Abstracts
The purpose of this study was to evaluate the helminth fauna of Steindachnerina insculpta in the Taquari River, a tributary of the Jurumirim reservoir in the state of São Paulo, and to investigate whether some helminth species may act as a bioindicator of environmental impact. The host fish sample was composed of 60 specimens, with 30 individuals sampled in a lotic stretch and 30 in a lentic stretch. The following were found: the monogeneans Anacanthoroides mizelli, Diaphorocleidus kabatai, Urocleidoides sp. and Euryhaliotrema chaoi; the digenean Sphincterodiplostomum musculosum and unidentified metacercariae; the nematode Travnema travnema and unidentified larvae; and the acanthocephalan Gorytocephalus plecostomorum. The total prevalence of monogeneans (z = 3.14; p = 0.002) and A. mizelli (z = 3.879; p ≤ 0.001), as well as the mean abundance of total monogeneans (U = 642.0; p ≤ 0.001) and A. mizelli (U = 623.5; p ≤ 0.001), were higher in the lentic stretch. Steindachnerina insculpta and its monogeneans comprise a potential group that could be used to investigate environmental impact, as demonstrated in this study.
Neotropical fish; teleost; parasites; bioindicator; reservoir
O objetivo desde estudo foi avaliar a helmintofauna de Steindachnerina insculpta no rio Taquari, um tributário do reservatório de Jurumirim, estado de São Paulo, e verificar se alguma espécie pode agir como bioindicadora de impacto ambiental. A amostragem de peixes foi composta de 60 espécimes, com 30 indivíduos amostrados no trecho lótico e 30 no trecho lêntico. Foram encontrados os monogenóides: Anacanthoroides mizelli, Diaphorocleidus kabatai, Urocleidoides sp. e Euryhaliotrema chaoi; o digenético Sphincterodiplostomum musculosum e metacercárias não identificadas, o nematoide Travnema travnema e larvas não identificadas; e o acantocéfalo Gorytocephalus plecostomorum. A prevalência total de monogenóides (z = 3,14; p = 0,002) e A. mizelli (z = 3,879; p ≤ 0,001) e também a abundância média de monogenóides total (U = 642,0; p ≤ 0,001) e A. mizelli (U = 623,5; p ≤ 0,001) foram maiores no trecho lêntico. Steindachnerina insculpta e seus monogenóides compõem um grupo em potencial que pode ser usado para investigar impactos ambientais, como mostrado neste estudo.
Peixes Neotropicais; teleósteos; parasitas; bioindicador; reservatório
Introduction
Steindachnerina insculpta, popularly known as “saguiru”, belongs to the Curimatidae family and has wide geographical distribution in South America (REIS et al., 2003Reis RE, Kullander SO, Ferraris CJ. Check list of the freshwater fishes of South and Central America. Porto Alegre: EdiPUCRS; 2003.). It is especially abundant in the Paranapanema River and is well-adjusted to artificial reservoirs. The main characteristics of this species are: body slightly silver, terminal mouth without teeth, sedentary with external fertilization, no parental care, and iliophagous feeding habit (DUKE ENERGY, 2008; BRANDãO et al., 2009Brandão H, Vidotto-Magnoni AP, Ramos IP, Carvalho ED. Assessment of the ichthyofauna in stretches under the influence of Salto Grande Reservoir (Middle Paranapanema River, SP/PR, Brazil). Acta Limnol Bras 2009; 21(4): 451-463.).
The construction of dams is one of the most damaging activities in the
Parana river-basin, affecting the main course of the river (AGOSTINHO et al.,
2008). Fish assemblages suffer drastically due to the impact of reservoirs,
which leads to predominance of generalist fish species (FREEMAN et al.,
2001), depletion of migratory species (BRITTO; CARVALHO, 2006Britto SGC, Carvalho ED. Ecological attributes of fish fauna in the
Taquaruçu Reservoir, Paranapanema River (Upper Paraná, Brazil):
composition and spatial distribution. Acta Limnol Bras 2006; 18(4):
377-388.) and transference of pathogens and parasites
(GABRIELLI; ORSI, 2000Gabrielli MA, Orsi ML. Dispersão de Lernea cyprinacea
(Linnaeus) (Crustacea, Copepoda) na região norte do estado
do Paraná, Brasil. Rev Bras Zool 2000; 17(2): 395-399.
http://dx.doi.org/10.1590/S0101-81752000000200010
http://dx.doi.org/10.1590/S0101-81752000...
).
Considering the impact of reservoirs on fish abundance, the prevalence and size of
fish parasite infracommunities may also be influenced (PAVANELLI; TAKEMOTO,
2000). The Taquari River is the second biggest tributary of the Jurumirim
reservoir (HENRY; NOGUEIRA, 1999Henry R, Nogueira MG. A Represa de Jurumirim (São Paulo):
Primeira síntese sobre o conhecimento limnológico e uma proposta
preliminar de manejo ambiental. In: Henry R. Ecologia de reservatórios:
estrutura, função e aspectos sociais. Botucatu: Fapesp/Fundibio;
1999. p. 651-685.)
and is one of the main tributaries of the Paranapanema River, which has been
significantly affected by the creation of the reservoir. There is strong local
interest in artisanal fishing in the Taquari River because of the great diversity
and abundance of fish. To date, there is no record of parasitological studies in
this river.
Fish parasites are considered to be bioindicator species since they can
reflect environmental impacts through their response to habitat alterations, such as
changes in physiology, chemical composition and prevalence or intensity (VIDAL-MARTÍNEZ et al., 2010Vidal-Martínez VM, Pech D, Sures B, Purucker T, Poulin R. Can
parasites really reveal environmental impact? Trends Parasitol 2010;
26(1): 44-51. PMid:19945346.
http://dx.doi.org/10.1016/j.pt.2009.11.001
http://dx.doi.org/10.1016/j.pt.2009.11.0...
). In
this context, monogeneans of fish have been used to assess environmental impacts,
since they have been shown to be suitable indicators (MADI; UETA, 2009Madi RR, Ueta MT. O papel de Ancyrocephalinae (Monogenea:
Dactylogyridae), parasito de Geophagus brasiliensis (Pisces:
Cichlidae), como indicador ambiental. Rev Bras Parasitol Vet 2009;
18(2): 38-41.
http://dx.doi.org/10.4322/rbpv.01802008
http://dx.doi.org/10.4322/rbpv.01802008...
). Steindachnerina
insculpta is a host that fits the criteria proposed by Overstreet (1997)Overstreet RM. Parasitological data as monitors of environmental
health. Parassitologia 1997; 39(3): 169-175.
