Abstracts
In this study, we report a case of pulmonary cryptococcosis in a patient presenting respiratory symptoms and a lung mass on the chest X-ray. The patient had no concomitant diseases, was seronegative for human immunodeficiency virus and was not receiving immunosuppressive therapy of any kind. The diagnosis was confirmed through transbronchial biopsy and bronchoalveolar lavage. The patient was treated as an outpatient with fluconazole (300 mg/day for six months), evolving to clinical improvement and partial regression of the lung mass, as seen on a second chest X-ray. The current case illustrates an unusual presentation of pulmonary cryptococcosis and raises questions regarding the therapeutic approaches proposed in the literature.
Cryptococcosis; Lung diseases, fungal; Lung; Immunocompetence
O presente trabalho relata um caso de criptococose pulmonar isolada em paciente com sintomas respiratórios, sem imunossupressão e sorologia negativa para o vírus da imunodeficiência humana, com massa pulmonar no radiograma de tórax. O diagnóstico foi confirmado pela biópsia transbrônquica e lavado broncoalveolar. A paciente recebeu tratamento ambulatorial com fluconazol, na dose de 300 mg/dia por seis meses, evoluindo com melhora clínica e regressão parcial da imagem radiológica. O presente caso ilustra uma apresentação não freqüente da criptococose pulmonar e faz considerações sobre a abordagem terapêutica com base na literatura.
Criptococose; Pneumopatias fúngicas; pulmonar; Pulmão; Imunocompetência
CASE REPORT
Isolated pulmonary cryptococcosis in an immunocompetent patient* * Study carried out at the Universidade Estadual de Montes Claros (UNIMONTES, State University of Montes Claros) - Montes Claros, Brazil.
Ana Teresa Fernandes BarbosaI; Fernando Antônio ColaresII; Edson da Silva GusmãoI; Amanda Araújo BarrosIII; Cristiane Gonçalves CordeiroIII; Maria Cecília Tolentino AndradeIII
IAssistant Professor at the Universidade Estadual de Montes Claros (UNIMONTES, State University of Montes Claros) - Montes Claros, Brazil
IIFull Professor of Pulmonology at the Universidade Estadual de Montes Claros (UNIMONTES, State University of Montes Claros) School of Medicine - Montes Claros, Brazil
IIIMedical student at the Universidade Estadual de Montes Claros (UNIMONTES, State University of Montes Claros) - Montes Claros, Brazil
Correspondence to Correspondence to: Ana Teresa Fernandes Barbosa Av. Deputado Esteves Rodrigues, 616, Sala 601, Centro Montes Claros, MG, Brazil Phone: 55 38 3211-1169 E-mail: anateresaf@terra.com.br
ABSTRACT
In this study, we report a case of pulmonary cryptococcosis in a patient presenting respiratory symptoms and a lung mass on the chest X-ray. The patient had no concomitant diseases, was seronegative for human immunodeficiency virus and was not receiving immunosuppressive therapy of any kind. The diagnosis was confirmed through transbronchial biopsy and bronchoalveolar lavage. The patient was treated as an outpatient with fluconazole (300 mg/day for six months), evolving to clinical improvement and partial regression of the lung mass, as seen on a second chest X-ray. The current case illustrates an unusual presentation of pulmonary cryptococcosis and raises questions regarding the therapeutic approaches proposed in the literature.
Keywords: Cryptococcosis; Lung diseases, fungal; Lung/radiography; Immunocompetence
INTRODUCTION
Cryptococcal infection has global distribution without any specific endemic area.(1) Cryptococcosis can be caused by two varieties of Cryptococcus neoformans: C. neoformans var. neoformans and C. neoformans var. gatti. The former is more prevalent.
C. neoformans is a saprophyte fungus with a characteristic polysaccharide capsule.(2) Its ecological niche is poorly defined but it has been associated with the feces of pigeons and other birds.(3)
In humans, cryptococcosis normally occurs when fungal allergens are aerosolized and inhaled. Presentations range from asymptomatic pulmonary colonization to meningeal involvement and disseminated disease.(1) C. neoformans var. neoformans has strong tropism for the central nervous system, and most of the infections caused by the fungus involve the meninges,(3) causing higher morbidity and mortality.
