Abstract
INTRODUCTION: Pseudomonas aeruginosa (P. aeruginosa) is one of the primary opportunistic pathogens responsible for nosocomial infections. Aminoglycosides are an import ant component of antipseudomonal chemotherapy. The inactivation of drugs by modifying enzymes is the most common mechanism of aminoglycoside resistance. OBJECTIVES: The inactivation of aminoglycosides by modifying enzymes is the primary resistance mechanism employed by P. aeruginosa. The aim of the present study was to investigate the occurrence of aminoglycoside resistance and the prevalence of four import ant modifying enzyme genes (aac (6')-I, aac (6')-II, ant (2")-I, aph (3')-VI) in P. aeruginosa in Iran. METHODS: A total of 250 clinical isolates of P. aeruginosa were collected from several hospitals in seven cities in Iran. Antimicrobial susceptibility tests (using the disk diffusion method and E-tests) were performed for all 250 isolates. In addition, all isolates were screened for the presence of modifying enzyme genes by polymerase chain reaction. RESULTS: The resistance rates, as determined by the disk diffusion method, were as follows: gentamicin 43%, tobramycin 38%, and amikacin 24%. Of the genes examined, aac (6')-II (36%) was the most frequently identified gene in phenotypic resist ant isolates, followed by ant (2")-I, aph (3')-VI, and aac (6')-I. CONCLUSIONS: Aminoglycoside resistance in P. aeruginosa remains a signific ant problem in Iran. Therefore, there is considerable local surveillance of aminoglycoside resistance.
Pseudomonas aeruginosa; Antibiotic; Resistance; aac (69)-II; ant (20)-I
CLINICAL SCIENCE
The prevalence of aminoglycoside-modifying enzyme genes (aac (6')-I, aac (6')-II, ant (2")-I, aph (3')-VI) in Pseudomonas aeruginosa
Farzam Vaziri; Shahin Najar Peerayeh; Qorban Behzadian Nejad; Abbas Farhadian
Department of Bacteriology, School of Medical Sciences, Tarbiat Modares University, Tehran, Iran
ABSTRACT
INTRODUCTION: Pseudomonas aeruginosa (P. aeruginosa) is one of the primary opportunistic pathogens responsible for nosocomial infections. Aminoglycosides are an import ant component of antipseudomonal chemotherapy. The inactivation of drugs by modifying enzymes is the most common mechanism of aminoglycoside resistance.
OBJECTIVES: The inactivation of aminoglycosides by modifying enzymes is the primary resistance mechanism employed by P. aeruginosa. The aim of the present study was to investigate the occurrence of aminoglycoside resistance and the prevalence of four import ant modifying enzyme genes (aac (6')-I, aac (6')-II, ant (2")-I, aph (3')-VI) in P. aeruginosa in Iran.
METHODS: A total of 250 clinical isolates of P. aeruginosa were collected from several hospitals in seven cities in Iran. Antimicrobial susceptibility tests (using the disk diffusion method and E-tests) were performed for all 250 isolates. In addition, all isolates were screened for the presence of modifying enzyme genes by polymerase chain reaction.
RESULTS: The resistance rates, as determined by the disk diffusion method, were as follows: gentamicin 43%, tobramycin 38%, and amikacin 24%. Of the genes
examined, aac (6')-II (36%) was the most frequently identified gene in phenotypic resist ant isolates, followed by ant (2")-I, aph (3')-VI, and aac (6')-I. CONCLUSIONS: Aminoglycoside resistance in P. aeruginosa remains a signific ant problem in Iran. Therefore, there is considerable local surveillance of aminoglycoside resistance.
Keywords:Pseudomonas aeruginosa; Antibiotic; Resistance; aac (69)-II; ant (20)-I.