PMid:9802064. and Sures (2004)Sures B. Environmental parasitology: relevancy of parasites in
monitoring environmental pollution. Trends Parasitol 2004; 20(4):
170-177. PMid:15099556.
http://dx.doi.org/10.1016/j.pt.2004.01.014
http://dx.doi.org/10.1016/j.pt.2004.01.0...
to serve as a
bioindicator fish species: restricted home range, parasites needing more than one
host in the life cycle, easy to sample, small size and wide geographical
distribution.
Thus, the aim of this study was to assess the helminth fauna of S. insculpta in the Taquari River, São Paulo, and investigate whether some helminth species in the parasite community are potential bioindicator of environmental impacts.
Materials and Methods
Study area
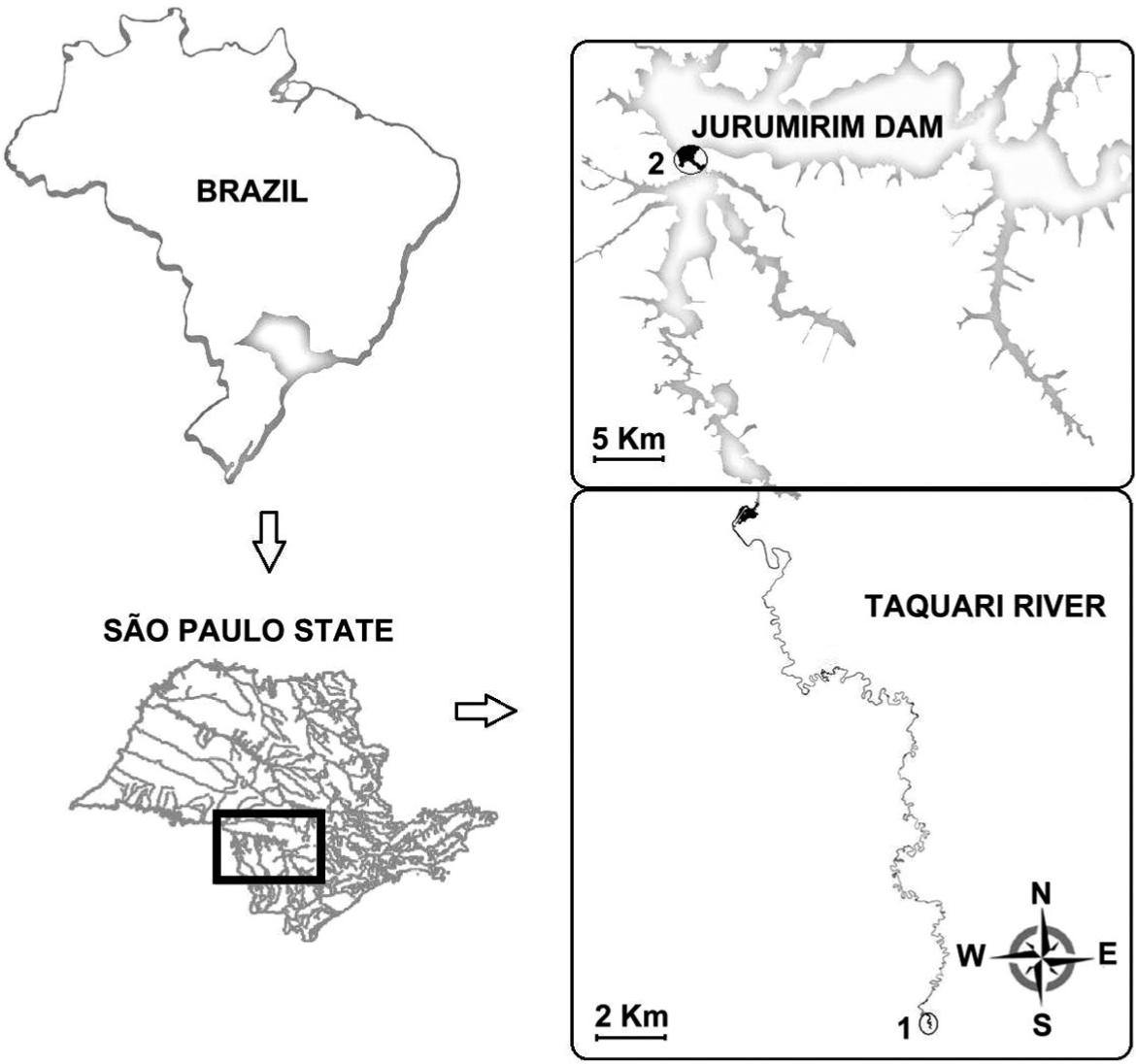
The Jurumirim reservoir constitutes a hydric complex formed by a water body with a surface area of 449 km2 and three main tributaries: Paranapanema, Veados and Taquari rivers. The Taquari River (Figure 1) is in confluence with the left bank of the Upper Paranapanema River, located in a tropical region in the southeastern portion of the state of São Paulo (23° 15′ 11,9″ S and 49° 12′ 34,2 ″W). The fluctuations of the water level are determined either by natural events (rainy and dry seasons) or artificial events (the reservoir operation system).
Map of the location of the Taquari River, a tributary of the Jurumirim Reservoir, state of São Paulo, Brazil: 1: upper stretch (lotic); 2: reservoir stretch (lentic).
Steindachnerina insculpta specimens were sampled in two stretches: a lotic site (upper stretch) located upstream (23° 40′ 2.90″ S and 49° 7′ 56.85″ W) with riparian forest, swamps and macrophytes; and a lentic site (reservoir stretch) located at the river mouth, flooded by the Jurumirim reservoir and located approximately 10 km away from the dam (23° 17′ 2.80″ S and 49° 12′ 6.90″ W). At the lentic site, there is an absence of riparian forest and macrophytes, and the banks are occupied by pasture lands, agricultural activity, and sand extraction activities. The physicochemical characteristics of the lentic and lotic stretches are presented in Table 1.
Mean values and standard deviations of the limnological parameters of the water surface for the two stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil.
Field and laboratory procedures
The fish were sampled in November and December 2011, using gill nets exposed for approximately 14 hours in two distinct areas: the upper stretch of the Taquari River (lotic site) and the reservoir stretch of the Taquari River (lentic site). The fish sample was composed of 60 specimens, with 30 specimens sampled in the lotic stretch, and 30 in the lentic stretch. After sampling, the fish were placed in individual plastic bags, frozen and taken to the laboratory for parasitological analyses. The standard length in centimeters (Ls) and total weight in grams (Wt) of the fish were measured using an ichthyometer and a precise scale in centigrams.
Fish voucher specimens were deposited in the Fish Biology and Genetics Laboratory, Institute of Biosciences (LBP 13313), Universidade Estadual Paulista (UNESP), municipality of Botucatu, state of São Paulo, Brazil.
