Abstract
Background:
The early symptoms of leptospirosis and dengue fever are difficult to distinguish and can cause diagnostic confusion. Due to the large dengue epidemics that has occurred in Brazil in recent years, it is possible that cases of leptospirosis were unreported. Therefore, we performed a retrospective study to detect leptospirosis in patients who were tested for dengue, but whose laboratory diagnoses were negative.
Methods:
Sera samples from 2,017 patients from 48 cities located in the central region of São Paulo state, Brazil, were studied. All samples were subjected to the microscopic agglutination test (MAT), 305 of which were taken from patients five days or less since the onset of symptoms, and were additionally subjected to real-time polymerase chain reaction (PCR).
Results:
The overall prevalence of leptospirosis cases was 21 (1.04%), with 20 through MAT (18 for Icterohaemorrhagiae and two for the Cynopteri serogroup) and one through PCR (amplicon sequencing compatible with Leptospira interrogans). According to previously established criteria, eight cases of leptospirosis were classified as “confirmed” and 13 as “probable”. The Brazilian notification system for health surveillance had no records for 16 patients positive for leptospirosis and, thus, they were considered unreported cases. Statistical analyses revealed that the prevalence of leptospirosis was higher in men (1.56%) than in women (0.56%), and the mean age was higher in positive patients (43.7 years) than in negative ones (32.3 years).
Conclusion:
The results indicated that patients suspected of dengue fever had evidence of leptospirosis or Leptospira infection, and most of these cases were unreported in the Brazilian notification system. The high burden of dengue may contribute to the misdiagnosis of leptospirosis, and health professionals should increase their awareness of leptospirosis as an important differential diagnosis of patients with suspicion of dengue.
Keywords:
Leptospira; Microscopic agglutination test; PCR; Unreported disease; Zoonosis
Background
Leptospirosis is among the leading zoonotic causes of morbidity and mortality worldwide, with annual estimates of 1.03 million cases and 59,900 deaths [11. Costa F, Hagan JE, Calcagno J, Kane M, Torgerson P, Martinez-Silveira MS, et al. Global morbidity and mortality of leptospirosis: A systematic review. PLoS Negl Trop Dis. 2015 Sep 17;9(9):e0003898. ]. In tropical areas, leptospirosis can be indistinguishable from other febrile illnesses, such as dengue fever [22. LaRocque RC, Breiman RF, Ari MD, Morey RE, Janan FA, Hayes JM, et al. Leptospirosis during dengue outbreak, Bangladesh. Emerg Infect Dis. 2005 May;11(5):766-9.]. Early-phase leptospirosis and classic dengue have unspecific and overlapping symptoms, including fever, headache, and muscle pain, which can lead to delayed or missed diagnosis of leptospirosis. This confusion between the early symptoms of both diseases can contribute to delayed diagnosis of leptospirosis cases, which leads to higher mortality rates [33. Flannery B, Pereira MM, Velloso FF, Carvalho CC, Codes LG, Orrico GS, et al. Referral pattern of leptospirosis cases during a large urban epidemic of dengue. Am J Trop Med Hyg. 2001 Nov;65(5):657-63.]. Few studies have been performed in Brazil comparing dengue and leptospirosis diagnosis. In Fortaleza state, approximately 20% of dengue-like cases may be leptospirosis cases in areas where the two diseases are endemic; and the actual number of leptospirosis cases may be 29 to 49 times higher than those diagnosed and reported by the health services [44. Fontes RM, Cavalcanti LPG, Oliveira ACA, Bezerra LFM, Gomes AMM, Colares JKB, et al. A new possibility for surveillance: Do we identify all cases of leptospirosis? Rev Inst Med Trop Sao Paulo. 2015 Sept-Oct;57(5):443-6.].
Brazil has experienced a large epidemic of dengue in recent years, reaching a high incidence between 2015 and 2016, when more than three million cases were estimated [55. Health Surveillance Secretariat of São Paulo state. Boletim Epidemiológico, Vigilância em Saúde no Brasil 2003-2019. Available from: https://portalarquivos2.saude.gov.br/images/pdf/2019/setembro/25/boletim-especial-21ago19-web.pdf
https://portalarquivos2.saude.gov.br/ima...