The most common radiological finding in cryptococcosis is single or multiple nodules, usually subpleural, ranging from 0.5 to 3 cm in diameter. The presence of cavitations is relatively uncommon when compared to other mycoses.(4)
Frequently seen as an opportunistic disease affecting immunocompromised patients, specifically those who suffer from acquired immunodeficiency syndrome,(4) cryptococcosis is rare in immunocompetent patients.(5)
CASE REPORT
A 69-year-old female homemaker from the city of Montes Claros, Brazil was admitted to the Clemente de Faria University Hospital with a clinical profile that had been established 30 days prior to admission. The patient presented with a history of fever, chills, asthenia, jaundice, cough with scant production of sputum, which, when produced, was yellowish and mucoid. Over the preceding 30 days, the patient had lost weight (7 kg). The patient had experienced episodes of hemoptysis and presented chest pain upon drawing a breath, more severely in the left infraclavicular and scapular regions. The patient was submitted to antibiotic therapy (amoxicillin, procaine penicillin and ceftriaxone), but there was no clinical improvement.
The patient had been a smoker for 30 years (four cigarettes/day) and had quit smoking two months prior. She had had a hysterectomy fifteen years prior. She denied having had contact with birds or pigeons.
The physical examination revealed a good nutritional state, with normal respiration and no lymph node enlargement. In addition, there were no alterations in the neurological examination. The patient presented symmetric chest expansion, and auscultation revealed reduced vocal fremitus and breath sounds, together with underlying fluid density in the left apex upon percussion.
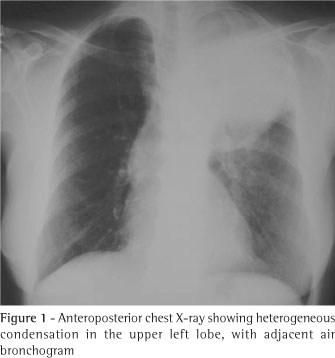
The chest X-ray revealed a dense mass in the upper left lobe (Figure 1). The computed tomography scan of the chest (Figure 2) confirmed the presence of a heterogeneous mass in the left upper lobe, with air bronchogram adjacent to the mass, suggesting an expansive neoplastic lesion. The patient was unable to produce a sputum sample for testing.
In order to reach a specific diagnosis, the patient was submitted to fiberoptic bronchoscopy, which showed no endobronchial alterations. Transbronchial biopsy and bronchoalveolar lavage revealed C. neoformans (Figure 3), with negative results for acid-fast bacilli and malignancy.
Proceeding with the evaluation, the patient presented reactive direct agglutination in the blood (1:40,000) and in urine (1:1000). Serology for human immunodeficiency virus was negative, and there were no other causes of immunosuppression. The patient was submitted to a spinal tap. In the study of the spinal fluid, the direct testing for C. neoformans was negative, and the direct agglutination revealed a reactive titer of 1:2. The blood culture was negative.
The patient was discharged on 300 mg/day of fluconazole and was in outpatient treatment for six months, after which there was clinical improvement and partial regression of the lung mass, as seen on a second chest X-ray.
DISCUSSION
The incidence of cryptococcosis in patients with negative serology for the human immunodeficiency virus reported in a recent population study(5) varied with the geographic location studied, ranging from 0.2 to 0.9 per 100,000 inhabitants. These rates are similar to those described for meningococcal meningitis (1:100.000 inhabitants). There are no data available regarding the incidence of cryptococcosis in Brazil.
Isolated pulmonary cryptococcosis is rare. Some authors(1) reported a series of 143 cases of cryptococcosis, 42 of which were in immunocompetent patients (29%). In contrast to the high dissemination rate seen in immunocompromised patients, 36 (86%) of the 42 immunocompetent patients were diagnosed with isolated pulmonary cryptococcosis.
Unlike some findings reported in the literature,(6-7) which showed the infection predominated in males, some studies(1,3,8) have found no predominance of either gender.
The literature indicates that pulmonary cryptococcosis affects patients in the 24-62 age bracket.(1,3,8) Some studies(6) have shown a predominance of elderly patients, such as in the present case.
Pulmonary cryptococcosis is prevalent in patients with a malfunction in the immunity mediated by cells, such as in acquired immunodeficiency syndrome, transplant-related immunosuppression, corticosteroid therapy, chemotherapy, neoplasms and lymphoproliferative diseases.(2,5,9) In some individuals, smoking may predispose to infection with C. neoformans.(5)
C. neoformans var. neoformans has been isolated form trees, as well as from bird feces, especially pigeon feces. The infection can be acquired by inhaling infected propagules, although it is not definitively clear how this happens.(5,10) The patient in the present case was not immunosuppressed, and there was no evidence of exposure to the fungus.