Parasitological analyses
The body, fins, nasal cavity, eyes and inner face of the operculum of the fish hosts were examined to find ectoparasites. The gills were subsequently removed and washed using 53 and 75 micrometer sieves and placed in Petri dishes. The parasites were collected using a stereomicroscope. After the external analyses, a longitudinal incision in the ventral surface was made and all inner organs were removed and separated. The visceral cavity and all organs were examined using a stereomicroscope to locate endoparasites. All helminths collected were preserved in 70% alcohol. For species identification, the monogeneans were cleared with Hoyer or Grey & Wess to view the sclerotized structures; the digeneans were stained with carmine and cleared with creosote; and the nematodes were diaphanized with lactophenol (EIRAS et al., 2006Eiras JC, Takemoto RM, Pavanelli GC. Métodos de estudo e técnicas laboratoriais em parasitologia de peixes. 2nd ed. Maringá: Eduem; 2006.).
The parasites were identified based mainly on identification keys and reference guides and were analyzed using the Qwin Lite 3.1 (Leica) computerized system for image analysis. Representatives of the helminth species were deposited in the Helminthological Collection of the Institute of Biosciences (CHIBB), UNESP, municipality of Botucatu, state of São Paulo, Brazil.
Statistical analysis
The ecological terminology, prevalence, infection/infestation
mean intensity, and mean abundance followed Bush
et al. (1997)Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets
ecology on its own terms: Margolis et al. revisited. J Parasitol 1997;
83(4): 575-583. PMid:9267395.
http://dx.doi.org/10.2307/3284227
http://dx.doi.org/10.2307/3284227...
. The infracommunity components were
classified in accordance with Bush and Holmes
(1986)Bush AO, Holmes JC. Intestinal helminths of lesser scaup ducks: an
interactive community. Can J Zool 1986; 64(1): 142-152.
http://dx.doi.org/10.1139/z86-023
http://dx.doi.org/10.1139/z86-023...
into core species (prevalence higher than
66.6%), secondary species (prevalence between 33.3% and
66.6%) and satellite species (prevalence less than
33%).
To determine the ecological attributes of infracommunities, the Shannon-Wiener diversity index was calculated: H′= -Σ(pi.In pi). This was used to measure the order or disorder in a system, by attributing greater weight to rare species, and was relatively independent of sample size. In the formula, H′ = species diversity; pi = proportion of species i in the community, in which pi = (ni/N); ni = individual numbers of i species and N = total number of individuals (KREBS, 1989Krebs CJ. Ecological methodology. New York: Harper & Row; 1989.; BEGON et al., 2007Begon M, Townsend CR, Harper JL. Ecologia de Indivíduos a Ecossistemas. 4th ed. Porto Alegre: Artmed; 2007.). Pielou's evenness, which is a derivate from the Shannon-Wiener index, was determined using the following formula: (J′=H′/Log2S), in which J′= evenness (ranging from 0 to 1) (KREBS, 1989Krebs CJ. Ecological methodology. New York: Harper & Row; 1989.).
Similarity analysis on parasites communities between the stretches studied was carried out using the Jaccard coefficient, which is based on the presence or absence of the local species sampled. This index ranged from 0 (dissimilar) to 1 (similar) and was calculated from the equation (Q = c/(a + b – c). 100), in which Q = Jaccard similarity coefficient; c = number of species in common between a and b; a = number of species sampled in a; and b = number of species sampled in b (KREBS, 1989Krebs CJ. Ecological methodology. New York: Harper & Row; 1989.).
The discrepancy index (D), described by Poulin (1993)Poulin R. The disparity between observed and uniform distributions
– A new look at parasite aggregation. Int J Parasitol 1993;
23(7): 937-944.
http://dx.doi.org/10.1016/0020-7519(93)90060-C
http://dx.doi.org/10.1016/0020-7519(93)9...
, was used to
evaluate the spatial distribution of parasites based on their abundance between
lotic and lentic stretches. This index ranges from 0 to 1, and can be
interpreted as: D = 0, all hosts harboring the same number of parasites;
D = 1, all parasites found in a single host. This analysis was calculated
in accordance with Rózsa et al.,
(2000)Rózsa L, Reiczigel J, Majoros G. Quantifying parasites in
samples of hosts. J Parasitol 2000; 86(2): 228-232.
PMid:10780537. using the Quantitative Parasitology 3.0
software.
The prevalence of parasites in the lotic and lentic stretches was compared using Z-test calculations. The mean intensity of infection/infestation and mean abundance between the two stretches were calculated using the Mann-Whitney U test. Principal component analysis (PCA) was carried out to compare parasite communities in the lotic and lentic stretches.
All values that corresponded to the variable mean were followed by their respective standard error. Statistical comparisons were made using the SigmaStat 3.1 software. The multivariate analysis was calculated using the MVSP software. The statistical significance level used was P < 0.05.
Results
Parasite communities
In the lotic stretch, all hosts analyzed were parasitized by at least one helminth species (total prevalence = 100%). A total of 1,527 parasites were collected, with a mean of 50.9 parasite/fish. The richness varied from 1 to 4 parasites, and the mean richness was 2.7 parasites/host. Nine helminth parasite taxa were found: the monogenean species Anacanthoroides mizelli (CHIBB 089L; 090L), Diaphorocleidus kabatai (CHIBB 091L), Urocleidoides sp. (CHIBB 093L; 094L) and Euryhaliotrema chaoi (CHIBB 092L); metacercariae of Sphincterodiplostomum musculosum (CHIBB 7064) and another digenean metacercaria that could not be identified; the nematode species Travnema travnema (CHIBB 7066) and unidentified larvae (CHIBB 7067); and the acanthocephalan Gorytocephalus plecostomorum (CHIBB 7065) (Table 2).
Number of parasites (N); prevalence (P); mean intensity of infection/infestation (MII ± SE); mean abundance (MA ± SE) and infection/infestation sites (IS) of the helminth parasites of Steindachnerina insculpta collected in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil.
Metacercariae of S. musculosum and the acanthocephalan G. plecostomorum were classified as core species. However, only metacercariae of S. musculosum showed high abundance and mean intensity of infection. All the other taxa were classified as satellite species, with low rates of abundance and mean intensity of infection/infestation (Table 2).
In the lentic stretch, all hosts analyzed were parasitized by at least one helminth species (total prevalence = 100%). A total of 1,372 parasites were collected, with a mean of 45.7 parasites/fish. The richness varied from 2 to 5 parasites with a mean richness of 3.3 parasites/host. Seven helminth parasite taxa were found: the monogenean species A. mizelli and D. kabatai; metacercariae of S. musculosum and another digenean metacercaria that could not be identified; nematode species T. travnema and unidentified larvae; and the acanthocephalan G. plecostomorum (Table 2).
