade e produção forrageira, resistente às cigarrinhas e adaptada a solos tropicais ácidos. Cali: CIAT. 22 p.).
Grass Mulato II and other cultivars were released as options for U. brizantha cv. Marandu, which has been widely used in Brazilian tropical pastures since the 1980s for its productivity and maintenance cost (Dias-Filho, 2007DIAS-FILHO, M.B., 2007. Degradação de pastagens: processo, causas e estratégias de recuperação. 3rd ed. Belém: Embrapa Amazônia Oriental. 190 p.).
A perennial, with short rhizomes, erect culms, with little branching and leaves ranging from glabrous to pubescent, U. brizantha it is one of the brachiarias more resistant to climatic variations, being able to vegetate in some stressful situations (Brito and Rodella, 2002BRITO C.J.F.A. and RODELLA, R.A., 2002. Caracterização morfo-anatômica da folha e do caule de (Hochst. ex A. Rich.) Stapf e (Rendle) Schweick. (Poaceae). Brachiaria brizanthaB. humidicolaRevista Brasileira de Botanica. Brazilian Journal of Botany, vol. 25, no. 2, pp. 221-228. http://dx.doi.org/10.1590/S0100-84042002000200010.
http://dx.doi.org/10.1590/S0100-84042002...
).
A causative factor of losses reported since the early 1990s in Costa Rica (Zuñiga-Pereira et al., 1998ZUÑIGA-PEREIRA, C., GONZÁLEZ-QUESADA, R., BUSTAMANTE-ROJAS, E. and ARGEL, P.J., 1998. Influencia de La humedad del suelo sobre la susceptibilidad de Brachiaria a hongos patógenos. Manejo Integrado de Plagas, vol. 49, pp. 51-57.) and in Brazil, especially in the North (Dias-Filho, 2006DIAS-FILHO, M.B., 2006. Respostas morfofisiológicas de Brachiaria spp. ao alagamento do solo e síndrome da morte do capim-marandu. In: R.A. BARBOSA, org. Morte de pastos de Braquiárias. Campo Grande: Embrapa Gado de Corte, pp. 83-101.) and later in the Midwest, is the death of the Marandu grass in specific regions of pastures. These regions are associated with areas of poor soil drainage and flooding, which are known for not only their low tolerance of the cultivar to waterlogging, but also the proliferation of pathogenic microorganisms by the excess of humidity.
Agronomic alterations caused by the excess of water in pastures have already been widely discussed by several authors in the recent decades (Zuñiga-Pereira et al., 1998ZUÑIGA-PEREIRA, C., GONZÁLEZ-QUESADA, R., BUSTAMANTE-ROJAS, E. and ARGEL, P.J., 1998. Influencia de La humedad del suelo sobre la susceptibilidad de Brachiaria a hongos patógenos. Manejo Integrado de Plagas, vol. 49, pp. 51-57.; Dias-Filho, 2007DIAS-FILHO, M.B., 2007. Degradação de pastagens: processo, causas e estratégias de recuperação. 3rd ed. Belém: Embrapa Amazônia Oriental. 190 p.), which led to the conclusion that grass death is caused by a number of factors that interact (e.g., low soil drainage capacity, compaction, nutritional deficit, excessive proliferation of fungi), characterizing the problem as a syndrome (Death of Pastures Syndrome - DPS).
There is no information in the literature about the tissue alterations suffered by plants affected by diseases such as DPS, and few are the records of the occurrence of DPS in other species or cultivars. The current information is that this syndrome began in Brachiaria pastures from the Marandu cultivar and some cultivars of the same genus are tolerant, but not resistant.
Studies of anatomical alterations generated by water deficit in forage were carried out by Melo et al. (2007)MELO, H.C., CASTRO, E.M., SOARES, A.M., MELO, L.A. and ALVES, J.D., 2007. Alterações anatômicas e fisiológicas em Stapf ex Massey e . Setaria ancepsPaspalum paniculatum L. sob condições de déficit hídricoHoehnea, vol. 34, no. 2, pp. 145-153. http://dx.doi.org/10.1590/S2236-89062007000200003.
http://dx.doi.org/10.1590/S2236-89062007...
, reporting the variation of proportions of chlorophyll parenchyma and the volume of bulliform cells. Caetano and Dias-Filho (2008)CAETANO, L.P. and DIAS-FILHO, M.B., 2008. Responses of six . Brachiaria spp. accessions to root rose floodingRevista Brasileira de Zootecnia, vol. 37, no. 5, pp. 795-801. http://dx.doi.org/10.1590/S1516-35982008000500003.
http://dx.doi.org/10.1590/S1516-35982008...
discussed the physiological responses of six Urochloa cultivars to soil waterlogging and the consequent decrease in productivity. However, they did not mention tissue alterations.
In this context, this study aimed to describe and compare individuals in different morphological stages of the DPS development by identifying its impact on plant tissues of root, rhizome, and leaves of U. brizantha cv. Marandu and U. hybrida cv. Mulato II.
2 Material and Methods
Individuals of Urochloa brizantha cv. Marandu and Urochloa hybrida cv. Mulato II were collected from three farms with the occurrence of death of pastures syndrome (DPS) in the Southern Amazon, located to the extreme north of Mato Grosso.
The region is part of the South American Amazon depression, with humid continental equatorial climate and dry season from May to September. The annual rainfall index can exceed 2,300 mm m-2 and the average temperature is 24.8 °C (Amazonas, 2006AMAZONAS. Governo do Estado. Secretaria de Estado de Planejamento e Coordenação Geral – SEPLAN, 2006. Unidades Climáticas do Estado de Mato Grosso: Atlas Climatológico de Mato Grosso. Cuiabá: Laboratório de Climatologia/UFMT. CD-ROM.).
The collection sites were determined from random visits to farms for conversations about the DPS. Ranches or farms that presented different production characteristics (utilization and different planting time) were selected until the sample size reached the necessary number to proceed with the anatomical studies.
The samples of the Marandu were collected in the area 1, in the municipality of Carlinda-MT situated between the coordinates 9° 50' 13” of South Latitude and 55° 49' 34” of West Longitude, is a rural property with 50 hectares used for extensive beef cattle livestock, with pasture planted eleven years ago, with the occurrence of DPS for about two years.. Area 2, in the municipality of Alta Floresta-MT, situated between the coordinates 9° 47' 56” of South Latitude and 55° 55' 05” of West Longitude, is a rural property with approximately 120 hectares with dairy cattle and rotational grazing, and the implanted species for nearly 14 years. Area 3, also in the municipality of Alta Floresta-MT, situated between the coordinates 9° 51' 03” of South Latitude and 56° 12' 35” of West Longitude, is a rural property with an area of 520 hectares with practices of rearing extensive beef cattle livestock. Marandu grass and Mulato II grass were replanted in this area a year ago in different pickets after the grazing reform process (with chemical soil correction and disking), where U. brizantha had already been cultivated, with productivity losses due to many focuses of DPS. In all three areas, small depressions in the relief indicated areas liable to waterlogging in short periods or the superficial flow of water (flood) after soil saturation at heavier rains.
Sample of root, rhizome, and leaves of plants were collected, and it uses as reference the spot (small areas in the pasture where the grass is dead). In each of the farms it was selected a spot for the collection of the Marandu, and in the area 3 there were collected, from different pickets, samples of the cultivar Mulato II with DPS in spots. From the spot, clumps were selected in three distinct morphological stages: 1- Individuals that had no sudden death syndrome, with green leaves and near the spot; 2 - Individuals that had some yellowed leaves located at the edge of the spot; and 3 - Individuals in the center of the spot with yellow and/or dry leaves.
