ABSTRACT
This study investigated the anesthetic effect of the essential oils (EOs) from the peel of Citrus x aurantium (EOCA) and Citrus x latifolia (EOCL) on silver catfish Rhamdia quelen. Fish were exposed to different concentrations of EOCA and EOCL to determine time of anesthesia induction and recovery. Induction of anesthesia was observed in all fish exposed to 400, 600 or 800 μL L−1 EOCA and 300, 400 or 500 μL L−1 EOCL. Another group of fish were exposed for 8 h to 50, 100, or 200 μL L−1 of either EOs. Overall, fish exposed to ethanol and both EOs presented higher ventilatory frequencies (VF) than the control group throughout the 8 h of exposure. Net ion (Na+, K+ and Cl−) effluxes and ammonia excretion were significantly lower in fish exposed to 50, 100 or 200 μL L−1 of either EOs compared to control fish. Mortality was 37% in fish exposed to 200 μL L−1 of either EOs after 8 h. These findings suggest that EOCA and EOCL are useful anesthetics and sedatives for Rhamdia quelen, but their usefulness as alternatives to reduce stress in fish transportation at the lower concentrations tested (50-100 µL L−1) deserves further study.
Keywords:
Anesthesia; Ion fluxes; Lemon oil; Orange oil; Ventilatory frequency
RESUMO
O efeito anestésico dos óleos essenciais (OEs) da casca de Citrus x aurantium (OECA) e Citrus x latifolia (OECL) em jundiá Rhamdia quelen foi investigado. Os peixes foram expostos a diferentes concentrações de OECA e OECL para determinar o tempo de indução e recuperação da anestesia. Todos peixes expostos a 400, 600 ou 800 μL L−1 OECA e 300, 400 ou 500 μL L−1 OECL foram anestesiados. Outro grupo de peixes foi exposto aos OEs durante 8 h a 50, 100 ou 200 μL L−1. Peixes expostos ao etanol e aos OEs apresentaram VF maior que o grupo controle durante as 8 h de exposição. Os efluxos líquidos de Na+, K+, Cl− e a excreção de amônia foram significativamente menores nos peixes expostos a 50, 100 ou 200 μL L−1 dos OEs em comparação com o grupo controle. A mortalidade foi de 37% nos peixes expostos a 200 μL L−1 de ambos os OEs após 8 h. Os resultados sugerem que OECA e OECL são anestésicos e sedativos úteis para o jundiá, mas sua utilidade como alternativa para reduzir o estresse no transporte de peixes nas concentrações mais baixas testadas (50-100 μL L−1) necessita de estudos adicionais.
Palavras-chave:
Anestesia; Fluxo de íons; Frequência ventilatória; Óleo de laranja; Óleo de limão
Introduction
Anesthetics have been used to reduce stress effects in animals caused by capture, handling and transportation (in this situation, at sedative concentrations), avoiding the occurrence of physiological, biochemical and molecular changes (Ross, Ross, 2008Ross LG, Ross B. Anaesthetic and sedative techniques for aquatic animals. 3rd ed. Oxford: Blackwell Science; 2008.; Gressler et al., 2012Gressler LT, Riffel APK, Parodi TV, Saccol EMH, Koakoski G, Costa ST et al. Silver catfish Rhamdia quelen immersion anaesthesia with essential oil of Aloysia triphylla (L’Herit) Britton or tricaine methanesulfonate: effect on stress response and antioxidant status. Aquacult Res [serial on the Internet]. 2012; 45(6):1061-72. Available from: http://dx.doi.org/10.1111/are.12043
http://dx.doi.org/10.1111/are.12043...
; Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
; Benovit et al., 2015Benovit SC, Silva LL, Salbego J, Loro VL, Mallmann CA, Baldisserotto B, Flores EMM, Heinzmann BM. Anesthetic activity and bio-guided fractionation of the essential oil of Aloysia gratissima (Gillies & Hook.) Tronc. in silver catfish Rhamdia quelen. An Acad Bras Cienc [serial on the Internet]. 2015; 87(3):1675-89. Available from: http://dx.doi.org/10.1590/0001-3765201520140223
http://dx.doi.org/10.1590/0001-376520152...
; Souza et al., 2017Souza CF, Baldissera MD, Salbego J, Lopes JM, Vaucher RA, Mourão VRH, Caron BO, Heinzmann BM, Silva LVF, Baldisserotto B. Physiological responses of Rhamdia quelen (Siluriformes: Heptapteridae) to anesthesia with essential oils from two different chemotypes of Lippia alba. Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160083. Available from: http://dx.doi.org/10.1590/1982-0224-20160083
http://dx.doi.org/10.1590/1982-0224-2016...
). The transportation of live fish, routinely performed in aquaculture, is one of the main factors that cause stress in fish (Barcellos et al., 2001Barcellos LJG, Woehl VM, Wassermann GF, Quevedo RM, Ittzés I, Krieger MH. Plasma levels of cortisol and glucose in response to capture and tank transference in Rhamdia quelen (Quoy and Gaimard), a South American catfish. Aquacult Res [serial on the Internet]. 2001; 32(2):121-23. Available from: https://doi.org/10.1046/j.1365-2109.2001.00539.x
https://doi.org/10.1046/j.1365-2109.2001...
; Becker et al., 2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
). When stress is induced by this factor, fish react by consuming more energy, which impacts considerably on the maintenance of homeostasis (Park et al., 2008Park MO, Hur WJ, Im SY, Seol DW, Lee J, Park IS. Anaesthetic efficacy and physiological responses to clove oil-anaesthetized kelp grouper Epinephelus bruneus. Aquacult Res [serial on the Internet]. 2008; 39(8):877-84. Available from: https://doi.org/10.1111/j.1365-2109.2008.01941.x
https://doi.org/10.1111/j.1365-2109.2008...
). Several studies have examined the effect of natural products with anesthetic properties, such as plant extracts and EOs, that coupled with their antioxidant properties are a promising alternative to traditional synthetic drugs (Guénette et al., 2007Guénette SA, Uhland FC, Helie P, Beaudry F, Vachon P. Pharmacokinetics of eugenol in rainbow trout (Onchorhynchus mykiss). Aquaculture [serial on the Internet]. 2007; 266(1-4):262-5. Available from: https://doi.org/10.1016/j.aquaculture.2007.02.046
https://doi.org/10.1016/j.aquaculture.20...
; Becker et al., 2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
; Zeppenfeld et al., 2014Zeppenfeld CC, Toni C, Becker AG, Miron DS, Parodi TV, Heinzmann BM, Barcellos LJG, Koakoski G, Da Rosa JGS, Loro VL, Cunha MA, Baldisserotto B. Physiological and biochemical responses of silver catfish, Rhamdia quelen, after transport in water with essential oil of Aloysia triphylla (L’Herit) Britton. Aquaculture [serial on the Internet]. 2014; 418-19:101-07. Available from: https://doi.org/10.1016/j.aquaculture.2013.10.013
https://doi.org/10.1016/j.aquaculture.20...
) and can help to minimize stress effects (Azambuja et al., 2011Azambuja CR, Mattiazzi J, Riffel APK, Finamor IA, Garcia LO, Heldwein CG, Heinzmann BM, Baldisserotto B, Pavanato MA, Llesuy SF. Effect of the essential oil of Lippia alba on oxidative stress parameters in silver catfish (Rhamdia quelen) subjected to transport. Aquaculture [serial on the Internet]. 2011; 319(1-2):156-61. Available from: https://doi.org/10.1016/j.aquaculture.2011.06.002
https://doi.org/10.1016/j.aquaculture.20...
; Toni et al., 2014Toni C, Becker AG, Simões LN, Pinheiro CG, de Lima Silva L, Heinzmann BM, Caron BO, Baldisserotto B. Fish anesthesia: effects of the essential oils of Hesperozygis ringens and Lippia alba on the biochemistry and physiology of silver catfish (Rhamdia quelen). Fish Physiol Biochem [serial on the Internet]. 2014; 40(3):701-14. Available from: http://dx.doi.org/10.1007/s10695-013-9877-4
http://dx.doi.org/10.1007/s10695-013-987...
; Becker et al., 2016Becker AG, Parodi TV, Zeppenfeld CC, Salbego J, Cunha MA, Heldwein CG, Loro VL, Heinzmann BM, Baldisserotto B. Pre-sedation and transport of Rhamdia quelen in water containing essential oil of Lippia alba: metabolic and physiological responses. Fish Physiol Biochem [serial on the Internet]. 2016; 42(1):73-81. Available from: http://dx.doi.org/10.1007/s10695-015-0118-x
http://dx.doi.org/10.1007/s10695-015-011...
). In addition, EOs have been recommended for fish anesthesia due to their low costs, easy accessibility, efficacy and environmental safety (Iversen et al., 2003Iversen M, Finstad B, Mckinley RS, Eliassen RA. The efficacy of metomidate, clove oil, Aqui-S™ and Benzoak® as anaesthetics in Atlantic salmon (Salmo salar L.) smolts, and their potential stress-reducing capacity. Aquaculture [serial on the Internet]. 2003; 221(1-4):549-66. Available from: https://doi.org/10.1016/S0044-8486(03)00111-X
https://doi.org/10.1016/S0044-8486(03)00...
; Cunha et al., 2011Cunha MA, Silva BF, Delunardo FAC, Benovit SC, Gomes LC, Heinzmann BM, Baldisserotto B. Anesthetic induction and recovery of Hippocampus reidi exposed to the essential oil of Lippia alba. Neotrop Ichthyol [serial on the Internet]. 2011; 9(3):683-88. Available from: http://dx.doi.org/10.1590/S1679-62252011000300022
http://dx.doi.org/10.1590/S1679-62252011...
; Pedrazzani et al., 2016Pedrazzani AS, Ostrensky A Neto. The anaesthetic effect of camphor (Cinnamomum camphora), clove (Syzygium aromaticum) and mint (Mentha arvensis) essential oils on clown anemonefish, Amphiprion ocellaris (Cuvier 1830). Aquacult Res [serial on the Internet]. 2016; 47(3):769-76. Available from: https://doi.org/10.1111/are.12535
https://doi.org/10.1111/are.12535...
).
Citrus (Rutaceae) includes several species of plants that produce some of the most cultivated fruits in the world, including oranges, bergamots and lemons, which have an appreciable content of essential oil in their peel (Kummer et al., 2013Kummer R, Fachini-Queiroz FC, Estevão-Silva CF, Grespan R, Silva EL, Bersani-Amado CA, Cuman RKN. Evaluation of anti-inflammatory activity of Citrus latifolia Tanaka essential oil and limonene in experimental mouse models. Evid-Based Compl Alt [serial on the Internet]. 2013; 2013:859083 Available from: http://dx.doi.org/10.1155/2013/859083
http://dx.doi.org/10.1155/2013/859083...
). Essential oils (EO) are extracted from the pericarp or peel of the fruits and consist mainly of monoterpenes and sesquiterpenes and phenylpropanoids, metabolites that confer their organoleptic characteristics (Astani et al., 2010Astani A, Reichling J, Schnitzler P. Comparative study on the antiviral activity of selected monoterpenes derived from essential oils. Phytother Res [serial on the Internet]. 2010; 24(5):673-79. Available from: https://doi.org/10.1002/ptr.2955
https://doi.org/10.1002/ptr.2955...
; Acar et al., 2015Acar U, Kesbiç OS, Yılmaz S, Gültepe N, Türker A. Evaluation of the effects of essential oil extracted from sweet orange peel (Citrus sinensis) on growth rate of tilapia (Oreochromis mossambicus) and possible disease resistance against Streptococcus iniae. Aquaculture [serial on the Internet]. 2015; 437(1):282-86. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.015
https://doi.org/10.1016/j.aquaculture.20...