The monogenean A. mizelli, metacercariae of S. musculosum and the acanthocephalan G. plecostomorum were classified as core species, T. travnema was classified as a secondary species and the other taxa were classified as satellite species. Only the metacercariae of S. musculosum showed high rates of abundance and mean intensity of infection (Table 2).
Comparison between lotic and lentic stretches
The total prevalence (z = −1.297; p = 0.194), total abundance and mean intensity of infection/infestation (U = 956.0; p = 0.549) were similar between the stretches. The mean richness was greater in the lentic stretch (U = 756.0; p = 0.019), but the number of species was greater in the lotic stretch.
The prevalences of total monogenean species (z = 3.14; p = 0.002) and A. mizelli (z = 3.879; p ≤ 0.001) (Figure 2), as well as the mean abundance of total monogenean species (U = 642.0; p ≤ 0.001) and A. mizelli (U = 623.5; p ≤ 0.001), were significantly higher in the lentic stretch (Figure 3).
Prevalence of helminth parasites of Steindachnerina insculpta sampled in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil. Caption: Tmono – total monogeneans; Amize - Anacanthoroides mizelli; Dkaba - Diaphorocleidus kabatai; Urocl - Urocleidoides sp.; Echai - Euryhaliotrema chaoi; Smusc - Sphincterodiplostomum musculosum metacercariae; Nim – unidentified metacercariae; Ttrav - Travnema travnema; Nil – unidentified larvae; Gplec - Gorytocephalus plecostomorum. *p < 0.05. Black bars: lotic stretch; White bars: lentic stretch.
Mean abundance of helminth parasites of Steindachnerina insculpta sampled in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil. Caption: Tmono – total monogeneans; Amize - Anacanthoroides mizelli; Dkaba - Diaphorocleidus kabatai; Urocl - Urocleidoides sp.; Echai - Euryhaliotrema chaoi; Smusc - Sphincterodiplostomum musculosum metacercariae; Nim – unidentified metacercariae; Ttrav - Travnema travnema; Nil – unidentified larvae; Gplec - Gorytocephalus plecostomorum. *p < 0.05. Black bars: lotic stretch; White bars: lentic stretch.
The Jaccard index (SJ = 8.0) showed high similarity among the parasite communities of the two stretches. However, the Shannon-Wiener index showed that the parasite communities in the lentic stretch were greater in diversity than in the lotic stretch. Pielou's evenness indexes were low for both stretches, suggesting the existence of a dominant species (Table 3). The Berger-Park index showed that in both stretches, metacercariae of S. musculosum were the dominant species with similar values (Table 4).
Ecological indexes for the parasite communities of Steindachnerina insculpta sampled in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil.
Berger-Parker index for the helminth species of Steindachnerina insculpta sampled in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil.
It was also observed that most of the parasite species showed aggregated distribution patterns for both stretches. Only species that occurred at high prevalence and mean abundance (metacercariae of S. musculosum and the acanthocephalan G. plecostomorum) showed a medium aggregation index (Table 5).
Aggregation index for the helminth species of Steindachnerina insculpta sampled in the lotic and lentic stretches of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil.
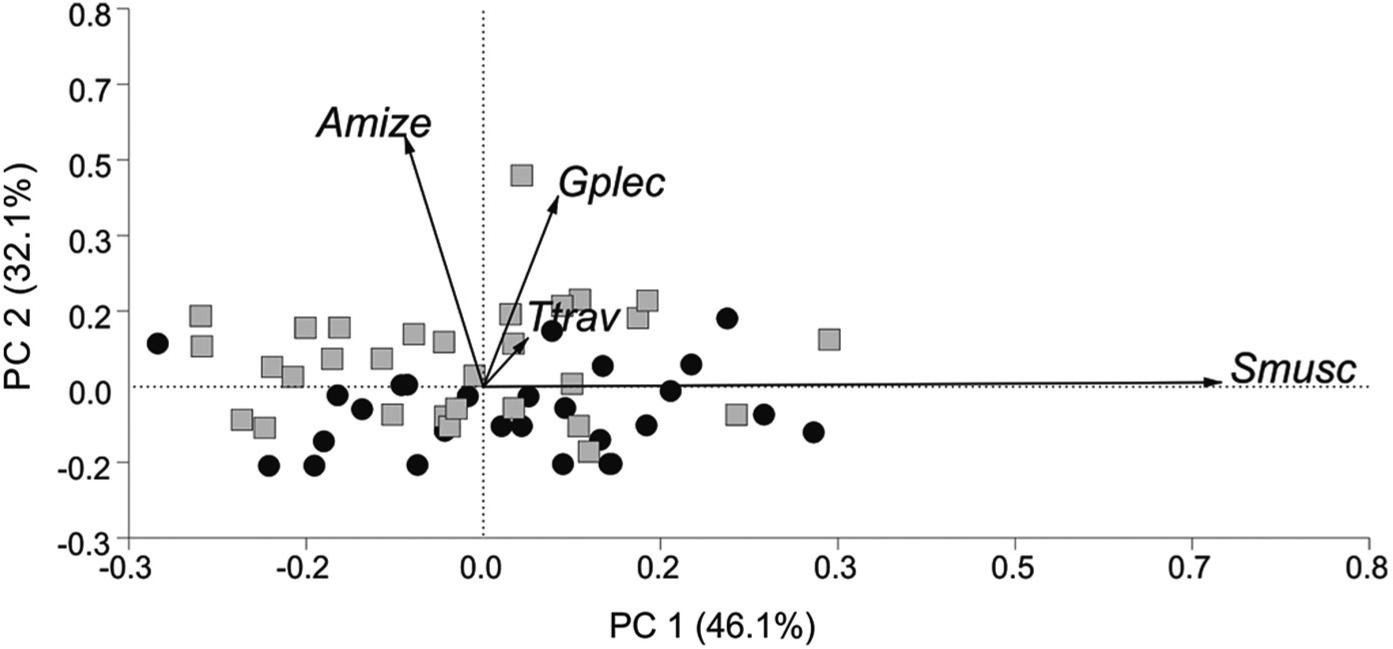
The multivariate analysis based on component communities of helminth parasites of S. insculpta led to the observation that there was relative separation between the specimens from the lotic stretch (mostly distributed in the upper squares) and lentic stretch (mostly distributed in the lower squares). In this analysis, it was observed that A. mizelli, G. plecostomorum and T. travnema were the main species responsible for this separation, in vectorial importance order, respectively (Figure 4).