Four clumps were selected for each stage of the syndrome, totaling 12 clumps per collecting area. The rhizomes with roots were dismembered and the second leaf fully expanded, from the base of the tillers, was removed for study. This material was fixed in FAA50 (formaldehyde, glacial acetic acid, and ethanol 50% 5: 5: 90, v/v) in field; they were kept for 48 hours in a vacuum pump and then stored in ethanol 70% (Johansen, 1940JOHANSEN, D.A., 1940. Plant microtechnique. New York: McGraw-Hill Book Co. Inc., 523 p.) in the Vegetal Biology Laboratory of the University of Mato Grosso State, campus of Alta Floresta.
Samples of rhizomes were processed through the method of separation by Kraus and Arduin (1997)KRAUS, J.E. and ARDUIN, M., 1997. Manual básico de métodos em morfologia vegetal. Rio de Janeiro: EDUR. 198 p. modified, where the samples were placed in test tubes with hydrogen peroxide (30 volumes) and glacial acetic acid in the ratio 1: 1 and maintained in oven at 60°C for about 24 hours. After this period, the samples were washed in distilled water, transversal cut freehand with a razor blade, stained with double staining, astra blue and basic fuchsin (Roeser, 1962ROESER, K.R., 1962. Die nadel der schwarzkiefer-massenprodukt und kunstwerk der natur. Microkosmos, vol. 61, pp. 33-36.), and mounted on semipermanent histological slides.
From each clump, four roots and four leaves were used, constituting 4 repetitions for each morphological stage per treatments for each organ of each cultivar. Transverse and longitudinal sections of roots and leaves were obtained freehand, with a razor blade, stained with double staining, astra blue and basic fuchsin (Roeser, 1962ROESER, K.R., 1962. Die nadel der schwarzkiefer-massenprodukt und kunstwerk der natur. Microkosmos, vol. 61, pp. 33-36.), and mounted on semipermanent histological slides.
For the analysis of the epidermis, three leaves fully expanded from each clump were selected, totaling 36 repetitions for each morphological stage in each cultivar. The method of separation by Kraus and Arduin (1997)KRAUS, J.E. and ARDUIN, M., 1997. Manual básico de métodos em morfologia vegetal. Rio de Janeiro: EDUR. 198 p. modified was used in which the leaf portions were placed in test tubes with hydrogen peroxide (30 volumes) and glacial acetic acid in the ratio 1: 1 and kept in oven at 60 °C for about 48 hours. After this period, the samples were washed in distilled water and ethanol 50%, and with a brush the two epidermal surfaces were separated, stained with basic fuchsin (Roeser, 1962ROESER, K.R., 1962. Die nadel der schwarzkiefer-massenprodukt und kunstwerk der natur. Microkosmos, vol. 61, pp. 33-36.), and mounted in glycerine jelly.
The sections were observed and photographed using a photonic binocular microscope (Leica ICC 50), coupled to a computer with the LAZ EZ 1.7.0 software. In the images, qualitative and quantitative anatomical characters were analyzed. Quantitatively evaluated were the following: (1) thickness of the foliar mesophyll; (2) thickness of the group of fibers from the foliar rib; (3) average diameter of the central vascular bundle of the foliar blade; (4) thickness of the cuticle; (5) size of the bulliform cells; (6) root diameter; (7) thickness of the root epidermis; (8) count of the poles of protoxylem; and (9) the diameter of the vascular cylinder of the root. Qualitatively evaluated were the following: (1) idioblasts; (2) epidermic cells; (3) cuticle; (4) form of the stomata; (5) arrangement of the stomata; (6) form of the guard cells; (7) subsidiary cells; (8) hypodermis; (9) trichomes; (10) mesophyll; (11) vascular and extravascular fibers; (12) vascular bundles; and (13) fungi.
Based on the photomicrographs and by means of the Anati Quanti 2® UFV program (Aguiar et al., 2007AGUIAR, T.V., SANT’ANNA-SANTOS, B.F., AZEVEDO, A.A. and FERREIRA, R.S., 2007. Anati Quanti: software de análises quantitativas para estudos em anatomia vegetal. Planta Daninha, vol. 25, no. 4, pp. 649-659. http://dx.doi.org/10.1590/S0100-83582007000400001.
http://dx.doi.org/10.1590/S0100-83582007...
), the tissues from 144 transversal cuts of roots and 144 transversal cuts of leaves were measured, and the epidermal cells of 216 paradermal images were counted.
The data were subjected to analysis of variance by the F test and the averages were compared by the Tukey test at 5% of probability (Ferreira, 2011FERREIRA, D.F., 2011. Sisvar: a computer statistical analysis system. Ciência e Agrotecnologia, vol. 35, no. 6, pp. 1039-1042.). Later, boards with the images were made to show general anatomic patterns for the species, seeking to identify the evolution of the disease in the affected tissues.
3 Results and Discussion
By identifying the spot in the field, individuals of the Urochloa hybrida cv. Mulato II affected by the death of pastures syndrome (DPS) presented yellowing and/or dried leaves, with visible decrease in width and size in relation to healthy plants. Similar changes were visualized in U. brizantha cv. Marandu, except for the changes in the width of the leaves.
Urochloa hybrida had short rhizomes, as well as U. brizantha; however, the structure of the first cultivar was thicker, as well as its roots. The amount of lateral roots in U. hybrida was markedly smaller, and they had greater variations in shape, predominantly cylindrical shapes to oval shapes. In this cultivar, there was rarely any kind of substance coating the roots or clumps (Figure 1A), different from what was seen in U. brizantha, where lateral roots and also an outer crust to the epidermis were often found. Although it is not a common characteristic of the Poaceae, Saccharum angustifolium (Nees) Trin. may also present roots in ovoid shape (Chaves, 2012CHAVES, B.E., 2012. Aspectos anatômicos da raiz e lâmina foliar de Saccharum L. (POACEAE) nativas do Brasil. Brasília: Universidade de Brasília, 260 p. Dissertação de Mestrado em Botânica.).