]. During this period, it is possible that many patients with leptospirosis were misdiagnosed due to the greater attention directed toward dengue. Another aggravating factor is that leptospirosis is a highly neglected disease in Brazil, affecting marginalized populations marked by poverty, low education levels, and racial segregation [66. Martins MHM, Spink MJP. Human leptospirosis as a doubly neglected diseases in Brazil. Cien Saude Colet. 2020 Mar;25(3):919-28. ]. National tenders do not make mentions of research or intervention investments for this disease [66. Martins MHM, Spink MJP. Human leptospirosis as a doubly neglected diseases in Brazil. Cien Saude Colet. 2020 Mar;25(3):919-28. ], and the Brazilian neglected disease program does not include leptospirosis as a priority area [77. Ministry of Health, Brazil. [Neglected diseases: strategies of the Brazilian Ministry of Health]. Rev Saude Publica. 2010 Feb;44(11):200-1. [Article in Portuguese].]. Thus, physicians and other healthcare professional may overlook leptospirosis more often than other febrile illnesses.
The aim of the present study is to investigate whether leptospirosis was the cause of illness in patients with suspected dengue who yielded negative results in laboratory tests. In addition, unreport of leptospirosis cases was investigated in the Brazilian notification system.
Methods
Study design
A retrospective survey was conducted using serum samples stored (-80°C) at the Adolfo Lutz Institute (IAL), Regional Laboratory Center II Bauru, São Paulo state (SP), Brazil, which performs laboratory diagnosis for healthcare units from 38 neighboring cities. The cities with the largest number of studied samples (more than 100) were Agudos, Barra Bonita, Bauru, Igaraçu do Tietê, Jaú and Lins (Additional file 1
Additional file 1.
Leptospirosis diagnosis among patients suspected of dengue fever in Brazil. Number of patients tested and positive results for leptospirosis diagnosis according to the municipality of origin in the central region of São Paulo state, Brazil.
), which together comprises a total population of 707,957 habitants [88. Brazilian Institute of Geography and Statistics - IBGE [Internet, cited 2021 January]. Available from: Available from: https://www.ibge.gov.br/cidades-e-estados.
https://www.ibge.gov.br/cidades-e-estado...
]. Samples of patients tested for dengue between 2014 and 2017 with negative results were included in the study. Dengue diagnosis was performed using enzyme-linked immunosorbent assays (Panbio® Dengue Early ELISA or Panbio® Dengue IgM Capture ELISA). Most patients tested in the IAL were from the central region of SP, where leptospirosis reporting in the majority of cities were below six cases during the study period (Additional file 2
Additional file 2.
Number of leptospirosis cases reported to the Notifiable Diseases Information System (SINAN) between 2014 and 2017 in São Paulo state (SP), Brazil. High reporting rates were concentrated mostly in the central-east and the southeast regions of the state. The central region of SP, where the study was conducted, presented less than six cases in most cities. Data were obtained from www.portalsinan.saude.gov in May 2020. The map was created in the QGIS 2.18 software using graduated style with Natural Breaks algorithm.
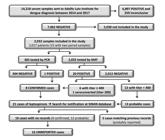
). The study design is shown in Figure 1.
Patients and samples
Among 14,218 samples that were tested for dengue in the intended period, we randomly selected 2,032 belonging to 2,017 patients (15 patients had acute and convalescent samples) (Figure 1). The minimum sample size was estimated as 1,367 patients considering an anticipated prevalence of 3.7% [99. Souza AI, Nogueira JMR, Pereira MM. Anti-Leptospira antibodies in patients in the Mato Grosso do Sul with clinical suspicion of dengue or viral hepatitis. Rev Soc Bras Med Trop. 2007 Jul-Aug;40(4):431-5.], 1% confidence limit, and a population of one million. The OpenEpi platform was used for the statistical calculation [1010. Sullivan KM, Dean A, Soe MM. OpenEpi: a web-based epidemiologic and statistical calculator for public health. Public Health Rep. 2009 May-Jun;123(3):471-4. ].
Flowchart of the study design, methodology, and results of leptospirosis diagnosis among patients suspected of dengue fever in São Paulo state, Brazil.
