Abstract
In order to evaluate the pathogenicity of yeasts isolated from vaginal secretion of pregnant and non-pregnant women - stored in mineral oil at the URM Mycology Collection, Department of Mycology, Federal University of Pernambuco - 30 samples belonging to the genera Candida, Rhodotorula, Trichosporon, and Kloeckera, were studied regarding their pathogenic characteristics, ability to grow at room temperature (28°C ± 1°C), 37°C, and 42°C for 72 hours, and production of both phospholipase and proteinase. Results showed that all 30 isolates (100%) were able to grow at room temperature and 37°C, and that 17 samples (57%) were able to grow at 42°C. Evaluation of enzymatic activity showed protease activity in only two isolates (7%), namely C. maritima and C. obtusa. Phospholipase activity was detected in 20 isolates (67%) using soy lecithin as substrate at different temperatures. The characterization of yeasts isolated from vaginal secretion and determination of their enzymatic activity may contribute to understanding the epidemiology of vulvovaginitis and assist in the treatment of patients.
pathogenicity; yeasts; enzymes
ORIGINAL PAPERS
Pathogenic characteristics of yeasts isolated from vaginal secretion preserved under mineral oil
Severo Gomes BI; Souza Motta CMI; Lima ANI; Porto ALFII
IDepartment of Mycology, Federal University of Pernambuco, Recife, Pernambuco State, Brazil
IIDepartment of Morphology and Physiology, Federal Rural University of Pernambuco, Recife, Pernambuco State, Brazil
Correspondence to Correspondence to: Bruno Severo Gomes Av. Engenheiro Domingos Ferreira, 4403, ap. 702 Boa Viagem, Recife, PE, 51020-040, Brasil Phone: +55 81 2126 8948 Email: bseverogomes@gmail.com
ABSTRACT
In order to evaluate the pathogenicity of yeasts isolated from vaginal secretion of pregnant and non-pregnant women - stored in mineral oil at the URM Mycology Collection, Department of Mycology, Federal University of Pernambuco - 30 samples belonging to the genera Candida, Rhodotorula, Trichosporon, and Kloeckera, were studied regarding their pathogenic characteristics, ability to grow at room temperature (28°C ± 1°C), 37°C, and 42°C for 72 hours, and production of both phospholipase and proteinase. Results showed that all 30 isolates (100%) were able to grow at room temperature and 37°C, and that 17 samples (57%) were able to grow at 42°C. Evaluation of enzymatic activity showed protease activity in only two isolates (7%), namely C. maritima and C. obtusa. Phospholipase activity was detected in 20 isolates (67%) using soy lecithin as substrate at different temperatures. The characterization of yeasts isolated from vaginal secretion and determination of their enzymatic activity may contribute to understanding the epidemiology of vulvovaginitis and assist in the treatment of patients.
Key words: pathogenicity, yeasts, enzymes.
INTRODUCTION
Yeasts are unicellular fungi that primarily reproduce by budding or fission. They are widely distributed in nature and have been isolated from humans and other animals, food, plants, air, soil, and water (1). Vulvovaginal infection by yeasts presents as an inflammation of genital mucosa, mainly affecting the vulva and vagina, and is developed as a result of yeast infection which may be part of the microbiota (2, 3). However, the factors responsible and mechanisms resulting in pathological effects are poorly understood in vulvovaginal infection by yeast (3, 4).
The main etiological agent of candidiasis is Candida albicans. Studies show that this species constitutes at least 60% of all Candida isolated from clinical samples. Since this yeast is a part of the normal human microbiota, candidiasis can be considered an opportunistic infection (1). However, other Candida species have also been frequently reported in the literature as etiological agents for candidiasis, including: C. tropicalis, C. parapsilosis, C. krusei, C. guilliermondii, C. glabrata, C. kefyr, C. lusitaniae, C. viswanathii and C. famata. All these have already been isolated from clinical samples (5).
Studies reinforce the need to evaluate the vulvovaginal infection by yeasts as a major cause of health impairment in women (4). The ability of some fungi to grow at up to 37°C, and also to produce enzymes such as phospholipases and proteinases permit establishment of a relationship with pathogenicity (5).
