Abstract
Currently, medically significant scorpion species belong to the Buthidae family and are represented by the genera Androctonus, Buthus, Mesobuthus, Hottentotta, Parabuthus, Tityus, Centruroides, Leiurus. Although Leiurus was originally considered a monotypic genus, four additional species have since been described. Leiurus abdullahbayrami (previously identified as L. quinquestriatus in Turkey) was classified as a new Leiurus species. This is the first report conducted on the lethality and biologic effects of L. abdullahbayrami scorpion venom in mice. In this study, the electrophoretic protein pattern of its venom was also determined. Two protein bands with molecular masses of 4 and 6 kDa were more strongly detected than other protein bands in the venom sample. Electrophoresis showed that L. abdullahbayrami scorpion venom possesses both short- and long-chain neurotoxins. The median lethal dose of this venom was found to be 0.19 mg/kg by subcutaneous (SC) injection in mice. Animals experimentally envenomed with L. abdullahbayrami venom exhibited hyperexcitability, agitation, aggressive behavior, squeaking and fighting, tachypnea, weakness, convulsions, and death due to cardiac and respiratory failure. In further studies, the potency of antivenom should be investigated in relation to the scorpion venom. Molecular and pharmacological studies are also required to identify and characterize L. abdullahbayrami scorpion venom.
scorpions; Leiurus spp.; scorpion venom; lethality; in vivo effects
ORIGINAL PAPERS
A newly described scorpion species, Leiurus abdullahbayrami (Scorpion: Buthidae), and the lethal potency and in vivo effects of its venom
Ozkan OI; Yagmur EAII; Ark MIII
IRefik Saydam Public Health Agency, Ankara, Turkey
IIZoology Section, Department of Biology, Faculty of Science, Ege University, Izmir, Turkey
IIIDepartment of Pharmacology, Faculty of Pharmacy, Gazi University, Ankara, Turkey
Correspondence to Correspondence to: Ozcan Ozkan Refik Saydam Hygiene Center 06100, Ankara, Turkey Phone: 00 90 312 498 2150 Email: ozcanozkan_62@hotmail.com.
ABSTRACT
Currently, medically significant scorpion species belong to the Buthidae family and are represented by the genera Androctonus, Buthus, Mesobuthus, Hottentotta, Parabuthus, Tityus, Centruroides, Leiurus. Although Leiurus was originally considered a monotypic genus, four additional species have since been described. Leiurus abdullahbayrami (previously identified as L. quinquestriatus in Turkey) was classified as a new Leiurus species. This is the first report conducted on the lethality and biologic effects of L. abdullahbayrami scorpion venom in mice. In this study, the electrophoretic protein pattern of its venom was also determined. Two protein bands with molecular masses of 4 and 6 kDa were more strongly detected than other protein bands in the venom sample. Electrophoresis showed that L. abdullahbayrami scorpion venom possesses both short- and long-chain neurotoxins. The median lethal dose of this venom was found to be 0.19 mg/kg by subcutaneous (SC) injection in mice. Animals experimentally envenomed with L. abdullahbayrami venom exhibited hyperexcitability, agitation, aggressive behavior, squeaking and fighting, tachypnea, weakness, convulsions, and death due to cardiac and respiratory failure. In further studies, the potency of antivenom should be investigated in relation to the scorpion venom. Molecular and pharmacological studies are also required to identify and characterize L. abdullahbayrami scorpion venom.
Key words: scorpions, Leiurus spp., scorpion venom, lethality, in vivo effects.
INTRODUCTION
Scorpions are venomous arthropods, members of the Arachnida class and order Scorpiones (1). The scorpion species that are of current medical importance belong to the Buthidae family, and are known to cause considerable public health problems in North Africa, Asia, the Middle East, India, Mexico, and Central and South America (2, 3).
The genus Leiurus was considered to be monotypic for many years, comprising the one species Leiurus quinquestriatus containing subspecies L. quinquestriatus quinquestriatus and L. quinquestriatus hebraeus (4-6). As of 2009, four species were added: L. jordanensis from Jordan, L. savanicola from Cameroon, L. nasheri from Yemen, and L. abdullahbayrami from Turkey (6-9).
In Turkey, Tulga (10) was the first to record L. quinquestriatus from Ad1yaman Province in the southeastern region, which was identified by Prof. A. Shulov. It was later reported in Gaziantep, Hatay, Kilis, Sanl1urfa, and Mardin provinces in Turkey (6, 11). Recently, Yagmur et al. (6) described Leiurus abdullahbayrami, which had previously been identified as L. quinquestriatus in the Turkish scorpiofauna and has also been reported in Syria (12).