Diagnosis of leptospirosis
Serological diagnosis of leptospirosis was performed in all samples using a microscopic agglutination test (MAT) executed with standard protocols [1111. Fornazari F, Costa da Silva R, Richini-Pereira VB, Beserra HEO, Luvizotto MCR, Langoni H. Comparison of conventional PCR, quantitative PCR, bacteriological culture and the Warthin Starry technique to detect Leptospira spp. in kidney and liver samples from naturally infected sheep from Brazil. J Microbiol Methods. 2012 Sep;90(3):321-6.]. Five reference strains, representing the major Leptospira serogroups, were initially selected according to their good performance in identifying MAT-confirmed cases of leptospirosis in previous studies conducted in Brazil [1212. Reis RB, Ribeiro GS, Felzemburgh RDM, Santana FS, Mohr S, Melendez AX, et al. Impact of environment and social gradient on Leptospira infection in urban slums. PLoS Negl Trop Dis. 2008 Apr 23;2(4):e228.]. The antigens were kindly provided by the Oswaldo Cruz Foundation (Salvador, Brazil), and included the following serovars and strains: Copenhageni (M20), Canicola (Hond Utrecht IV), Autumnalis (Akyiami), Ballum (Mus 127) and Cynopteri (3522 C). An additional strain named RGA (Icterohaemorrhagiae serogroup) was included due to its good performance in our laboratory tests. The maintenance of the serovar Ballum in the culture media used in our laboratory was extremely difficult, thus, we later excluded this antigen from the study.
Molecular diagnosis was performed in samples from 305 patients who had five days or less between the onset of symptoms and blood collection, which is the period with higher chances of detecting leptospires in blood [1313. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001 Apr;14(2):296-326.]. Serum DNA was extracted using an IllustraTM Blood Genomic Prep Mini spin kit according to the manufacturer’s instructions. To optimize the concentration of the assay template, DNA extraction was performed with 400 µL of serum instead of 200 µL. Samples were subjected to real-time polymerase chain reaction (PCR) using StepOneTM Plus Real Time PCR System equipment, Power SYBR® Green mix, and Lipl32 primers [1414. Stoddard RA, Gee JE, Wilkins PP, McCaustland K, Hoffmaster AR. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn Microbiol Infect Dis. 2009 Jul;64(3):247-55.]. Cycling conditions consisted of initial denaturation at 95˚C for 10 min, followed by 45 cycles of amplification (95˚C for 10 s and 58˚C for 30 min) and melting curve analysis. PCR amplicons were sequenced using the Sanger method as previously described [1515. Fornazari F, Langoni H, Marson PM, Nóbrega DB, Teixeira CR. Leptospira reservoirs among wildlife in Brazil: Beyond rodents. Acta Trop. 2018 Feb;178:205-12.] and compared to the Leptospira sequences available from the NCBI collection.
Since early symptoms of dengue and leptospirosis are very similar and difficult to distinguish [22. LaRocque RC, Breiman RF, Ari MD, Morey RE, Janan FA, Hayes JM, et al. Leptospirosis during dengue outbreak, Bangladesh. Emerg Infect Dis. 2005 May;11(5):766-9., 33. Flannery B, Pereira MM, Velloso FF, Carvalho CC, Codes LG, Orrico GS, et al. Referral pattern of leptospirosis cases during a large urban epidemic of dengue. Am J Trop Med Hyg. 2001 Nov;65(5):657-63.], we considered that patients tested for dengue had clinical signs consistent with leptospirosis. Thus, positive cases in leptospirosis diagnosis were classified as “probable” or “confirmed” according to criteria established by the Leptospirosis Burden Epidemiology Reference Group. Probable cases were defined when MAT titre were 100 or 200 in single samples, while confirmed cases were defined in any one of the following: MAT titre ≥ 400; fourfold increase in MAT titre in acute and convalescent samples; detection of pathogenic Leptospira DNA by PCR [1616. World Health Organization. Report of the Second Meeting of the Leptospirosis Burden Epidemiology Reference Group. 2011. Available from: https://apps.who.int/iris/handle/10665/44588.
https://apps.who.int/iris/handle/10665/4...
].