). Brazil is one of the main suppliers of the EOs of orange, lemon and other citrus fruits, with Citrus x aurantium L., Citrus x sinensis and Citrus x latifolia being the species of major commercial importance (Bizzo et al., 2009Bizzo HR, Hovell AMC, Rezende CM. Essential oils in Brazil: general aspects, development and prospects. New Chem [serial on the Internet]. 2009; 32(3):588-94. Available from: http://dx.doi.org/10.1590/S0100-40422009000300005
http://dx.doi.org/10.1590/S0100-40422009...
; Mattos Jr et al., 2010Mattos D Jr, Milaneze TF, Azevedo FA, Quaggio JA. Soil nutrient availability and its impact on fruit quality of Tahiti acid lime. Rev Bras Frutic [serial on the Internet]. 2010; 32(1):335-42. Available from: http://dx.doi.org/10.1590/S0100-29452010005000032
http://dx.doi.org/10.1590/S0100-29452010...
). EOs of plants of this genus have been used in the treatment of anxiety or insomnia in humans (Lehrner et al., 2005Lehrner J, Marwinski G, Lehr S, Johren P, Deecke L. Ambient odor of orange and lavender reduce anxiety and improve mood in a dental office. Physiol Behav [serial on the Internet]. 2005; 86(1-2):92-95. Available from: http://dx.doi.org/10.1016/j.physbeh.2005.06.031
http://dx.doi.org/10.1016/j.physbeh.2005...
), and oils of different species have shown promising results in preclinical trials using rodents. The most noteworthy among these are C. x sinensis (Faturi et al., 2010Faturi CB, Leite JR, Alves PB, Canton AC, Teixeira-Silva F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog Neuro-Psychopha [serial on the Internet]. 2010; 34(4):605-09. Available from: http://dx.doi.org/10.1016/j.pnpbp.2010.02.020
http://dx.doi.org/10.1016/j.pnpbp.2010.0...
) and C. x limon (Ceccarelli et al., 2004Ceccarelli I, Lariviere WR, Fiorenzani P, Sacerdote P, Aloisi AM. Effects of long-term exposure of lemon essential oil odor on behavioral, hormonal and neuronal parameters in male and female rats. Brain Res [serial on the Internet]. 2004; 1001(1-2):78-86. Available from: http://dx.doi.org/10.1016/j.brainres.2003.10.063
http://dx.doi.org/10.1016/j.brainres.200...
) in rats, and C. x latifolia (Gargano et al., 2008Gargano AC, Almeida CAR, Costa M. Essential oils from Citrus latifolia and Citrus reticulata reduced anxiety and prolong ether sleeping time in mice. Tree For Sci Biotechnol. 2008; 2(1):121-24.) and C. x aurantium (Carvalho-Freitas, Costa, 2002Carvalho-Freitas MIR, Costa M. Anxiolytic and sedative effects of extracts and essential oil from Citrus aurantium L. Biol Pharm Bull. 2002; 25(12):1629-33.; Pultrini et al., 2006Pultrini AM, Galindo LA, Costa M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci [serial on the Internet]. 2006; 78(15):1720-25. Available from: http://dx.doi.org/10.1016/j.lfs.2005.08.004
http://dx.doi.org/10.1016/j.lfs.2005.08....
) in mice. Sedative and anxiolytic-like effects have been described for the EO obtained from the peel of Citrus x aurantium in rats and mice (Leite et al., 2008Leite MP, Fassin J Jr, Baziloni EMF, Almeida RN, Mattei R, Leite JR. Behavioral effects of essential oil of Citrus aurantium L. inhalation in rats. Braz J Pharmacogn [serial on the Internet]. 2008; 18:661-66. Available from: http://dx.doi.org/10.1590/S0102-695X2008000500003
http://dx.doi.org/10.1590/S0102-695X2008...
; Costa et al., 2013Costa CARA, Cury TC, Cassettan BO, Takahira RK, Flório JC, Costa M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. BMC Complem Altern M [serial on the Internet]. 2013; 13:42. Available from: http://dx.doi.org/10.1186/1472-6882-13-42
http://dx.doi.org/10.1186/1472-6882-13-4...
). However, the literature does not present reports of their anesthetic effects on fish.
Mindful of this, the aim of this study was to evaluate the effectiveness of the EOs of C. x aurantium (EOCA) and C. x latifolia (EOCL) as anesthetics and sedatives for the silver catfish, Rhamdia quelen (Quoy & Gaimard). In addition, we investigated ion regulatory balance, survival rates, ventilatory frequency (VF) and water parameters during long-term exposure to EOs, to analyze their possible use as sedative in transport.
Materials and Methods
Animals. The experiments were conducted in the Fish Physiology Laboratory at the Universidade Federal de Santa Maria (UFSM), Rio Grande do Sul (RS), Brazil. Silver catfish (mean ± SEM, 17.88 ± 3.42 g; 12.86 ± 1.35 cm) (voucher UFRGS 22661) were obtained from a fish culture near Santa Maria, southern Brazil and acclimated to the laboratory conditions for seven days. Dissolved oxygen and temperature were measured with a Y5512 oxygen meter (YSI Inc., Yellow Springs, OH, USA). The pH was verified with a DMPH-2 pH meter (Digimed, São Paulo, SP, Brazil). Total ammonia nitrogen (TAN) and un-ionized ammonia (NH3) levels were determined as described by Zeppenfeld et al. (2014Zeppenfeld CC, Toni C, Becker AG, Miron DS, Parodi TV, Heinzmann BM, Barcellos LJG, Koakoski G, Da Rosa JGS, Loro VL, Cunha MA, Baldisserotto B. Physiological and biochemical responses of silver catfish, Rhamdia quelen, after transport in water with essential oil of Aloysia triphylla (L’Herit) Britton. Aquaculture [serial on the Internet]. 2014; 418-19:101-07. Available from: https://doi.org/10.1016/j.aquaculture.2013.10.013
https://doi.org/10.1016/j.aquaculture.20...
).
The experimental protocol was approved by the Ethical and Animal Welfare Committee of the UFSM under registration number 074/2014.
Essential oil extraction, analysis and identification of constituents. The fruits were purchased from a local market in Santa Maria. Immediately after purchase, fruits were washed in tap water and peeled. The peels were processed for extracting EOCA and EOCL by hydrodistillation for 3 h in a Clevenger apparatus (Council of Europe, 2007Council of Europe. European Pharmacopeia. 6th ed. Strassbourg: European Directorate for the Quality of Medicines; 2007.) and stored at −4 °C until utilization. The qualitative analysis of the EOs was performed by gas chromatography-mass spectrometry-total ion chromatogram using an Agilent 7890A gas chromatograph coupled with an Agilent 5975C mass selective detector and employing a HP-5MS column (5% phenyl, 95% methylsiloxane, 30 m × 0.25 mm i.d. × 0.25 mm) as described by Pinheiro et al. (2016Pinheiro CG, Machado CM, Amaral LP, Silva DT, Almeida CAA, Longhi SJ, Mallmann CA, Heinzmanna BM. Seasonal variability of the essential oil of Hesperozygis ringens (Benth.) Epling. Braz J Biol [serial on the Internet]. 2016; 76(1):176-84. Available from: http://dx.doi.org/10.1590/1519-6984.16314
http://dx.doi.org/10.1590/1519-6984.1631...
). The constituents were identified by comparison of the Kovats retention index and their mass spectra with data from the mass spectral library (NIST, 2010NIST/ EPA/ NIH mass spectral library and search/ analysis programs. John Wiley and Sons. Hoboken, 2010.) and the literature (Adams, 2009Adams RP. Identification of essential oil components by gas chromatography / quadrupole mass spectroscopy. Allured Publishing Corporation. 2009.). The quantitative analysis was performed with an Agilent 7890A gas chromatograph with flame ionization detection using the same parameters described by Pinheiro et al. (2016Pinheiro CG, Machado CM, Amaral LP, Silva DT, Almeida CAA, Longhi SJ, Mallmann CA, Heinzmanna BM. Seasonal variability of the essential oil of Hesperozygis ringens (Benth.) Epling. Braz J Biol [serial on the Internet]. 2016; 76(1):176-84. Available from: http://dx.doi.org/10.1590/1519-6984.16314
http://dx.doi.org/10.1590/1519-6984.1631...
). The percentage of the chemical components was based on peak area normalization. The yields of EOCA and EOCL were 0.92% (v/w) and 0.45% (v/w), respectively.
Experiment 1: Anesthetic induction and recovery. Juvenile fish were individually transferred to aquaria containing 1 L of continuously aerated water and the EOs previously diluted in ethanol (1:10). Concentrations of 100, 200, 400, 600 or 800 μL L−1 of EOCA and 100, 200, 300, 400 or 500 μL L−1 of EOCL were used. These concentrations were determined after a pre-treatment trial. Ethanol controls were also tested at the same concentrations used for the dilutions corresponding to the highest EO concentrations. To evaluate the time required for anesthesia induction, 10 juveniles were used for each concentration and EO tested, and each juvenile was used only once, according to Silva et al. (2013aSilva LL, Garlet QI, Benovit SC, Dolci G, Mallmann CA, Bürger ME, Baldisserotto B, Longhi SJ, Heinzmann BM. Sedative and anesthetic activities of the essential oils of Hyptis mutabilis (Rich.) Briq. and their isolated components in silver catfish (Rhamdia quelen). Braz J Med Biol Res [serial on the Internet]. 2013a; 46(9):771-79. Available from: http://dx.doi.org/10.1590/1414-431X20133013
http://dx.doi.org/10.1590/1414-431X20133...
). All fish were fasted the day prior to sampling. The stages of anesthesia observed in the present study are an adaptation of those detailed by Small (2003Small BC. Anesthetic efficacy of metomidate and comparison of plasma cortisol responses to tricaine methanesulfonate, quinaldine and clove oil anesthetized channel catfish Ictalurus punctatus. Aquaculture [serial on the Internet]. 2003; 218(1-4):177-85. Available from: https://doi.org/10.1016/S0044-8486(02)00302-2
https://doi.org/10.1016/S0044-8486(02)00...
). The induction times were recorded during sedation, which were characterized by the decreased reactivity to external stimuli, and anesthesia, in which the total loss of equilibrium, cessation of locomotion and a lack of response to tactile stimuli occurred. The maximum observation time was 30 min. After induction, juveniles were transferred to anesthetic free aquaria to measure recovery time. Animals were considered to be recovered when they showed normal swimming behavior and response to external stimuli. After recovery, fish were grouped according to the anesthetic protocol and transferred into continuously aerated 250 L aquaria, where they were observed for 48 h for any signs of abnormal behavior, disease or mortality.
Experiment 2: Ventilatory frequency (VF) and ion fluxes. This experiment was conducted to evaluate the ventilatory frequency (per minute) of the fish exposed to EOCA or EOCL at concentrations of 50, 100 or 200 μL L−1 for 0, 0.5, 1, 2, 4 or 8 h, to evaluate the efficacy of these EOs to be used for fish transport at sedative concentrations. This experiment lasted 8 h because it is the usual time of transport of juveniles in South Brazil (Tondolo et al., 2013Tondolo JSM, Amaral LP, Simoes LN, Garlet QI, Schindler B, Oliveira TM, Silva BF, Gomes LC, Baldisserotto B, Mallmann CA, Heinzmann BM. Anesthesia and transport of fat snook Centropomus parallelus with the essential oil of Nectandra megapotamica (Spreng.) Mez. Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):667-74. Available from: http://dx.doi.org/10.1590/S1679-62252013000300020
http://dx.doi.org/10.1590/S1679-62252013...
; Cunha et al., 2017Cunha JA, Scheeren CA, Salbego S, Gressler LT, Madaloz LM, Bandeira Junior G, Bianchini AE, Pinheiro CG, Bordignon SAL, Heinzmann BM, Baldisserotto B. Essential oils of Cunila galioides and Origanum majorana as anesthetics for Rhamdia quelen: efficacy and effects on ventilation and ionoregulation. Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160076 DOI: 10.1590/1982-0224-20160076
https://doi.org/10.1590/1982-0224-201600...