Several substances produced by yeasts have been associated with infection, or regarded as virulence factors. Infections caused by these microorganisms are directly related to the production of exoenzymes. Currently, these enzymes have been considered one of the most important virulence factors for yeasts (6-11). It has been reported that enzymes like phospholipase and proteinases are involved in the virulence of C. albicans. Nevertheless, little is known about the role these exoenzymes play in the pathogenesis of vulvovaginal candidiasis (7, 12, 13).
The pathogenesis of vulvovaginal infection by yeasts is still not completely understood, but consensus is that it is currently a significant problem in women's health. Professionals working in this area have a need to understand current issues addressed on its pathogenesis and to characterize host influences and virulence factors of causative agents to identify their importance in this pathology (14-16).
In the diagnostic laboratory, C. albicans is the species most frequently isolated from the female genital tract, since 20 to 25% of asymptomatic women have a positive vaginal culture. About 75% of adult women have at least one episode of vulvovaginal candidiasis in their lifetime, with a prevalence of C. albicans in 70 to 90% (17). Some of these patients, about 5%, are likely to have new episodes and suffer recurrences (18, 19).
The ability to produce hydrolytic enzymes is considered an important pathogenic factor that interferes with fungus invasion in the host cell, causes the destruction of cellular components, contributes to tissue damage and causes partial or total cell dysfunction. Moreover, proteinases hydrolyze peptide bonds whereas phospholipases hydrolyze phosphoglycerides (20-22).
Another factor also considered characteristic of pathogenicity is the ability of some fungi to grow at 37°C; however, growth at 37°C and 42°C allow species delimitation (23).
Determinants of pathogenicity to specific yeasts have been raised with the aim of producing new antifungal drugs, including growth at 37°C and enzymatic activities of phospholipase and protease (24, 25).
Protease production has been associated with the pathogenicity of yeasts (26). According to some authors, these enzymes are responsible for the persistence and colonization of the yeasts in the vaginal tract (7). They also facilitate tissue penetration and immunoglobulin A (IgA) cleavage, an important factor in vaginal immunity (26, 27).
The aim of this work was to detect extracellular enzymes, proteases and phospholipases produced by yeast species isolated from vaginal secretions as a biotechnological technique to establish the identity and relationship of yeast species.
MATERIALS AND METHODS
Yeasts
Thirty yeasts strains isolated from vaginal secretion samples were selected from stocks stored in mineral oil at the URM Mycology Collection of the Department of Mycology, Center of Biological Sciences, Federal University of Pernambuco, Brazil. The project was approved by the Ethics Research Committee of the Health Science Center, Pernambuco Federal University.
The following pathogenic characteristics were studied: the ability to grow at room temperature (RT = 28°C ± 1°C), 37°C and 42°C for 72 hours, and the production of both phospholipase and proteinase. All cultures were from the URM Mycology Collection and were subjected to taxonomic confirmation according to traditional criteria which are based on morphological and physiological evidence (28-30).
Sample Reactivation
The stock samples preserved in mineral oil were reactivated by growing in glycoside broth and then transferred to slants containing Sabouraud agar plus yeast extract (SAB+YE).
Detection of Pathogenic Characteristics
Growth temperature
Yeast growth was analyzed at room temperature (RT = 28°C ± 1°C), 37°C, and 42°C for 72 hours. For each temperature, each yeast isolate was streaked in quadruplicate on Sabouraud agar in test tubes.
Detection of enzymes
Tests were performed in triplicate for detection of protease enzymes and phospholipases. Seeding was performed by moving fragments of 72-hour growth culture onto medium surface at the center of culture plates. The three plates were incubated at room temperature (RT = 28°C ± 1°C) and 37°C. Protease and phospholipase production was detected by the formation of halos around the culture (31).
Detection of Protease
Protease activity was verified using milk casein (plate) and gelatin (in tubes) as substrates (1). Protease activity on agar plates was observed by casein hydrolysis in a medium containing skimmed milk; then seeding samples were observed for growth and enzyme activity by halo formation over a 10- to 15-day period. Protease was detected by the formation of transparent halos around the colonies. Production of this enzyme was confirmed with acidified mercuric chloride solution (HgCl2, 12 g; concentrated HCl, 16 mL; distilled water, 80 mL). Enzyme activity was estimated by the difference between circle and colony diameter. The absence of a halo indicated the absence of protease activity. Protease activity was verified by gelatin liquefaction using a gelatin substrate kept in tubes stored at room temperature (RT = 28°C ± 1°C).