Scatterplot scores of the principal component analysis (PCA) on parasite communities of Steindachnerina insculpta in the lotic stretch (•) and lentic stretches (▪) of the Taquari River, Jurumirim reservoir, state of São Paulo, Brazil. The values shown on the ordinate and abscissa axes represent the greatest quantity and variation of the data set. Vectorial scale = 0.71. Amize - Anacanthoroides mizelli; Gplec - Gorytocephalus plecostomorum; Smusc - Sphincterodiplostomum musculosum metacercariae; Ttrav - Travnema travnema.
Discussion
Considering the analyses of the limnological parameters, it was possible to confirm that the lentic and lotic stretches of the Taquari River are distinct. Downstream flooding areas are more abruptly affected by impoundments and flood pulses, thus altering the natural process. Additionally, variations in the flood pulse may affect floodplains through altering the water levels that define the limits of riparian vegetation (JUNK et al., 1989Junk WJ, Bayley PB, Sparks RE. The flood pulse concept in river-floodplain systems. Can Spec Publ Fish Aquat Sci 1989; 106: 110-127.).
There have been a few recent studies about the helminth parasite fauna of
S. insculpta. Takemoto et al. (2009) found the
monogenean Urocleidoides sp., metacercariae of
Diplostomum sp. (Digenea: Diplostomatidae) and the
nematodes T. travnema and Cosmoxynema vianai in
S. insculpta samples from the Upper Paraná River floodplain. In
a study on new host and distribution records of nematodes in the Peixe River, state
of São Paulo, Abdallah et al.
(2012)Abdallah VD, Azevedo RK, Carvalho ED, Silva RJ. New hosts and
distribution records for nematode parasites of freshwater fishes from São Paulo
State, Brazil. Neotrop Helminthol 2012; 6(1): 43-57. reported occurrences of C. vianai,
Guyanema sp., Ichthyouris sp., and T.
travnema. Finally, Zago et al.
(2013)Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
reported for the first time metacercariae of S.
musculosum in S. insculpta in the Chavantes reservoir,
state of São Paulo. Furthermore, Ceschini et al.
(2010)Ceschini TL, Takemoto RM, Yamada FH, Moreira LHA, Pavanelli GC.
Endoparasites of Steindachinerina brevipinna (Eigenmann and Eigenmann,
1889), collected in the tributaries Corvo and Guairacá of Paranapanema
River, Paraná State, Brazil. Acta Sci Biol Sci 2010; 32(2):125-130.
http://dx.doi.org/10.4025/actascibiolsci.v32i2.4102
http://dx.doi.org/10.4025/actascibiolsci...
studied the endoparasite fauna of the congeneric species
Steindachnerina brevipinna sampled in the Paranapanema River,
state of Paraná, Brazil, and found metacercariae of S. musculosum
and the nematode T. travnema.
The helminth parasites found in S. insculpta are generalists and had been already reported in other freshwater fish hosts (Table 6). The monogeneans A. mizelli, D. kabatai and E. chaoi, and the acanthocephalan G. plecostomorum found in the present study are new host records for S. insculpta. Nevertheless, all the records found represent new geographical distribution for these parasites, given that there were no previous parasitological studies conducted in the Taquari River, São Paulo. One monogenean specimen found was identified only at genus level (Urocleidoides), due to difficulties in viewing the structures, and thus it was not possible to assess host-parasite interactions for it.
Metacercariae of S. musculosum were the dominant species
in the lotic and lentic stretches, occurring in 100% of the hosts, and at
high abundance. Zago et al. (2013)Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
reported lower prevalence (96.67%) for these metacercariae than in
the present study. However the mean intensity of infection (96.6 versus
41.5) and mean abundance (93.3 versus 41.5) were higher. Moreover,
Zago et al. (2013)Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
found
S. musculosum also parasitizing the coelomic cavity. Ceschini et al. (2010)Ceschini TL, Takemoto RM, Yamada FH, Moreira LHA, Pavanelli GC.
Endoparasites of Steindachinerina brevipinna (Eigenmann and Eigenmann,
1889), collected in the tributaries Corvo and Guairacá of Paranapanema
River, Paraná State, Brazil. Acta Sci Biol Sci 2010; 32(2):125-130.
http://dx.doi.org/10.4025/actascibiolsci.v32i2.4102
http://dx.doi.org/10.4025/actascibiolsci...
found these
metacercariae in S. brevipinna, but they were in the ovary of the
hosts, with lower prevalence (90.47%) than in the present study,
but with higher infection intensity (52.63 versus 41.5).
Thus far, metacercariae of S. musculosum have been found
in a few fish species (CESCHINI et al.,
2010Ceschini TL, Takemoto RM, Yamada FH, Moreira LHA, Pavanelli GC.
Endoparasites of Steindachinerina brevipinna (Eigenmann and Eigenmann,
1889), collected in the tributaries Corvo and Guairacá of Paranapanema
River, Paraná State, Brazil. Acta Sci Biol Sci 2010; 32(2):125-130.
http://dx.doi.org/10.4025/actascibiolsci.v32i2.4102
http://dx.doi.org/10.4025/actascibiolsci...
; TAKEMOTO et al., 2009; ABDALLAH et
al., 2005Abdallah VD, Azevedo RK, Luque JL. Ecologia da comunidade de
metazoários parasitos do sairú Cyphocharax gilbert (Quoy e Gaimard,
1824) (Characiformes: Curimatidae) do Rio Guandu, estado do Rio
de Janeiro, Brasil. Rev Bras Parasitol Vet 2005; 14(4): 154-159.
PMid:16445872.). In the present study, the Berger-Parker index showed
that this digenean was the dominant species in S. insculpta,
corroborating the findings of Zago et al.
(2013)Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
and suggesting that this fish species is highly
susceptible to infections with this parasite. Moreover, this parasite species was
found in the larval stage, suggesting that S. insculpta is in an
intermediate position in the food chain and that it might be part of the diet of
some piscivorous birds that are the definitive hosts of S.
musculosum (ZAGO et al.,
2013Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
).
All the specimens of metacercariae of S. musculosum
found in the present study were in the eyes of S. insculpta. A fish
parasitized with approximately 40 metacercariae is likely to have cataracts or even
blindness, depending on the size of the fish (EVANS et al., 1976 apud ZAGO et al., 2013Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
). Thus, high infection
rates by these metacercariae in the eyes reinforce this information, since the fish
are weakened and become easy prey for the definitive host, thereby allowing this
parasite to finish its life cycle (ABDALLAH et
al., 2005Abdallah VD, Azevedo RK, Luque JL. Ecologia da comunidade de
metazoários parasitos do sairú Cyphocharax gilbert (Quoy e Gaimard,
1824) (Characiformes: Curimatidae) do Rio Guandu, estado do Rio
de Janeiro, Brasil. Rev Bras Parasitol Vet 2005; 14(4): 154-159.