) and the concentrations chosen were those which only induced sedation in the anesthetic induction experiment. Groups of fish were also exposed to water (control) and ethanol (1800 μL L−1). One fish per aquarium (11 cm x 16 cm x 11 cm, n = 8 aquaria each concentration and EO; fish 16.59 ± 3.18 g; 12.70 ± 0.80 cm) was maintained with 1 L of water and the respective anesthetic concentrations. The VF was quantified by visually counting 20 successive opercular movements and measuring the elapsed time with a chronometer (adapted from Alvarenga, Volpato, 1995Alvarenga CMD, Volpato GL. Agonistic profile and metabolism in alevins of the Nile tilapia. Physiol Behav. 1995; 57(1):75-80.).
Water samples (5 mL) were collected at the beginning and at the end of exposure. Chloride levels were determined according to Zall et al. (1956Zall DM, Fisher MD, Garner QM. Photometric determination of chlorides in water. Anal Chem [serial on the Internet]. 1956; 28(11):1665-68. Available from: http://dx.doi.org/10.1021/ac60119a009
http://dx.doi.org/10.1021/ac60119a009...
) and Na+ and K+ levels with a B262 flame spectrophotometer (Micronal, São Paulo, Brazil). Net ion fluxes (jnet) and ammonia excretion were calculated according to the equation:
Where [ion1] and [ion2] are the ion or ammonia concentrations in the water at the beginning and end of the exposure, respectively, V is the water volume (in L), M is the mass of the fish (in kg) and t is the duration of the experiment (h).
Statistical analysis. All data are represented as mean ± SEM, and were subjected to Levene’s test to check homogeneity of variances. Evaluation of the anesthetic activity was performed by regression analysis (concentration × times of sedation, anesthesia induction and recovery). The comparisons of anesthetic activity between EOs (concentration 400 μL L−1), ventilatory frequencies and net ion fluxes were analyzed using Kruskal-Wallis ANOVA test, followed by the multiple comparison of mean ranks for all groups. Differences between means were tested at the 95% probability level. Analysis was performed using the software STATISTICA ver. 7.0 (SPSS, Chicago, IL, USA).
Results
Essential oil analysis and identification of constituents. The major chemical components of EOCA were limonene (93.89%), linalool (2.60%) and β-pinene (1.71%). A total of 20 substances were identified in EOCL, the major constituents were limonene (49.73%), γ-terpinene (11.12%), α and β-citral (9.17%) and β-pinene (9.14%).
Experiment 1: Anesthesia induction and recovery in R. quelen exposed to EOCA and EOCL.
No mortality was registered throughout exposure to the EOs nor up to 48 h after the exposure. The concentrations of EOCA at 200-300 μL L−1 or EOCL at 100-200 μL L−1 caused only light sedation in 30% of the fish throughout the 30 min evaluation period. Increasing the concentration of the EOs proportionally decreased the time required for sedation and anesthesia induction, which occurred in all animals at 400-800 μL L−1 EOCA or 300-500 μL L−1 EOCL. An inverse relationship between the concentration of EO and the time to recover from anesthesia was observed with EOCA, but not with EOCL. All EOCL concentrations tested induced anesthesia within 7 min or less and recovery in 10 min or less. The lowest time of anesthesia induction (less than 3 min) and recovery (less than 5 min) was observed at 500 μL L−1 EOCL (Tab. 1).
Time (s) required for induction and recovery from anesthesia using the essential oils of Citrus x aurantium (EOCA) and Citrus x latifolia (EOCL) in Rhamdia quelen (n = 10 each concentration). 1Y = 103.043−0.00097X (r2 = 0.83); 2Y = 103.361−0.00082X (r2 = 0.92); 3Y = 103.243−0.00087X (r2 = 0.89); 4Y = 102.672−0.0013X (r2 = 0.97); 5Y = 103.070−0.0017X (r2 = 0.92); 6No significant relationship or difference between concentrations (P > 0.05). Equations represent relationships between the times of sedation, anesthesia or recovery and concentrations of EOs, where y = time to reach the stages (seconds) and x = EO concentrations (μL L−1). *significantly different from the same concentration of EOCA (P < 0.05). Values are means ± SEM.
Sedation and anesthesia with 400 μL L−1 EOCL was faster than with the same concentration of EOCA. Ethanol at the highest concentration used (7200 μL L−1) to dilute the EOs did not produce any anesthetic effect when applied alone.
Experiment 2: Water parameters, net ion fluxes and ammonia excretion.
In the second set of experiments, the dissolved oxygen levels, temperature and NH3 levels in the water did not exhibit any significant difference between the treatments before and after 8 h exposure to EOCA and EOCL. However, pH levels were significantly lower in groups exposed to all concentrations of either EOs (Tab. 2).
Water parameters after 8 h exposure of Rhamdia quelen to the essential oils of Citrus x aurantium (EOCA) and Citrus x latifolia (EOCL) added to the water. *significantly different from control (P < 0.05). Values are means ± SEM. Dissolved oxygen and un-ionized ammonia were expressed as mg/L-1 and temperature as °C.
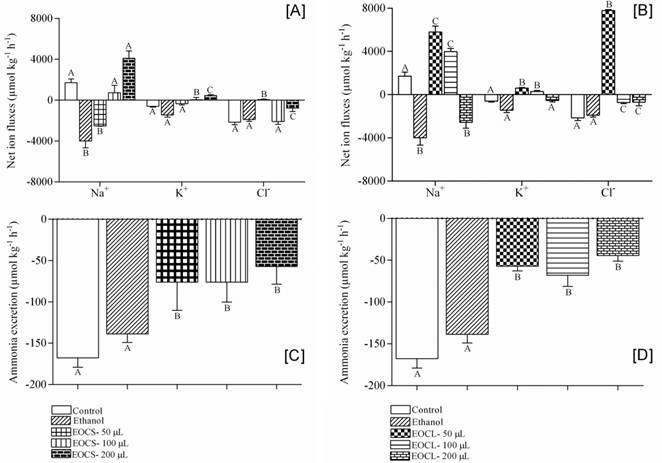
Fish of the control group presented net Na+ influx and net K+ and Cl− effluxes. Exposure to ethanol, 50 μL L−1 EOCA or 200 μL L−1 EOCL provoked significantly higher net Na+ efflux compared to control fish, while 50 or 100 μL L−1 EOCL significantly increased net Na+ influx compared to control and ethanol-exposed fish. Fish exposed to 100 or 200 μL L−1 EOCA and 50 or 100 μL L−1 EOCL significantly increased net K+ influx compared to control and ethanol-exposed fish. Fish exposed to 50 or 200 μL L−1 EOCA and 100 or 200 μL L−1 EOCL reduced net Cl− effluxes and 50 μL L−1 EOCL significantly increased net Cl− effluxes compared to control and ethanol-exposed fish. Ethanol did not significantly change net K+ or Cl− fluxes (Figs. 1a-b). Ammonia excretion was significantly lower in fish exposed to all concentrations of either EOs than in the control and ethanol groups throughout the 8 h exposure (Figs. 1c-d).
Net ion (Na+, K+, Cl−) fluxes (a-b) and ammonia excretion (c-d) in silver catfish through 8 h of exposure to essential oils of Citrus x aurantium (EOCA) and Citrus x latifolia (EOCL). Values are means ± SEM. Different letters indicate significant differences between treatments (P < 0.05). Positive values indicate net influxes and negative values net effluxes.
No mortality was recorded in fish exposed to 50μL L−1 or 100 μL L−1 EOCA or EOCL throughout or after exposure. However, mortality was 37% after 8 h exposure to 200 μL L−1 of either EOs.
Ventilatory frequency (VF). Ventilatory frequency decreased with time in all treatments. Fish exposed to ethanol, 50, 100 or 200 μL L−1 EOCA presented higher VF compared to the control group throughout the first 4 h of exposure, however at 200 μL L−1 EOCA after 8 h of exposure reduced VF was observed. Fishes exposed to 50 or 100μL L−1 EOCL had increased VF between 0.5-8 h compared to control fish, while those exposed to 200 μL L−1 EOCL had increased VF at the moment of exposure and after 8 h, but this was reduced between 0.5-2 h compared to control group (Tab. 3).
Ventilatory frequency (opercular or buccal movements min−1) of Rhamdia quelen maintained in water with the essential oils of Citrus x aurantium (EOCA) or Citrus x latifolia (EOCL). Values are means ± SEM. Different capital letters in the rows indicate significant difference between treatments at the same time (P < 0.05). Different lowercase letters in the columns indicate significant difference between times in the same treatment (P < 0.05).
Discussion
Yields of both EOs obtained during this study can be considered good (EOCA was 0.92% v/w and EOCL 0.45% v/w) when compared to literature reports on the yield of Citrus oils, of 0.4% (Bizzo et al., 2009Bizzo HR, Hovell AMC, Rezende CM. Essential oils in Brazil: general aspects, development and prospects. New Chem [serial on the Internet]. 2009; 32(3):588-94. Available from: http://dx.doi.org/10.1590/S0100-40422009000300005
http://dx.doi.org/10.1590/S0100-40422009...
). Citrus oils represent a viable economic alternative considering their potential, yield and low acquisition cost. It is important to consider that the use of waste originating from the manufacture of juices of tropical and subtropical fruits reduces environmental contamination (Kobori, Jorge, 2005Kobori CN, Jorge N. Characterization of some seed oils of fruits for utilization of industrial residues. Cienc Agrotec [serial on the Internet]. 2005; 29(5):1008-14. Available from: http://dx.doi.org/10.1590/S1413-70542005000500014
http://dx.doi.org/10.1590/S1413-70542005...
).
Both EOCA and EOCL were shown to contain limonene as the major component, in agreement with previous analysis of Sharma, Tripathi (2008Sharma N, Tripathi A. Effects of Citrus sinensis (L.) Osbeck epicarp essential oil on growth and morphogenesis of Aspergillus niger (L.) Van Tieghem. Microbiol Res [serial on the Internet]. 2008; 163(3):337-44. Available from: http://dx.doi.org/10.1016/j.micres.2006.06.009
http://dx.doi.org/10.1016/j.micres.2006....
) and Kummer et al. (2013Kummer R, Fachini-Queiroz FC, Estevão-Silva CF, Grespan R, Silva EL, Bersani-Amado CA, Cuman RKN. Evaluation of anti-inflammatory activity of Citrus latifolia Tanaka essential oil and limonene in experimental mouse models. Evid-Based Compl Alt [serial on the Internet]. 2013; 2013:859083 Available from: http://dx.doi.org/10.1155/2013/859083
http://dx.doi.org/10.1155/2013/859083...
). Besides EOCA and EOCL, other EOs that showed anxiolytic-like effects in animal models, such as C. x aurantium, C. x reticulata, Foeniculum vulgare and Lippia alba (limonene-carvone chemotype), have limonene as a major component (Gargano et al., 2008Gargano AC, Almeida CAR, Costa M. Essential oils from Citrus latifolia and Citrus reticulata reduced anxiety and prolong ether sleeping time in mice. Tree For Sci Biotechnol. 2008; 2(1):121-24.; Faturi et al., 2010Faturi CB, Leite JR, Alves PB, Canton AC, Teixeira-Silva F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog Neuro-Psychopha [serial on the Internet]. 2010; 34(4):605-09. Available from: http://dx.doi.org/10.1016/j.pnpbp.2010.02.020
http://dx.doi.org/10.1016/j.pnpbp.2010.0...
; Saiyudthong, Marsden, 2011Saiyudthong S, Marsden C. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phytother Res [serial on the Internet]. 2011; 25(6):858-62. Available from: http://dx.doi.org/10.1002/ptr.3325
http://dx.doi.org/10.1002/ptr.3325...
