ABSTRACT
INTRODUCTION:
The cochlear implant device has the capacity to measure the electrically evoked compound action potential of the auditory nerve. The neural response telemetry is used in order to measure the electrically evoked compound action potential of the auditory nerve.
OBJECTIVE:
To analyze the electrically evoked compound action potential, through the neural response telemetry, in children with bilateral cochlear implants.
METHODS:
This is an analytical, prospective, longitudinal, historical cohort study. Six children, aged 1-4 years, with bilateral cochlear implant were assessed at five different intervals during their first year of cochlear implant use.
RESULTS:
There were significant differences in follow-up time (p = 0.0082) and electrode position (p = 0.0019) in the T-NRT measure. There was a significant difference in the interaction between time of follow-up and electrode position (p = 0.0143) when measuring the N1-P1 wave amplitude between the three electrodes at each time of follow-up.
CONCLUSION:
The electrically evoked compound action potential measurement using neural response telemetry in children with bilateral cochlear implants during the first year of follow-up was effective in demonstrating the synchronized bilateral development of the peripheral auditory pathways in the studied population.
Keywords:
Child; Cochlear nerve; Cochlear implant; Evoked potentials
RESUMO
INTRODUÇÃO:
O implante coclear tem a capacidade de medir o potencial de ação composto eletricamente evocado do nervo auditivo (ECAP). Para esta verificação utiliza-se uma medida chamada telemetria de respostas neurais.
OBJETIVO:
Analisar o potencial de ação composto evocado eletricamente, por meio da neurotelemetria de respostas neurais, em crianças usuárias de implante coclear bilateral.
MÉTODO:
Trata-se de um estudo analítico, prospectivo, de coorte histórica longitudinal. Foram recrutadas seis crianças, com idades entre de 1-4 anos, usuárias de implante coclear bilateral. Estas crianças foram avaliadas em cinco momentos durante o primeiro ano de uso do implante coclear.
RESULTADOS:
Houve diferença significativa no tempo de acompanhamento (p = 0,0082) e posição do eletrodo (p = 0,0019) na medida de T-NRT. Houve diferença significativa na interação entre tempo de acompanhamento e posição do eletrodo (p = 0,0143) na medida da amplitude das ondas N1-P1 entre os três eletrodos a cada tempo de acompanhamento.
CONCLUSÃO:
A mensuração do ECAP por meio da NRT nas crianças com implante coclear bilateral durante o primeiro ano de acompanhamento foi uma medida importante para apresentar o desenvolvimento bilateral da via auditiva periférica de forma sincronizada nesta população estudada.
Palavras-chave:
Criança; Nervo coclear; Implante coclear; Potenciais evocados
Introduction
The cochlear implant (CI) device is widely accepted and has been considered one of the most important therapeutic options for patients with severe and/or profound bilateral sensorineural hearing loss, in cases who did not achieve satisfactory auditory perception benefits with the use of individual sound amplification device (ISAD). The CIs have been indicated at progressively younger ages due to advances in early audiological diagnosis and new technologies in the CI devices.11. Profant M, Kabatová Z, Simková L. From hearing screening to cochlear implantation: cochlear implants in children under 3 years of age. Acta Otolaryngol. 2008;128:369-72.
Over the last decades, bilateral CI surgery began to be performed, a procedure that can be performed simultaneously or sequentially. The simultaneous technique is used when the patient receives the two internal components in a single surgical procedure, and the sequential one, when the patient receives the two internal components in different surgical procedures.
Patients who require a CI have been increasingly choosing the bilateral procedure. Some studies have shown that these patients benefit from improved speech in the perception with noise22. Litovsky R, Parkinson A, Arcaroli J, Sammeth C. Simultaneous bilateral cochlear implantation in adults: a multicenter clinical study. Ear Hear. 2006;27:714-31. and improved sound location.33. Litovsky RY, Parkinson A, Arcaroli J. Spatial hearing and speech intelligibility in bilateral cochlear implant users. Ear Hear. 2009;30:419-31.and44. Papsin BC, Gordon KA. Bilateral cochlear implants should be the standard for children with bilateral sensorineural deafness. Curr Opin Otolaryngol Head Neck Surg. 2008;16:69-74.