Five decades ago, Tulga (10) obtained venom from the telson of L. quinquestriatus using the maceration technique. The author observed that toxicity was directly related to the telson portion. In addition, Ozkan et al. (13) evaluated the neutralizing capacity of Androctonus crassicauda antivenom against L. quinquestriatus from Iraq. Apart from these studies, no information has been found regarding the toxicity, in vivo effects, and venom proteins of Turkish Leiurus specimens (14).
Since there are no data on lethality and the effects of Leiurus abdullahbayrami scorpion venom, research must be done to find out its effects on population health. Therefore, the main objective of this study was to determine the protein profiles (molecular weight), lethality, and in vivo effects of venom from a new Leiurus scorpion species.
MATERIALS AND METHODS
Scorpion Origin
During summer 2009, scorpions (n = 27) were collected using an ultraviolet (UV) lamp at night in the Southeastern region of the Gaziantep and Sanl1urfa provinces in Turkey (Figure 1). The animals were housed in individual plastic boxes at the Department of Entomology, Faculty of Veterinary Medicine, Ankara University, Turkey. The scorpions were fed crickets or cockroaches and received water daily. Taxonometric identification followed Yagmur et al. (6).
Experimental Animals
Swiss mice of both genders (20 ± 2 g) - bred in the animal facilities of Refik Saydam Public Health Agency (RSPHA) - were employed to determine the median lethal dose (LD50) via SC administration. The animals were housed under controlled temperature (20 ± 2°C), in 12-hour light/dark cycle and fed commercial rodent pellets and water ad libitum throughout the experiment.
Venoms
The venom was obtained from mature L. abdullahbayrami scorpions by electrical stimulation of the telson. It was dissolved with sterile double-distilled water and centrifuged at 14,000 rpm for 15 minutes at 4°C. The supernatant was stored at -20°C until use. Protein concentration was determined using a BCA kit (Pierce, USA) according to the manufacturer's instructions. Bovine serum albumin was employed as standard.
Lethality and In Vivo Venom Effects
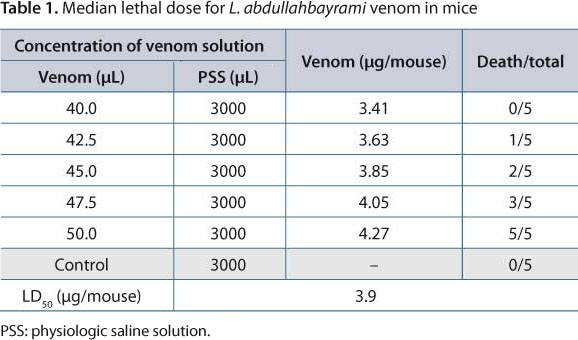
Experimental protocols for the animal experiments were approved by RSPHA Ethics Committee. Venom LD50 was assessed by SC injections in mice (20 ± 2 g) whereas lethality was determined as described by Behrens and Karber (15). For both lethality and in vivo effects, five mice per dose group were SC injected with various doses (3.41, 3.63, 3.85, 4.05, and 4.27 µg/mouse) of L. abdullahbayrami venom, diluted in a 0.5-mL physiologic saline solution (0.85% NaCl). An equivalent volume of saline was injected into five mice as negative control group (Table 1). The animals were observed for 24 hours after venom injection to determine LD50. Deaths occurring after 24 hours were recorded. In terms of in vivo effects, the mice injected with venom for the lethality assay were monitored and the evolution of their signs was recorded for up to 24 hours.
Gel Electrophoresis of Venom
The venoms were analyzed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) according to Laemmli (16). Venom of the L. abdullahbayrami scorpion was run on NuPAGE® Novex Bis-Tris 4-12% gel in MES SDS running buffer (Invitrogen, USA, NP0002; 50 mM MES, 50 mM Tris-HCl, 1% SDS, 1.025 Mm EDTA) using Xcell SureLock® Mini-cell (Invitrogen, USA) following standard manufacturer protocol. SeeBlue® Plus2 Pre-Stained Standard (Invitrogen, USA) was run in parallel in order to calculate molecular weights of the proteins. The gel was stained with Coomassie blue, then scanned and molecular weights of the proteins were calculated with Molecular Imaging Software® (Kodak MIS, USA).