Unreported leptospirosis cases
Leptospirosis cases were verified if they were unreported by accessing the Notifiable Diseases Information System database (SINAN) [1717. Notifiable Diseases Information System - SINAN. Available from: http://tabnet.datasus.gov.br/cgi/deftohtm.exe?sinannet/cnv/leptosp.def.
http://tabnet.datasus.gov.br/cgi/deftoht...
]. Since the incidence of reported leptospirosis was below six cases in most of the studied cities (Additional file 2
Additional file 2.
Number of leptospirosis cases reported to the Notifiable Diseases Information System (SINAN) between 2014 and 2017 in São Paulo state (SP), Brazil. High reporting rates were concentrated mostly in the central-east and the southeast regions of the state. The central region of SP, where the study was conducted, presented less than six cases in most cities. Data were obtained from www.portalsinan.saude.gov in May 2020. The map was created in the QGIS 2.18 software using graduated style with Natural Breaks algorithm.
), notification of each case was possible by searching for records using the variables “city” and “year of diagnosis”. If one or more cases were registered with the same data, the variables “age”, “gender”, and “month of symptoms” of the investigated patient were included in the search. Leptospirosis cases with no matching records in SINAN were classified as “unreported”, and those for which all the variables matched previous records were classified as “probably reported” (Figure 1).
Data analysis
Frequencies were calculated with a 5% significance level. The results of leptospirosis diagnosis were compared with patient’s sex using the Chi-square test and, if significance was detected, the odds ratio (OR) was calculated. The age of the patients was tested for normality of data using the Shapiro-Wilk test and, in case of non-normal distribution, comparison with the results of leptospirosis diagnosis was made through the Kruskal-Wallis test followed by the post-hoc Dunn’s test. The variables were considered significant when the P value was < 0.05. All analyses were performed using the R software.
Results
Subjects were from 48 municipalities located mainly in the central region of SP (Figure 2 and Additional file 1 Additional file 1. Leptospirosis diagnosis among patients suspected of dengue fever in Brazil. Number of patients tested and positive results for leptospirosis diagnosis according to the municipality of origin in the central region of São Paulo state, Brazil. ). Most patients were female (52.5%), ages ranged from six days to 88 years (mean of 32.4 years), and the time between onset of symptoms and blood sampling ranged from one to 73 days (mean of 8.4 days). The 15 patients with paired samples had a mean of 8.8 days between sample collection.
The diagnosis of leptospirosis revealed 21 (1.04%; CI 95% 0.6 - 1.5) positive patients distributed in 11 cities, of which 20 were positive via MAT (18 for Icterohaemorrhagiae and 2 for Cynopteri) and one via PCR (Table 1 and Additional file 1 Additional file 1. Leptospirosis diagnosis among patients suspected of dengue fever in Brazil. Number of patients tested and positive results for leptospirosis diagnosis according to the municipality of origin in the central region of São Paulo state, Brazil. ). The PCR amplicon sequencing hit the expected region of the Lipl32 gene of Leptospira interrogans with 100% identity and a 4e-94 e-value (Additional file 3 Additional file 3. Multiple sequence alignment of the PCR amplicon (FG1453) with 10 best hits obtained in the BLAST search. The consensus sequence and the identity percentage are at the top of the figure. The sequencing chromatogram is above the amplicon sequence. The NCBI accession numbers of the BLAST search are at the left side indicated by the positions 2 to 11 in the alignment. The sequences for the alignment were obtained from www.ncbi.nlm.nih.gov, and the figure was created in the Geneious v2019.2.1 software. ). Among the 21 positive cases, eight were classified as “confirmed” and 13 as “probable”. No records were found for 16 cases (four confirmed and 12 probable), thus, these were considered “unreported”. The remaining five cases matched previous records from SINAN and were, thus, considered “probably reported”. These results are summarized in Figure 1 and Table 1. Statistical analyses revealed that the prevalence of leptospirosis was higher in men (1.56%; OR = 2.8) than in women (0.56%), and the mean age was higher in positive patients (43.7 years) than in negative ones (32.3 years).
Number of patients by municipality tested for leptospirosis in the central region of São Paulo state, Brazil (the map was created using the QGIS 2.18 software with graduated style and Natural Breaks algorithm).