The process known as "Programming" or "Mapping" of the CI speech processor is performed at regular intervals postoperatively. The Mapping process aims to determine the appropriate dynamic range of electrical stimulation for each electrode channel. The dynamic range is the difference between the detection perception threshold (T-level) and loudness - maximum comfort (level C).55. Thai- Van H, Truy E, Charasse B, Boutitie F, Chanal JM, Cochard N, et al. Modeling the relationship between psychophysical perception and electrically evoked compound action poten- tial threshold in young cochlear implant recipients: clinical implications for implant fitting. Clin Neurophysiol. 2004;115: 2811-24.
The measurement of the electrode impedance telemetry can provide an indication of the electrode interface status in the tissues, as well as the appropriate electrode function. Significant changes in these measures can be indicative of changes in the surrounding tissue and/or changes in electrode function. Initial changes in electrode impedance can be expected due to physical changes in the electrode-tissue interface.66. Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000;21: 164-74.
The CI has the capacity to measure the electrically evoked compound action potential (ECAP) of the auditory nerve. The system applies an electrical pulse to a specific intracochlear electrode and the evoked neural response is recorded in an adjacent electrode. A measure called Neural Response Telemetry (NRT) is used to assess this potential. The system elicits a valid neural response and robust recordings. These responses are recorded and returned to the programming interface system for clinical analysis.77. Abbas PJ, Brown CJ, Shallop JK, Firszt JB, Hughes ML, Hong SH, et al. Summary of results using the Nucleus CI24M implant to record the electrically evoked compound action potential. Ear Hear. 1999;20:45-59.and88. Brown CJ, Abbas PJ, Gantz BJ. Preliminary experience with neu- ral response telemetry in the Nucleus CI24M cochlear implant. Am J Otol. 1998;19:320-7.
The ECAP provides a relatively direct measurement of the auditory nerve response after electrical stimulation and it is measured in current units (CUs).99. Lai W. An NRT cookbook: guidelines for making NRT measurements. 1st ed. Zürich: Cochlear AG; 1999. p. 1-44. The ECAP waveform typically consists of an initial negative peak followed by a positive peak, called N1 and P1, respectively.77. Abbas PJ, Brown CJ, Shallop JK, Firszt JB, Hughes ML, Hong SH, et al. Summary of results using the Nucleus CI24M implant to record the electrically evoked compound action potential. Ear Hear. 1999;20:45-59.and88. Brown CJ, Abbas PJ, Gantz BJ. Preliminary experience with neu- ral response telemetry in the Nucleus CI24M cochlear implant. Am J Otol. 1998;19:320-7.
The NRT threshold (T-NRT) is defined as the smallest amount of electric current that can evoke these physiological responses. Studies have shown that the T-NRT measured intraoperatively or at postoperative intervals can be correlated with the psychophysical detection of the threshold (T level) and the maximum level of comfort (C level) in patients with CIs. The amplitude of the response (measured between N1 and P1) varies with increasing stimulus intensity and it is measured in millivolts (µV).1010. Thai- Van H, Chanal JM, Coudert C, Veuillet E, Truy E, Collet L. Relationship between NRT measurements and behavioral levels in children with the Nucleus 24 cochlear implant may change over time: preliminary report. Int J Pediatr Otorhinolaryngol. 2001;58:153-62.,1111. Gordon KA, Ebinger KA, Gilden JE, Shapiro WH. Neural response telemetry in 12- to 24-month-old children. Ann Otol Rhinol Laryngol Suppl. 2002;189:42-8.and1212. Brown CJ, Hughes ML, Luk B, Abbas PJ, Wolaver A, Gervais J. The relationship between EAP and EABR thresholds and levels used to program the nucleus 24 speech processor: data from adults. Ear Hear. 2000;21:151-63.
Considering the abovementioned facts, this study aims to analyze the ECAP through NRT in children who received bilateral cochlear implants. The ECAP will be analyzed in relation to the T-NRT visual threshold and the amplitude of N1-P1 peak during the first year of CI use.
Methods
The study was approved by the Research Ethics Committee (REC) of Faculdade de Ciências da Saúde, Opinion number 571,432/2014. The participants' parents or guardians signed the free and informed consent form (FICF) accepting their participation in the study.
This is an analytical, prospective, longitudinal contemporary cohort study.