PMid:16445872.; ZAGO et al.,
2013Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho
ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae)
infecting Steindachnerina insculpta (Characiformes, Curimatidae) in
the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013;
22(1): 98-103. PMid:24252954.
http://dx.doi.org/10.1590/S1984-29612013000100018
http://dx.doi.org/10.1590/S1984-29612013...
).
Anacanthoroides mizelli and G.
plecostomorum were also found at high prevalence, but low abundance
(2.15 and 3.4, respectively). There is only one record of A.
mizelli in freshwater fish, which was in Prochilodus
reticulatus (KRITSKY; BOEGER,
2002Kritsky DC, Boeger WA. Neotropical Monogenoida. 41. New and
previously described species of Dactylogyridae (Platyhelminthes) from
the gills of marine and freshwater perciform fishes (Teleostei) with
proposal of a new genus and a hypothesis on phylogeny. Zoosystema 2002;
24(1): 7-40.), whose biology is similar to S. insculpta,
since they are both iliophagous species (DUKE ENERGY, 2008). For
G. plecostomorum, there is also only one record in fresh water
fish, which was Plecostomus plecostomus (NICKOL; THATCHER, 1971Nickol BB, Thatcher VE. Two new acanthocephalans from Neotropical
fishes: Neoechinorhynchus prochilodorum sp. n. and Gorytocephalus plecostomorum
gen. et sp. n. J Parasitol 1971; 57(3): 576-581. PMid:5090966.
http://dx.doi.org/10.2307/3277919
http://dx.doi.org/10.2307/3277919...
). This fish is demersal (DUKE
ENERGY, 2008) feeding on the substrate, like iliophagous species. Therefore, as
shown in the present study, there is the possibility that P. reticulatus, P.
plecostomus and S. insculpta may acquire the same
parasite, since they might be ingesting the same intermediate hosts that dwell in
the substrate, and also share the same habitat.
Vidal-Martínez et al.
(2010)Vidal-Martínez VM, Pech D, Sures B, Purucker T, Poulin R. Can
parasites really reveal environmental impact? Trends Parasitol 2010;
26(1): 44-51. PMid:19945346.
http://dx.doi.org/10.1016/j.pt.2009.11.001
http://dx.doi.org/10.1016/j.pt.2009.11.0...
affirmed that bioindicators are species that reflect
environmental impacts. In a review of parasites (helminths, crustaceans and
protozoans) as potential indicators, these authors compiled pertinent data from
1997 to 2008 and grouped some environmental impact variables with parasites that
showed significant interaction. These analyses demonstrated, through field and
laboratory observations, that these interactions between parasites and
eutrophication, pulp-mill effluents, crude oil, PCBs (polychlorinated
biphenyls), pesticides or heavy metals can be positive, negative or null.
Monogeneans are fish ectoparasites with a direct life cycle that are
found on the skin, in the nasal operculum and on the gills of the hosts (THATCHER, 2006Thatcher VE. Aquatic Biodiversity in Latin America Biodiversidade
Acuática en América Latina – Amazon Fish Parasites. 2nd ed. Moscow:
Pensoft; 2006.). Thus, these parasites are
in direct contact with the environment (NACHEV,
2010Nachev M. Bioindication capacity of fish parasites for the
assessment of water quality in the Danube River [Thesis]. Germany:
Universität Duisburg-Essen; 2010.) and are very sensitive to any change in water parameters
(BAYOUMY et al., 2008Bayoumy EM, Osman HAM, El-Bana LF, Hassanain MA. Monogenean
Parasites as Bioindicators for Heavy Metals Status in Some Egyptian Red Sea
Fishes. Global Vet 2008; 2(3): 117-122.).
Therefore, studies on monogeneans as biomarkers have been conducted by taking
different approaches: Siddall et al.
(1997)Siddall R, Koskivaara M, Valtonen ET. Dactylogyrus
(Monogenea) infections on the gills of roach (Rutilus rutilus
L.) experimentally exposed to pulp and paper mill effluent. Parasitology
1997; 114(5): 439-446.
http://dx.doi.org/10.1017/S003118209600875X
http://dx.doi.org/10.1017/S0031182096008...
assessed the effects of exposure to relatively high
concentrations of effluents from a pulp and paper mill on the prevalence, abundance
and distribution of Dactylogyrus spp. on the gills of
Rutilus rutilus that was sampled in Saravesi Lake, Finland.
They found a significant discrepancy between ectoparasite levels and effluent
concentration. Vital (2008)Vital JF. Diversidade de Parasitas de Pygocentrus nattereri
(Kner, 1858) (Characiformes: Characidae) durante o ciclo
hidrológico em um lago de várzea e seu potencial como indicadora de
qualidade ambiental [Dissertação]. Manaus: Universidade Federal
do Amazonas, Instituto Nacional de Pesquisas da Amazônia;
2008.
compared the parasitism indexes of the host Pygocentrus nattereri
sampled in different phases of the hydrological cycle of Piranha Lake, Amazonas,
Brazil, and pointed out that there was a significant difference in intensity of
Dactylogyridae between the rainy and dry seasons. Bayoumy et al. (2008)Bayoumy EM, Osman HAM, El-Bana LF, Hassanain MA. Monogenean
Parasites as Bioindicators for Heavy Metals Status in Some Egyptian Red Sea
Fishes. Global Vet 2008; 2(3): 117-122. studied monogeneans of some fish in the
Gulf of Suez, Red Sea, as bioindicators for heavy metals, and found highly
significant positive relationships between water temperature and prevalence of
monogeneans, as well as between metal concentrations and prevalence. Madi and Ueta (2009)Madi RR, Ueta MT. O papel de Ancyrocephalinae (Monogenea:
Dactylogyridae), parasito de Geophagus brasiliensis (Pisces:
Cichlidae), como indicador ambiental. Rev Bras Parasitol Vet 2009;
18(2): 38-41.
http://dx.doi.org/10.4322/rbpv.01802008
http://dx.doi.org/10.4322/rbpv.01802008...
analyzed the
role of monogeneans belonging to the Ancyrocephalinae family parasitizing
Geophagus brasiliensis, as environmental bioindicators in
reservoirs with distinct trophic characteristics in the state of São Paulo, Brazil
(Juqueri reservoir – eutrophic; Jaguari reservoir –
oligomesotrophic), and showed that variations in the prevalence and intensity
of infection of these parasites were associated with variations in the amount of
suspended material in the water of each reservoir.
It was observed in the present study that the prevalence and mean
abundance of total monogeneans and A. mizelli were significantly
greater in the lentic stretch. This significant difference corroborates previous
studies that reported the use of monogeneans as indicators of environmental impact
(SIDDALL et al., 1997Siddall R, Koskivaara M, Valtonen ET. Dactylogyrus
(Monogenea) infections on the gills of roach (Rutilus rutilus
L.) experimentally exposed to pulp and paper mill effluent. Parasitology
1997; 114(5): 439-446.
http://dx.doi.org/10.1017/S003118209600875X
http://dx.doi.org/10.1017/S0031182096008...