; Hatano et al., 2012Hatano VY, Torricelli AS, Giassi ACC, Coslope LA,Viana MB. Anxiolytic effects of repeated treatment with an essential oil from Lippia alba and (R)-(-)-carvone in the elevated T-maze. Braz J Med Biol Res. 2012; 45(3):238-43.; Mesfin et al., 2014Mesfin M, Asres K, Shibeshi W. Evaluation of anxiolytic activity of the essential oil of the aerial part of Foeniculum vulgare Miller in mice. BMC Complem Altern M [serial on the Internet]. 2014; 14:310. Available from: https://doi.org/10.1186/1472-6882-14-310
https://doi.org/10.1186/1472-6882-14-310...
). Limonene directly binds to the adenosine A2A receptor, which may induce sedative effects (Park et al., 2011Park HM, Lee JH, Yaoyao J, Jun HJ, Lee SJ. Limonene, a natural cyclic terpene, is an agonistic ligand for adenosine A(2A) receptors. Biochem Bioph Res Co [serial on the Internet]. 2011; 404(1):345-48. Available from: http://dx.doi.org/10.1016/j.bbrc.2010.11.121
http://dx.doi.org/10.1016/j.bbrc.2010.11...
) and also demonstrated direct vasorelaxant effects (Kang et al., 2013Kang P, Suh SH, Min SS, Seol GH. The essential oil of Citrus bergamia Risso induces vasorelaxation of the mouse aorta by activating K + channels and inhibiting Ca 2+ influx. J Pharm Pharmacol [serial on the Internet]. 2013; 65(5):745-49. Available from: http://dx.doi.org/10.1111/jphp.12031
http://dx.doi.org/10.1111/jphp.12031...
). Vale et al. (2002Vale GT, Furtado EC, Santos JG, Viana GSB. Central effects of citral, myrcene and limonene, constituents of essential oil chemotypes from Lippia alba (Mill.) N.E. Brown. Phytomedicine [serial on the Internet]. 2002; 9(8):709-14. Available from: https://doi.org/10.1078/094471102321621304
https://doi.org/10.1078/0944711023216213...
) also reported the sedative and motor relaxant effects of limonene as a result of direct activity on the central nervous system (CNS). Additionally, the intraperitoneal injection of EOCA promoted a depressant activity in the CNS of mice, and this activity was attributed to limonene (Carvalho-Freitas, Costa, 2002Carvalho-Freitas MIR, Costa M. Anxiolytic and sedative effects of extracts and essential oil from Citrus aurantium L. Biol Pharm Bull. 2002; 25(12):1629-33.).
β-pinene, another major component of EOCA and EOCL, may contribute to the anesthetic effects of these EOs because D- and L-enantiomers of α- and β-pinene induced sedation and anesthesia in mice (Kasanen et al., 1998Kasanen JP, Pasanen AL, Pasanen P, Liesivuori J, Kosma VM, Alarie Y. Stereospecificity of the sensory irritation receptor for nonreactive chemicals illustrated by pinene enantiomers. Arch Toxicol. 1998; 72(8):514-23.; Mercier et al., 2009Mercier B, Prost J, Prost M. The essential oil of turpentine and its major volatile fraction (alpha- and betapinenes): a review. Int J Occup Med Env [serial on the Internet]. 2009; 22(4):331-42. DOI: 10.2478/v10001-009-0032-5
https://doi.org/10.2478/v10001-009-0032-...
). In addition, the EO of the leaves of Hyptis mutabilis, containing 7.9% β-pinene, induced anesthesia in silver catfish (Silva et al., 2013aSilva LL, Garlet QI, Benovit SC, Dolci G, Mallmann CA, Bürger ME, Baldisserotto B, Longhi SJ, Heinzmann BM. Sedative and anesthetic activities of the essential oils of Hyptis mutabilis (Rich.) Briq. and their isolated components in silver catfish (Rhamdia quelen). Braz J Med Biol Res [serial on the Internet]. 2013a; 46(9):771-79. Available from: http://dx.doi.org/10.1590/1414-431X20133013
http://dx.doi.org/10.1590/1414-431X20133...
). α-pinene is reported as a positive modulator of GABAA receptors (Komiya et al., 2006Komiya M, Takashi T, Harada E. Lemon oil vapor causes an anti-stress effect via modulating the 5-HT and DA activities in mice. Behav Brain Res [serial on the Internet]. 2006; 172(2):240-49. Available from: http://dx.doi.org/10.1016/j.bbr.2006.05.006). These receptors are recognized as important targets for the modulation of sedative, anxiolytic and general anesthetic agents. Additionally, linalool, found in EOCA, has anesthetic effects in silver catfish (both S-(+) and R-(−) linalool isomers) (Silva et al., 2017Silva LL, Balconi LS, Gressler LT, Garlet QI, Sutili FJ, Vargas APC, Baldisserotto B, Morel AF , Heinzmann BM. S-(+)- and R-(-)-linalool: a comparison of the in vitro anti-Aeromonas hydrophila activity and anesthetic properties in fish. An Acad Bras Cienc [serial on the Internet]. 2017; 89(1):203-12. Available from: http://dx.doi.org/10.1590/0001-3765201720150643
http://dx.doi.org/10.1590/0001-376520172...
) and common carp, Cyprinus carpio Linnaeus (Mirghaed et al., 2016Mirghaed AT, Ghelichpour T, Hoseini SM. Myrcene and linalool as new anesthetic and sedative agents in common carp, Cyprinus carpio - comparison with eugenol. Aquaculture [serial on the Internet]. 2016; 464(1):165-70. Available from: https://doi.org/10.1016/j.aquaculture.2016.06.028
https://doi.org/10.1016/j.aquaculture.20...
), though its mechanism of action seems not to involve the benzodiazepine site of the GABAergic system (Heldwein et al., 2014Heldwein CG, Silva LL, Gai EZ, Roman C, Parodi TV, Burger ME, Baldisserotto B, Flores EM, Heinzmann BM. S-(+)-Linalool from Lippia alba: sedative and anesthetic for silver catfish (Rhamdia quelen). Vet Anaesth Analg [serial on the Internet]. 2014; 41(6):621-9. Available from: https://doi.org/10.1111/vaa.12146
https://doi.org/10.1111/vaa.12146...
). Citral, another major component of EOCL, increased barbituric sleeping time of mice (Vale et al., 2002Vale GT, Furtado EC, Santos JG, Viana GSB. Central effects of citral, myrcene and limonene, constituents of essential oil chemotypes from Lippia alba (Mill.) N.E. Brown. Phytomedicine [serial on the Internet]. 2002; 9(8):709-14. Available from: https://doi.org/10.1078/094471102321621304
https://doi.org/10.1078/0944711023216213...
) and blocks the excitability of rat sciatic nerves (Sousa et al., 2015Sousa DG, Sousa SD, Silva RE, Silva-Alves KS, Ferreira-da-Silva FW, Kerntopf MR, Menezes IR, Leal-Cardoso JH, Barbosa R. Essential oil of Lippia alba and its main constituent citral block the excitability of rat sciatic nerves. Braz J Med Biol Res [serial on the Internet]. 2015; 48(8):697-702. Available from: https://doi.org/10.1590/1414-431X20154710
https://doi.org/10.1590/1414-431X2015471...
). Citral is also the main component of other EOs that have anesthetic effects in fish: Aloysia triphylla (Gressler et al., 2012Gressler LT, Riffel APK, Parodi TV, Saccol EMH, Koakoski G, Costa ST et al. Silver catfish Rhamdia quelen immersion anaesthesia with essential oil of Aloysia triphylla (L’Herit) Britton or tricaine methanesulfonate: effect on stress response and antioxidant status. Aquacult Res [serial on the Internet]. 2012; 45(6):1061-72. Available from: http://dx.doi.org/10.1111/are.12043
http://dx.doi.org/10.1111/are.12043...
; Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
; Santos et al., 2017Santos AC, Junior GB, Zago DC, Zeppenfeld CC, da Silva DT, Heinzmann BM, da Cunha MA. Anesthesia and anesthetic action mechanism of essential oils of Aloysia triphylla and Cymbopogon flexuosus in silver catfish (Rhamdia quelen). Vet Anaesth Analg [serial on the Internet]. 2017; 44(1):106-13. Available from: http://dx.doi.org/ 10.1111/vaa.12386
http://dx.doi.org/ 10.1111/vaa.12386...
Cymbopogon flexuosus (Limma-Neto et al., 2016Limma-Netto JD, Sena AC, Copatti CE. Essential oils of Ocimum basilicum and Cymbopogon flexuosus in the sedation, anesthesia and recovery of tambacu (Piaractus mesopotamicus male x Colossoma macropomum female). Bol Inst Pesca [serial on the Internet]. 2016; 42(3):727-33. Available from: http://dx.doi.org/10.20950/1678-2305.2016v42n3p727
http://dx.doi.org/10.20950/1678-2305.201...
) and L. alba citral chemotype (Souza et al., 2017Souza CF, Baldissera MD, Salbego J, Lopes JM, Vaucher RA, Mourão VRH, Caron BO, Heinzmann BM, Silva LVF, Baldisserotto B. Physiological responses of Rhamdia quelen (Siluriformes: Heptapteridae) to anesthesia with essential oils from two different chemotypes of Lippia alba. Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160083. Available from: http://dx.doi.org/10.1590/1982-0224-20160083
http://dx.doi.org/10.1590/1982-0224-2016...
). The anesthetic effects of citral in fish does not involve the benzodiazepine site of the GABAergic system γ-terpinene, also a major component of EOCL, increases dopamine release from rat brain striatum slices (Fukumoto et al., 2006Fukumoto S, Sawasaki E, Okuyama S, Miyake Y, Yokogoshi H. Flavor components of monoterpenes in citrus essential oils enhance the release of monoamines from rat brain slices. Nutr Neurosci [serial on the Internet]. 2006; 9(1-2):73-80. Available from: http://dx.doi.org/10.1080/10284150600573660
http://dx.doi.org/10.1080/10284150600573...
), but to our knowledge, no study so far has analyzed the sedative and/or anesthetic activities of this compound.
Silver catfish exposed to EOCA at 200-300 μL L−1 or to EOCL at 100-200 μL L−1 were not fully anesthetized, and presented only light sedation. The animals maintained the capacity of reaction to external stimuli, with reduced movements, but normal equilibrium, which would be recommended by Becker et al. (2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
) for fish transportation. However, when exposed to 400-600 or 800 μL L−1 EOCA or 300-400 or 500 μL L−1 EOCL, the increasing concentration proportionally decreased the time required for sedation and anesthesia induction. The inverse relationship between the concentration of EO and induction time verified in this study had previously been observed: in R. quelen anesthetized with the EOs of L. alba (Cunha et al., 2010Cunha MA, Barros FMC, Garcia LO, Veeck APL, Heinzmann BM, Loro VL, Emanuelli T, Baldisserotto B. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen. Aquaculture [serial on the Internet]. 2010; 306(1-4):403-06. Available from: https://doi.org/10.1016/j.aquaculture.2010.06.014
https://doi.org/10.1016/j.aquaculture.20...
) and A. triphylla (Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
); in Centropomus parallelus Poey, 1860 with the EO of Nectandra megapotamica (Tondolo et al., 2013Tondolo JSM, Amaral LP, Simoes LN, Garlet QI, Schindler B, Oliveira TM, Silva BF, Gomes LC, Baldisserotto B, Mallmann CA, Heinzmann BM. Anesthesia and transport of fat snook Centropomus parallelus with the essential oil of Nectandra megapotamica (Spreng.) Mez. Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):667-74. Available from: http://dx.doi.org/10.1590/S1679-62252013000300020
http://dx.doi.org/10.1590/S1679-62252013...