Sample
The study included six children, five females and one male. Ages ranged from 1 to 4 years. The children had congenital hearing loss and were Nucleus(r) bilateral cochlear implant users (Cochlear Corporation), having been submitted to simultaneous surgery and total electrode insertion. Data were analyzed between January (2012) and March (2014). Participants with unilateral cochlear implant, auditory neuropathy spectrum disorder (ANSD), partial insertion of the electrode array and those submitted to the sequential technique during internal component surgery was excluded.
Equipment
A speech processor, external antenna (coil) with magnet, connector cable between speech processor and the external antenna, programming interface - POD (Programming POD), and the computer used to send and receive neural information were used to evaluate impedance telemetry and neural responses.
Procedure
The impedance telemetry and neural response recordings were collected through an AutoNRT system, using the software Custom Sound EP 3.2 version to perform intraoperative measurements and Custom Sound 3.2 for postoperative measurements.
The impedance telemetry and NRT were first performed in the operating room, after the internal component insertion and while the child was still anesthetized, and these procedures also continued to be carried out in the postoperative period. During the intraoperative period, the neural responses were recorded in the 22 electrodes; however, for the analysis in this study only the electrodes E1, E11 and E22 were used. Postoperatively, the participants were evaluated at five moments (first, third, sixth and twelfth months) after surgery, and the responses were recorded in the electrodes E1, E11 and E22.
The impedances were measured in the MP1 monopolar, MP2 monopolar, MP1+2 monopolar and Common Ground (CG) modes. Values were considered normal when between 0.7 kO and 30 kO. Electrodes with electrical problems such as short circuits - "short" (<0.7 kO) and open circuits - "open" (>30 kO) were not selected.
The parameters for ECAP recording were: interpulse interval of 400 µs, stimulation rate of 80 Hz with series of 25 µs of pulse width, the number of presentations varied between 100 and 200 pulses per second for amplifier gains in, respectively, 50 dB, the window for recording was 1600 µs. The current level of the masking noise was fixed at 10 units above the stimulation level.
All participants received the same parameters in CI programming in the postoperative period. The parameters used were in accordance with the standardization of Cochlear Corporation: ACE (advanced combination encoder) speech coding strategy, mode of stimulation MP1 + 2, stimulation rate of 900 pulses per second per channel, eight maximas and pulse width of 25 µs.
For each electrode, the characteristics of the T-NRT threshold and peak amplitude of N1-P1 waves were compared between the returns. These measurements were separately compared for: the right and left ear, the intra and postoperative periods and electrode position. Electrode 01 (E01) was called basal, electrode 11 (E11) was called middle and electrode 22 (E22) was called apical. These nominations are in accordance with their positions in the cochlear region.
The "activation" of the CI electrodes occurred within one month after the intraoperative period, which would be the first time the device was activated, thus sending the electrical signal to the auditory nerve through the 22 intracochlear electrodes; after this period, the postoperative measurements were started.
From each measured electrode, only the valid responses were selected, that is, the NRT responses had to be identifiable, even though it is known that responses are increasingly less robust in the electrical threshold.
The answers were analyzed through the automatic software function. The corresponding recording location was chosen for the electrodes to be analyzed in basal, middle and apical form (E1, E11 and E22 electrodes). When this was not possible, that is, when these electrodes did not respond or when the children showed some discomfort, the closest electrodes' responses were not considered.
Statistical analysis
The results of this study were analyzed by a mixed-effect model of analysis of variance for repeated measures. Once the measurements of each individual were obtained in both ears, in the three assessed electrodes and during follow-up, a factorial design structure was used in the model.
When the global p-value of any factor was less than 0.05, a Bonferroni correction was used to adjust the multiple tested comparisons. Analyses were performed using the SAS 9.3 software.
Results
Table 1 shows the participants' characterization. All children had profound sensorineural hearing loss, of idiopathic etiology.
Table 2 shows the mean age of participants at the CI surgery and the time of CI use (months). The mean age at surgery was 23.50 months (standard deviation 5.43) and the time of cochlear implant use was 14.67 months (standard deviation 6.50).
The results showed the ECAP response measured by NRT. After that, the responses of two measures used in this study, the visual T-NRT and the amplitude of N1-P1 waves, will be shown.
T-NRT visual measurement
Initially, we adjusted the model of mixed-effect analysis of variance with repeated measures. We evaluated the factors: ear (right and left), time of follow-up between intraoperative and postoperative periods and position of electrodes in the cochlea. All these measures were also correlated with each other. The results are shown in Table 3.