INTRODUCTION
Pseudomonas aeruginosa (P. aeruginosa) is one of the pri mary opportunistic pathogens responsible for nosocomial infection s. The most import ant problem in the eradication of P. aeruginosa is the frequently observed multi-drug resistanc e of the species.1 In addition, P. aeruginosa can also acquire resistance to vario us antimicrobial agents, such as aminoglycosides, β-lactams2 and fluoroquinolones.3 Aminoglycosides are an import ant component of antipseu domonal chemotherapy,4 and they exhibit synergy with β-lactams.5 Resistance to aminoglycosides occurs via enzy matic modification, impermeability, the activity of efflux pumps (MexXY-OprM),6 the PhoP-PhoQ system,7 ndvB dependent biofilm formation,8 and the activity of 16s rRNA methylases.9 Among these mechanisms, the inactivation of drugs b y plasmid- or chromosome-encoded modifying enzymes is the most common. These modifying enzymes include aminoglycoside phosphoryl transferase (aph), ami nogl ycoside acetyltransferase (aac), and aminoglycoside (ant).10-12 nucleoti dyl transferase Four of these enzymes, encoded by aac (6')-I, aac (6')-II, ant (2")-I, and aph (3')-VI, are of particular significance because they are among the most common modifying enzymes present in P. aeruginosa, and their substrates are the most import ant antipseudomonal aminoglycosides. aac (6')-I confers resistance to tobramycin and amikacin, aac (6')-II and ant (2")-I inactivate tobramycin and gentamicin, and amikacin is the substrate of aph (3')-VI.13,14
The aim of the present nationwide study was to investigate the occurrence of aminoglycoside resistance and the prevalence of the resistance-modifying enzyme genes, aac (6')-I, aac (6')-II, ant (2")-I and aph (3')-VI, in P. aeruginosa isolated from several hospitals in seven Iranian cities.
METHODS AND MATERIALS
Collection of bacterial isolates
A total of 250 non-duplicate, clinical isolates of P. aeruginosa were collected from a nationwide distribution of several hospitals in seven cities in Iran (Tehran, Shiraz, Zahedan, Tabriz, Sannandaj, Sari, and Ahvaz) between May 2007 and January 2008. Strain data and the demographic and clinical data for each patient were regularly forwarded to our laboratory. The study population was 62% male and 38% female. The specimens were isolated from urine (38%), wounds (18%), the trachea (18%), blood (10%), sputum (9%), and other sources (7%). Isolate confirmations were conducted using conventional biochemical tests, and then the isolates were stored at 76˚C in glycerol skim milk broth.
Antimicrobial susceptibility testing
Antim icrobial susceptibility tests were performed using the disk diffusion method according to Clinical and Laboratory Standards Institute (CLSI) guidelines15 for three aminoglycosides [gentamicin (10 μg), amikacin (30 μg), and tobramy cin (10 μg)] and for five other antibiotics [imipenem (10 μg), piperacillin (100 μg), ticarcillin (75 μg), ceftazidime (30 μg), and ciprofloxacin (5 μg)]. All drugs were obtained from M ast laboratories (Merseyside, United Kingdom). For all 250 isolates, the minimum inhibitory concentrations (MIC) of amikacin, gentamicin, and tobramycin were determined using the E-test (Biodisk, Dalvagen, Sweden) according to CLSI guidelines. P. aeruginosa (ATCC 27853) served as a control for the disk diffusion and E-tests.
Polymerase chain reaction amplification
Polymerase chain reaction (PCR) was used to screen all 250 isolates for the presence of the modifying enzyme genes, aac (6')-I, aac (6')-II, ant (2")-I and aph (39)-VI. The total template DNA for the PCR amplification was extracted from the supernat ant of a mixture of P. aeruginosa cells produced by the boiling method. PCR amplification was performed using 2.5 μL of the template DNA, 1 mL of each primer,16 19.5 μL master mix, and 1 μL of Taq DNA polymerase (CinnaGen) in a total volume of 25 μL. A thermocycler (Mastercycler gradient; Eppendorf, Hamburg, Germany) was programmed with the appropriate conditions. Then, 5 ml of each PCR product was analyzed by electrophoresis on a 1% (w/v) TAE agarose gel (Fermentas UAB, Vilnius, Lithuania) containing 0.1 μl/ml ethidium bromide. The amplicons were then visualized on a UV transilluminator and photographed (BioDoc-Analyse; Biometra, Goettingen, Germany).
Statistical analysis
All statistical analyses were performed using Statistical Package for Social Sciences (SPSS) software (version 11.5) for Windows (χ 2-test and Fisher's exact test). P-values of ,0.05 were considered significant.