RESULTS
The new species was distinguished from other members of the genus according to size of pedipalp, chela, metasomal V segment, and location of trichobothrium db (dorsal basal) on the fixed finger of the pedipalp. The scorpions were then identified as L. abdullahbayrami (Figure 2) under stereomicroscope (Stemi 2000-C®, Zeiss, Germany).
Lethality Assay and In Vivo Effects on Mice
A reduced number of mice were used in order to avoid causing unnecessary pain and suffering to the animals. Therefore, both lethality and in vivo venom effects on the same Swiss mouse were determined. The lethal toxicity of L. abdullahbayrami venom was determined after SC injections of different venom doses and the death/survival ratio (Table 1) was collected over a 24-hour period. The LD50 of L. abdullahbayrami scorpion venom was found to be 0.19 mg/kg by SC injection (Table 1).
Experimental mice showed the following intoxication symptoms after injection: hyperexcitability, agitation, aggressive behavior, squeaking and fighting, tachypnea, convulsions, weakness, paralysis, coma, and death. However, not all animals exhibited hypersalivation or lacrymation.
SDS-PAGE Analysis of Venom
Venom protein content was found to be 0.513 mg protein/mL. The protein profile of L. abdullahbayrami venom was analyzed using NuPAGE® Novex 4-12% Bis-Tris gradient gel (Invitrogen, USA) followed by Coomassie blue staining. Proteins in the venom (10 µg) were found to be between 3 and 188 kDa by gradient gel electrophoresis (Figure 3). Four different protein bands with molecular masses of 4, 6, 31, and 46 kDa were detected in the venom sample by Kodak MIS.
DISCUSSION
About 1700 different species of scorpion distributed over fourteen families exist in the world (17). Up until now, in Turkey, Buthidae is the family of medical significance, comprising four genera (Androctonus, Leiurus, Mesobuthus, and Hottentotta) with eight different species.
The L. quinquestriatus scorpion is considered one of the most dangerous, and is blamed for many scorpion envenomation cases in countries from North Africa and the Middle East. The species is therefore considered most significant to humans and an influential cause of morbidity, especially in children (18-22).
To the best of our knowledge, there have been no reports on the toxicity and biological effects of Leiurus species from Turkey since 1960. This work is the first to determine the electrophoretic protein pattern, lethality, and effects of the venom of the new scorpion species, L. abdullahbayrami.
In a study, Tulga (10) declared that the scorpion venom toxicity was related to the telson portion per mouse. The author emphasized that L. abdullahbayrami venom is four to five times more toxic than A. crassicauda venom. However, Ozkan et al. (13) stated that venom from the Iraqi L. abdullahbayrami is double the toxicity of A. crassicauda scorpion venom. Ismail (18) also reported that smaller quantities of L. quinquestriatus venom are 26 to 28 times more lethal than the largest quantity of L. quinquestriatus scorpion venom. Conversely, even if the venom was extracted using a single method, lethal dose depends on many factors such as genus, species, gender, geographic origin of the scorpion, and structure and quantity of the venom (23). The median lethal dose of L. quinquestriatus venom was previously reported as 0.25 mg/kg, which makes it one of the most toxic species of scorpion in the world (22, 24-26). In the current work, the LD50 of L. abdullahbayrami venom was calculated to be 0.19 mg/kg in mice.
The venom of scorpions contains neurotoxic polypeptides that consist of low molecular weight peptides, which can cause a variety of symptoms, ranging from pain at the sting site to death (27, 28). The neurotoxic peptides responsible for the envenomation symptoms interact with ion channels and have the potential to cause massive damage to the nervous system of both vertebrates and invertebrates (29, 30). The short-chain neurotoxins with 3 to 4.4 kDa act on potassium or chloride channels. Long-chain neurotoxins, which have 6.5 to 8.5 kDa, mostly act on sodium channels (29, 31-33). Fatani et al. (22) stated that sodium and calcium channel blockers ameliorated the effects of L. quinquestriatus quinquestriatus scorpion venom. Hence, they said that these ion channel blockers could be useful in scorpion sting cases if antivenoms were not readily available. In our study, we showed the electrophoretic protein pattern of the scorpion venom to be between 3 and 188 kDa by gradient gel. Two protein bands with molecular masses between 4 and 6 kDa were more strongly detected than other protein bands in our venom samples. As a result, electrophoresis analysis indicates that L. abdullahbayrami scorpion venom possesses both short and long-chain neurotoxins according to electrophoretic protein patterns.