); and in C. carpio with the EOs of Melaleuca alternifolia and Mentha spicata (Hajek, 2011Hajek GJ. The anaesthetic-like effect of tea tree oil in common carp Cyprinus carpio L. Aquacult Res [serial on the Internet]. 2011; 42(2):296-300. Available from: https://doi.org/10.1111/j.1365-2109.2010.02625.x
https://doi.org/10.1111/j.1365-2109.2010...
; Roohi, Imanpoor, 2015Roohi Z, Imanpoor MR. The efficacy of the oils of spearmint and methyl salicylate as new anesthetics and their effect on glucose levels in common carp (Cyprinus carpio L., 1758) juveniles. Aquaculture [serial on the Internet]. 2015; 437:327-32. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.019
https://doi.org/10.1016/j.aquaculture.20...
).
Only the highest concentration of EOCL (500 µL L−1) presented ideal times for induction (less than 3 min) and recovery (less than 5 min) according to Marking, Meyer (1985Marking LL, Meyer FP. Are better anesthetics needed in fisheries? Fisheries. 1985; 10(6):2-5.). However, 300-400 µL L−1 EOCL or 800 µL L−1 EOCA may also be suitable for anesthesia (up to 8 min). In addition, anesthetic concentrations that showed a longer recovery time may be appropriate for surgical procedures that require a delayed recovery after anesthetic exposure (Prince, Powell, 2000Prince A, Powell C. Clove oil as an anaesthetic for invasive field procedures on adult rainbow trout. N Am J Fish Manage. 2000; 20(4):1029-32.), and the lowest concentrations of EOCA (400-600 μL L−1) would suit this purpose. The EOCL, which contains a lower percentage of limonene (49.7%) than EOCA (93.89%), showed better anesthetic effects, indicating that the other components (γ-terpinene, citral and β-pinene) may be more effective in inducing anesthesia in R. quelen. Therefore, it is possible that the activity of the major component is modulated by other minor molecules (Hoet et al., 2006Hoet S, Stévigny C, Herent MF, Quetin-Leclercq J. Antitrypanosomal compounds from leaf essential oil of Strychnos spinosa. Planta Med [serial on the Internet]. 2006; 72(5):480-2. Available from: http://dx.doi.org/10.1055/s-2005-916255
http://dx.doi.org/10.1055/s-2005-916255...
).
The concentration range of EOCL required to induce anesthesia is similar to the EO of Cinnamomum camphora (30% limonene) in Amphiprion ocellaris Cuvier, 1830 (Pedrazzani, Ostrensky Neto, 2016Pedrazzani AS, Ostrensky A Neto. The anaesthetic effect of camphor (Cinnamomum camphora), clove (Syzygium aromaticum) and mint (Mentha arvensis) essential oils on clown anemonefish, Amphiprion ocellaris (Cuvier 1830). Aquacult Res [serial on the Internet]. 2016; 47(3):769-76. Available from: https://doi.org/10.1111/are.12535
https://doi.org/10.1111/are.12535...
) but overall, both EOCL and EOCA are less effective than other EOs tested in R. quelen, such as those from L. alba (Cunha et al., 2010Cunha MA, Barros FMC, Garcia LO, Veeck APL, Heinzmann BM, Loro VL, Emanuelli T, Baldisserotto B. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen. Aquaculture [serial on the Internet]. 2010; 306(1-4):403-06. Available from: https://doi.org/10.1016/j.aquaculture.2010.06.014
https://doi.org/10.1016/j.aquaculture.20...
), Ocimum gratissimum (Silva et al., 2012Silva LL, Parodi TV, Reckziegel P, Garcia VO, Bürger ME, Baldisserotto B, Malmann CA, Pereira AMS, Heinzmann BM. Essential oil of Ocimum gratissimum: anesthetic effect, mechanism of action and tolerance in silver catfish (Rhamdia quelen). Aquaculture [serial on the Internet]. 2012; 350-353:91-97. Available from: https://doi.org/10.1016/j.aquaculture.2012.04.012
https://doi.org/10.1016/j.aquaculture.20...
), H. mutabilis (Silva et al., 2013aSilva LL, Garlet QI, Benovit SC, Dolci G, Mallmann CA, Bürger ME, Baldisserotto B, Longhi SJ, Heinzmann BM. Sedative and anesthetic activities of the essential oils of Hyptis mutabilis (Rich.) Briq. and their isolated components in silver catfish (Rhamdia quelen). Braz J Med Biol Res [serial on the Internet]. 2013a; 46(9):771-79. Available from: http://dx.doi.org/10.1590/1414-431X20133013
http://dx.doi.org/10.1590/1414-431X20133...
), Hesperozygis ringens (Silva et al., 2013bSilva LL, Silva TS, Garlet QI, Cunha MA, Mallmann CA, Baldisserotto B, Longhi SJ, Pereira AMS, Heinzmann BM. Anesthetic activity of Brazilian native plants in silver catfish (Rhamdia quelen). Neotrop Ichthyol [serial on the Internet]. 2013b; 11(2):443-51. Available from: http://dx.doi.org/10.1590/S1679-62252013000200014
http://dx.doi.org/10.1590/S1679-62252013...
), A. triphylla (Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
) and Ocimum americanum (Silva et al., 2015Silva LD, Garlet QI, Koakoski G, Abreu MS, Mallmann CA, Baldisserotto B, Barcellos LJG, Heinzmann BM. Anesthetic activity of the essential oil of Ocimum americanum in Rhamdia quelen (Quoy & Gaimard, 1824) and its effects on stress parameters. Neotrop Ichthyol [serial on the Internet]. 2015; 13(4):715-22. Available from: http://dx.doi.org/10.1590/1982-0224-20150012
http://dx.doi.org/10.1590/1982-0224-2015...
).
Most fish anesthetics have an inhibitory effect on the respiratory system, which results in a lower ventilatory frequency (Keene et al., 1998Keene JL, Noakes DLG, Moccia RD, Soto CG. The efficacy of clove oil as an anaesthetic for rainbow trout, Oncorhynchus mykiss (Walbaum). Aquacult Res . 1998; 29(2):89-101.), but this effect can vary depending on the concentration of the anesthetic (Roohi, Imanpoor, 2015Roohi Z, Imanpoor MR. The efficacy of the oils of spearmint and methyl salicylate as new anesthetics and their effect on glucose levels in common carp (Cyprinus carpio L., 1758) juveniles. Aquaculture [serial on the Internet]. 2015; 437:327-32. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.019
https://doi.org/10.1016/j.aquaculture.20...
). Ethanol increased VF in silver catfish, and EOCA and EOCL avoided this increase only for some periods in fish exposed to 200 µL L−1, but these concentrations also provoked mortality. In C. carpio anesthetized with spearmint oil, VF first increased and then gradually decreased with increasing concentration of anesthetic (Roohi, Imanpoor, 2015Roohi Z, Imanpoor MR. The efficacy of the oils of spearmint and methyl salicylate as new anesthetics and their effect on glucose levels in common carp (Cyprinus carpio L., 1758) juveniles. Aquaculture [serial on the Internet]. 2015; 437:327-32. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.019
https://doi.org/10.1016/j.aquaculture.20...
) compared to control fish. Eugenol and the EO of L. alba (Becker et al., 2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
; Hohlenwerger et al., 2016Hohlenwerger JC, Copatti CE, Sena AC, Couto RD, Baldisserotto B, Heinzmann BM, Caron BO, Schmidt D. Could the essential oil of Lippia alba provide a readily available and cost-effective anaesthetic for Nile tilapia (Oreochromis niloticus)? Mar Freshw Behav Phy [serial on the Internet] . 2016; 49(2):119-26. Available from: https://doi.org/10.1080/10236244.2015.1123869
https://doi.org/10.1080/10236244.2015.11...
) also reduced VF in fish, but the EO of H. ringens did not change this parameter compared to control group (Toni et al., 2015Toni C, Martos-Sitcha JA, Jarabo IJ, Mancera JM, Rodriguez GM, Pinheiro CG, Heinzmann BM, Baldisserotto B. Stress response in silver catfish (Rhamdia quelen) exposed to the essential oils of Hesperozygis ringens. Fish Physiol Biochem [serial on the Internet]. 2015; 41(1):129-38. Available from: http://dx.doi.org/10.1007/s10695-014-0011-z
http://dx.doi.org/10.1007/s10695-014-001...
).
Overall, the sedative concentrations of EOCA and EOCL that did not provoke mortality in silver catfish increased VF compared to control and ethanol groups. The higher VF could indicate higher stress and/or metabolism and the lower water pH observed at the end of 8 h exposure to all EOs concentrations could be related to carbon dioxide excretion by the fish, corroborating this hypothesis. Although water carbon dioxide concentration was not measured in the present study, aquaria were aerated all the time, and in this situation carbon dioxide usually remains low (Colt, Orwicz, 1991Colt J, Orwicz K. Modeling production capacity of aquatic culture systems under freshwater conditions. Aquacult Eng [serial on the Internet]. 1991; 10(1):1-29. Available from: https://doi.org/10.1016/0144-8609(91)90008-8
https://doi.org/10.1016/0144-8609(91)900...
; Colt, Kroeger, 2013Colt J, Kroeger E. Impact of aeration and alkalinity on the water quality and product quality of transported tilapia - A simulation study. Aquacult Eng [serial on the Internet]. 2013; 55:46-58. Available from: https://doi.org/10.1016/j.aquaeng.2013.03.002
https://doi.org/10.1016/j.aquaeng.2013.0...
). However, CO2 removal is not immediate and therefore can still react with carbonates from the water, reducing water pH (Moran, 2010Moran D. Carbon dioxide degassing in fresh and saline water. II: Degassing performance of an air-lift. Aquacult Eng [serial on the Internet]. 2010; 3(3):120-27. Available from: https://doi.org/10.1016/j.aquaeng.2010.09.001
https://doi.org/10.1016/j.aquaeng.2010.0...
; Colt et al., 2012Colt J, Watten B, Pfeiffer T. Carbon dioxide stripping in aquaculture. Part 1: Terminology and reporting. Aquacult Eng [serial on the Internet]. 2012; 47:27-37. Available from: https://doi.org/10.1016/j.aquaeng.2011.12.008
https://doi.org/10.1016/j.aquaeng.2011.1...
), as observed in the present study. In addition, the decrease of water pH could be due, at least partially, to higher net acidic equivalent loss, which could be provoked by stimulation of gill H+-ATPase, as observed by Toni et al. (2014Toni C, Becker AG, Simões LN, Pinheiro CG, de Lima Silva L, Heinzmann BM, Caron BO, Baldisserotto B. Fish anesthesia: effects of the essential oils of Hesperozygis ringens and Lippia alba on the biochemistry and physiology of silver catfish (Rhamdia quelen). Fish Physiol Biochem [serial on the Internet]. 2014; 40(3):701-14. Available from: http://dx.doi.org/10.1007/s10695-013-9877-4
http://dx.doi.org/10.1007/s10695-013-987...
) in silver catfish exposed to the EO of H. ringens, or of the Na+/H+ antiporter, which would explain the higher net Na+ influx induced by some concentrations of EOCA and EOCL.
Stress increases ion branchial efflux (McDonald et al., 1991McDonald DG, Cavdek V, Ellis R. Gill design in freshwater fishes - Interrelationships among gas exchange, ion regulation, and acid-base regulation. Physiol Zool. 1991; 64(1):103-23.), but both EOs reduced ion loss at some concentrations and also reduced ammonia excretion, which could be also related to a reduction of the metabolism. Net ion fluxes provided a good correlation with the stress of transport for silver catfish (Becker et al., 2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
, 2013Becker AG, Cunha MA, Garcia LO, Zeppenfeld CC, Parodi TV, Maldaner G, Morel AF, Baldisserotto B. Efficacy of eugenol and the methanolic extract of Condalia buxifolia during the transport of the silver catfish Rhamdia quelen. Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):675-81. Available from: http://dx.doi.org/10.1590/S1679-62252013000300021
http://dx.doi.org/10.1590/S1679-62252013...
; Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
; Zeppenfeld et al., 2014Zeppenfeld CC, Toni C, Becker AG, Miron DS, Parodi TV, Heinzmann BM, Barcellos LJG, Koakoski G, Da Rosa JGS, Loro VL, Cunha MA, Baldisserotto B. Physiological and biochemical responses of silver catfish, Rhamdia quelen, after transport in water with essential oil of Aloysia triphylla (L’Herit) Britton. Aquaculture [serial on the Internet]. 2014; 418-19:101-07. Available from: https://doi.org/10.1016/j.aquaculture.2013.10.013
https://doi.org/10.1016/j.aquaculture.20...
; Garcia et al., 2015Garcia LO, Barcellos LJG, Baldisserotto B. Net ion fluxes and ammonia excretion during transport of Rhamdia quelen juveniles. Cienc Rural [serial on the Internet]. 2015; 45(10):1854-58. Available from: http://dx.doi.org/10.1590/0103-8478cr20141420
http://dx.doi.org/10.1590/0103-8478cr201...
). Sedation with eugenol and the EOs of L. alba and A. triphylla also reduced ammonia excretion and ion loss in silver catfish (Becker et al., 2012Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba. Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
http://dx.doi.org/10.1007/s10695-011-956...
, 2016Becker AG, Parodi TV, Zeppenfeld CC, Salbego J, Cunha MA, Heldwein CG, Loro VL, Heinzmann BM, Baldisserotto B. Pre-sedation and transport of Rhamdia quelen in water containing essential oil of Lippia alba: metabolic and physiological responses. Fish Physiol Biochem [serial on the Internet]. 2016; 42(1):73-81. Available from: http://dx.doi.org/10.1007/s10695-015-0118-x
http://dx.doi.org/10.1007/s10695-015-011...
; Parodi et al., 2014Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen. Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
http://dx.doi.org/10.1007/s10695-013-984...
; Zeppenfeld et al., 2014Zeppenfeld CC, Toni C, Becker AG, Miron DS, Parodi TV, Heinzmann BM, Barcellos LJG, Koakoski G, Da Rosa JGS, Loro VL, Cunha MA, Baldisserotto B. Physiological and biochemical responses of silver catfish, Rhamdia quelen, after transport in water with essential oil of Aloysia triphylla (L’Herit) Britton. Aquaculture [serial on the Internet]. 2014; 418-19:101-07. Available from: https://doi.org/10.1016/j.aquaculture.2013.10.013
https://doi.org/10.1016/j.aquaculture.20...
), methanolic extract of Condalia buxifolia and the EOs of Cunila galioides and Origanum majorana decreased ion loss in silver catfish (Becker et al., 2013Becker AG, Cunha MA, Garcia LO, Zeppenfeld CC, Parodi TV, Maldaner G, Morel AF, Baldisserotto B. Efficacy of eugenol and the methanolic extract of Condalia buxifolia during the transport of the silver catfish Rhamdia quelen. Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):675-81. Available from: http://dx.doi.org/10.1590/S1679-62252013000300021
http://dx.doi.org/10.1590/S1679-62252013...
; Cunha et al., 2017Cunha JA, Scheeren CA, Salbego S, Gressler LT, Madaloz LM, Bandeira Junior G, Bianchini AE, Pinheiro CG, Bordignon SAL, Heinzmann BM, Baldisserotto B. Essential oils of Cunila galioides and Origanum majorana as anesthetics for Rhamdia quelen: efficacy and effects on ventilation and ionoregulation. Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160076 DOI: 10.1590/1982-0224-20160076
https://doi.org/10.1590/1982-0224-201600...
). Eugenol also reduced Cl− efflux during and after transport of Salmo salar (Iversen et al., 2003Iversen M, Finstad B, Mckinley RS, Eliassen RA. The efficacy of metomidate, clove oil, Aqui-S™ and Benzoak® as anaesthetics in Atlantic salmon (Salmo salar L.) smolts, and their potential stress-reducing capacity. Aquaculture [serial on the Internet]. 2003; 221(1-4):549-66. Available from: https://doi.org/10.1016/S0044-8486(03)00111-X
https://doi.org/10.1016/S0044-8486(03)00...
). The addition of linalool, a compound found in EOCA composition (2.60%), to the water of transport, also reduced ammonia levels in the water post-transport of C. carpio, but as there was also an increase on serum urea levels in the linalool-treated fish, Mazandarani et al. (2017Mazandarani M, Hoseini SM, Ghomshani MD. Effects of linalool on physiological responses of Cyprinus carpio (Linnaeus, 1758) and water physico-chemical parameters during transportation. Aquac Res [serial on the Internet]. 2017; 48(12):5775-81. Available from: https://doi.org/10.1111/are.13400
https://doi.org/10.1111/are.13400...
) supposed that this compound may provoke improper ammonia excretion in the fish and conversion of ammonia to urea. It is not possible to rule out that the lower ammonia excretion induced in silver catfish by the EOs tested in the present study may cause the same effect because plasma ammonia or urea levels were not determined. Additional studies analyzing ammonia and urea excretion and plasma levels in fish exposed to the tested EOs must be performed to clarify this doubt.
The results suggest that EOCA and EOCL are effective anesthetics for silver catfish, but their usefulness as alternatives to reduce stress in fish transportation at the lower concentrations tested (50-100 µL L−1) deserves further study because some results indicate that these EOs may increase metabolism.
Acknowledgments
The authors offer thanks for the financial support and research fellowships awarded to B.M. Heinzmann and B. Baldisserotto received by Conselho Nacional de Desenvolvimento Tecnológico (CNPq), the postdoctoral fellowship to J.M. Lopes from Fundação de Amparao à Pesquisa do Estado do Maranhão (FAPEMA) and the PhD scholarship to C.F. Souza from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
References
- Acar U, Kesbiç OS, Yılmaz S, Gültepe N, Türker A. Evaluation of the effects of essential oil extracted from sweet orange peel (Citrus sinensis) on growth rate of tilapia (Oreochromis mossambicus) and possible disease resistance against Streptococcus iniae Aquaculture [serial on the Internet]. 2015; 437(1):282-86. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.015
» https://doi.org/10.1016/j.aquaculture.2014.12.015 - Adams RP. Identification of essential oil components by gas chromatography / quadrupole mass spectroscopy. Allured Publishing Corporation. 2009.
- Alvarenga CMD, Volpato GL. Agonistic profile and metabolism in alevins of the Nile tilapia. Physiol Behav. 1995; 57(1):75-80.
- Astani A, Reichling J, Schnitzler P. Comparative study on the antiviral activity of selected monoterpenes derived from essential oils. Phytother Res [serial on the Internet]. 2010; 24(5):673-79. Available from: https://doi.org/10.1002/ptr.2955
» https://doi.org/10.1002/ptr.2955 - Azambuja CR, Mattiazzi J, Riffel APK, Finamor IA, Garcia LO, Heldwein CG, Heinzmann BM, Baldisserotto B, Pavanato MA, Llesuy SF. Effect of the essential oil of Lippia alba on oxidative stress parameters in silver catfish (Rhamdia quelen) subjected to transport. Aquaculture [serial on the Internet]. 2011; 319(1-2):156-61. Available from: https://doi.org/10.1016/j.aquaculture.2011.06.002
» https://doi.org/10.1016/j.aquaculture.2011.06.002 - Barcellos LJG, Woehl VM, Wassermann GF, Quevedo RM, Ittzés I, Krieger MH. Plasma levels of cortisol and glucose in response to capture and tank transference in Rhamdia quelen (Quoy and Gaimard), a South American catfish. Aquacult Res [serial on the Internet]. 2001; 32(2):121-23. Available from: https://doi.org/10.1046/j.1365-2109.2001.00539.x
» https://doi.org/10.1046/j.1365-2109.2001.00539.x - Becker AG, Cunha MA, Garcia LO, Zeppenfeld CC, Parodi TV, Maldaner G, Morel AF, Baldisserotto B. Efficacy of eugenol and the methanolic extract of Condalia buxifolia during the transport of the silver catfish Rhamdia quelen Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):675-81. Available from: http://dx.doi.org/10.1590/S1679-62252013000300021
» http://dx.doi.org/10.1590/S1679-62252013000300021 - Becker AG, Parodi TV, Heldwein CG, Zeppenfeld CC, Heinzmann BM, Baldisserotto B. Transportation of silver catfish, Rhamdia quelen, in water with eugenol and the essential oil of Lippia alba Fish Physiol Biochem [serial on the Internet]. 2012; 38 (3):789-96. Available from: http://dx.doi.org/10.1007/s10695-011-9562-4
» http://dx.doi.org/10.1007/s10695-011-9562-4 - Becker AG, Parodi TV, Zeppenfeld CC, Salbego J, Cunha MA, Heldwein CG, Loro VL, Heinzmann BM, Baldisserotto B. Pre-sedation and transport of Rhamdia quelen in water containing essential oil of Lippia alba: metabolic and physiological responses. Fish Physiol Biochem [serial on the Internet]. 2016; 42(1):73-81. Available from: http://dx.doi.org/10.1007/s10695-015-0118-x
» http://dx.doi.org/10.1007/s10695-015-0118-x - Benovit SC, Silva LL, Salbego J, Loro VL, Mallmann CA, Baldisserotto B, Flores EMM, Heinzmann BM. Anesthetic activity and bio-guided fractionation of the essential oil of Aloysia gratissima (Gillies & Hook.) Tronc. in silver catfish Rhamdia quelen An Acad Bras Cienc [serial on the Internet]. 2015; 87(3):1675-89. Available from: http://dx.doi.org/10.1590/0001-3765201520140223
» http://dx.doi.org/10.1590/0001-3765201520140223 - Bizzo HR, Hovell AMC, Rezende CM. Essential oils in Brazil: general aspects, development and prospects. New Chem [serial on the Internet]. 2009; 32(3):588-94. Available from: http://dx.doi.org/10.1590/S0100-40422009000300005
» http://dx.doi.org/10.1590/S0100-40422009000300005 - Carvalho-Freitas MIR, Costa M. Anxiolytic and sedative effects of extracts and essential oil from Citrus aurantium L. Biol Pharm Bull. 2002; 25(12):1629-33.
- Ceccarelli I, Lariviere WR, Fiorenzani P, Sacerdote P, Aloisi AM. Effects of long-term exposure of lemon essential oil odor on behavioral, hormonal and neuronal parameters in male and female rats. Brain Res [serial on the Internet]. 2004; 1001(1-2):78-86. Available from: http://dx.doi.org/10.1016/j.brainres.2003.10.063
» http://dx.doi.org/10.1016/j.brainres.2003.10.063 - Colt J, Kroeger E. Impact of aeration and alkalinity on the water quality and product quality of transported tilapia - A simulation study. Aquacult Eng [serial on the Internet]. 2013; 55:46-58. Available from: https://doi.org/10.1016/j.aquaeng.2013.03.002
» https://doi.org/10.1016/j.aquaeng.2013.03.002 - Colt J, Orwicz K. Modeling production capacity of aquatic culture systems under freshwater conditions. Aquacult Eng [serial on the Internet]. 1991; 10(1):1-29. Available from: https://doi.org/10.1016/0144-8609(91)90008-8
» https://doi.org/10.1016/0144-8609(91)90008-8 - Colt J, Watten B, Pfeiffer T. Carbon dioxide stripping in aquaculture. Part 1: Terminology and reporting. Aquacult Eng [serial on the Internet]. 2012; 47:27-37. Available from: https://doi.org/10.1016/j.aquaeng.2011.12.008
» https://doi.org/10.1016/j.aquaeng.2011.12.008 - Council of Europe. European Pharmacopeia. 6th ed. Strassbourg: European Directorate for the Quality of Medicines; 2007.