Detection of Phospholipase
A modified method was adopted to verify phospholipase presence, in which seeding was performed by placing culture samples on the center of Petri dishes containing Sabouraud agar supplemented with 1 M sodium chloride, calcium chloride 0.005 M and two egg yolks (the modification consisted of replacing the egg yolk with a emulsion from Difco Laboratories, USA) and kept at room temperature (RT = 28°C ± 1°C) and 37°C (32). Concurrently, phospholipase activity was investigated by the same method but replacing egg yolks with soy lecithin (31). Observations were made on the 5th, 10th, and 15th day to check and measure the formation of an opaque halo around the colony. Activity zone (ZA) was calculated by dividing colony diameter by the sum of the colony diameter and size of precipitation zone.
Interpretation: ZA equal to or greater than 1.0 detects no phospholipase activity, ZA lower than 1.0 detects phospholipase activity. Statistical analysis consisted of presenting events in absolute numbers, percentages, and averages, and used the Fisher's exact test for association of variables with significance set at p < 0.05.
RESULTS
Stocked yeasts strains from the URM Mycology Collection were viable regardless of preservation time, presenting growth when transferred from glycoside broth to SAB+YE (30). Confirmation of strain identification: all studied yeasts strains displayed sugar assimilation and fermentation patterns that corresponded to previously identified species (15).
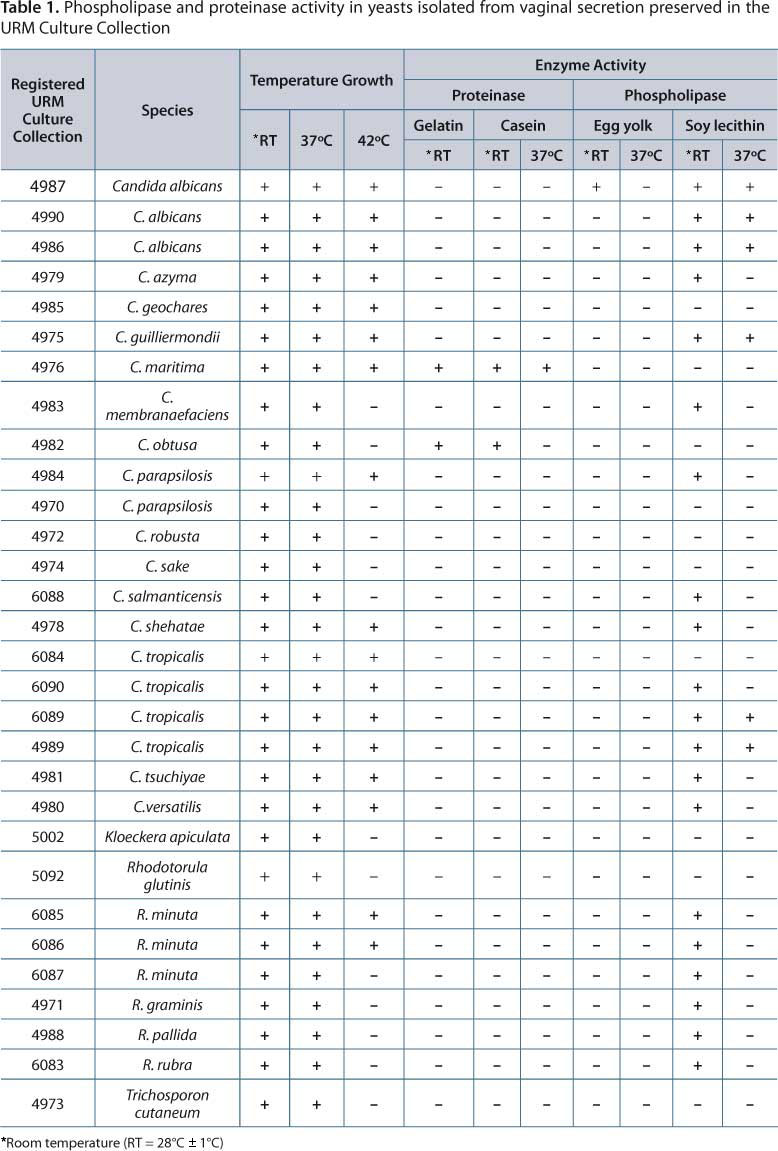
All 30 isolates showed growth at room temperature (RT = 28°C ± 1°C) and 37°C, and 17 (57%) grew at 42°C (Table 1). To detect enzyme activity, protease activity was observed in two isolates (7%) C. maritima and C. obtusa. C. maritima URM 4976 presented proteolytic activity both in medium containing gelatin (RT = 28°C ± 1°C) and medium containing casein (RT = 28°C ± 1°C and 37°C) as substrate. C. maritima URM 4976 also grew at all temperatures tested (RT = 28°C ± 1°C, 37°C and 42°C). The yeasts were isolated from vaginal secretions of non-pregnant women without vulvovaginal yeast infection symptoms. At room temperature only, C. obtusa URM4982 also showed proteolytic activity on gelatin and casein. Growth was observed at two temperatures (RT = 28°C ± 1°C and 37° C).