Once we detected significant differences in the time of follow-up (p = 0.0082) and electrode position (p = 0.0019), the analysis was continued by comparing the mean T-NRT values, separately for each factor, using multiple comparisons two by two between the levels of each factor with Bonferroni correction.
Table 4 shows the mean values and standard deviations for the factors: ear, position of the electrodes in the cochlea and time of follow-up between intraoperative and postoperative periods. These factors were assessed separately.
Fig. 1 shows the mean values of the T-NRT threshold for each ear, separately.
Fig. 2 shows the mean values for T-NRT measured in each electrode, separately. To analyze these results, three electrodes were assessed in each ear, at five different times, during the first year of CI use.
Thus, a total of 180 measurements were performed (60 for each electrode). The results obtained were 51 measurements in basal electrodes, 49 in medial electrodes and 55 in apical electrodes, totaling 155 measurements. It is noteworthy that participant 1 was assessed only up to the first three months of CI use, because she had used the device for only 5 months, and thus 12 measurements were lost (2 in electrode E01, 2 in electrode E011 and 2 in electrode E22). Excluding this measurement in this participant, 168 measurements remained. Of this number, 13 measurements showed absence in the NRT measurement.
Table 5 shows multiple comparisons for the electrode factor and p-values with Bonferroni correction.
The mean value of T-NRT in electrode E01 was significantly higher than in electrode E22 (p = 0.0075); the mean value of T-NRT in electrode 11 was significantly higher than in electrode 22 (p = 0.0035), whereas the mean values of T-NRT did not differ significantly between electrode E01 and electrode E11.
Table 6 shows multiple comparisons for the time factor (p-values) with Bonferroni correction.
The mean value of T-NRT intraoperatively was significantly higher than the mean T-NRT values in the first (p = 0.0238) and third (p = 0.0203) months of follow-up. There is no significant difference in other comparisons between times of follow-up (6th month and 12th month).
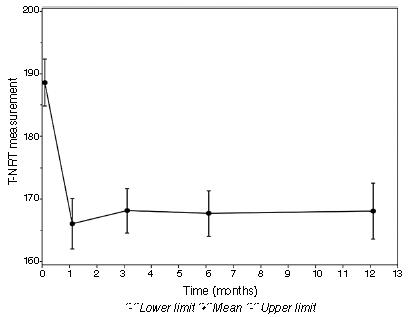
Fig. 3 shows this significant difference of p-value between the intraoperative period and the first and third months of follow-up. At 6 months and 12 months of follow-up, the measured T-NRT values were not significant.
Difference in T-NRT (current units) between the intraoperative and the postoperative follow-up periods.
Measurement of the N1-P1 wave amplitude
In the results of the N1-P1 wave amplitude measurements, initially the adjustment of the model of mixed-effect analysis of variance with repeated measures was carried out. We evaluated the factors: ear (right and left), time of follow-up between intraoperative and postoperative periods and position of electrodes in the cochlea. All these measures were also correlated with each other. The results are shown in Table 7.
Once a significant difference was detected in the interaction between time of follow-up and electrode position (p = 0.0143), the analysis was continued by comparing the mean values of N1-P1 wave amplitude, between the three electrodes at each time of follow-up, with the use of multiple comparisons two by two between the levels of each electrode at each time, with Bonferroni correction.
Table 8 shows the mean values and the respective standard deviations for each electrode and time combination.
Table 9 shows multiple comparisons between the electrodes at each time, adjusted by Bonferroni correction factor.
In the third month of follow-up in the postoperative period, the mean N1-P1 wave amplitude (µV) value in electrode E11 was significantly lower than the mean N1-P1 wave amplitude value in electrode E22 (p = 0.0285).
Fig. 4 shows this significance of the p-value in the 3rd month of follow-up between E11 and E22.
Mean value of N1-P1 wave amplitude (µV) in the third month of follow-up between E11 and E22.
Discussion
While measuring impedance telemetry during the 12 months, normal values were observed for all the assessed electrodes. Thus, it was not necessary to exclude any electrode for the measurements used in this study. These results also showed integrity of the cochlear implant internal component. These data agree with the study by Hughes et al.,66. Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000;21: 164-74. who reported that this measure provides an appropriate indication about the electrode interface status in tissue, as well as the appropriate function of the electrode.