C. neoformans principally affects the lungs and the central nervous system, but it can also affect the skin, prostate and eyes, as well as potentially having a disseminated, multiple-organ presentation. In immunocompetent patients, approximately one-third of all cases of pulmonary cryptococcosis are asymptomatic, and it is typically an accidental finding on an X-ray. Symptomatic patients can present symptoms of infection, such as fever, cough, chest pain, weight loss and purulent sputum.(1,5,9,11-12)
Some authors reported that 24 (67%) of 42 cases as symptomatic, showing respiratory and/or constitutional symptoms such as cough, dyspnea, fever, night sweats and hemoptysis.(1) The authors of that study also highlighted the fact that, in immunocompromised patients, cryptococcosis can be severe and rapidly progressive, requiring prolonged antifungal treatment. However, in immunocompetent patients, pulmonary cryptococcosis can resolve spontaneously, without the need for antifungal treatment.
There is no characteristic radiological profile, although the presence of single or multiple nodules that are well defined and not calcified is more common and can mimic neoplasia on the X-ray.(5,9) Radiographic alterations have been reported, including infiltrates (62%), nodules (38%), masses (19%), cavitations (14%) and pleural effusion (3%), without anatomical distribution patterns and without any accompanying hilar or mediastinal lymph node enlargement. Empyema, pneumothorax and pleural involvement suggestive of Pancoast tumor,(13) as well as lung masses invading the chest wall,(14) have also been reported in the literature. A computed tomography scan of the chest corroborates the radiological findings, providing better details for the differential diagnosis,(11) and, when necessary, helps program the invasive diagnostic techniques. In the present case, the patient presented a lung mass that mimicked neoplasia.
There is no consensus in the literature regarding criteria that could be used to distinguish among proven cryptococcal infection, probable cryptococcal infection and colonization only.(1,2,8-9)
Since the clinical presentation of isolated pulmonary cryptococcosis is unspecific and the radiological profile is non-pathognomonic, it is important to clarify the differential diagnosis with other pulmonary mycoses and primary or metastatic lung neoplasias.(4-5)
Cryptococcosis can be diagnosed by the direct study of the fungus in sputum, bronchoalveolar lavage, spinal fluid and histologic samples. It is confirmed by cultures from these samples. C. neoformans can be isolated in most of the routinely used mycological or bacteriological culture media. Spinal tap for cerebrospinal fluid testing should be carried out in cases presenting symptoms suggestive of central nervous system involvement or asymptomatic pulmonary cryptococcosis, with positive serum antigens that indicate its dissemination.(5)
Serologic tests for the detection of Cryptococcus are specific and sensitive (90%). The most common method for the detection of cryptococcal antigen is latex agglutination. A positive test with titers equal to or greater than 1:4 in biological fluid samples strongly suggests infection. There is more extensive clinical experience with the serology testing of cerebrospinal fluid and blood, although detection of the antigen in urine and bronchoalveolar lavage fluid has also been used. Some authors emphasized the fact that any positive test, regardless of the titer obtained, must be carefully considered within the clinical context of the case.(5) Serology still has a prognostic characteristic reported by some authors: high titers (1:1.024) usually reflect a larger fungal population, low immune response and probable treatment failure. Positive serologic results in the serum are associated with systemic dissemination, which increases the risk of evolving to central nervous system involvement, even in immunocompetent patients.(1,5,8,13)
In the present study, the patient, being unable to provide a sputum sample for testing, presented positive direct testing in the bronchoalveolar lavage and in the transbronchial biopsy, with high titers in the serum and urine samples. The patient presented no clinical neurological involvement. However, since there were high titers in the serum serology, she was submitted to spinal tap with direct study and serology of the spinal fluid, the results of both of which were negative. Therefore, she was diagnosed with isolated pulmonary cryptococcosis.
Although complications are rare in the isolated pulmonary form, incapacitating symptoms, fungal meningitis and respiratory insufficiency have been reported, even in immunocompetent patients.(1,6)
Although the treatment for cryptococcal meningitis has been established in the literature, there are no randomized, controlled studies defining the best therapeutic approach for immunocompetent patients diagnosed with isolated pulmonary cryptococcosis. For immunosuppressed patients, symptomatic immunocompetent patients and patients presenting positive serology for Cryptococcus,(8) even for those presenting multiple nodules or extensive infiltrate, treatment is recommended.(1,7,13) Clinical observation is reserved for isolated pulmonary cryptococcosis in asymptomatic immunocompetent patients presenting negative serology. The objectives of the treatment would be to promote rapid clinical resolution and to prevent the development of potentially fatal complications.