Discussion
Leptospirosis diagnosis was confirmed among patients suspected of dengue fever in the central region of SP. Half of the confirmed cases and almost all probable cases were not recorded in the SINAN database, indicating misdiagnosis and/or misreporting. The early symptoms of dengue and leptospirosis are difficult to distinguish and are the most likely origin of diagnostic confusion, as discussed in previous studies [22. LaRocque RC, Breiman RF, Ari MD, Morey RE, Janan FA, Hayes JM, et al. Leptospirosis during dengue outbreak, Bangladesh. Emerg Infect Dis. 2005 May;11(5):766-9., 33. Flannery B, Pereira MM, Velloso FF, Carvalho CC, Codes LG, Orrico GS, et al. Referral pattern of leptospirosis cases during a large urban epidemic of dengue. Am J Trop Med Hyg. 2001 Nov;65(5):657-63., 1818. Ellis T, Imrie A, Katz AR, Effler PV. Underrecognition of leptospirosis during a dengue fever outbreak in Hawaii, 2001-2002. Vector Borne Zoonotic Dis. 2008 Aug;8(4):541-7.]. The dengue epidemics in Brazil have possibly contributed to misdiagnosis, since health professionals were more prone to suspect dengue. Another aggravating factor may be the low familiarity and awareness of physicians with leptospirosis in the studied region.
SP is the most developed and populous state of Brazil, possessing broad access to free public health services. Therefore, one would expect high efficiency in detecting and reporting diseases whose notification is mandatory. However, the results suggest that leptospirosis remains unreported even in the most developed state of Brazil. Considering the social and economic status of other Brazilian states, mainly those located in the North and Northeast regions, we strongly believe that they have higher rates of unreported leptospirosis than SP. Infectious diseases are considered particularly prone to underestimation due to their specific characteristics, such as asymptomatic or self-limiting course. Therefore, they are represented inadequately by raw surveillance data [1919. Gibbons CL, Mangen MJJ, Plass D, Havelaar AH, Brooke RJ, Kramarz P, et al. Measuring underreporting and under-ascertainment in infectious diseases datasets: a comparison of methods. BMC Public Health. 2014 Feb 11;14:147.], corroborating our results. In order to improve the measurement of a specific disease and promote the best use of resources, public health surveillance systems should be evaluated periodically. Such improvement should assess a broad system of attributes, such as data quality, representativeness, flexibility, sensitivity, timeliness, among others [2020. German RR, Lee LM, Horan JM, Milstein RL, Pertowski CA, Waller MN, et al. Updated guidelines for evaluating public health surveillance systems: recommendations from the Guidelines Working Group. MMWR Recomm Rep. 2001 Jul;50(RR-13):1-35.].
The number of leptospirosis cases in the population studied was probably higher than that detected in our study. Although MAT is considered the immunological reference standard for the diagnosis of leptospirosis [2121. Rajapakse S, Rodrigo C, Handunnetti SM, Fernando SD. Current immunological and molecular tools for leptospirosis: diagnostic, vaccine design, and biomarkers for predicting severity. Ann Clin Microbiol Antimicrob. 2015 Jan 16;14:2.], it has several limitations, and its performance largely depends on the availability of local strains [2222. Hartskeerl RA, Smythe LD. The role of leptospirosis reference laboratories. In: Adler B, editor. Leptospira and leptospirosis. Current Topics in Microbiology and Immunology. Springer-Verlag Berlin Heidelberg. p. 273-88. 2015.]. We were not able to isolate or obtain isolated strains of leptospires from humans in the studied region, and this may have limited the efficiency of our serological tests. Regarding the molecular diagnosis, it is possible that the use of sera instead of blood may have affected the PCR detection rate, since leptospires may adhere to leucocytes and/or red blood cells and incorporate into the clot [1414. Stoddard RA, Gee JE, Wilkins PP, McCaustland K, Hoffmaster AR. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn Microbiol Infect Dis. 2009 Jul;64(3):247-55.]. However, serum may be better than whole blood as a specimen type for the molecular diagnosis of acute leptospirosis, possibly because blood contains more PCR inhibitors [2323. Agapondi SB, Matthias MA, Moreno AC, Vinetz JM. Utility of quantitative polymerase chain reaction in leptospirosis diagnosis: association of level of leptospiremia and clinical manifestations in Sri Lanka. Clin Infect Dis. 2012 May;54(9):1249-55.]. It is uncertain whether the type of sample affected our molecular diagnosis. Other flaws that may have limited the detection of leptospirosis were the low availability of patients with acute and convalescent samples, and the failure to employ the Ballum serovar.