T-NRT visual threshold
In Fig. 1, there was no significant difference between the ears (p = 0.1202) in T-NRT measure and in the measured N1-P1 wave amplitude (p = 0.2429) (Table 7). These findings allow us to infer that bilateral CI with simultaneous surgical technique, for this study population, was effective for synchronized neural stimulation in both ears. The stimulation provided by a cochlear implant is single, and a large electric pulse can promote greater stimulation on neurons for the auditory nerve to respond to the stimulus in a highly synchronized manner, allowing benefits for children with severe hearing loss.1313. Gordon KA, Papsin BC, Harrison RV. Activity-dependent devel- opmental plasticity of the auditory brain stem in children who use cochlear implants. Ear Hear. 2003;24:485-90.
Gordon et al.1414. Gordon KA, Valero J, Hoesel RV, Papsin BC. Abnormal timing delays in auditory brainstem responses evoked by bilateral cochlear implant use in children. Otol Neurotol. 2008;29:193-8. performed a study with EABR (Electrically Evoked Auditory Brainstem Response) and concluded that the reduction of latencies in the waves may reflect an increase in neural synchrony or a more rapid nerve conduction in the brain stem promoted by bilateral stimulation versus unilateral stimulation.
The ECAP through the NRT measure only peripherally assesses the auditory pathway (auditory nerve), but it is important to emphasize that this electrical stimulation triggered in the auditory pathway is one of the precursors to make auditory information effectively arrive at the auditory cortex.1010. Thai- Van H, Chanal JM, Coudert C, Veuillet E, Truy E, Collet L. Relationship between NRT measurements and behavioral levels in children with the Nucleus 24 cochlear implant may change over time: preliminary report. Int J Pediatr Otorhinolaryngol. 2001;58:153-62.
Fig. 2 shows the mean values for T-NRT measurement in each electrode separately. Postoperatively, absences were observed in the recordings of this measure, which can be explained by some discomfort during the procedure (4 measurements) and the remaining by the absence of neural response per se (9 measurements, that is, 4 in electrode E01, 4 in electrode E11 and 1 in electrode E22). These data are similar to other data from literature. 1515. Smoorenburg GF, Willeboer C, van Dijk JE. Speech perception in Nucleus CI24M cochlear implant users with processor settings based on electrically evoked compound action potential thresholds. Audiol Neurootol. 2002;7:335-47. Lai et al.1616. Lai WK, Akist M, Akdas F, Diller N. Longitudinal behaviour of neural response telemetry (NRT) data and clinical implications. Int J Audiol. 2004;43:252-63. state that, over time, the trend of NRT levels improve and generally become stable. These responses to electrical stimulation of electrodes show variations according to their position within the cochlea and the density and integrity of the neural population.1717. Gantz BJ, Brown CJ, Abbas PJ. Intraoperative measurements of electrically evoked auditory nerve compound action potential. Am J Otol. 1994;15:137-44.
In this study, the mean T-NRT measurement (Table 4) during the intraoperative period was 188.64 CUs, whereas in the postoperative period it was 166.03 CUs in the first month; 168.12 CUs in the third month; 167.68 CUs in the sixth month, and 171.50 CUs in the 12th month. The mean value of T-NRT in the intraoperative period was significantly higher than the mean T-NRT values in the first (p = 0.0238) and third (p = 0.0203) months of follow-up. There is no significant difference in other comparisons between periods (6, 9, 12 months).
However, in the studies of Tanamati et al.,1818. Tanamati LF, Bevilacqua MC, Costa AO. Avaliação longitudinal do ECAP registrado em crianças usuárias de implante coclear. Braz J Otorhinolaryngol. 2009;75:90-6. Hughes et al.,66. Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000;21: 164-74. Thai Van et al.,1010. Thai- Van H, Chanal JM, Coudert C, Veuillet E, Truy E, Collet L. Relationship between NRT measurements and behavioral levels in children with the Nucleus 24 cochlear implant may change over time: preliminary report. Int J Pediatr Otorhinolaryngol. 2001;58:153-62. and Muhaimeed et al.1919. Muhaimeed HA, Anazy FA, Hamed O, Shubair E. Correlation between NRT measurement level and behavioral levels in pedi- atrics cochlear implant patients. Int J Pediatr Otorhinolaryngol. 2010;74:356-60. there were no statistically significant differences in the T-NRT threshold during the first year of device programming. Lai et al.1616. Lai WK, Akist M, Akdas F, Diller N. Longitudinal behaviour of neural response telemetry (NRT) data and clinical implications. Int J Audiol. 2004;43:252-63. also found no significant difference in this measure in the first 15 months of device use. In these literature findings, the CI surgery was performed with participants from different age groups. Only in the study of Tanamati et al.1818. Tanamati LF, Bevilacqua MC, Costa AO. Avaliação longitudinal do ECAP registrado em crianças usuárias de implante coclear. Braz J Otorhinolaryngol. 2009;75:90-6. a younger age at surgery was observed, before 3 years, but with no statistical significance. The age at CI surgery (23.50 months) and the bilateral stimulation of the hearing nerve can justify the significant finding during the first three months in our population.