In earlier epidemiological and clinical studies, L. quinquestriatus venom was capable of provoking cardiac complications such as pulmonary edema, myocarditis, changes in heart rate and rhythm, and cardiac failure (19, 34-39). In Turkey, Söker and Haspolat (40) reported 64 cases of scorpion stings in children from the provinces of Mardin, Sirnak, Batman, and Siirt in the southeastern Anatolia region, who were admitted to the Pediatric Emergency Department in Dicle University Hospital at Diyarbakir between 1995 and 1999 (Figure 1). Those authors also recorded a 12.5% lethality rate, which was due to cardiac and respiratory failure during the first 24 hours of hospitalization. Recently, Bosnak et al. (41) showed that 2.2% of all cases of scorpion stings admitted to a referral hospital in Turkey were caused by L.quinquetriatus.
In addition, Bosnak et al. (41) stated that pulmonary edema developed in 9.6% and dyspnea in 23.0% of victims; the predominant signs of cardiovascular system compromise were tachycardia (36.5%), dyspnea (23.0%), paleness (15.3%), hypertension (7.6%), and hypotension (3.8%), and manifestations of cholinergic stimulation, including excessive sweating (32.6%) and vomiting (3.8%), were seen after scorpion stings in children in southeast Turkey. However Al et al. (42) reported 120 cases of scorpion envenomation in the Batman province, but cardiac dysfunction, myocardial damage and death were not recorded as secondary symptoms to the major systemic envenomations. Scorpion toxins cause the direct release of neurotransmitters, such as acetylcholine and catecholamines, which produce signs of autonomic system over-activity or autonomic storms (43, 44).
In our study, mice experimentally envenomed with L. abdullahbayrami venom displayed hyperexcitability, agitation, aggressive behavior, squeaking and fighting, tachypnea, convulsions, weakness, paralysis, and death due to cardiac and respiratory failure. Fatani et al. (22) reported that animal symptoms resulting from L. quinquestriatus scorpion envenomation included agitation, aggressive behavior, hyperventilation, hypersalivation, lachrymation, micturition, defecation, shivering, periodic spastic contractions, gasping, convulsions, and in fatal cases, cardiac and respiratory arrest. Although we also observed these sympathetic signs such as agitation, excitability, aggressive behavior, tachypnea, convulsions and death due to cardiac and respiratory failure, we did not observe any hypersalivation or lachrymation (parasympathetic signs) in envenomed mice. Results of the present study showed that L. abdullahbayrami venom manifested sympathetic effects that were more marked than the parasympathetic effects according to the signs of intoxication.
As far as we know, the Leiurus species was only found in Ad1yaman province, but has now been recorded in Hatay, Kilis, Gaziantep, and Mardin provinces in the southeastern region of Turkey, where scorpionism is a known significant medical problem (Figure 1). On the other hand, monovalent antivenom (A. crassicauda) has been administered in the treatment of all scorpion species sting cases in Turkey (45). Therefore, further studies should investigate the potency of antivenom against the scorpion venom. Molecular and pharmacological studies are also required to identify and characterize L. abdullahbayrami scorpion venom.
ACKNOWLEDGEMENTS
This study was supported by Refik Saydam Public Health Agency of Health Ministry, Ankara, Turkey. We wish to thank Juliana Simionato for her valuable comments on this manuscript. We also thank Mehmet Özkörük for his help in field trips.
6. Yagmur EA, Koç H, Kunt KB. Description of a new species of Leiurus Ehrenberg, 1828 (Scorpiones: Buthidae) from SouthЕastЕrn Turkey. Euscorpius. 2009;85(85):1-20.
Submission status
Received: February 28, 2011.
Accepted: June 13, 2011.
Abstract:published online: June 17, 2011.
Full paper published online: November 30, 2011.
Conflicts of interest
There is no conflict.
Financial source
The present study was supported by Refik Saydam Public Health Agency, Ankara, Turkey.
Ethics committee approval
The present study was approved by the Ethics Committee of Refik Saydam Public Health Agency (under protocol number 2009/3).
- 1. Petricevich VL. Scorpion venom and the inflammatory response. Mediators Inflamm. 2010;2010:1-16.
- 2. Efrati P. Venoms of Buthinae. Epidemiology symptomatology and treatment of Buthinae stings. In: Bettini S, editor. Handbook of experimental pharmacology. Arthropod venoms. Berlin: Spinger-Verlag; 1978. p. 312-7.
- 3. Chippaux JP, Goyffon M. Epidemiology of scorpionism: a global appraisal. Acta Trop. 2008;107(2):71-9.