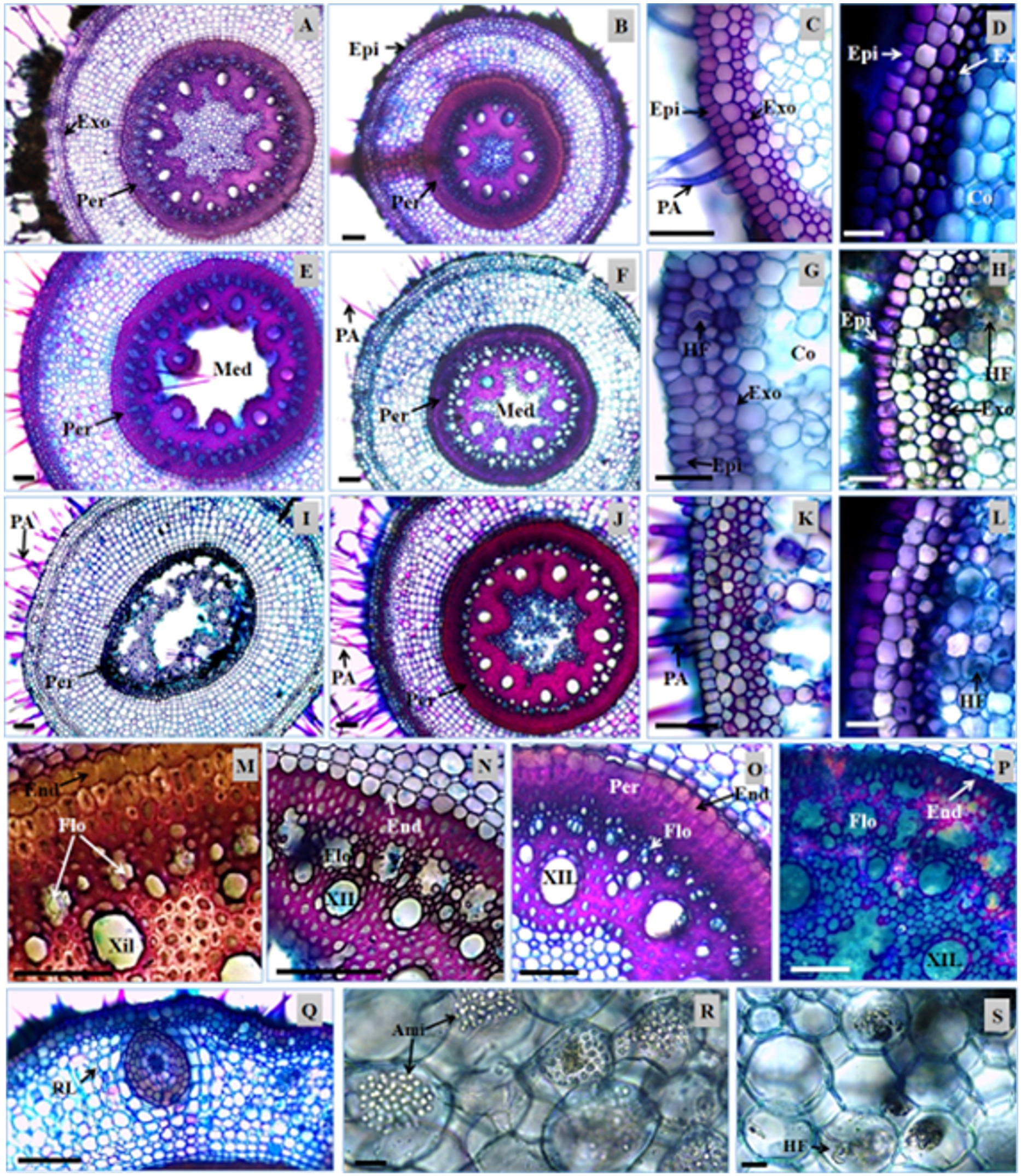
Transversal sections of the roots of Urochloa hybrida and Urochloa brizantha. A, C, E, G, I, K, M, N, Q, R and S - U. hybrida; B, D, F, H, J, L, O and P – U. brizantha. Epidermis (Ep), exodermis (Ex), absorbent hair (AH), lateral root (LR), endoderm (End), pericycle (Per), phloem (Phl), xylem (Xyl), parenchymatic marrow (PaM), starch (Sta), and fungal hyphae (FH). Bars: 100 µm (A to P); 10 µm (Q and S).
No significant differences were found between the collection areas for each cultivar. Therefore, the morphological comparison between the stages and the cultivars was prioritized.
The presence of absorbent hairs on the roots of both cultivars was registered (Figure 1A, 1B, 1C, 1D, 1E). Based on the transversal sections, it was found that the hairs were unicellular. In U. brizantha they had greater changes in shape, and in U. hybrida they were more rectilinear (Figure 1C). Variations in the amount of hair were not observed. However, it was noted by visual comparison that the length of the roots were diminished with the progress of the DPS in U. hybrida.
By analyzing the U. decumbens contaminated with heavy metals, Silva (2006)SILVA, S., 2006. Fungos micorrízicos arbusculares no crescimento, extração de metais e características anatômicas de Brachiaria decumbens Stapf em solo contaminado com metais pesados. Lavras: Universidade Federal de Lavras, 82 p. Tese de Doutorado em Solos e Nutrição de Plantas. found a drastic decrease in absorbent hairs in plant roots from contaminated areas. Chaves (2012)CHAVES, B.E., 2012. Aspectos anatômicos da raiz e lâmina foliar de Saccharum L. (POACEAE) nativas do Brasil. Brasília: Universidade de Brasília, 260 p. Dissertação de Mestrado em Botânica. observed simple unicellular root hairs, and usually short, in species of Saccharum, and argued that their presence and shape is related to the environment in which the individual lives. It is possible that the similarity between the amount of hair of the analyzed samples from both cultivars of this study was due to the analysis of plants that live in the same environment, and the quantity of these can be related to abiotic factors (water availability, soil compaction) in each collection area, or even the gene expression.
The epidermis of the root of the cultivars is unistratified (Figure 1C, 1D), and no anatomical changes or significant statistic difference was found for the wall thickness of cells between the DPS stages (Table 1).
Morphometry of the roots of the Urochloa brizantha Marandu and U. hybrida Mulato II in three morphological stages death syndrome pastures.
Below the epidermis, there was a layer of cells bulkier than the epidermis. The exodermis was well defined in both cultivars (Figure 1C, 1D), with greater wall thickness in U. brizantha. The thickening of the cell walls is a barrier to water loss and the entrance of pathogens, and the main compounds of this structure are lignin and suberin (Larcher, 2000LARCHER, W.,2000. Ecofisiologia vegetal. São Carlos: Rima Editora. 531 p.). Plants in more advanced stages of the syndrome exhibited exodermis cells with thinner walls (Figure 1G, 1H, 1K, 1L).
In healthy plants, the cortex was arranged symmetrically, with cells of various shapes in U. hybrida (Figure 1A, 1C) and spherical shapes of thin cell wall in U. Brizantha (Figure 1B, 1D). With the evolution of the DPS, the region exhibited cell degradation, with the emergence of gaps generated by cell lysis (Figure 1E, 1F, 1G, 1H). These spaces were higher in U. hybrida, starting to form aerenchyma (Figure 1G, 1K).
Although there is no significant difference among the DPS stages for the root diameter measures (Table 1), a tissue degradation that occurred in the cortical region was visible. Dias-Filho (2006)DIAS-FILHO, M.B., 2006. Respostas morfofisiológicas de Brachiaria spp. ao alagamento do solo e síndrome da morte do capim-marandu. In: R.A. BARBOSA, org. Morte de pastos de Braquiárias. Campo Grande: Embrapa Gado de Corte, pp. 83-101. observed a decrease in the concentration of soluble sugars in the roots of Urochloa that are little tolerant to flooding, and he also registered the increase in exudation of compounds which attract fungi (e.g. ethanol). Fungal hyphae were found in the cortical region of both cultivars in the later stages of the DPS (Figure 1G, 1H).
The metabolic changes caused by flooding probably provided lower efficiency of the plant defense mechanisms in relation to pathogens, as described by Ramos et al. (2010)RAMOS, T. J. N., CARVALHO, C. J. R., SOUZA, C. M. A. and VASCONCELOS, S. S., 2010. Alterações morfológicas e crescimento de duas espécies gramíneas sob alagamento. Revista de Ciências Agrárias, vol. 53, no. 1, pp. 5-11., which could explain the occurrence of fungal hyphae within the cortical cells of the cultivars analyzed in this study.
In tests carried out with U. brizantha and Paspalum fasciculatum, Ramos et al. (2010)RAMOS, T. J. N., CARVALHO, C. J. R., SOUZA, C. M. A. and VASCONCELOS, S. S., 2010. Alterações morfológicas e crescimento de duas espécies gramíneas sob alagamento. Revista de Ciências Agrárias, vol. 53, no. 1, pp. 5-11. found that the root biomass of the flooded plants was significantly lower compared with not flooded plants. It is possible that the lysis of the cells from the cortex and the decrease in production of sugars by the leaves are directly related to the changes in metabolism (decrease of cell replacement and starch) of the individuals, resulting from the action of biotic and abiotic factors, external to the plant, such as the occurrence of fungi and waterlogged soil.