To justify the absence of significant correlation after 3 months of CI use, it should be considered that the NRT measurement only allows an investigation of the neural responsiveness capability in the peripheral auditory system. Thus, one can infer that the first three months were essential for the stimulation of the auditory nerve in the children followed in this study and that the neural response remained in the presence of electrical stimulation after this period. The NRT measurement cannot guarantee that the cognitive mechanisms involved in the auditory perception are activated.1010. Thai- Van H, Chanal JM, Coudert C, Veuillet E, Truy E, Collet L. Relationship between NRT measurements and behavioral levels in children with the Nucleus 24 cochlear implant may change over time: preliminary report. Int J Pediatr Otorhinolaryngol. 2001;58:153-62.
Table 5 shows that the mean value of T-NRT in electrode E01 was significantly higher than in electrode E22 (p = 0.0075); the mean value of T-NRT in electrode E11 was significantly higher than in electrode E22 (p = 0.0035), and the mean values of T-NRT did not differ significantly between electrodes E01 and E11.
These data are similar to those reported by Dees et al.,2020. Cafarelli-Dees D, Dillier N, Lai WK, Wallenberg EV, Dijk BV, Akdas F, et al. Normative findings of electrically evoked action potential measurements using the neural response telemetry of the Nucleus CI24M cochlear implant system. Audiol Neurootol. 2005;10:105-16. Vlahovic et al.2121. Vlahovic S, Sindija B, Aras I, Gluncic M, Trotic R. Differences between electrically evoked compound action potential (ECAP) and behavioral measures in children with cochlear implants operated in the school age vs, operated in the first years of life. Int J Pediatr Otorhinolaryngol. 2012;76:731-9. and Brown et al.,2222. Brown CJ, Abbas PJ, Etler CP, O'Brien S, Oleson JJ. Effects of long-term use of a cochlear implant on the electrically evoked compound action potential. J Am Acad Audiol. 2010;21:5-15. who observed a significant effect between the apical and basal electrodes. Considering the literature findings correlated to the present study data, we can assume that chronic stimulation of the low-frequency cochlear region during the preoperative period may have contributed to improve neuronal survival and/or improve neuronal synchrony of peripheral neurons in the apical portion of the cochlea, or that performing hearing rehabilitation preoperatively, and the activation and development of the central auditory pathways contribute to the functional maintenance of spiral ganglion cells in the apical portion of the cochlea.2121. Vlahovic S, Sindija B, Aras I, Gluncic M, Trotic R. Differences between electrically evoked compound action potential (ECAP) and behavioral measures in children with cochlear implants operated in the school age vs, operated in the first years of life. Int J Pediatr Otorhinolaryngol. 2012;76:731-9.
N1-P1 peak amplitude
In Table 7, the time of CI use and the electrode position were correlated, and there was a significant difference (p = 0.0143) in the N1-P1 amplitude with these factors. Gordon et al.1313. Gordon KA, Papsin BC, Harrison RV. Activity-dependent devel- opmental plasticity of the auditory brain stem in children who use cochlear implants. Ear Hear. 2003;24:485-90. also found significance in the increase of the N1 peak amplitude in the ECAP and in the V wave in EABR, during the time of CI use. Tanamati et al.18 evaluated the first year of CI use and found that in all electrodes there was an increase in the N1 peak amplitude between the second and third returns.