; VITAL, 2008Vital JF. Diversidade de Parasitas de Pygocentrus nattereri
(Kner, 1858) (Characiformes: Characidae) durante o ciclo
hidrológico em um lago de várzea e seu potencial como indicadora de
qualidade ambiental [Dissertação]. Manaus: Universidade Federal
do Amazonas, Instituto Nacional de Pesquisas da Amazônia;
2008.; BAYOUMY et al., 2008Bayoumy EM, Osman HAM, El-Bana LF, Hassanain MA. Monogenean
Parasites as Bioindicators for Heavy Metals Status in Some Egyptian Red Sea
Fishes. Global Vet 2008; 2(3): 117-122.; MADI; UETA,
2009Madi RR, Ueta MT. O papel de Ancyrocephalinae (Monogenea:
Dactylogyridae), parasito de Geophagus brasiliensis (Pisces:
Cichlidae), como indicador ambiental. Rev Bras Parasitol Vet 2009;
18(2): 38-41.
http://dx.doi.org/10.4322/rbpv.01802008
http://dx.doi.org/10.4322/rbpv.01802008...
). Furthermore, this study evaluated the use of these parasites
as environmental indicators for the effect of an impoundment (Jurumirim
dam) on parasite communities, an approach that has not previously been reported
in the literature.
Monogeneans are more easily found in lentic environments, since it is less difficult for their free-swimming larvae to reach the hosts (DOGEL, 1961Dogel VA. Ecology of the parasites of freshwater fishes. In: Dogel VA, Petrushevski GK, Polyanski YI. Parasitology of fishes. Leningrad: University Press; 1961. p. 1-47.). Therefore, this greater number of monogeneans may be due to the decreased water flow in the lentic environment, which enables more exchange of parasites among the hosts.
Steindachnerina insculpta is a fish species with
iliophagous feeding habit that is important for food chain maintenance (REIS et al., 2003Reis RE, Kullander SO, Ferraris CJ. Check list of the freshwater
fishes of South and Central America. Porto Alegre: EdiPUCRS;
2003.). Nevertheless, high
monogenean infestations on the gills may threaten the breathing capacity of the
fish, leading to death. Thus, a reduction in the number of these fish specimens may
bring about impacts for other fish species that feed on S.
insculpta, thus corroborating affirmations about the impacts of
impoundments upon fish assemblages and parasite fauna (GABRIELLI; ORSI, 2000Gabrielli MA, Orsi ML. Dispersão de Lernea cyprinacea
(Linnaeus) (Crustacea, Copepoda) na região norte do estado
do Paraná, Brasil. Rev Bras Zool 2000; 17(2): 395-399.
http://dx.doi.org/10.1590/S0101-81752000000200010
http://dx.doi.org/10.1590/S0101-81752000...
; AGOSTINHO et al., 2007Agostinho AA, Gomes LC, Pelicice FM. Ecologia e Manejo de Recursos
Pesqueiros em Reservatórios do Brasil. Maringá: Eduem;
2007.).
This study contributes particularly towards initial characterization of the helminth fauna of S. insculpta in the Taquari River, in which nine taxa were found, thereby widening the knowledge of the geographical distribution of these parasites. Moreover, this study reports new records for S. inscultpa as a host for the monogeneans A. mizelli, D. kabatai and E. chaoi, and the acanthocephalan G. plecostomorum. Furthermore, this study indicates that the total monogenean and A. mizelli parasitism of S. insculpta could be used as potential bioindicators, due to their greater prevalence and abundance in the lentic gradient.
The authors thank FAPESP (2010/19543-6) and CAPES (AUX-PE-PNPD 3005/2010) for funding the project, and CAPES for the scholarship granted to the first author.
References
- Abdallah VD, Azevedo RK, Luque JL. Metazoários parasitos dos lambaris Astyanax bimaculatus (Linnaeus, 1758), A.parahybae Eigenman, 1908 e Oligosarcus hepsetus (Cuvier, 1829) (Osteichthyes: Characidae), do Rio Guandu, Estado do Rio de Janeiro, Brasil. Rev Bras Parasitol Vet 2004; 13(2): 57-63.
- Abdallah VD, Azevedo RK, Luque JL. Ecologia da comunidade de metazoários parasitos do sairú Cyphocharax gilbert (Quoy e Gaimard, 1824) (Characiformes: Curimatidae) do Rio Guandu, estado do Rio de Janeiro, Brasil. Rev Bras Parasitol Vet 2005; 14(4): 154-159. PMid:16445872.
- Abdallah VD, Azevedo RK, Carvalho ED, Silva RJ. New hosts and distribution records for nematode parasites of freshwater fishes from São Paulo State, Brazil. Neotrop Helminthol 2012; 6(1): 43-57.
- Agostinho AA, Gomes LC, Pelicice FM. Ecologia e Manejo de Recursos Pesqueiros em Reservatórios do Brasil. Maringá: Eduem; 2007.
- Agostinho AA, Pelicice FM, Gomes LC. Dams and the fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz J Biol 2008; 68(4 Suppl): 1119-1132. PMid:19197482. http://dx.doi.org/10.1590/S1519-69842008000500019
» http://dx.doi.org/10.1590/S1519-69842008000500019 - Almeida KSS, Cohen SC. Diversidade de Monogenea (Platyhelminthes) Parasitos de Astyanax altiparanae do Reservatório da Usina Hidrelétrica de Itaipu. Saúde & Amb Rev, Duque de Caxias 2011; 6(1): 31-41.
- Bayoumy EM, Osman HAM, El-Bana LF, Hassanain MA. Monogenean Parasites as Bioindicators for Heavy Metals Status in Some Egyptian Red Sea Fishes. Global Vet 2008; 2(3): 117-122.
- Begon M, Townsend CR, Harper JL. Ecologia de Indivíduos a Ecossistemas. 4th ed. Porto Alegre: Artmed; 2007.
- Brandão H, Vidotto-Magnoni AP, Ramos IP, Carvalho ED. Assessment of the ichthyofauna in stretches under the influence of Salto Grande Reservoir (Middle Paranapanema River, SP/PR, Brazil). Acta Limnol Bras 2009; 21(4): 451-463.
- Britto SGC, Carvalho ED. Ecological attributes of fish fauna in the Taquaruçu Reservoir, Paranapanema River (Upper Paraná, Brazil): composition and spatial distribution. Acta Limnol Bras 2006; 18(4): 377-388.