- Costa CARA, Cury TC, Cassettan BO, Takahira RK, Flório JC, Costa M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. BMC Complem Altern M [serial on the Internet]. 2013; 13:42. Available from: http://dx.doi.org/10.1186/1472-6882-13-42
» http://dx.doi.org/10.1186/1472-6882-13-42 - Cunha MA, Barros FMC, Garcia LO, Veeck APL, Heinzmann BM, Loro VL, Emanuelli T, Baldisserotto B. Essential oil of Lippia alba: a new anesthetic for silver catfish, Rhamdia quelen Aquaculture [serial on the Internet]. 2010; 306(1-4):403-06. Available from: https://doi.org/10.1016/j.aquaculture.2010.06.014
» https://doi.org/10.1016/j.aquaculture.2010.06.014 - Cunha JA, Scheeren CA, Salbego S, Gressler LT, Madaloz LM, Bandeira Junior G, Bianchini AE, Pinheiro CG, Bordignon SAL, Heinzmann BM, Baldisserotto B. Essential oils of Cunila galioides and Origanum majorana as anesthetics for Rhamdia quelen: efficacy and effects on ventilation and ionoregulation. Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160076 DOI: 10.1590/1982-0224-20160076
» https://doi.org/10.1590/1982-0224-20160076 - Cunha MA, Silva BF, Delunardo FAC, Benovit SC, Gomes LC, Heinzmann BM, Baldisserotto B. Anesthetic induction and recovery of Hippocampus reidi exposed to the essential oil of Lippia alba Neotrop Ichthyol [serial on the Internet]. 2011; 9(3):683-88. Available from: http://dx.doi.org/10.1590/S1679-62252011000300022
» http://dx.doi.org/10.1590/S1679-62252011000300022 - Faturi CB, Leite JR, Alves PB, Canton AC, Teixeira-Silva F. Anxiolytic-like effect of sweet orange aroma in Wistar rats. Prog Neuro-Psychopha [serial on the Internet]. 2010; 34(4):605-09. Available from: http://dx.doi.org/10.1016/j.pnpbp.2010.02.020
» http://dx.doi.org/10.1016/j.pnpbp.2010.02.020 - Fukumoto S, Sawasaki E, Okuyama S, Miyake Y, Yokogoshi H. Flavor components of monoterpenes in citrus essential oils enhance the release of monoamines from rat brain slices. Nutr Neurosci [serial on the Internet]. 2006; 9(1-2):73-80. Available from: http://dx.doi.org/10.1080/10284150600573660
» http://dx.doi.org/10.1080/10284150600573660 - Garcia LO, Barcellos LJG, Baldisserotto B. Net ion fluxes and ammonia excretion during transport of Rhamdia quelen juveniles. Cienc Rural [serial on the Internet]. 2015; 45(10):1854-58. Available from: http://dx.doi.org/10.1590/0103-8478cr20141420
» http://dx.doi.org/10.1590/0103-8478cr20141420 - Gargano AC, Almeida CAR, Costa M. Essential oils from Citrus latifolia and Citrus reticulata reduced anxiety and prolong ether sleeping time in mice. Tree For Sci Biotechnol. 2008; 2(1):121-24.
- Gressler LT, Riffel APK, Parodi TV, Saccol EMH, Koakoski G, Costa ST et al Silver catfish Rhamdia quelen immersion anaesthesia with essential oil of Aloysia triphylla (L’Herit) Britton or tricaine methanesulfonate: effect on stress response and antioxidant status. Aquacult Res [serial on the Internet]. 2012; 45(6):1061-72. Available from: http://dx.doi.org/10.1111/are.12043
» http://dx.doi.org/10.1111/are.12043 - Guénette SA, Uhland FC, Helie P, Beaudry F, Vachon P. Pharmacokinetics of eugenol in rainbow trout (Onchorhynchus mykiss). Aquaculture [serial on the Internet]. 2007; 266(1-4):262-5. Available from: https://doi.org/10.1016/j.aquaculture.2007.02.046
» https://doi.org/10.1016/j.aquaculture.2007.02.046 - Hajek GJ. The anaesthetic-like effect of tea tree oil in common carp Cyprinus carpio L. Aquacult Res [serial on the Internet]. 2011; 42(2):296-300. Available from: https://doi.org/10.1111/j.1365-2109.2010.02625.x
» https://doi.org/10.1111/j.1365-2109.2010.02625.x - Hatano VY, Torricelli AS, Giassi ACC, Coslope LA,Viana MB. Anxiolytic effects of repeated treatment with an essential oil from Lippia alba and (R)-(-)-carvone in the elevated T-maze. Braz J Med Biol Res. 2012; 45(3):238-43.
- Heldwein CG, Silva LL, Gai EZ, Roman C, Parodi TV, Burger ME, Baldisserotto B, Flores EM, Heinzmann BM. S-(+)-Linalool from Lippia alba: sedative and anesthetic for silver catfish (Rhamdia quelen). Vet Anaesth Analg [serial on the Internet]. 2014; 41(6):621-9. Available from: https://doi.org/10.1111/vaa.12146
» https://doi.org/10.1111/vaa.12146 - Hoet S, Stévigny C, Herent MF, Quetin-Leclercq J. Antitrypanosomal compounds from leaf essential oil of Strychnos spinosa Planta Med [serial on the Internet]. 2006; 72(5):480-2. Available from: http://dx.doi.org/10.1055/s-2005-916255
» http://dx.doi.org/10.1055/s-2005-916255 - Hohlenwerger JC, Copatti CE, Sena AC, Couto RD, Baldisserotto B, Heinzmann BM, Caron BO, Schmidt D. Could the essential oil of Lippia alba provide a readily available and cost-effective anaesthetic for Nile tilapia (Oreochromis niloticus)? Mar Freshw Behav Phy [serial on the Internet] . 2016; 49(2):119-26. Available from: https://doi.org/10.1080/10236244.2015.1123869
» https://doi.org/10.1080/10236244.2015.1123869 - Iversen M, Finstad B, Mckinley RS, Eliassen RA. The efficacy of metomidate, clove oil, Aqui-S™ and Benzoak® as anaesthetics in Atlantic salmon (Salmo salar L.) smolts, and their potential stress-reducing capacity. Aquaculture [serial on the Internet]. 2003; 221(1-4):549-66. Available from: https://doi.org/10.1016/S0044-8486(03)00111-X
» https://doi.org/10.1016/S0044-8486(03)00111-X - Kang P, Suh SH, Min SS, Seol GH. The essential oil of Citrus bergamia Risso induces vasorelaxation of the mouse aorta by activating K + channels and inhibiting Ca 2+ influx. J Pharm Pharmacol [serial on the Internet]. 2013; 65(5):745-49. Available from: http://dx.doi.org/10.1111/jphp.12031
» http://dx.doi.org/10.1111/jphp.12031 - Kasanen JP, Pasanen AL, Pasanen P, Liesivuori J, Kosma VM, Alarie Y. Stereospecificity of the sensory irritation receptor for nonreactive chemicals illustrated by pinene enantiomers. Arch Toxicol. 1998; 72(8):514-23.
- Keene JL, Noakes DLG, Moccia RD, Soto CG. The efficacy of clove oil as an anaesthetic for rainbow trout, Oncorhynchus mykiss (Walbaum). Aquacult Res . 1998; 29(2):89-101.
- Kobori CN, Jorge N. Characterization of some seed oils of fruits for utilization of industrial residues. Cienc Agrotec [serial on the Internet]. 2005; 29(5):1008-14. Available from: http://dx.doi.org/10.1590/S1413-70542005000500014
» http://dx.doi.org/10.1590/S1413-70542005000500014 - Komiya M, Takashi T, Harada E. Lemon oil vapor causes an anti-stress effect via modulating the 5-HT and DA activities in mice. Behav Brain Res [serial on the Internet]. 2006; 172(2):240-49. Available from: http://dx.doi.org/10.1016/j.bbr.2006.05.006
- Kummer R, Fachini-Queiroz FC, Estevão-Silva CF, Grespan R, Silva EL, Bersani-Amado CA, Cuman RKN. Evaluation of anti-inflammatory activity of Citrus latifolia Tanaka essential oil and limonene in experimental mouse models. Evid-Based Compl Alt [serial on the Internet]. 2013; 2013:859083 Available from: http://dx.doi.org/10.1155/2013/859083
» http://dx.doi.org/10.1155/2013/859083 - Lehrner J, Marwinski G, Lehr S, Johren P, Deecke L. Ambient odor of orange and lavender reduce anxiety and improve mood in a dental office. Physiol Behav [serial on the Internet]. 2005; 86(1-2):92-95. Available from: http://dx.doi.org/10.1016/j.physbeh.2005.06.031
» http://dx.doi.org/10.1016/j.physbeh.2005.06.031 - Leite MP, Fassin J Jr, Baziloni EMF, Almeida RN, Mattei R, Leite JR. Behavioral effects of essential oil of Citrus aurantium L. inhalation in rats. Braz J Pharmacogn [serial on the Internet]. 2008; 18:661-66. Available from: http://dx.doi.org/10.1590/S0102-695X2008000500003
» http://dx.doi.org/10.1590/S0102-695X2008000500003 - Limma-Netto JD, Sena AC, Copatti CE. Essential oils of Ocimum basilicum and Cymbopogon flexuosus in the sedation, anesthesia and recovery of tambacu (Piaractus mesopotamicus male x Colossoma macropomum female). Bol Inst Pesca [serial on the Internet]. 2016; 42(3):727-33. Available from: http://dx.doi.org/10.20950/1678-2305.2016v42n3p727
» http://dx.doi.org/10.20950/1678-2305.2016v42n3p727 - Marking LL, Meyer FP. Are better anesthetics needed in fisheries? Fisheries. 1985; 10(6):2-5.
- Mattos D Jr, Milaneze TF, Azevedo FA, Quaggio JA. Soil nutrient availability and its impact on fruit quality of Tahiti acid lime. Rev Bras Frutic [serial on the Internet]. 2010; 32(1):335-42. Available from: http://dx.doi.org/10.1590/S0100-29452010005000032
» http://dx.doi.org/10.1590/S0100-29452010005000032 - Mazandarani M, Hoseini SM, Ghomshani MD. Effects of linalool on physiological responses of Cyprinus carpio (Linnaeus, 1758) and water physico-chemical parameters during transportation. Aquac Res [serial on the Internet]. 2017; 48(12):5775-81. Available from: https://doi.org/10.1111/are.13400
» https://doi.org/10.1111/are.13400 - McDonald DG, Cavdek V, Ellis R. Gill design in freshwater fishes - Interrelationships among gas exchange, ion regulation, and acid-base regulation. Physiol Zool. 1991; 64(1):103-23.
- Mercier B, Prost J, Prost M. The essential oil of turpentine and its major volatile fraction (alpha- and betapinenes): a review. Int J Occup Med Env [serial on the Internet]. 2009; 22(4):331-42. DOI: 10.2478/v10001-009-0032-5
» https://doi.org/10.2478/v10001-009-0032-5 - Mesfin M, Asres K, Shibeshi W. Evaluation of anxiolytic activity of the essential oil of the aerial part of Foeniculum vulgare Miller in mice. BMC Complem Altern M [serial on the Internet]. 2014; 14:310. Available from: https://doi.org/10.1186/1472-6882-14-310
» https://doi.org/10.1186/1472-6882-14-310 - Mirghaed AT, Ghelichpour T, Hoseini SM. Myrcene and linalool as new anesthetic and sedative agents in common carp, Cyprinus carpio - comparison with eugenol. Aquaculture [serial on the Internet]. 2016; 464(1):165-70. Available from: https://doi.org/10.1016/j.aquaculture.2016.06.028
» https://doi.org/10.1016/j.aquaculture.2016.06.028 - Moran D. Carbon dioxide degassing in fresh and saline water. II: Degassing performance of an air-lift. Aquacult Eng [serial on the Internet]. 2010; 3(3):120-27. Available from: https://doi.org/10.1016/j.aquaeng.2010.09.001
» https://doi.org/10.1016/j.aquaeng.2010.09.001 - NIST/ EPA/ NIH mass spectral library and search/ analysis programs. John Wiley and Sons. Hoboken, 2010.