Icterohaemorrhagiae serogroup prevailed among MAT-positive patients. An important retrospective study conducted in SP found that 54.8% of 9,335 seropositive patients reacted to the Icterohaemorrhagiae serogroup [2424. Romero EC, Bernardo CCDM, Yasuda PH. Human leptospirosis: a twenty-nine-year serological study in São Paulo, Brazil. Rev Inst Med Trop S Paulo. 2003 Sep-Oct;45(5):245-8.], corroborating our findings. In humans, severe leptospirosis is frequently but not invariably caused by serovars of the Icterohaemorrhagiae serogroup [1313. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001 Apr;14(2):296-326.], making it one of the most notorious leptospires important to public health. Serovar Icterohaemorrhagiae is the most prevalent serovar reported in rats [2525. Boey K, Shiokawa K, Rajeev S. Leptospira infection in rats: A literature review of global prevalence and distribution. PLoS Negl Trop Dis. 2019 Aug 9;13(8):e0007499.], indicating that this animal group was the possible origin of infection among the MAT-positive patients. Cynopteri was the only reactive serovar of two patients and, curiously, they were from the same city and their onset of symptoms differed by only 72 days (data not shown). According to these patients’ addresses, the distance between their houses was only 1.1 km. Since this was a retrospective study, we could not obtain more detailed information on these two patients, but their close date of symptoms and residences suggest a common source of infection by the Cynopteri serogroup. Data on the importance of Cynopteri causing leptospirosis is very scarce in Brazil, with only one well-documented case [2626. Lupi O, Netto MAC, Avelar K, Romero C, Bruniera R, Brasil P. Cluster of leptospirosis cases among military personnel in Rio de Janeiro, Brazil. Int J Infect Dis. 2013 Feb;17(2):129-31.].
Men had a higher prevalence than women, which is in accordance with most leptospirosis studies, reflecting higher exposure levels to leptospires due to occupational and outdoor activities [2727. Hagan JE, Moraga P, Costa F, Capian N, Ribeiro GS, Wunder EA, et al. Spatiotemporal determinants of urban leptospirosis transmission: Four-year prospective cohort study of slum residents in Brazil. PLoS Negl Trop Dis. 2016 Jan 15;10(1): e0004275., 2828. Lau CL, Watson CH, Lowry JH, David MC, Craig SB, Wynwood SJ, et al. Human leptospirosis infection in Fiji: An eco-epidemiological approach to identifying risk factors and environmental drivers for transmission. PLoS Negl Trop Dis. 2016 Jan 28;10(1):e0004405. ]. The age of positive patients was higher than that of negative ones, suggesting greater risks of infection in individuals above 40 years of age. Age-specific behaviors can be related to Leptospira exposure [2828. Lau CL, Watson CH, Lowry JH, David MC, Craig SB, Wynwood SJ, et al. Human leptospirosis infection in Fiji: An eco-epidemiological approach to identifying risk factors and environmental drivers for transmission. PLoS Negl Trop Dis. 2016 Jan 28;10(1):e0004405. ]; however, high incidences are observed in many age groups above 20 years of age [1616. World Health Organization. Report of the Second Meeting of the Leptospirosis Burden Epidemiology Reference Group. 2011. Available from: https://apps.who.int/iris/handle/10665/44588.
https://apps.who.int/iris/handle/10665/4...
].
Conclusions
In conclusion, approximately 1% of patients suspected of dengue fever had evidence of Leptospira infection, with most of these cases being unreported to the Brazilian notification system. This may imply underreporting leptospirosis in the study area. Health professionals should increase their awareness of leptospirosis among patients presenting unspecific symptoms compatible with dengue, especially during periods with a high burden of dengue.