RESULTS AND DISCUSSION
The resistance rates as determined using the disk diffusion method are presented in Figure 1. According to the disk diffusion method, 135 isolates were resist ant to aminogl ycosides. The resistance rates according to the E-test (base on MICs) were as follows: gentamicin 40%, tobramy cin 36%, and amikacin 21%. The aminoglycoside susceptibility profiles according to the disk diffusion and E-test results for the 250 isolates are listed in Table 1.
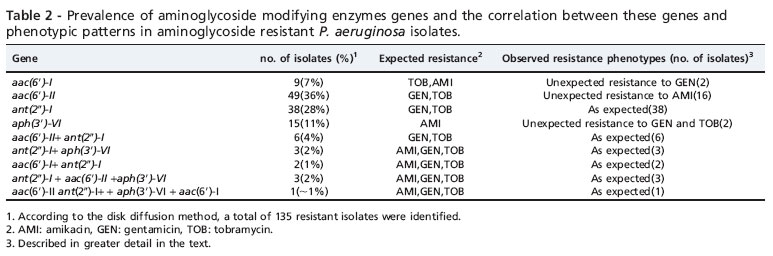
PCR analysis revealed the absence of resistance genes in susceptible isolates. The prevalence of aminoglycoside resistance genes in the 135 resist ant isolates (as determined by the disk diffusion method) was as follows: aac (6')-II was detected in 36% of the resist ant isolates, ant (2")-I was detected in 28%, aph (3')-VI in 11%, and aac (6')-I was found in 7% of the resist ant isolates (Figure 2). Interestingly, individu al aminoglycoside-resist ant isolates carried multi ple (two to four) modifying enzyme genes. Six isolates harbored ant (2")-I and aac(6')-II; three harbored aph(3')-VI and ant (2")-I; two harbored ant (2")-I and aac (6')-I; and three isolates harbored ant (2")-I, aac (6')-II and aph(3')-VI. Only one isolate harbored all four genes.
Several previous studies have examined the occurrence of aminoglycoside resistance mechanisms in P. aeruginosa isolated from different countries. The overall incidence of aminoglycoside resistance found in our study (according to the disk diffusion test and the E-test) was much higher than the incidence that has been reported previously in different countrie s worldwide.14,16,17 However, Estahbanati and co workers reported that 53.3% of clinical isolates from Iranian burn patients were resist ant to amikacin, and 90.7% were resist ant to gentamicin, a result that reveals a high level of aminoglycoside resistance in their study.18
The aminoglycoside resistance rate was almost as high in our isolates, and most of the resist ant isolates harbored modifying enzyme genes. In addition, none of the susceptible isolates harbored these resistance genes. These results highlight the importance of aminoglycoside-modification-related mechanisms in aminoglycoside resistance in P. aeruginosa. Two genes, aac (6')-II and ant (20)-I, were the most frequent resistanc e genes observed in these isolates. These results are similar to what has been observed in different studies in other countries.19-21 All isolates harboring the aac (6')-II gene were resist ant to gentamicin and tobramycin (100% concordance), which indicates that aac (6')-II is a signific ant determin ant of gentamicin and tobramycin resistance in P. aeruginosa.Itis import ant to mention that we encountered an unexpected phenoty pe in some isolates (Table 2). For example, when an isolate harbored only the aph (3')-VI gene, which has amikacin as a substrate, resistance to gentamicin, tobramycin, and amikacin was observed. We presume that the reason for this phe nomenon might be the action of other resistance mechani sms, such as impermeability, efflux pumps, or other types of modifying enzymes. On the other hand, we detected 15 isolates which co-harbored two, three, or four aminoglyco side-modifying enzyme genes simultaneously, which is in contrast to several studies conducted in the USA and Europe, which reported that the majority of isolates exhibit only a single aminoglycoside modifying gene.22
Miller et al. brought together the results of several studies of aminoglycoside resistance in P. aeruginosa carried out worldwide.22 Their results indicated that in Europe, aac(6')II was the most prevalent resistance gene (32.5%), followed by ant(2")-I (16.9%). These results are in concordance with our study. In contrast to our results, a Korean nationwide study of 250 isolates of P. aeruginosa reported that aph(3')-VI, ant(2")-I , and aac(6')-I were all prevalent, but none harbored aac(6')-II.16 The difference in the distribution of modifying enzymes may derive from differences in aminoglycoside prescription patterns, the selection of bacterial population or geographical differences in the occurrence of aminoglycoside resistance genes.