The phospholipase activity was detected in 20 isolates (67%) using soy lecithin as substrate at different temperatures:
-
C. albicans URM4987 showed phospholipase activity in egg yolk (
Table 1).
-
C. albicans (URM4987, URM4990, URM4986),
C. tropicalis (URM6089, URM4989), and
C. guilliermondii URM4975 showed phospholipase activity in soy lecithin substrate at room temperature and 37°C.
-
C. parapsilosis 4984URM,
C. tropicalis URM6090,
C. salmanticensis URM6088,
C. tsuchiyae URM4981,
C. shehatae URM4978,
C. versatilis URM4980,
C. membranaefaciens URM 4983,
C. azyma URM4979,
R. minuta (URM6085, URM6086, URM6087)
R. graminis URM4971,
R. pallida URM 4988,
R. rubra URM6083 showed phospholipase activity in lecithin substrate at room temperature (
Table 1).
-
Phospholipase activity was not observed in
C. parapsilosis 4970URM,
C. tropicalis URM6084,
C. maritima URM4976,
C. geochares URM4985,
C. obtusa URM4982,
C. robusta URM4972,
C. sake URM4974,
K. apiculata URM5002,
R. glutinis URM5092 and
T. cutaneum URM4973.
In relation to pregnancy C. albicans URM4987 was isolated from vaginal secretions of pregnant women with symptoms, and showed phospholipase activity at the two temperatures studied, while C. robusta URM4972, also isolated from pregnant women with symptoms, was inactive. The other yeast isolated from vaginal secretions of pregnant women, but without symptoms, was R. minuta URM6086. This one only showed phospholipase activity at ambient temperature.
Of the 30 samples analyzed, 19 were isolated from vaginal secretions of pregnant women without symptoms. Phospholipase activity (at 37°C) was detected in C. albicans URM4990, C. tropicalis URM6089, and C. guilliermondii URM4975. In C. albicans URM4986 and C. tropicalis URM4989 the activity was also observed; however, the yeasts were isolated from vaginal secretions of non-pregnant women without symptoms.
DISCUSSION
Host predisposing factors have been well studied and are therefore known, at least in part, by professionals working in women's health. Nevertheless, aspects related to pathogens, which are yeasts, are little known. Thus, updating and knowledge of basic concepts and clinical trials related to this pathology are very important in helping professionals monitor the infection (14). The importance of understanding the process of virulence, particularly at the bacterial level has been recognized in recent years, and that further studies are required on yeasts, especially those isolated from vaginal secretion (17).
Agar diffusion techniques using solid media are commonly employed to verify the activity of different enzymes. Then, results are expressed by the formation of halos resulting from the hydrolysis of specific substrates or indicator color change in culture medium (32).
Detection of protease activity in just two of a large number of studied samples has been frequently reported by some authors. Some studies have found no protease activity in any of the isolates (33). Several authors have reported detecting phospholipase in yeast species, which are mentioned as being able to express this enzyme (34).