The endoderm is a layer of cells with thickened and smaller walls in relation to other cells from the cortex in cultivars. In both cultivars, the thickening of the periclinal and anticlinal walls of the endoderm cells presented a noticeable decrease in the more advanced stages of the DPS (Figure 1M, 1N, 1O, 1P). The endoderm is able to reduce the reflux of ions accumulated in the vascular cylinder and in the cortex, hindering its loss to the soil solution (Steudle, 2000STEUDLE, E., 2000. Water uptake by roots: effects of water deficit. Journal of Experimental Botany, vol. 51, no. 350, pp. 1531-1542. http://dx.doi.org/10.1093/jexbot/51.350.1531. PMid:11006304.
http://dx.doi.org/10.1093/jexbot/51.350....
).
The pericycle, a set of sclerenchymatic fibers in two or three layers that involve the vascular cylinder, presented great thickness of the cell walls (Figure 1M, 1O). In U. hybrida the greater thickness occurred in healthy plants (Figure 1A, 1E, 1I), while in U. brizantha, this region rarely exhibited anatomical differences between DPS stages (Figure 1B, 1F, 1J).
Silva (2006)SILVA, S., 2006. Fungos micorrízicos arbusculares no crescimento, extração de metais e características anatômicas de Brachiaria decumbens Stapf em solo contaminado com metais pesados. Lavras: Universidade Federal de Lavras, 82 p. Tese de Doutorado em Solos e Nutrição de Plantas. reported that plants contaminated with heavy metals have a decrease in the thickness of the pericycle, a variation that is attributed to probable alterations in photosynthetic rates. With the advancement of the DPS, the samples of U. hybrida exhibited similar anatomical alterations, which may also be due to the lower metabolic production.
The vascular cylinder of the cultivars is polyarc, and the average is 36 poles of protoxylem in U. hybrida, and 35 in U. brizantha (Table 1). The phloem and xylem groups are usually functional in healthy plants. Observed in the more advanced stages of the DPS were lysis of tracheal elements and, subsequently, lysis of the vessel elements (Figure 1N, 1P), promoting the interruption of transport of substances between the organs of the plant. In U. hybrida with the progress of the syndrome, the complete disruption of the internal structures to the pericycle has been observed in some roots (Figure 1I). In U. brizantha a hollow structure was observed in the location of the phloem, as well as obstructed elements of metaxylem and protoxylem (Figure 1J, 1P).
With the lysis and obstruction of the conducting vessels, the roots lose their ability to absorb water and nutrients, causing depletion of the plant's energy reserves (Bibiano et al., 2012BIBIANO, C., BRAGA, R., OLIVEIRA, M., FERREIRA, E. and SANTOS, J., 2012. Efeitos de subdoses de picloram em pré e pós emergência do feijão. In XXVIII Congresso Brasileiro da Ciência Das Plantas Daninhas, 2012. Campo Grande. Campo Grande: Sociedade Brasileira da Ciência das Plantas Daninhas, pp. 1-3.). This is a justification for the reduction of the structural polysaccharides and the reserve of plants in an advanced stage of the DPS.
In the center of the root of the cultivars, a medullary parenchyma, composed of small cells with thin cell wall, was found (Figure 1A, 1B, 1O). Also found were the presence of large amounts of starch in healthy plants (Figure 1R) and the presence of fungi in some samples in this region (Figure 1S). There was a higher proportion of parenchymatic marrow in U. hybrida, and these cells were the first to collapse in the samples with the advanced stage of the DPS (Figure 1E, 1F). In U. hybrida, it was common to observe this obstructed structure; in U. brizantha, it was more common to find the structure to remain hollow (Figure 1E, 1F, 1I, 1J). However, the two characteristics were found in both cultivars.
The prolonged stress situation to which the individuals studied were subjected probably led to the deterioration of the medullary tissue. The parenchyma is one of the root tissues more sensitive to environmental changes (Pereira et al., 2008PEREIRA, F.J., CASTRO, E.M., SOUZA, T.C. and MAGALHÃES, P.C., 2008. Evolução da anatomia radicular do milho ‘Saracura’ em ciclos de seleção sucessivos. Pesquisa Agropecuaria Brasileira, vol. 43, no. 12, pp. 1649-1656. http://dx.doi.org/10.1590/S0100-204X2008001200002.
http://dx.doi.org/10.1590/S0100-204X2008...
). In this context, the probable condition of hypoxia of the radicular tissues may have been instrumental in the degradation of this structure. Anatomically, the rhizomes of the cultivars showed a small amount of tector trichomes, unicellular, not homogeneously distributed (Figure 2C, 2D, 2H). The unistratified epidermis, composed of juxtaposed cells and varied forms, presented a thickening in the periclinal and anticlinal walls, which was also extended to the first and second layers of the cortex (Figure 2G, 2K). In U. brizantha, the epidermis and subepidermic layer of the cortex showed themselves sclerified, forming a thick and compact barrier in healthy plants of the species (Figure 2D).
Transversal sections of rhizomes of Urochloa hybrida and Urochloa brizantha. A, C, E, G, I, K, M and O – U. hybrida; B, D, F, H, J, L, N and P – U. brizantha. Epidermis (Ep), tector trichomes (TT), vascular bundle (VB), cortex (Co), and pericycle (Per). Bars: 200 µm.
Unistratified epidermis for rhizomes, as noted in U. brizantha and U. hybrida, has also been described for other species of Poaceae such as Paspalum barretoi, P. minus (Eichemberg, 2012EICHEMBERG, M.T., 2012. Morfologia e anatomia de Paspalum L. (Poaceae, Poales) e suas implicações taxonômicas. Rio Claro: Universidade Estadual Paulista, 103 p. Tese de Doutorado em Botânica.), and for Digitaria insularis (Machado et al., 2008MACHADO, A.F.L., MEIRA, R.M.S., FERREIRA, L.R., FERREIRA, F.A., TUFFI SANTOS, L.D., FIALHO, C.M.T. and MACHADO, M.S., 2008. Caracterização anatômica de folha, colmo e rizoma de Digitaria insularis.Planta Daninha, vol. 26, no. 1, pp. 1-8. http://dx.doi.org/10.1590/S0100-83582008000100001.
http://dx.doi.org/10.1590/S0100-83582008...
). U. brizantha exhibited sclerification of this layer, which may be a physiological response of the cultivar to stress, being able to prevent or hinder the entry of pathogens, especially because it is the most striking characteristic in samples in a more advanced stage of the DPS.
The cortex region of the cultivars showed varied proportions in relation to the other tissues of the organ. In U. hybrida, 8 to 19 layers of parenchymatic isodiametric cells (Figure 2A, 2E, 2I) were observed, while 9 to 16 layers were observed in U. brizantha (Figure 2B, 2F, 2J). At some points, vascular bundles representing vascular traces were found (Figure 2A, 2B, 2D, 2F, 2I, 2J, 2K).
With the evolution of the DPS and because of the cell lysis, the parenchyma of the cortical region presented a disorganization, which was more evident in U. hybrida (Figure 2I).
The cultivars, when healthy, could be distinguished by the structure of the pericycle and the vascular cylinder. Urochloa brizantha, regardless of the stage of the disease, presented a continuous pericycle, forming a fibrous and sclerotic cord, where the vascular bundles were immersed. Samples of U. hybrida, when healthy, presented a discontinuous pericycle (Figure 2A), which was not visible in samples at more advanced stages of the DPS (Figure 2E, 2I).
Urochloa brizantha and U. hybrida presented isolated collateral vascular bundles, with homogeneous distribution in the cylinder; peripheral vascular bundles immersed in the sclerotic cord and far from each other were recorded (Figure 2B, 2E, 2I, 2J). In the sample of individuals in a more advanced stage of the DPS, the vascular bundles exhibited lysis of the phloem and obstruction of the xylem (Figure 2O, 2P).