According to these results of the N1-P1 peak amplitude in ECAP, it can be assumed that changes in the ECAP amplitude suggest an improvement in neural synchrony in the primary auditory nerve during the time of CI use. A greater synchronization could, in theory, be due to a change in the way the stimulation reaches and activates the primary nerve fibers and/or a reduced variation in the onset of neuronal response times. The increase in wave amplitude suggests, therefore, that the continuous stimulation activates more neurons, and thus a greater number can be recruited to establish a response with synchrony.1313. Gordon KA, Papsin BC, Harrison RV. Activity-dependent devel- opmental plasticity of the auditory brain stem in children who use cochlear implants. Ear Hear. 2003;24:485-90.
Table 9 and Fig. 4 show that, in the third month of postoperative follow-up, the mean value of N1-PI wave amplitude (µV) in electrode E11 was significantly lower than the mean N1-P1 wave amplitude in electrode E22 (p = 0.0285). There were no significant differences for other comparisons between the electrodes at each time period.
Gordon et al.,2323. Gordon KA, Ebinger KA, Gilden JE, Shapiro WH. Neural response telemetry in 12- to 24-month-old children. Ann Otol Rhinol Laryngol. 2002;189:42-8. Hughles et al.66. Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000;21: 164-74. and Vlahovic et al.2121. Vlahovic S, Sindija B, Aras I, Gluncic M, Trotic R. Differences between electrically evoked compound action potential (ECAP) and behavioral measures in children with cochlear implants operated in the school age vs, operated in the first years of life. Int J Pediatr Otorhinolaryngol. 2012;76:731-9. also found higher amplitude of ECAP in the apical electrodes than in the medial and basal electrodes. These results suggest that electrical stimulation of the basal electrodes may involve a smaller number of spiral ganglion cells, when compared to stimulation in the intermediate or apical electrodes.2323. Gordon KA, Ebinger KA, Gilden JE, Shapiro WH. Neural response telemetry in 12- to 24-month-old children. Ann Otol Rhinol Laryngol. 2002;189:42-8. The amplitudes reflect the sum of the neuron activities, that is, the number of neurons that respond to a stimulus, which in turn indicate better preservation of neuronal function.2121. Vlahovic S, Sindija B, Aras I, Gluncic M, Trotic R. Differences between electrically evoked compound action potential (ECAP) and behavioral measures in children with cochlear implants operated in the school age vs, operated in the first years of life. Int J Pediatr Otorhinolaryngol. 2012;76:731-9.
Considering the data shown in this study, we observed that the auditory pathways in implanted children become more efficient during the first year of cochlear implant use, probably because the groups of nerves respond faster and with a greater degree of synchrony. These activity-dependent processes are probably due to increased synaptic networks, and perhaps to an increase in myelination.1313. Gordon KA, Papsin BC, Harrison RV. Activity-dependent devel- opmental plasticity of the auditory brain stem in children who use cochlear implants. Ear Hear. 2003;24:485-90.
Conclusion
Based on the analysis of neural responses in six children evaluated in the first year of use of bilateral cochlear implants, we concluded that the measurement of ECAP by NRT was an important measure to assess the bilateral development of the peripheral auditory pathway in a synchronized manner in this assessed population.
References
-
1Profant M, Kabatová Z, Simková L. From hearing screening to cochlear implantation: cochlear implants in children under 3 years of age. Acta Otolaryngol. 2008;128:369-72.
-
2Litovsky R, Parkinson A, Arcaroli J, Sammeth C. Simultaneous bilateral cochlear implantation in adults: a multicenter clinical study. Ear Hear. 2006;27:714-31.
-
3Litovsky RY, Parkinson A, Arcaroli J. Spatial hearing and speech intelligibility in bilateral cochlear implant users. Ear Hear. 2009;30:419-31.
-
4Papsin BC, Gordon KA. Bilateral cochlear implants should be the standard for children with bilateral sensorineural deafness. Curr Opin Otolaryngol Head Neck Surg. 2008;16:69-74.
-
5Thai- Van H, Truy E, Charasse B, Boutitie F, Chanal JM, Cochard N, et al. Modeling the relationship between psychophysical perception and electrically evoked compound action poten- tial threshold in young cochlear implant recipients: clinical implications for implant fitting. Clin Neurophysiol. 2004;115: 2811-24.
-
6Hughes ML, Brown CJ, Abbas PJ, Wolaver AA, Gervais JP. Comparison of EAP thresholds with MAP levels in the Nucleus 24 cochlear implant: data from children. Ear Hear. 2000;21: 164-74.