In our results we observed phospholipase in C. parapsilosis in soy lecithin substrate. This finding differs from other authors and corroborates the results of other studies in which phospholipase activity was detected in C. albicans, C. parapsilosis, C. tropicalis, and C. guilliermondii (35, 36). Enzymatic activity has been detected in yeasts isolated from biological samples from different anatomical origins (anus, vagina, and mouth) in 17 patients (37, 38). The detection of phospholipase and proteinase enzymes on solid media has been described, especially in C. albicans samples. Some studies also found that certain species of yeasts can produce proteases whereas others cannot, which may contribute to the increased virulence.However, in numerous cases of vaginal infection caused by yeasts proteinase activity is not detected. This may indicate that the secretion of protease is not the only factor involved in the ability of causing vulvovaginitis (35, 36).
Regarding the fact that we detected phospholipase production in 67% of samples among 14 different species, our results differ from those found in isolates from the oral cavity, in which it was found only in C. albicans samples (35).
Previous studies have shown that 30 to 100% of C. albicans isolates are phospholipase secretory (36). In our study we observed phospholipase activity in C. tropicalis and C. parapsilosis, which differs from another research that using 41 Candida strains, detected it only in C. albicans (39). However, a different study found that, in addition to C. albicans and C. parapsilosis strains, C. tropicalis and C. guilliermondii also showed phospholipase activity, which confirms our results (36).
In Brazil, studies found that 51.1% of analyzed C. albicans samples showed phospholipase activity (40, 41). Percentages are variable in relation to activity detection, with values of 73.3, 96.6, and even 100% of samples (36, 39, 42). Regarding C. albicans, the present findings showed varying degrees of phospholipase activity, within the parameters found by other authors (32, 39).
For enzyme activity in C. albicans, the three strains tested were positive for phospholipase production (100%), but none was positive for protease. Our results differ from other authors that observed 84.61% of the samples producing phospholipase and 61.83%, proteinase (35). It also differs from findings from a study that showed a 71.9% frequency of phospholipase production and 68.7% of proteinase in C. albicans samples (43).
Unlike our results, the latter study did not detect any protease activity in other yeast species, except for C. tropicalis (43). Enzyme production by yeasts isolated from any anatomical site is of great epidemiological importance. In Candido et al. (35), 35.3% of isolates tested demonstrated both types of enzymatic activity, indicating the importance of performing correlation between the production of these enzymes and pathogenicity, especially if isolated from clinically healthy individuals, so that more accurate preventive methods can be adopted (35).
The proteinase-producing capacity was found in C. tropicalis, C. parapsilosis and C. glabrata isolates of samples from anal mucous secretion, sputum, oropharyngeal secretions and urine. According to some authors, the test is important for defining the microorganism as etiological agent or colonizer in sites where they are part of the normal microbiota. This explains the importance of collection even in the absence of symptoms (44).
Thus the characterization of yeasts isolated from vaginal secretion, and the determination of their enzymatic activity may contribute to the understanding of vulvovaginitis epidemiology and assist in patient treatment.
ACKNOWLEDGEMENTS
The authors are grateful to The National Council for Scientific and Technological Development (CNPq), Micoteca URM604 (URM Mycology Collection - Federal University of Pernambuco, Recife, PE, Brazil) and Prof. Lusinete Aciole de Queiroz (in memoriam).
Submission status
Received: April 18, 2011.
Accepted: July 11, 2011.
Abstract:published online: July 13, 2011.
Full paper published online: November 30, 2011.
Conflicts of interest
There is no conflict.
Financial source
The National Council for Scientific and Technological Development (CNPq) provided the financial grants.
Ethics committee approval
The present study was approved by the Ethics Research Committee of the Health Science Center, Federal University of Pernambuco, under the protocol number 304/2003-CEP/CCS.
- 1. Lacaz CS, Porto E, Martins JEC, Heins-Vaccari EM. Tratado de micologia médica. 9th ed. São Paulo: Sarvier; 2002. 1104 p.
- 2. Adad SJ, de Lima RV, Sawan ZT, Silva ML, de Souza MA, Saldanha JC, et al. Frequency of Trichomonas vaginalis, Candida sp. and Gardnerella vaginalis in cervical-vaginal smears in four different decades. São Paulo Med J. 2001;119(6):200-5.
- 3. Ziarrusta GB. Vulvovaginitis candidiásica. Rev Iberoam Micol. 2002;19(1): 22-4.