Vascular bundles changing direction were also recorded in rhizomes of Smilax goyazana (Smilacaceae), which was attributed to the addition of new bundles, which were distinguished from the development of aerial branches in the plant (Palhares and Silveira, 2005PALHARES, D. and SILVEIRA, C.E.S., 2005. Estudo anatômico do rizoma de A. DC. (Smilacaceae). Smilax goyazanaRevista Brasileira de Plantas Medicinais, vol. 8, pp. 52-62.).
By analyzing the leaves of U. hybrida and leaves of U. brizantha in paradérmica view, a very similar epidermal cell arrangement was observed. Based on the counts, significant variations were not detected in stomatal index nor in stomatal density in the stages of the DPS for each cultivar.
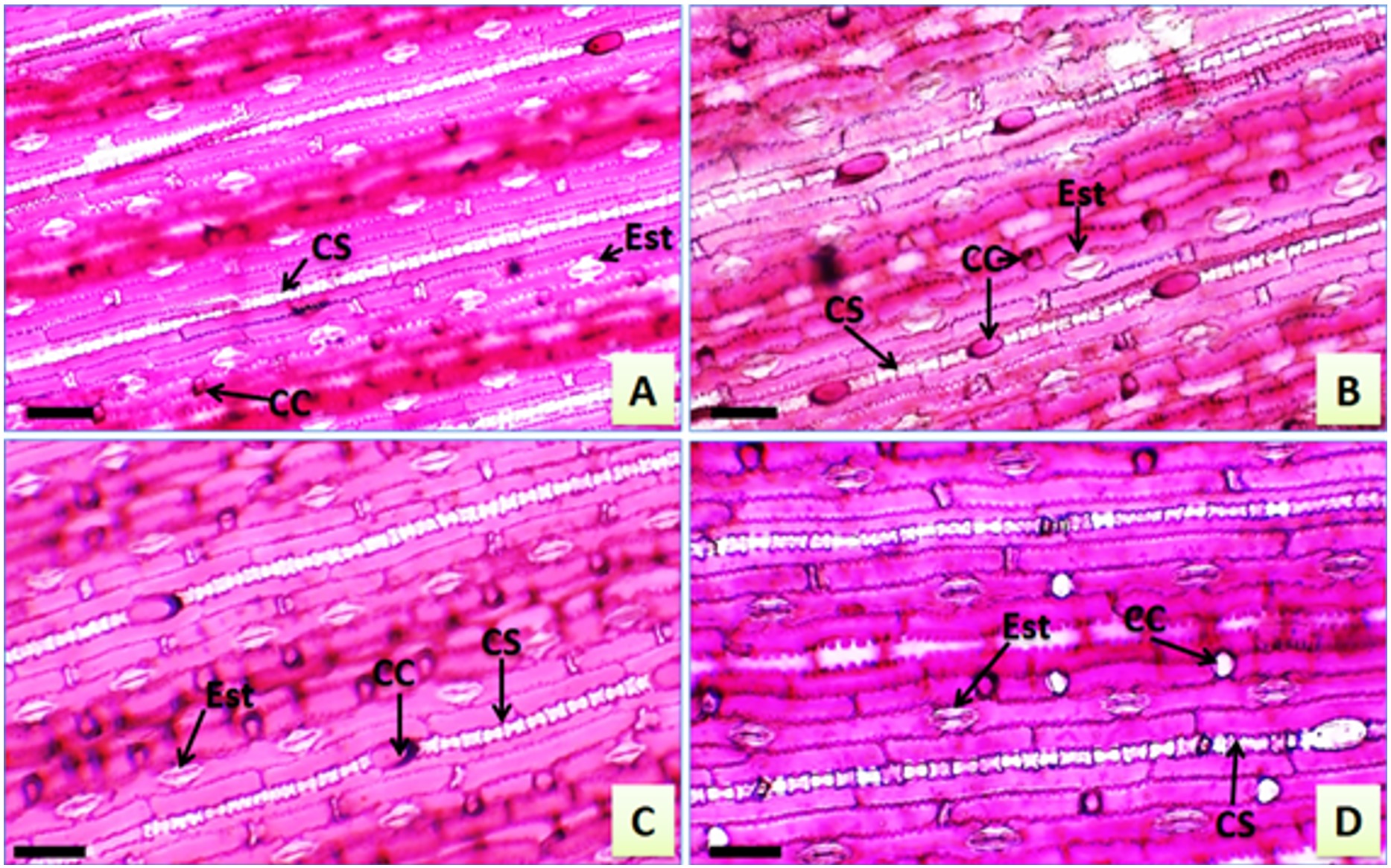
The epidermis presented cells in different shapes and sinuous anticlinal walls. Long cells, short intercostal cells, and silicified cells were observed for the genre (Figure 3A, 3B, 3C, 3D), as described by Ellis (1979)ELLIS, R.P., 1979. A procedure for standardizing comparative leaf anatomy in the Poaceae. The epidermis as seen in surface view. Bothalia, vol. 12, no. 4, pp. 641-671. http://dx.doi.org/10.4102/abc.v12i4.1441.
http://dx.doi.org/10.4102/abc.v12i4.1441...
and registered to other species of Poaceae by Jesus Junior et al. (2012)JESUS JUNIOR, L.A., OLIVEIRA, R.P., LEITE, K.R. and SILVA, L.B., 2012. Comparative analysis of the leaf anatomy in two Parodiolyra species (Poaceae: Olyreae) occurring on forests in Eastern Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 72, no. 1, pp. 205-210. http://dx.doi.org/10.1590/S1519-69842012000100025. PMid:22437403.
http://dx.doi.org/10.1590/S1519-69842012...
.
Paradermic sections of leaves of Urochloa hybrida cv. Mulato II and U. brizantha cv. Marandu. A – U. hybrida adaxial face; B – U. hybrida abaxial face; C – U. brizantha adaxial face; D – U. brizantha adaxial face. Siliceous cells (SC), stomata (Sto), short cells (SC). Bar: 80 µm.
Observed in transversal sections of the leaves of both cultivars were a thin cuticle on the entire surface of the foliar blade (Figure 4A, 4B) and a unistratified epidermis, composed of rectangular and small cells with rounded corners and thin walls (Figure 4M, 4N, 4O), as described by Metcalfe and Chalk (1960)METCALFE, C.R. and CHALK, L., 1960. Anatomy of monocotyledons: Gramíneae. Oxford: Clarendon Press, 1960, vol. 1. and Ellis (1976)ELLIS, R.P., 1976. A procedure for standardizing comparative leaf anatomy in the Poaceae. I. The leaf-blade as viewed in transverse section. Bothalia, vol. 12, no. 1, pp. 65-109. http://dx.doi.org/10.4102/abc.v12i1.1382.
http://dx.doi.org/10.4102/abc.v12i1.1382...
for Poaceae. Variations in the shape of these cells were noticed, although most of them followed this pattern. In samples in advanced stages of the DPS, less bulky epidermal cells were observed on the abaxial surface, particularly in U. hybrida (Figure 4I).