-
7Abbas PJ, Brown CJ, Shallop JK, Firszt JB, Hughes ML, Hong SH, et al. Summary of results using the Nucleus CI24M implant to record the electrically evoked compound action potential. Ear Hear. 1999;20:45-59.
-
8Brown CJ, Abbas PJ, Gantz BJ. Preliminary experience with neu- ral response telemetry in the Nucleus CI24M cochlear implant. Am J Otol. 1998;19:320-7.
-
9Lai W. An NRT cookbook: guidelines for making NRT measurements. 1st ed. Zürich: Cochlear AG; 1999. p. 1-44.
-
10Thai- Van H, Chanal JM, Coudert C, Veuillet E, Truy E, Collet L. Relationship between NRT measurements and behavioral levels in children with the Nucleus 24 cochlear implant may change over time: preliminary report. Int J Pediatr Otorhinolaryngol. 2001;58:153-62.
-
11Gordon KA, Ebinger KA, Gilden JE, Shapiro WH. Neural response telemetry in 12- to 24-month-old children. Ann Otol Rhinol Laryngol Suppl. 2002;189:42-8.
-
12Brown CJ, Hughes ML, Luk B, Abbas PJ, Wolaver A, Gervais J. The relationship between EAP and EABR thresholds and levels used to program the nucleus 24 speech processor: data from adults. Ear Hear. 2000;21:151-63.
-
13Gordon KA, Papsin BC, Harrison RV. Activity-dependent devel- opmental plasticity of the auditory brain stem in children who use cochlear implants. Ear Hear. 2003;24:485-90.
-
14Gordon KA, Valero J, Hoesel RV, Papsin BC. Abnormal timing delays in auditory brainstem responses evoked by bilateral cochlear implant use in children. Otol Neurotol. 2008;29:193-8.
-
15Smoorenburg GF, Willeboer C, van Dijk JE. Speech perception in Nucleus CI24M cochlear implant users with processor settings based on electrically evoked compound action potential thresholds. Audiol Neurootol. 2002;7:335-47.
-
16Lai WK, Akist M, Akdas F, Diller N. Longitudinal behaviour of neural response telemetry (NRT) data and clinical implications. Int J Audiol. 2004;43:252-63.
-
17Gantz BJ, Brown CJ, Abbas PJ. Intraoperative measurements of electrically evoked auditory nerve compound action potential. Am J Otol. 1994;15:137-44.
-
18Tanamati LF, Bevilacqua MC, Costa AO. Avaliação longitudinal do ECAP registrado em crianças usuárias de implante coclear. Braz J Otorhinolaryngol. 2009;75:90-6.
-
19Muhaimeed HA, Anazy FA, Hamed O, Shubair E. Correlation between NRT measurement level and behavioral levels in pedi- atrics cochlear implant patients. Int J Pediatr Otorhinolaryngol. 2010;74:356-60.
-
20Cafarelli-Dees D, Dillier N, Lai WK, Wallenberg EV, Dijk BV, Akdas F, et al. Normative findings of electrically evoked action potential measurements using the neural response telemetry of the Nucleus CI24M cochlear implant system. Audiol Neurootol. 2005;10:105-16.
-
21Vlahovic S, Sindija B, Aras I, Gluncic M, Trotic R. Differences between electrically evoked compound action potential (ECAP) and behavioral measures in children with cochlear implants operated in the school age vs, operated in the first years of life. Int J Pediatr Otorhinolaryngol. 2012;76:731-9.
-
22Brown CJ, Abbas PJ, Etler CP, O'Brien S, Oleson JJ. Effects of long-term use of a cochlear implant on the electrically evoked compound action potential. J Am Acad Audiol. 2010;21:5-15.
-
23Gordon KA, Ebinger KA, Gilden JE, Shapiro WH. Neural response telemetry in 12- to 24-month-old children. Ann Otol Rhinol Laryngol. 2002;189:42-8.
-
☆
Please cite this article as: Caldas FF, Cardoso CC, Barreto MASC, Teixeira MS, Hilgenberg AMS, Serra LSM, et al. Analysis of electrically evoked compound action potential of the auditory nerve in children with bilateral cochlear implants. Braz J Otorhinolaryngol. 2016;82: 123-30.
Publication Dates
-
Publication in this collection
Mar-Apr 2016
History
-
Received
31 May 2014 -
Accepted
24 Dec 2014