- 4. Cavalcante VLN, Miranda AT, Portugal GMP. Rastreamento de candidose vaginal durante a prevenção do câncer cérvico-uterino. DST J Bras Doenças Sex Transm. 2005;17(1):44-8.
- 5. Neder RN. Microbiologia: manual de laboratório. São Paulo: Nobel; 1992. 98 p.
- 6. Wain WH, Price MF, Cawson RA. Factors affecting plague formation by Candida albicans infecting chick chorio-allantoic membrane. Sabouraudia. 1976;14(2):149-51.
- 7. Ghannoum MA. Potential role of phospholipases in virulence and fungal pathogenesis. Clin Microbiol Rev. 2000;13(1):122-43.
- 8. De Bernardis F, Sullivan PA, Cassone A. Aspartyl proteinases of Candida albicans and their role in pathogenicity. Med Mycol. 2001;39(4):303-13.
- 9. Mardh PA, Rodrigues AG, Genç M, Novikova N, Martinez-de-Oliveira J, Guaschino S. Facts and myths on recurrent vulvovaginal candidosis: a review on epidemiology, clinical manifestations, diagnosis, pathogenesis and therapy. Int J STD AIDS. 2002;13(1):522-39.
- 10. Marrazzo JM. Vulvovaginal candidiasis: over the counter doesn't seem to lead to resistance. Br Med J. 2003;326(7397):993-4.
- 11. Nardin ME. Prevalencia de la candidiasis vulvovaginal y su relación con algunos factores de riesgo. Rev Argent Microbiol. 2000.
- 12. Dostal J, Hamal P, Pavlícková L, Soucek M, Ruml T, Pichová I, et al. Simple method for screening Candida species isolates for the presence of secreted proteinases: a tool for the prediction of successful inhibitory treatment. J Clin Microbiol. 2003;41(2):712-6.
- 13. Schaller M, Bein M, Korting HC, Baur S, Hamm G, Monod M, et al. The secreted aspartyl proteinases Sap 1 and Sap 2 cause tissue damage in an in vitro model of vaginal candidiasis based on reconstituted human vaginal epithelium. Infect Immun. 2003;71(6):3227-34.
- 14. Álvares CA, Svidzinski TIE, Consolaro MEL. Candidíase vulvovaginal: fatores predisponentes do hospedeiro e virulência das leveduras. J Bras Patol Med Lab. 2007;43(5):319-27.
- 15. Shinobu CS, Ogatta SFY, Bizerra F, Furlaneto L, Peralta RM, Svidzinski TIE, et al. Ausência de associação entre genótipos e fatores de virulência em C. albicans isoladas de secreção vaginal. Braz J Microbiol. 2007;38(3):467-71.
- 16. Ferrer J. Vaginal candidosis: epidemiological and etiological factors. Int J Gynaecol Obstet. 2000;71 Suppl 1:S21-7.
- 17. Calderone RA, Fonzi WA. Virulence factors of Candida albicans. Trends Microbiol. 2001;9(7):327-35.
- 18. Cotran RS, Kunar K, Robbins SL. Robbins: Patologia estrutural e funcional. 6th ed. Rio de Janeiro: Guanabara-Koogan; 2000. 515 p.
- 19. Dan M, Poch F, Levin D. High rate of vaginal infections caused by non-C. albicans Candida species among asymptomatic women. Med Mycol. 2002;40(4):383-6.
- 20. Sidrim JJC, Rocha MFG. Micologia médica à luz de autores contemporâneos. Rio de Janeiro: Guanabara-Koogan; 2004. 388 p.
- 21. Ghannoum MA, Abu-Elteen KH. Pathogenicity determinants of Candida Mycoses. 1990;33(6):265-82.
- 22. Barrett-Bee K, Hayes Y, Wilson RG, Ryley JF. A comparison of phospholipase activity, cellular adherence and pathogenicity of yeasts. J Gen Microbiol. 1985;131(1):1217-21.
- 23. Trabulsi LR, Alterthum F, Gompertz OF, Candeias JAN. Microbiologia. São Paulo: Ateneu; 2005. 586 p.
- 24. Ruchel R. A variety of Candida proteinases and their possible targets of proteolytic attack in the host. Zentralbl Bakteriol Mikrobiol Hyg Ser A. 1984;257(1):266-74.