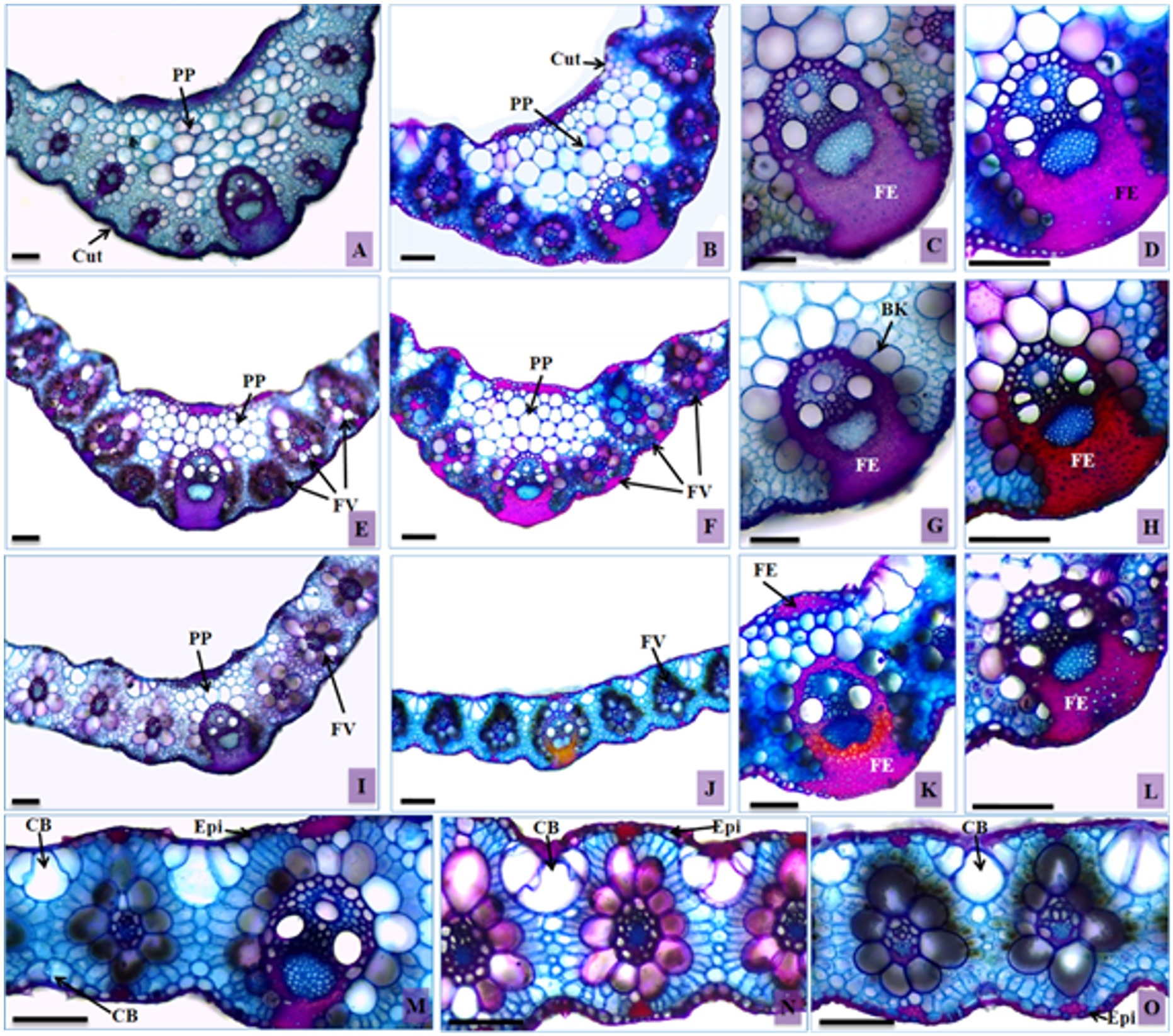
Transversal sections of leaves of Urochloa hybrida and Urochloa brizantha. A, C, E, G, I, K and M – U. hybrida; B, D, F, H, J, L, N and O – U. brizantha. Bulliform cells (BC), epidermis (Ep), cuticle (Cut), filling parenchyma (FP), sclerenchymatic fibers (SF), sheath Kranz (SK), and vascular bundle (VB). Bars: 100 µm.
Bulliform cells in groups of three to five cells, forming an array structure in the leaf mesophyll, were registered in both cultivars (Figure 4M, 4N). There was a significant difference in the volume of the bulliform cells in the adaxial surface of the leaf blade epidermis. An increase in the diameter of these cells in U. brizantha was observed in the samples with the evolution of DPS, which came to occupy a larger area in relation to the other tissues (Table 2).
Morphometry of the leaves of Urochloa brizantha Marandu and U. hybrida Mulato II in three morphological stages death syndrome pastures.
The bulliform cell clusters showed very similar characteristics among the cultivars, as described for the genre and also for other genera of grasses such as Cynodon and Paspalum (Metcalfe and Chalk, 1960METCALFE, C.R. and CHALK, L., 1960. Anatomy of monocotyledons: Gramíneae. Oxford: Clarendon Press, 1960, vol. 1.; Eichemberg, 2012EICHEMBERG, M.T., 2012. Morfologia e anatomia de Paspalum L. (Poaceae, Poales) e suas implicações taxonômicas. Rio Claro: Universidade Estadual Paulista, 103 p. Tese de Doutorado em Botânica.). Paspalum plants submitted to water deficiency conditions presented groups of bulliform cells with an increase in volume (Melo et al., 2007MELO, H.C., CASTRO, E.M., SOARES, A.M., MELO, L.A. and ALVES, J.D., 2007. Alterações anatômicas e fisiológicas em Stapf ex Massey e . Setaria ancepsPaspalum paniculatum L. sob condições de déficit hídricoHoehnea, vol. 34, no. 2, pp. 145-153. http://dx.doi.org/10.1590/S2236-89062007000200003.
http://dx.doi.org/10.1590/S2236-89062007...
). This characteristic is similar to that seen in U. brizantha in an advanced stage of DPS (Figure 4O), but in reverse environmental conditions. The bad conduction of the xylem and the lack of water in the aerial organs of the plant, even in flooded soil, can justify the morphological similarity among the studies.
In healthy plants of U. brizantha, the bulliform cells of the adaxial face were visualized at a level below the other epidermal cells, forming furrows. In samples of more advanced stages of DPS, it was common to find them at the same level of the other epidermal cells (Figure 4O). This feature was rarely observed in U. hybrida.
On the abaxial surface, the bulliform cells were slightly larger than the other epidermal cells in healthy plants, being larger in U. hybrida (Figure 4M). They did not stand out when compared with other epidermal cells in the samples in more advanced stages of the DPS in both cultivars (Figure 4O).
In both cultivars the observed mesophyll was homogeneous, and no differentiation between parenchymas occurred. Both are classified as C4 plants of Kranz anatomy (Metcalfe and Chalk, 1960METCALFE, C.R. and CHALK, L., 1960. Anatomy of monocotyledons: Gramíneae. Oxford: Clarendon Press, 1960, vol. 1.), as described for D. insularis, which also showed a simple hem and a sclerenchymatic cap around the bundles, which was facing the epidermis (Machado et al., 2008MACHADO, A.F.L., MEIRA, R.M.S., FERREIRA, L.R., FERREIRA, F.A., TUFFI SANTOS, L.D., FIALHO, C.M.T. and MACHADO, M.S., 2008. Caracterização anatômica de folha, colmo e rizoma de Digitaria insularis.Planta Daninha, vol. 26, no. 1, pp. 1-8. http://dx.doi.org/10.1590/S0100-83582008000100001.
http://dx.doi.org/10.1590/S0100-83582008...
).