- Park MO, Hur WJ, Im SY, Seol DW, Lee J, Park IS. Anaesthetic efficacy and physiological responses to clove oil-anaesthetized kelp grouper Epinephelus bruneus Aquacult Res [serial on the Internet]. 2008; 39(8):877-84. Available from: https://doi.org/10.1111/j.1365-2109.2008.01941.x
» https://doi.org/10.1111/j.1365-2109.2008.01941.x - Park HM, Lee JH, Yaoyao J, Jun HJ, Lee SJ. Limonene, a natural cyclic terpene, is an agonistic ligand for adenosine A(2A) receptors. Biochem Bioph Res Co [serial on the Internet]. 2011; 404(1):345-48. Available from: http://dx.doi.org/10.1016/j.bbrc.2010.11.121
» http://dx.doi.org/10.1016/j.bbrc.2010.11.121 - Parodi TV, Cunha MA, Becker AG, Zeppenfeld CC, Martins DI, Koakoski G, Barcellos LG, Heinzmann BM, Baldisserotto B. Anesthetic activity of the essential oil of Aloysia triphylla and effectiveness in reducing stress during transport of albino and gray strains of silver catfish, Rhamdia quelen Fish Physiol Biochem [serial on the Internet]. 2014; 40(2):323-34. Available from: http://dx.doi.org/10.1007/s10695-013-9845-z
» http://dx.doi.org/10.1007/s10695-013-9845-z - Pedrazzani AS, Ostrensky A Neto. The anaesthetic effect of camphor (Cinnamomum camphora), clove (Syzygium aromaticum) and mint (Mentha arvensis) essential oils on clown anemonefish, Amphiprion ocellaris (Cuvier 1830). Aquacult Res [serial on the Internet]. 2016; 47(3):769-76. Available from: https://doi.org/10.1111/are.12535
» https://doi.org/10.1111/are.12535 - Pinheiro CG, Machado CM, Amaral LP, Silva DT, Almeida CAA, Longhi SJ, Mallmann CA, Heinzmanna BM. Seasonal variability of the essential oil of Hesperozygis ringens (Benth.) Epling. Braz J Biol [serial on the Internet]. 2016; 76(1):176-84. Available from: http://dx.doi.org/10.1590/1519-6984.16314
» http://dx.doi.org/10.1590/1519-6984.16314 - Prince A, Powell C. Clove oil as an anaesthetic for invasive field procedures on adult rainbow trout. N Am J Fish Manage. 2000; 20(4):1029-32.
- Pultrini AM, Galindo LA, Costa M. Effects of the essential oil from Citrus aurantium L. in experimental anxiety models in mice. Life Sci [serial on the Internet]. 2006; 78(15):1720-25. Available from: http://dx.doi.org/10.1016/j.lfs.2005.08.004
» http://dx.doi.org/10.1016/j.lfs.2005.08.004 - Roohi Z, Imanpoor MR. The efficacy of the oils of spearmint and methyl salicylate as new anesthetics and their effect on glucose levels in common carp (Cyprinus carpio L., 1758) juveniles. Aquaculture [serial on the Internet]. 2015; 437:327-32. Available from: https://doi.org/10.1016/j.aquaculture.2014.12.019
» https://doi.org/10.1016/j.aquaculture.2014.12.019 - Ross LG, Ross B. Anaesthetic and sedative techniques for aquatic animals. 3rd ed. Oxford: Blackwell Science; 2008.
- Saiyudthong S, Marsden C. Acute effects of bergamot oil on anxiety-related behaviour and corticosterone level in rats. Phytother Res [serial on the Internet]. 2011; 25(6):858-62. Available from: http://dx.doi.org/10.1002/ptr.3325
» http://dx.doi.org/10.1002/ptr.3325 - Santos AC, Junior GB, Zago DC, Zeppenfeld CC, da Silva DT, Heinzmann BM, da Cunha MA. Anesthesia and anesthetic action mechanism of essential oils of Aloysia triphylla and Cymbopogon flexuosus in silver catfish (Rhamdia quelen). Vet Anaesth Analg [serial on the Internet]. 2017; 44(1):106-13. Available from: http://dx.doi.org/ 10.1111/vaa.12386
» http://dx.doi.org/ 10.1111/vaa.12386 - Sharma N, Tripathi A. Effects of Citrus sinensis (L.) Osbeck epicarp essential oil on growth and morphogenesis of Aspergillus niger (L.) Van Tieghem. Microbiol Res [serial on the Internet]. 2008; 163(3):337-44. Available from: http://dx.doi.org/10.1016/j.micres.2006.06.009
» http://dx.doi.org/10.1016/j.micres.2006.06.009 - Silva LL, Balconi LS, Gressler LT, Garlet QI, Sutili FJ, Vargas APC, Baldisserotto B, Morel AF , Heinzmann BM. S-(+)- and R-(-)-linalool: a comparison of the in vitro anti-Aeromonas hydrophila activity and anesthetic properties in fish. An Acad Bras Cienc [serial on the Internet]. 2017; 89(1):203-12. Available from: http://dx.doi.org/10.1590/0001-3765201720150643
» http://dx.doi.org/10.1590/0001-3765201720150643 - Silva LL, Garlet QI, Benovit SC, Dolci G, Mallmann CA, Bürger ME, Baldisserotto B, Longhi SJ, Heinzmann BM. Sedative and anesthetic activities of the essential oils of Hyptis mutabilis (Rich.) Briq. and their isolated components in silver catfish (Rhamdia quelen). Braz J Med Biol Res [serial on the Internet]. 2013a; 46(9):771-79. Available from: http://dx.doi.org/10.1590/1414-431X20133013
» http://dx.doi.org/10.1590/1414-431X20133013 - Silva LD, Garlet QI, Koakoski G, Abreu MS, Mallmann CA, Baldisserotto B, Barcellos LJG, Heinzmann BM. Anesthetic activity of the essential oil of Ocimum americanum in Rhamdia quelen (Quoy & Gaimard, 1824) and its effects on stress parameters. Neotrop Ichthyol [serial on the Internet]. 2015; 13(4):715-22. Available from: http://dx.doi.org/10.1590/1982-0224-20150012
» http://dx.doi.org/10.1590/1982-0224-20150012 - Silva LL, Parodi TV, Reckziegel P, Garcia VO, Bürger ME, Baldisserotto B, Malmann CA, Pereira AMS, Heinzmann BM. Essential oil of Ocimum gratissimum: anesthetic effect, mechanism of action and tolerance in silver catfish (Rhamdia quelen). Aquaculture [serial on the Internet]. 2012; 350-353:91-97. Available from: https://doi.org/10.1016/j.aquaculture.2012.04.012
» https://doi.org/10.1016/j.aquaculture.2012.04.012 - Silva LL, Silva TS, Garlet QI, Cunha MA, Mallmann CA, Baldisserotto B, Longhi SJ, Pereira AMS, Heinzmann BM. Anesthetic activity of Brazilian native plants in silver catfish (Rhamdia quelen). Neotrop Ichthyol [serial on the Internet]. 2013b; 11(2):443-51. Available from: http://dx.doi.org/10.1590/S1679-62252013000200014
» http://dx.doi.org/10.1590/S1679-62252013000200014 - Small BC. Anesthetic efficacy of metomidate and comparison of plasma cortisol responses to tricaine methanesulfonate, quinaldine and clove oil anesthetized channel catfish Ictalurus punctatus Aquaculture [serial on the Internet]. 2003; 218(1-4):177-85. Available from: https://doi.org/10.1016/S0044-8486(02)00302-2
» https://doi.org/10.1016/S0044-8486(02)00302-2 - Souza CF, Baldissera MD, Salbego J, Lopes JM, Vaucher RA, Mourão VRH, Caron BO, Heinzmann BM, Silva LVF, Baldisserotto B. Physiological responses of Rhamdia quelen (Siluriformes: Heptapteridae) to anesthesia with essential oils from two different chemotypes of Lippia alba Neotrop Ichthyol [serial on the Internet]. 2017; 15(1):e160083. Available from: http://dx.doi.org/10.1590/1982-0224-20160083
» http://dx.doi.org/10.1590/1982-0224-20160083 - Sousa DG, Sousa SD, Silva RE, Silva-Alves KS, Ferreira-da-Silva FW, Kerntopf MR, Menezes IR, Leal-Cardoso JH, Barbosa R. Essential oil of Lippia alba and its main constituent citral block the excitability of rat sciatic nerves. Braz J Med Biol Res [serial on the Internet]. 2015; 48(8):697-702. Available from: https://doi.org/10.1590/1414-431X20154710
» https://doi.org/10.1590/1414-431X20154710 - Tondolo JSM, Amaral LP, Simoes LN, Garlet QI, Schindler B, Oliveira TM, Silva BF, Gomes LC, Baldisserotto B, Mallmann CA, Heinzmann BM. Anesthesia and transport of fat snook Centropomus parallelus with the essential oil of Nectandra megapotamica (Spreng.) Mez. Neotrop Ichthyol [serial on the Internet]. 2013; 11(3):667-74. Available from: http://dx.doi.org/10.1590/S1679-62252013000300020
» http://dx.doi.org/10.1590/S1679-62252013000300020 - Toni C, Becker AG, Simões LN, Pinheiro CG, de Lima Silva L, Heinzmann BM, Caron BO, Baldisserotto B. Fish anesthesia: effects of the essential oils of Hesperozygis ringens and Lippia alba on the biochemistry and physiology of silver catfish (Rhamdia quelen). Fish Physiol Biochem [serial on the Internet]. 2014; 40(3):701-14. Available from: http://dx.doi.org/10.1007/s10695-013-9877-4
» http://dx.doi.org/10.1007/s10695-013-9877-4 - Toni C, Martos-Sitcha JA, Jarabo IJ, Mancera JM, Rodriguez GM, Pinheiro CG, Heinzmann BM, Baldisserotto B. Stress response in silver catfish (Rhamdia quelen) exposed to the essential oils of Hesperozygis ringens Fish Physiol Biochem [serial on the Internet]. 2015; 41(1):129-38. Available from: http://dx.doi.org/10.1007/s10695-014-0011-z
» http://dx.doi.org/10.1007/s10695-014-0011-z - Vale GT, Furtado EC, Santos JG, Viana GSB. Central effects of citral, myrcene and limonene, constituents of essential oil chemotypes from Lippia alba (Mill.) N.E. Brown. Phytomedicine [serial on the Internet]. 2002; 9(8):709-14. Available from: https://doi.org/10.1078/094471102321621304
» https://doi.org/10.1078/094471102321621304 - Zall DM, Fisher MD, Garner QM. Photometric determination of chlorides in water. Anal Chem [serial on the Internet]. 1956; 28(11):1665-68. Available from: http://dx.doi.org/10.1021/ac60119a009
» http://dx.doi.org/10.1021/ac60119a009 - Zeppenfeld CC, Toni C, Becker AG, Miron DS, Parodi TV, Heinzmann BM, Barcellos LJG, Koakoski G, Da Rosa JGS, Loro VL, Cunha MA, Baldisserotto B. Physiological and biochemical responses of silver catfish, Rhamdia quelen, after transport in water with essential oil of Aloysia triphylla (L’Herit) Britton. Aquaculture [serial on the Internet]. 2014; 418-19:101-07. Available from: https://doi.org/10.1016/j.aquaculture.2013.10.013
» https://doi.org/10.1016/j.aquaculture.2013.10.013
Edited by
Publication Dates
-
Publication in this collection
11 June 2018 -
Date of issue
2018
History
-
Received
29 Nov 2017 -
Accepted
11 Apr 2018