Abbreviations
CI: confidence interval; DNA: deoxyribonucleic acid; ELISA: enzyme-linked immunosorbent assay; IAL: Adolfo Lutz Institute; MAT: microscopic agglutination test; OR: odds ratio; PCR: real-time polymerase chain reaction; SINAN: Notifiable Diseases Information System; SP: São Paulo state.
Acknowledgments
We are thankful to Adolfo Lutz Institute and Oswaldo Cruz Foundation for providing the samples and the Leptospira strains, respectively.
References
- 1. Costa F, Hagan JE, Calcagno J, Kane M, Torgerson P, Martinez-Silveira MS, et al. Global morbidity and mortality of leptospirosis: A systematic review. PLoS Negl Trop Dis. 2015 Sep 17;9(9):e0003898.
- 2. LaRocque RC, Breiman RF, Ari MD, Morey RE, Janan FA, Hayes JM, et al. Leptospirosis during dengue outbreak, Bangladesh. Emerg Infect Dis. 2005 May;11(5):766-9.
- 3. Flannery B, Pereira MM, Velloso FF, Carvalho CC, Codes LG, Orrico GS, et al. Referral pattern of leptospirosis cases during a large urban epidemic of dengue. Am J Trop Med Hyg. 2001 Nov;65(5):657-63.
- 4. Fontes RM, Cavalcanti LPG, Oliveira ACA, Bezerra LFM, Gomes AMM, Colares JKB, et al. A new possibility for surveillance: Do we identify all cases of leptospirosis? Rev Inst Med Trop Sao Paulo. 2015 Sept-Oct;57(5):443-6.
- 5. Health Surveillance Secretariat of São Paulo state. Boletim Epidemiológico, Vigilância em Saúde no Brasil 2003-2019. Available from: https://portalarquivos2.saude.gov.br/images/pdf/2019/setembro/25/boletim-especial-21ago19-web.pdf
» https://portalarquivos2.saude.gov.br/images/pdf/2019/setembro/25/boletim-especial-21ago19-web.pdf - 6. Martins MHM, Spink MJP. Human leptospirosis as a doubly neglected diseases in Brazil. Cien Saude Colet. 2020 Mar;25(3):919-28.
- 7. Ministry of Health, Brazil. [Neglected diseases: strategies of the Brazilian Ministry of Health]. Rev Saude Publica. 2010 Feb;44(11):200-1. [Article in Portuguese].
- 8. Brazilian Institute of Geography and Statistics - IBGE [Internet, cited 2021 January]. Available from: Available from: https://www.ibge.gov.br/cidades-e-estados.
» https://www.ibge.gov.br/cidades-e-estados. - 9. Souza AI, Nogueira JMR, Pereira MM. Anti-Leptospira antibodies in patients in the Mato Grosso do Sul with clinical suspicion of dengue or viral hepatitis. Rev Soc Bras Med Trop. 2007 Jul-Aug;40(4):431-5.
- 10. Sullivan KM, Dean A, Soe MM. OpenEpi: a web-based epidemiologic and statistical calculator for public health. Public Health Rep. 2009 May-Jun;123(3):471-4.
- 11. Fornazari F, Costa da Silva R, Richini-Pereira VB, Beserra HEO, Luvizotto MCR, Langoni H. Comparison of conventional PCR, quantitative PCR, bacteriological culture and the Warthin Starry technique to detect Leptospira spp. in kidney and liver samples from naturally infected sheep from Brazil. J Microbiol Methods. 2012 Sep;90(3):321-6.
- 12. Reis RB, Ribeiro GS, Felzemburgh RDM, Santana FS, Mohr S, Melendez AX, et al. Impact of environment and social gradient on Leptospira infection in urban slums. PLoS Negl Trop Dis. 2008 Apr 23;2(4):e228.
- 13. Levett PN. Leptospirosis. Clin Microbiol Rev. 2001 Apr;14(2):296-326.
- 14. Stoddard RA, Gee JE, Wilkins PP, McCaustland K, Hoffmaster AR. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn Microbiol Infect Dis. 2009 Jul;64(3):247-55.
- 15. Fornazari F, Langoni H, Marson PM, Nóbrega DB, Teixeira CR. Leptospira reservoirs among wildlife in Brazil: Beyond rodents. Acta Trop. 2018 Feb;178:205-12.