CONCLUSIONS
In conclusion, although aminoglycosides remain useful antipseudomonal agents, resistance to these drugs conti nues to be a major issue, especially in Iran. Because these aminogl ycoside resistance genes are usually located on mobile genetic elements (i.e., plasmid, transposon, or integrons),23,24 there is a growing concern that they could easily spread and be disseminated among other bacteria. Integron s that carry gene cassettes encoding both aacs and carbapenemases will only exacerbate this problem.25 The design of novel aminoglycosides with stronger affinity for their targets and resistance to these modifying enzymes is inevitable,26 and the new generation of anti-Pseudomonas therapy is forthcoming.27 Aminoglycoside resistance among clinical isolates of P. aeruginosa promises to become a major clinical concern in the future, and continuous local surveillance of aminoglycoside resistance is crucial.
ACKNOWLEDGEMENTS
This study was funded by a M.Sc. gr ant from the Tarbiat Modares University, Tehran, Iran.
Received for publication on February 1, 2011; First review completed on March 3, 2011; Accepted for publication on May 16, 2011
E-mail: najarp_s@modares.ac.ir / vaziri@modares.ac.ir. Tel.: 00 98 021 82883870
- 1. Lambert PA. Mechanisms of antibiotic resista nce in Pseudomonas aeruginosa. J R Soc Med. 2002;95(suppl 41):226.
- 2. Park YJ. Prevalence of Ambler class A and D b-lactamases among clinical isolates of Pseudomonas aeruginosain Korea. J Antimicrob Chemother. 2005;56:122-7, doi: 10.1093/jac/dki160.
- 3. Jalal S, Ciofu O, Høiby N, Gotoh N, Wretlind B. Molecular mechanisms of fluoroquinolone resistance in Pseudomonas aeruginosaisolates from cystic fibrosis patients. Antimicrob Agents Chemother. 2000;44:7102.
- 4. Giamarello H. Therapeutic guidelines for Pseudomonas aeruginosa infections. Int J antimicrob Agen. 2000;16:103-6, doi: 10.1016/S0924 8579(00)00212-0.
- 5. Dubois V, Arpin C, Dupart V, Scavelli A, Coulange L, Andre C, et al. blactam and aminoglycoside resistance rates and mechanisms amongPseudomonas aeruginosain French general practice (community and private healthcare centres). J Antimicrob Chemother. 2008;62:316-23, doi: 1 0.1093/jac/dkn174.
- 6. Islam S, Oh H, Jalal S, Karpati F, Ciofu O, Høiby N, et al. Chromosomal mechanisms of aminoglycoside resistance in Pseudomonas aeruginosaisolates from cystic fibrosis patients. Clin Microbiol Infect. 2009;15:606, doi: 10.1111/j.1469-0691.2008.02097.x.
- 7. Macfarlan E, Kwasnicka A, Hancock R. Role of Pseudomonas aeruginosa PhoP-PhoQ in resistance to antimicrobial cationic peptides and aminoglycosides. Microbiol. 2000;146:254354.
- 8. Sadovskay I, Vinogradov E, Li J, Hachani A, Kowalska K, Filloux A. H igh-level antibiotic resistance in Pseudomonas aeruginosabiofilm: the n dvB gene is involved in the production of highly glycerol-phosphory lated b-(1 R3)-glucans, which bind aminoglycosides. Glycobiol. 2 010;20:895904, doi: 10.1093/glycob/cwq047.
- 9. Doi Y, Ghilardi AC, Ad ams J, de Oliveira GD, Paterson DL. High prevalence of metallo-b-lactamase and 16S rRNA methylase coproduction among imipenem-resistantPseudomonas aeruginosaisolates in Brazil. Antimicrob Agents Chemother. 2007;51:338890, doi: 10.1128/ A AC.00443-07.
- 10. Shaw KJ, Rather PN, Hare RS, Miller GH. Molecular genetics aminoglyc oside resistance genes and familial relationship of the aminoglycoside modifying enzymes. Microbiol Rev. 1993;57:13863.