Fluconazole (200-400 mg/day) has been used as an initial treatment and continued for 3 to 6 months, according to the radiologic resolution and symptom relief in immunocompetent patients, requiring prolonged follow-up.(1,5,8,13,15) Amphotericin B, due to its toxicity, should only be used in patients with isolated pulmonary cryptococcosis who do not respond to fluconazole, in those who are severely ill and in those presenting central nervous system involvement. Surgical resection of lung masses affected with cryptococcosis has been used as a means of ruling out accompanying malignancy.(5)
We conclude that further prospective controlled studies of isolated pulmonary cryptococcosis are necessary in order to mold a better definition of its natural evolution in immunocompetent patients, as well as to establish guidelines for the therapeutic approach to this disease.
REFERENCES
Submitted: 25 August 2005. Accepted, after review: 24 October 2005
- 1. Nadrous HF, Antonios VS, Terrell CL, Ryu JH. Pulmonary cryptococcosis in nonimmunocompromised patients. Chest. 2003;124(6):2143-7. Comment in: Chest. 2003;124(6):2049-51.
- 2. Vilchez RA, Linden P, Lacomis J, Costello P, Fung J, Kusne S. Acute respiratory failure associated with pulmonary cryptococcosis in non-aids patients. Chest. 2001;119(6):1865-9.
- 3. Sarosi GA. Cryptococcal lung disease in patients without HIV infection. Chest. 1999;115(3):610-1. Comment on: Chest. 1999;115(3):734-40.
- 4. Fraser RS, Colman N, Muller NL, Paré PD. Fungi and actinomyces. In: Fraser RS, Muller NL, Colman N, Paré PD. Fraser and Paré´s diagnosis of disease of the chest. 4th ed. Philadelphia: Saunders; 1999. p. 904-9.
- 5. Perfect JR, Casadevall A. Cryptococcosis. Infect Dis Clin North Am. 2002;16(4):837-74, v-vi.
- 6. Kerkering TM, Duma RJ, Shadomy S. The evolution of pulmonary cryptococcosis: clinical implications from a study of 41 patients with and without compromising host factors. Ann Intern Med. 1981;94(5):611-6.
- 7. Nunez M, Peacock JE Jr, Chin R Jr. Pulmonary cryptococcosis in the immunocompetent host. Therapy with oral fluconazole: a report of four cases and a review of the literature. Chest. 2000;118(2):527-34.
- 8. Aberg JA, Mundy LM, Powderly WG. Pulmonary cryptococcosis in patients without HIV infection. Chest. 1999;115(3):734-40. Comment in: Chest. 1999;115(3): 610-1.
- 9. Chen KY, Ko SC, Hsueh PR, Luh KT, Yang PC. Pulmonary fungal infection: emphasis on microbiological spectra, patient outcome, and prognostic factors. Chest. 2001;120(1):177-84.
- 10. Pappalardo MC, Melhem MS. Cryptococcosis: a review of the Brazilian experience for the disease. Rev Inst Med Trop Sao Paulo. 2003;45(6):299-305.
- 11. Silva ACG, Marchiori E, Souza Jr AS, Irion KL. Criptococose pulmonar: aspectos na tomografia computadorizada. Radiol Bras. 2003;36(5):277-82.
- 12. Holanda MA, Silva COS, Rocha RT, Salgado AR. Criptococose pleural em paciente com síndrome de imunodeficiência adquirida. J Pneumol. 1995;21(4): 201-4.
- 13. Wheat LJ, Goldman M, Sarosi G. State-of-the-art review of pulmonary fungal infections. Semin Respir Infect. 2002;17(2):158-81.
- 14. Mitsuoka S, Kanazawa H. Images in thorax. An unique case of primary pulmonary cryptococcosis with extensive chest wall invasion. Thorax. 2005;60(1):86.
- 15. Saag MS, Graybill RJ, Larsen RA, Pappas PG, Perfect JR, Powderly WG, et al. Practice guidelines for the management of cryptococcal disease. Infectious Diseases Society of America. Clin Infect Dis. 2000;30(4):710-8.
Publication Dates
-
Publication in this collection
15 Mar 2007 -
Date of issue
Oct 2006
History
-
Accepted
24 Oct 2005 -
Received
25 Aug 2005