- 16. World Health Organization. Report of the Second Meeting of the Leptospirosis Burden Epidemiology Reference Group. 2011. Available from: https://apps.who.int/iris/handle/10665/44588.
» https://apps.who.int/iris/handle/10665/44588. - 17. Notifiable Diseases Information System - SINAN. Available from: http://tabnet.datasus.gov.br/cgi/deftohtm.exe?sinannet/cnv/leptosp.def.
» http://tabnet.datasus.gov.br/cgi/deftohtm.exe?sinannet/cnv/leptosp.def. - 18. Ellis T, Imrie A, Katz AR, Effler PV. Underrecognition of leptospirosis during a dengue fever outbreak in Hawaii, 2001-2002. Vector Borne Zoonotic Dis. 2008 Aug;8(4):541-7.
- 19. Gibbons CL, Mangen MJJ, Plass D, Havelaar AH, Brooke RJ, Kramarz P, et al. Measuring underreporting and under-ascertainment in infectious diseases datasets: a comparison of methods. BMC Public Health. 2014 Feb 11;14:147.
- 20. German RR, Lee LM, Horan JM, Milstein RL, Pertowski CA, Waller MN, et al. Updated guidelines for evaluating public health surveillance systems: recommendations from the Guidelines Working Group. MMWR Recomm Rep. 2001 Jul;50(RR-13):1-35.
- 21. Rajapakse S, Rodrigo C, Handunnetti SM, Fernando SD. Current immunological and molecular tools for leptospirosis: diagnostic, vaccine design, and biomarkers for predicting severity. Ann Clin Microbiol Antimicrob. 2015 Jan 16;14:2.
- 22. Hartskeerl RA, Smythe LD. The role of leptospirosis reference laboratories. In: Adler B, editor. Leptospira and leptospirosis. Current Topics in Microbiology and Immunology. Springer-Verlag Berlin Heidelberg. p. 273-88. 2015.
- 23. Agapondi SB, Matthias MA, Moreno AC, Vinetz JM. Utility of quantitative polymerase chain reaction in leptospirosis diagnosis: association of level of leptospiremia and clinical manifestations in Sri Lanka. Clin Infect Dis. 2012 May;54(9):1249-55.
- 24. Romero EC, Bernardo CCDM, Yasuda PH. Human leptospirosis: a twenty-nine-year serological study in São Paulo, Brazil. Rev Inst Med Trop S Paulo. 2003 Sep-Oct;45(5):245-8.
- 25. Boey K, Shiokawa K, Rajeev S. Leptospira infection in rats: A literature review of global prevalence and distribution. PLoS Negl Trop Dis. 2019 Aug 9;13(8):e0007499.
- 26. Lupi O, Netto MAC, Avelar K, Romero C, Bruniera R, Brasil P. Cluster of leptospirosis cases among military personnel in Rio de Janeiro, Brazil. Int J Infect Dis. 2013 Feb;17(2):129-31.
- 27. Hagan JE, Moraga P, Costa F, Capian N, Ribeiro GS, Wunder EA, et al. Spatiotemporal determinants of urban leptospirosis transmission: Four-year prospective cohort study of slum residents in Brazil. PLoS Negl Trop Dis. 2016 Jan 15;10(1): e0004275.
- 28. Lau CL, Watson CH, Lowry JH, David MC, Craig SB, Wynwood SJ, et al. Human leptospirosis infection in Fiji: An eco-epidemiological approach to identifying risk factors and environmental drivers for transmission. PLoS Negl Trop Dis. 2016 Jan 28;10(1):e0004405.
-
Availability of data and materials
All data generated or analyzed during this study are included in this article. -
Funding
This study was supported by the São Paulo Research Foundation (FAPESP, process no. 2015/06446-6). -
Ethics approval
The study was approved by the ethics committees of the Adolfo Lutz Institute (protocol no. 59) and the Botucatu Medical School, São Paulo State University (UNESP - protocol no. 5411). -
Consent for publication
Not applicable.
Supplementary material
The following online material is available for this article:
Publication Dates
-
Publication in this collection
26 Mar 2021 -
Date of issue
2021
History
-
Received
05 Aug 2020 -
Accepted
23 Jan 2021