- 25. Fuzakawa Y, Kagaya K. Molecular bases adhesion of Candida albicans J Med Vet Mycol. 1997;35(2):87-99.
- 26. Ruchel R. Properties of a purified proteinase from the yeast Candida albicans Biochim Biophys Acta. 1981;659(1):99-113.
- 27. Ruchel R, Uhlemann K, Boning B. Secretion of acid proteinases by different species of genus Candida Zentralbl Bakteriol Mikrobiol Hyg Orig A. 1983;255(1):537-48.
- 28. Lodder J. The Yeast: a taxonomic study. Oxford: North Holland Publishing Company; 1970. 1385 p.
- 29. Kreger-Van Rij NJW. The yeast: a taxonomic study. 3rd ed. Amsterdam: Elsevier Sci Publication; 1984. 1082 p.
- 30. Barnett JA, Yamada T, Nozawa Y. Yeasts: characteristics and identification. 3rd ed. Cambridge: Cambridge University Press; 2000. 1002 p.
- 31. Price MF, Cawson RA. Phospholipase activity in Candida albicans Sabouraudia. 1977;15(1):179-85.
- 32. Price MF, Wilkinson ID, Gentry IO. Plate method for detection of phospholipase activity in Candida albicans Sabouraudia. 1982;20(1):7-14.
- 33. Muhsin TM, Aubaid AH, Al-Duboon AH. Extracellular enzyme activities of dermatophytes and yeast isolates on solid media. Mycoses. 1997;40(1): 465-9.
- 34. Chaves GM, Cavalcanti MAQ, Porto ALF. Pathogenicity characteristics of stocked and fresh yeasts strains. Braz J Microbiol. 2003;34(3):197-202.
- 35. Candido RC, Azevedo RVP, Komesu MC. Enzimotipagem de espécies do gênero Candida isoladas da cavidade bucal. Rev Soc Bras Med Trop. 2000;33(5):437-42.
- 36. Shimizu MT. Fosfolipase em espécies de Candida Rev Microbiol. 1989;20(1):338.
- 37. Silva JO, Ferreira JC, Candido RC. Atividade enzimática, produção de slime e sensibilidade a antifúngicos de Candida sp. Rev Soc Bras Med Trop. 2007;40(3):354-5.
- 38. Al-Hedaithy SSA. Spectrum and proteinase production of yeasts causing vaginitis in Saudi Arabian women. Med Sci Monit. 2002;8(7):CR498-501.
- 39. Samaranayake LP, Raeside JM, McFarlane TW. Factors affecting the phospholipase activity of Candida species in vitro Sabouraudia. 1984;22(1):201-7.
- 40. Menezes EA, Augusto KL, Freire CCF, Cunha FA, Montenegro RM, Montenegro Júnior RM. Frequência e atividade enzimática de Candida spp. na cavidade oral de pacientes diabéticos do serviço de endocrinologia de um hospital de Fortaleza-CE. J Bras Patol Med Lab. 2007;43(4):241-4.
- 41. Bonifácio e Souza EM, Paula CR, Purchio A, Gambale V, Corrêa B, Cury AE. Aspectos morfo-fisiológicos, fatores de virulência e sensibilidade a antifúngicos de amostras de Candida albicans, sorotipos A e B, isoladas em São Paulo, Brasil. Rev Microbiol.1990;21(3):247-53.
- 42. Carvalho LP, Bacellar O, Neves NA, Carvalho EM, Jesus AR. Avaliação da resposta imune celular em pacientes com candidíase recorrente. Rev Soc Bras Med Trop. 2003;36(5):571-6.
- 43. Camargo FP, Alves IA, Parlow MS, Goulart LS. Isolamento de Candida sp. da mucosa vaginal de mulheres atendidas em um serviço de ginecologia do município de Santo Ângelo - RS. NewsLab. 2008:87.
- 44. Chakrabarti A, Nayak N, Talwar P. In vitro proteinase production by Candida species. Mycopathologia. 1991;141(1):163-8.
Publication Dates
-
Publication in this collection
05 Dec 2011 -
Date of issue
2011
History
-
Received
18 Apr 2011 -
Accepted
11 July 2011