- 11. Smith CA, Baker EN. Aminoglycoside antibiotic resistance by enzymatic deactivation. Curr Drug Targets Infect Disord. 2002;2:14360, doi: 10. 2174/1568005023342533.
- 12. Yamane K, Wachino J, Doi Y, Kurokawa H, Arakawa Y. Global spread of multiple aminoglycoside resistance genes. Emerg Infect Dis. 2005;11: 951-3.
- 13. Miller GH, Sabatelli FJ, Naples L, Hare RS, Shaw KJ. The most frequently occurring aminoglycoside resistance mechanisms combined results of surveys in eight regions of the world. J Chemother. 1995;7(Suppl. 2):1730.
- 14. Poole K. Aminoglycoside resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2005;49:47987, doi: 10.1128/AAC.49.2. 4 79-487.2005.
-
15Clinical and Laboratory Standards Institute. Performance standards for antimicrob ial susceptibility testing; seventeenth informational supple ment. CLSI document M100-S17. CLSI, 2007 Wayne, PA.
- 16. Kim JY, Park YJ, Kwon HJ, Han K, Kang MW, Woo GJ. Occurrence and mechanisms of amikacin resistance and its association with b-lactamases i n Pseudomonas aeruginosa: a Korean nationwide study. J Antimicrob Chemother . 2008;62:479-83, doi: 10.1093/jac/dkn244.
- 17. Cavallo J, Hocquet D, Plesiat P, Fabre R, Roussel-Delvallez M. Susceptibil ity of Pseudomonas aeruginosato antimicrobials: a 2004 F rench multicentre hospital study. J Antimicrob Chemother. 2007;59: 10214, doi: 10.1093/jac/dkm076.
- 18. Estahbanati H, Kashani P, Ghanaatpisheh F. Frequency of Pseudomonas aeruginosaserotypes in burn wound infections and their resistance to antibiotics. Burns. 2002;28:3408, doi: 10.1016/S0305-4179(02)00024-4.
- 19. Busch-Sorensen C, Sonmezoglu M, Frimodt-Moller N, Hojbjerg T, Miller GH, Espersen F. Aminoglycoside resistance mechanisms in Enterobacteriaceae and Pseudomonas spp. from two Danish hospitals: correlation with type of aminoglycoside used. APMIS. 1996;104:7638.
- 20. Over U, Gur D, Unal S, Miller GH. The changing nature of aminoglycoside resistance mechanisms and prevalence of newly recognized resistance mechanisms in Turkey. Clin Microbiol Infect. 2001;7:470-8.
- 21. Phillips I, King A, Shannon K. Prevalence and mechanisms of aminoglyc oside resistance. A ten-year study. Am J Med. 1986;80:4855.
- 22. Miller GH, Sabatelli F J, Hare RS, Glupczynski Y, Mackey P, Shlaes D, et al. The most frequent aminoglycoside resistance mechanisms changes w ith time and geographic area: a reflection of aminoglycoside usage patterns. Clin Infect Dis. 1997;24:S46-62, doi: 10.1093/clinids/24. Supplement_1.S46.
- 23. Park YJ. Aminoglycoside Resistance in Gram-negative Bacilli. Korean J Clin Microbiol. 2009;12:57-61.
- 24. Ramirez MS, Tolmasky ME. Aminoglycoside modifying enzymes. Drug R esist. 2010;13:15171, doi: 10.1016/j.drup.2010.08.003.
- 25. Strateva T, Yordanov D.Pseudomonas aeruginosa a phenomenon of b acterial resistance. J Medic Microbiol. 2009;58:113348, doi: 10.1099/ j mm.0.009142-0.
- 26. Haddad J, Kotra LP, Llano-Sotelo B, Kim C, Azucena EF, Liu M, et al. Design of novel antibiotics that bind to the ribosomal acyltransfer site. J Am Chem Soc. 2002;124:3229-37, doi: 10.1021/ja011695m.
- 27. Page M, Heim J. Prospects for the next anti-Pseudomonas drug. Curr Opin Pharmacol. 2009;9:55865, doi: 10.1016/j.coph.2009.08.006.
Publication Dates
-
Publication in this collection
23 July 2012 -
Date of issue
2011
History
-
Accepted
16 May 2011 -
Reviewed
03 Mar 2011 -
Received
01 Feb 2011