Urochloa hybrida presented a mesophyll significantly thinner in the samples at more advanced stages of the DPS (Table 2). This characteristic may be attributable to the limited absorption of nutrients by the roots, due to malfunction of the conductive vessels. In Setaria anceps Stapf, the thickness of the foliar limbo of the vascular bundles and the fiber layer of the basal portion of the vascular bundle was lower in plants under hydric deficiency, compared to the control plants (Melo et al., 2007MELO, H.C., CASTRO, E.M., SOARES, A.M., MELO, L.A. and ALVES, J.D., 2007. Alterações anatômicas e fisiológicas em Stapf ex Massey e . Setaria ancepsPaspalum paniculatum L. sob condições de déficit hídricoHoehnea, vol. 34, no. 2, pp. 145-153. http://dx.doi.org/10.1590/S2236-89062007000200003.
http://dx.doi.org/10.1590/S2236-89062007...
).
In the cultivars under analysis, especially in U. hybrida, plants in the final stage of DPS presented vascular bundles of identical sizes (Figure 4I), a characteristic different from the one observed in healthy plant samples (Figure 4A, 4B, 4E, 4F). This variation is unusual; the different sizes of vascular bundles are peculiar characteristics of a species, and such characteristics are even used for taxonomy by authors such as Ellis (1976)ELLIS, R.P., 1976. A procedure for standardizing comparative leaf anatomy in the Poaceae. I. The leaf-blade as viewed in transverse section. Bothalia, vol. 12, no. 1, pp. 65-109. http://dx.doi.org/10.4102/abc.v12i1.1382.
http://dx.doi.org/10.4102/abc.v12i1.1382...
, Chaves (2012)CHAVES, B.E., 2012. Aspectos anatômicos da raiz e lâmina foliar de Saccharum L. (POACEAE) nativas do Brasil. Brasília: Universidade de Brasília, 260 p. Dissertação de Mestrado em Botânica. and Eichemberg (2012)EICHEMBERG, M.T., 2012. Morfologia e anatomia de Paspalum L. (Poaceae, Poales) e suas implicações taxonômicas. Rio Claro: Universidade Estadual Paulista, 103 p. Tese de Doutorado em Botânica..
The parenchyma of the filling of the central rib of the cultivars is composed of rounded cells with thin walls, covering all the areas; it has an irregular shape and larger volume when close to the center of the structure (Figure 4A, 4B). The samples of individuals affected by the DPS presented a small number of cells in this region (Figure 4E, 4F), with significant differences between the morphological stages of collection (Table 2). The more advanced stages started to present the keel of the leaf with a thickness similar to the rest of the foliar limbo (Figure 4I, 4J).
The groups of subepidermal sclerenchymatic fibers and the extensions of sheath of the vascular bundles from healthy individuals of U. brizantha were more pronounced than those of U. hybrida (Figure 4B, 4A). They were found in both limbo surfaces for both cultivars, being most commonly found on the abaxial surface, connecting the vascular bundles to the epidermis. In samples in advanced stages of DPS there was a lower proportion of these fibers (Figure 4C, 4D, 4G, 4H, 4K, 4L), with a significant difference among the analyzed stages (Table 2).
Regarding the production and accumulation of fibers, these are connected directly to the appropriate plant metabolism. If its functioning is modified, the production of structural polysaccharides such as lignin is reduced, and this tissue tends to be consumed for maintenance of vital functions (Taiz and Zeiger, 2013TAIZ, L. and ZEIGER, E., 2013. Fisiologia vegetal. 5th ed. Porto Alegre: Artmed. 954 p.). Caetano and Dias-Filho (2008)CAETANO, L.P. and DIAS-FILHO, M.B., 2008. Responses of six . Brachiaria spp. accessions to root rose floodingRevista Brasileira de Zootecnia, vol. 37, no. 5, pp. 795-801. http://dx.doi.org/10.1590/S1516-35982008000500003.
http://dx.doi.org/10.1590/S1516-35982008...
, while studying different species of grasses, concluded that U. brizantha cv. Marandu was more sensitive to flooding than other grasses because it had a more significant reduction in production of dry mass of leaves.
The decrease in the parenchyma tissue and the leaf fibers may be directly related to the sensitivity of the cultivars to the excess of water in the soil. Such stress, if prolonged, could have caused the decrease in the photosynthetic rate, the consumption of structural polysaccharides and reserve, and the decrease in resistance to the entry of pathogens, culminating in the death of the plants.
The evolution of the Death of the Pastures Syndrome (DPS) on the cultivars was similar; however, there were variations probably due to physiological responses to environmental stress. U. hybrida Mulato II distinguished itself by the appearance of aerenchyma and lower extension of roots in the more advanced stages of the DPS.
Overall, the changes start at the roots, with a disorganization of the cortical region, lysis, and obstruction of the conductor system. Thus, the tissue changes present themselves in the rhizomes and culminate in the reduction of the foliar mesophyll, reduction of the volume of epidermal cells, and a probable decrease in the photosynthetic rates of the plant.
-
(With 4 figures)
References
- AGUIAR, T.V., SANT’ANNA-SANTOS, B.F., AZEVEDO, A.A. and FERREIRA, R.S., 2007. Anati Quanti: software de análises quantitativas para estudos em anatomia vegetal. Planta Daninha, vol. 25, no. 4, pp. 649-659. http://dx.doi.org/10.1590/S0100-83582007000400001
» http://dx.doi.org/10.1590/S0100-83582007000400001 - AMAZONAS. Governo do Estado. Secretaria de Estado de Planejamento e Coordenação Geral – SEPLAN, 2006. Unidades Climáticas do Estado de Mato Grosso: Atlas Climatológico de Mato Grosso. Cuiabá: Laboratório de Climatologia/UFMT. CD-ROM.
- ARGEL, P.J., MILES, J.W., GUIOT, J.D., CUADRADO, H. and LASCANO, C.E., 2007. Cultivar Mulato II (Brachiaria hybrida CIAT 36087): Gramínea de alta qualidade e produção forrageira, resistente às cigarrinhas e adaptada a solos tropicais ácidos. Cali: CIAT. 22 p.
- BIBIANO, C., BRAGA, R., OLIVEIRA, M., FERREIRA, E. and SANTOS, J., 2012. Efeitos de subdoses de picloram em pré e pós emergência do feijão. In XXVIII Congresso Brasileiro da Ciência Das Plantas Daninhas, 2012. Campo Grande. Campo Grande: Sociedade Brasileira da Ciência das Plantas Daninhas, pp. 1-3.
- BRITO C.J.F.A. and RODELLA, R.A., 2002. Caracterização morfo-anatômica da folha e do caule de (Hochst. ex A. Rich.) Stapf e (Rendle) Schweick. (Poaceae). Brachiaria brizanthaB. humidicolaRevista Brasileira de Botanica. Brazilian Journal of Botany, vol. 25, no. 2, pp. 221-228. http://dx.doi.org/10.1590/S0100-84042002000200010
» http://dx.doi.org/10.1590/S0100-84042002000200010 - CAETANO, L.P. and DIAS-FILHO, M.B., 2008. Responses of six . Brachiaria spp. accessions to root rose floodingRevista Brasileira de Zootecnia, vol. 37, no. 5, pp. 795-801. http://dx.doi.org/10.1590/S1516-35982008000500003
» http://dx.doi.org/10.1590/S1516-35982008000500003 - CHAVES, B.E., 2012. Aspectos anatômicos da raiz e lâmina foliar de Saccharum L. (POACEAE) nativas do Brasil. Brasília: Universidade de Brasília, 260 p. Dissertação de Mestrado em Botânica.
- COSTA, K.A.P., ROSA, B. and OLIVEIRA, I.P., 2006. Efeito da estacionalidade na produção de matéria seca e composição bromatológica da cv. Marandu. Brachiaria brizanthaCiência Animal Brasileira, vol. 6, no. 3, pp. 187-193.