- 4. Levy G, Amitai P. Fauna Palaestina, Arachnida I: Scorpiones. Jerusalem: Israel Academy of Sciences and Humanities; 1980. p. 130.
- 5. Ross LK. Notes on gestation periods and litter size in the arenicolous buthid scorpion Leiurus quinquestriatus (Ehrenberg, 1828) (Scorpiones: Buthidae). J Venom Anim Toxins incl Trop Dis. 2009;15(2):347-52.
- 7. Lourenço WR, Modry D, Amr Z. Description of a new species of Leiurus Ehrenberg, 1828 (Scorpiones, Buthidae) from the South of Jordan. Revue Suisse de Zoologie. 2002:109(3):635-42.
- 8. Lourenço WR, Qi JX, Cloudsley-Thompson JL. The African species of the genus Leiurus Ehrenberg, 1828 (Scorpiones: Buthidae) with the description of a new species. Bol Soc Entomol Aragonesa. 2006:39;97-101.
- 9. Kovarik F. Leiurus nasheri sp. nov. from Yemen (Scorpiones, Buthidae). Acta Soc Zool Bohem. 2007;71:137-41.
- 10. Tulga T. Cross-reactions between anti-scorpion (Buthus quinquestriatus) and anti-scorpion (Prionurus crassicauda) sera. Türk Hij Tec Biol Derg. 1960;20(2):191-203.
- 11. Crucitti P, Vignoli V. Gli scorpioni (Scorpiones) dell'Anatolia sud-orientale (Turchia). Boll Mus Reg Sci Nat Torino. 2002;19(2):433-80
- 12. Khalil N, Yagmur EA. Leiurus abdullahbayrami (Scorpiones: Buthidae), a new species for the scorpion fauna of Syria. Serket. 2010;12(1):1-6.
- 13. Ozkan O, Adiguzel S, Inceoglu B, Lango J, Ertek M, Hammock BD. Evaluation of the neutralizing capacity of Androctonus crassicauda (Olivier, 1807) antivenom against Leiurus quinquestratius (Ehrenberg, 1928) venom (Scorpiones: Buthidae). J Venom Anim Toxins incl Trop Dis. 2008;14(3):481-96.
- 14. Adiguzel S. In vivo and in vitro effects of scorpion venoms in Turkey: a mini-review. J Venom Anim Toxins incl Trop Dis. 2010;16(2):198-211.
- 15. Behrens B, Karber C. Wie sind Reihenversuche fur biolgische Auswertungen am zwerkmabigsten Anzuordnen. Arch Exp Pathol Pharmakol. 1935;177:379-8.
- 16. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4 Nature. 1970;227(5259):680-5.
- 17. Fet V, Soleglad ME. Contributions to scorpion systematics. I. On recent changes in high-level taxonomy. Euscorpius. 2005;31:1-13.
- 18. Ismail M. Serotherapy of the scorpion envenoming syndrome is irrationally convicted without trail. Toxicon. 1993;31:1077-8.
- 19. Ismail M. The scorpion envenoming syndrome. Toxicon. 1995;33(7):825-58.
- 20. Dittrich K, Power AP, Smith NA. Scorpion sting syndrome - a ten year experience. Ann Saudi Med. 1995;15(2):148-5.
- 21. Al-Asmari AK, Al-Saif AA, Abdo NM. Morphological identification of scorpion species from Jazan and Al-Medina Al-Munawara regions, Saudi Arabia. J Venom Anim Toxins incl Trop Dis. 2007;13(4):821-3.
- 22. Fatani AJ, Ahmed AA, Abdel-Halim RM, Abdoon NA, Darweesh AQ. Comparative study between the protective effects of Saudi and Egyptian antivenoms, alone or in combination with ion channel modulators, against deleterious actions of Leiurus quinquestriatus scorpion venom. Toxicon. 2010; 55(4):773-86.
- 23. Krifi MN, Marrakchi N, El Ayeb M, Dellagi K. Effect of some variables on the in vivo determination of scorpion and viper venom toxicities. Biologicals. 1998;26(4):277-88.
- 24. Bücherl W. Scorpionism in the Old World. In: Bücherl W, Buckley EE, editors. Venomous animals and their venoms. New York: Academic Press; 1971. p. 317-47.
- 25. Zlotkin E, Miranda F, Rochat H. Chemistry and pharmacology of Buthidae scorpion venoms. In: Bettini S, editor. Handbook of experimental pharmacology. Arthropod Venoms. Berlin: Springer-Verlag; 1978. p. 317-29.
- 26. El-Alfy AT, Ahmed AA, Fatani AJ, Kader F. Amelioration of the cardiovascular manifestations of the yellow scorpion Leiurus quinquestriatus envenomation in rats by red grape seeds proanthocyanidins. Toxicon. 2008;51(3):321-33.
- 27. Loret E, Hammock BD. Structure and neurotoxicity of venoms. In: Brownell P, Polis G, editors. Scorpion biology and research. New York: Oxford University Press; 2001. p. 204-33.
- 28. Calvete JJ. Antivenomics and venom phenotyping: a marriage of convenience to address the performance and range of clinical use of antivenoms. Toxicon. 2010;56(7):1284-91.
- 29. Possani LD, Merino E, Corona M, Bolivar F, Becerril B. Peptides and genes coding for scorpion toxins that affect ion-channels. Biochimie. 2000;82(9-12):861-8.
- 30. Mebs D. Scorpions and snakes, such as cobras, mambas and vipers made the African continent famous for venomous animals. Bull Soc Pathol Exot. 2002;95(3):131.
- 31. Possani LD, Becerril B, Delepierre M, Tytgat J. Scorpion toxins specific for Na+- channels. Eur J Biochem. 1999;264(2):287-300.
- 32. Rodríguez de la Vega RC, Possani, LD. Current views on scorpion toxins specific for K +-channels. Toxicon. 2004;43(8):865-75.
- 33. Rodríguez de la Vega RC, Possani, LD. Overview of scorpion toxins specific for Na+ channels and related peptides: biodiversity, structure-function relationships and evolution. Toxicon. 2005;46(8):831-44.
- 34. Gueron M, Ilia R, Sofer S. The cardiovascular system after scorpion envenomation. A review. J Toxicol Clin Toxicol. 1992;30(2):245-58.
- 35. Gajre G, Dammas AS. Scorpion envenomation in children: should all stings be given antivenom? Ann Saudi Med. 1999;19(5):444-6.
- 36. Sofer S, Shahak, E, Slonim A, Gueron M. Myocardial injury without heart failure following envenomation by the scorpion Leiurus quinquestriatus in children. Toxicon. 1991;29(3):382-5.
- 37. Sofer S, Shalev H, Weizman Z, Shahak E, Gueron M. Acute pancreatitis in children following envenomation by the yellow scorpion Leiurus quinquestriatus. Toxicon. 1991;29(1)125-8.
- 38. Sofer S, Gueron M. Respiratory failure in children following envenomation by the scorpion Leiurus quinquestriatus: hemodynamics and neurological aspects. Toxicon. 1988;26(10):931-9.
- 39. Shalita EA, Wells RD. Treatment of yellow scorpion (Leiurus quinquestriatus) sting: a case report. J Am Pharm Assoc. 2007;47(5):616-9.
- 40. Söker M, Haspolat K. Güneydogu ve Anadolu bölgesinde çocuklarda akrep sokmasi: 64-vakanin degerlendirilmesi. Çocuk Sagl1g1 ve Hastal1klar1 Dergisi. 2000;43(1):43-50.
- 41. Bosnak M, Ece A, Yolbas I, Bosnak V, Kaplan M, Gurkan F. Scorpion sting envenomation in children in southeast Turkey. Wilderness Environ Med. 2009;20(2):118-24.
- 42. Al B, Y1lmaz DA, Sögut O, Örak M, Üstundag M, Bokurt S. Epidemiological, clinical characteristics and outcome of scorpion envenomation in Batman, Turkey: an analysis of 120 cases. Akademik Acil Tip Dergisi. 2009;8(3):9-14.
- 43. Gwee MC, Nirthanan S, Khoo HE, Gopalakrishnakone P, Kini RM, Cheah LS. Autonomic effects of some scorpion venoms and toxins. Clin Exp Pharmacol Physiol. 2002;29(9):795-801.
- 44. Dittrich K, Ahmed R, Ahmed QA. Cardiac arrest following scorpion envenomation. Ann Saudi Med. 2002;22(1-2):87-90.
- 45. Adiguzel S, Ozkan O, Inceoglu B. Epidemiological and clinical characteristics of scorpionism in children in Sanliurfa, Turkey. Toxicon. 2007;49(6):875-80.
Publication Dates
-
Publication in this collection
05 Dec 2011 -
Date of issue
2011
History
-
Accepted
13 June 2011 -
Received
28 Feb 2011