- DIAS-FILHO, M.B., 2006. Respostas morfofisiológicas de Brachiaria spp. ao alagamento do solo e síndrome da morte do capim-marandu. In: R.A. BARBOSA, org. Morte de pastos de Braquiárias. Campo Grande: Embrapa Gado de Corte, pp. 83-101.
- DIAS-FILHO, M.B., 2007. Degradação de pastagens: processo, causas e estratégias de recuperação. 3rd ed. Belém: Embrapa Amazônia Oriental. 190 p.
- EICHEMBERG, M.T., 2012. Morfologia e anatomia de Paspalum L. (Poaceae, Poales) e suas implicações taxonômicas. Rio Claro: Universidade Estadual Paulista, 103 p. Tese de Doutorado em Botânica.
- ELLIS, R.P., 1976. A procedure for standardizing comparative leaf anatomy in the Poaceae. I. The leaf-blade as viewed in transverse section. Bothalia, vol. 12, no. 1, pp. 65-109. http://dx.doi.org/10.4102/abc.v12i1.1382
» http://dx.doi.org/10.4102/abc.v12i1.1382 - ELLIS, R.P., 1979. A procedure for standardizing comparative leaf anatomy in the Poaceae. The epidermis as seen in surface view. Bothalia, vol. 12, no. 4, pp. 641-671. http://dx.doi.org/10.4102/abc.v12i4.1441
» http://dx.doi.org/10.4102/abc.v12i4.1441 - FERREIRA, D.F., 2011. Sisvar: a computer statistical analysis system. Ciência e Agrotecnologia, vol. 35, no. 6, pp. 1039-1042.
- JESUS JUNIOR, L.A., OLIVEIRA, R.P., LEITE, K.R. and SILVA, L.B., 2012. Comparative analysis of the leaf anatomy in two Parodiolyra species (Poaceae: Olyreae) occurring on forests in Eastern Brazil. Brazilian Journal of Biology = Revista Brasileira de Biologia, vol. 72, no. 1, pp. 205-210. http://dx.doi.org/10.1590/S1519-69842012000100025 PMid:22437403.
» http://dx.doi.org/10.1590/S1519-69842012000100025 - JOHANSEN, D.A., 1940. Plant microtechnique. New York: McGraw-Hill Book Co. Inc., 523 p.
- KRAUS, J.E. and ARDUIN, M., 1997. Manual básico de métodos em morfologia vegetal. Rio de Janeiro: EDUR. 198 p.
- LARCHER, W.,2000. Ecofisiologia vegetal. São Carlos: Rima Editora. 531 p.
- MACHADO, A.F.L., MEIRA, R.M.S., FERREIRA, L.R., FERREIRA, F.A., TUFFI SANTOS, L.D., FIALHO, C.M.T. and MACHADO, M.S., 2008. Caracterização anatômica de folha, colmo e rizoma de Digitaria insularis.Planta Daninha, vol. 26, no. 1, pp. 1-8. http://dx.doi.org/10.1590/S0100-83582008000100001
» http://dx.doi.org/10.1590/S0100-83582008000100001 - MELO, H.C., CASTRO, E.M., SOARES, A.M., MELO, L.A. and ALVES, J.D., 2007. Alterações anatômicas e fisiológicas em Stapf ex Massey e . Setaria ancepsPaspalum paniculatum L. sob condições de déficit hídricoHoehnea, vol. 34, no. 2, pp. 145-153. http://dx.doi.org/10.1590/S2236-89062007000200003
» http://dx.doi.org/10.1590/S2236-89062007000200003 - METCALFE, C.R. and CHALK, L., 1960. Anatomy of monocotyledons: Gramíneae. Oxford: Clarendon Press, 1960, vol. 1.
- PALHARES, D. and SILVEIRA, C.E.S., 2005. Estudo anatômico do rizoma de A. DC. (Smilacaceae). Smilax goyazanaRevista Brasileira de Plantas Medicinais, vol. 8, pp. 52-62.
- PEREIRA, F.J., CASTRO, E.M., SOUZA, T.C. and MAGALHÃES, P.C., 2008. Evolução da anatomia radicular do milho ‘Saracura’ em ciclos de seleção sucessivos. Pesquisa Agropecuaria Brasileira, vol. 43, no. 12, pp. 1649-1656. http://dx.doi.org/10.1590/S0100-204X2008001200002
» http://dx.doi.org/10.1590/S0100-204X2008001200002 - PEREIRA, R.C., DAVIDE, L.C., TECHIO, V.H. and TIMBÓ, A.L.O., 2012. Duplicação cromossômica de gramíneas forrageiras: uma alternativa para programas de melhoramento genético. Ciência Rural, vol. 42, no. 7, pp. 1278-1285. http://dx.doi.org/10.1590/S0103-84782012000700023
» http://dx.doi.org/10.1590/S0103-84782012000700023 - RAMOS, T. J. N., CARVALHO, C. J. R., SOUZA, C. M. A. and VASCONCELOS, S. S., 2010. Alterações morfológicas e crescimento de duas espécies gramíneas sob alagamento. Revista de Ciências Agrárias, vol. 53, no. 1, pp. 5-11.
- ROESER, K.R., 1962. Die nadel der schwarzkiefer-massenprodukt und kunstwerk der natur. Microkosmos, vol. 61, pp. 33-36.
- SANCHÊS, S.S.C., GALVÃO C.M.L., RODRIGUES, R.C., SIQUEIRA, J.C., JESUS A.P.R., ARAÚJO, J.S., SOUSA, T. V. R. and SILVA-JUNIOR, A. L., 2013. Produção de forragem e características morfofisiológicas do capim-mulato cultivado em latossolo do cerrado em função de doses de nitrogênio e potássio. Revista Brasileira de Agropecuária Sustentável, vol. 3, no. 1, pp. 81-89.
- SILVA, S., 2006. Fungos micorrízicos arbusculares no crescimento, extração de metais e características anatômicas de Brachiaria decumbens Stapf em solo contaminado com metais pesados. Lavras: Universidade Federal de Lavras, 82 p. Tese de Doutorado em Solos e Nutrição de Plantas.
- STEUDLE, E., 2000. Water uptake by roots: effects of water deficit. Journal of Experimental Botany, vol. 51, no. 350, pp. 1531-1542. http://dx.doi.org/10.1093/jexbot/51.350.1531 PMid:11006304.
» http://dx.doi.org/10.1093/jexbot/51.350.1531 - TAIZ, L. and ZEIGER, E., 2013. Fisiologia vegetal. 5th ed. Porto Alegre: Artmed. 954 p.
- TEODORO, M.S.R., PINHO COSTA, K.A., DIAS, F.J.S., SIMON, G.A., SAENZ, E.A.C., SEVERIANO, E.C. and CRUVINEL, W.S., 2012. Composição bromatológica dos capins Marandu e Mulato II submetidos a diferentes alturas de resíduo. Global Science and technology, vol. 5, no. 3, p. 37-146.
- VALLS, J.F.M. and PEÑALOZA, A.D.P., 2004. Recursos genéticos de gramíneas forrageiras para a pecuária. In: 41ª Reunião Anual da Sociedade Brasileira de Zootecnia, 2004. Campo Grande. Campo Grande: Sociedade Brasileira de Zootecnia. CD-ROM.
- ZUÑIGA-PEREIRA, C., GONZÁLEZ-QUESADA, R., BUSTAMANTE-ROJAS, E. and ARGEL, P.J., 1998. Influencia de La humedad del suelo sobre la susceptibilidad de Brachiaria a hongos patógenos. Manejo Integrado de Plagas, vol. 49, pp. 51-57.
Publication Dates
-
Publication in this collection
11 July 2016 -
Date of issue
Jan-Mar 2017
History
-
Received
16 July 2015 -
Accepted
25 Nov 2015