- Bush AO, Holmes JC. Intestinal helminths of lesser scaup ducks: an interactive community. Can J Zool 1986; 64(1): 142-152. http://dx.doi.org/10.1139/z86-023
» http://dx.doi.org/10.1139/z86-023 - Bush AO, Lafferty KD, Lotz JM, Shostak AW. Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1997; 83(4): 575-583. PMid:9267395. http://dx.doi.org/10.2307/3284227
» http://dx.doi.org/10.2307/3284227 - Ceschini TL, Takemoto RM, Yamada FH, Moreira LHA, Pavanelli GC. Endoparasites of Steindachinerina brevipinna (Eigenmann and Eigenmann, 1889), collected in the tributaries Corvo and Guairacá of Paranapanema River, Paraná State, Brazil. Acta Sci Biol Sci 2010; 32(2):125-130. http://dx.doi.org/10.4025/actascibiolsci.v32i2.4102
» http://dx.doi.org/10.4025/actascibiolsci.v32i2.4102 - Dogel VA. Ecology of the parasites of freshwater fishes. In: Dogel VA, Petrushevski GK, Polyanski YI. Parasitology of fishes. Leningrad: University Press; 1961. p. 1-47.
- Duke Energy. Peixes do rio Paranapanema. 2nd ed. Horizonte Geográfico: São Paulo; 2008.
- Eiras JC, Takemoto RM, Pavanelli GC. Métodos de estudo e técnicas laboratoriais em parasitologia de peixes. 2nd ed. Maringá: Eduem; 2006.
- Freeman MC, Bowen ZH, Bovee KD, Irwin ER. Flow and habitat effects on juvenile fish abundance in natural and altered flow regimes. Ecol Appl 2001; 11(1): 179-190. http://dx.doi.org/10.1890/1051-0761(2001)011[0179:FAHEOJ]2.0.CO;2
» http://dx.doi.org/10.1890/1051-0761(2001)011[0179:FAHEOJ]2.0.CO;2 - Gabrielli MA, Orsi ML. Dispersão de Lernea cyprinacea (Linnaeus) (Crustacea, Copepoda) na região norte do estado do Paraná, Brasil. Rev Bras Zool 2000; 17(2): 395-399. http://dx.doi.org/10.1590/S0101-81752000000200010
» http://dx.doi.org/10.1590/S0101-81752000000200010 - Henry R, Nogueira MG. A Represa de Jurumirim (São Paulo): Primeira síntese sobre o conhecimento limnológico e uma proposta preliminar de manejo ambiental. In: Henry R. Ecologia de reservatórios: estrutura, função e aspectos sociais. Botucatu: Fapesp/Fundibio; 1999. p. 651-685.
- Junk WJ, Bayley PB, Sparks RE. The flood pulse concept in river-floodplain systems. Can Spec Publ Fish Aquat Sci 1989; 106: 110-127.
- Krebs CJ. Ecological methodology. New York: Harper & Row; 1989.
- Kritsky DC, Boeger WA. Neotropical Monogenoida. 41. New and previously described species of Dactylogyridae (Platyhelminthes) from the gills of marine and freshwater perciform fishes (Teleostei) with proposal of a new genus and a hypothesis on phylogeny. Zoosystema 2002; 24(1): 7-40.
- Kritsky DC, Thatcher VE. New monogenetic trematodes from freshwater fishes of Western Colombia with the proposal of Anacanthoroides gen. n. (Dactylogyridae). Proc Helminthol Soc Wash 1976; 43(2): 129-134.
- Luque JL, Aguiar JC, Vieira FM, Gibson DI, Santos CP. Checklist of Nematoda associated with the fishes of Brazil. Zootaxa 2011; 3082: 1-88.
- Madi RR, Ueta MT. O papel de Ancyrocephalinae (Monogenea: Dactylogyridae), parasito de Geophagus brasiliensis (Pisces: Cichlidae), como indicador ambiental. Rev Bras Parasitol Vet 2009; 18(2): 38-41. http://dx.doi.org/10.4322/rbpv.01802008
» http://dx.doi.org/10.4322/rbpv.01802008 - Mendonza-Franco EF, Reina RG, Torchin ME. Dactylogyrids (Monogenoidea) parasitizing the gills of Astyanax spp. (Characidae) from Panama and Southeast Mexico, a new species of Diaphorocleidus and a proposal for Characithecium n. gen. J Parasitol 2009; 95(1): 46-55. PMid:19245277. http://dx.doi.org/10.1645/GE-1592.1
» http://dx.doi.org/10.1645/GE-1592.1 - Nachev M. Bioindication capacity of fish parasites for the assessment of water quality in the Danube River [Thesis]. Germany: Universität Duisburg-Essen; 2010.
- Nickol BB, Thatcher VE. Two new acanthocephalans from Neotropical fishes: Neoechinorhynchus prochilodorum sp. n. and Gorytocephalus plecostomorum gen. et sp. n. J Parasitol 1971; 57(3): 576-581. PMid:5090966. http://dx.doi.org/10.2307/3277919
» http://dx.doi.org/10.2307/3277919 - Overstreet RM. Parasitological data as monitors of environmental health. Parassitologia 1997; 39(3): 169-175. PMid:9802064.
- Pavanelli, GC, Takemoto RM. Aspects of the ecology of proteocephalid cestodes, parasites of Sorubim lima (Pimelodidae), of the Upper Paraná River, Brazil: II. Interspecific associations and distribution of gastrintestinal parasites. Braz J Biol 2000; 60(4): 585-590. PMid:11241956.
- Pereira C. Travnema travnema n. gen. n. sp. (Nematoda: Oxyuridae) parasito de Curimatus elegans (Pisces: Characinidae) no Nordeste Brasileiro. In: Livro Jubilar do Dr. Lauro Travassos. Rio de Janeiro; 1938. v. 3, p. 318-385.
- Poulin R. The disparity between observed and uniform distributions – A new look at parasite aggregation. Int J Parasitol 1993; 23(7): 937-944. http://dx.doi.org/10.1016/0020-7519(93)90060-C
» http://dx.doi.org/10.1016/0020-7519(93)90060-C - Reis RE, Kullander SO, Ferraris CJ. Check list of the freshwater fishes of South and Central America. Porto Alegre: EdiPUCRS; 2003.
- Rózsa L, Reiczigel J, Majoros G. Quantifying parasites in samples of hosts. J Parasitol 2000; 86(2): 228-232. PMid:10780537.
- Siddall R, Koskivaara M, Valtonen ET. Dactylogyrus (Monogenea) infections on the gills of roach (Rutilus rutilus L.) experimentally exposed to pulp and paper mill effluent. Parasitology 1997; 114(5): 439-446. http://dx.doi.org/10.1017/S003118209600875X
» http://dx.doi.org/10.1017/S003118209600875X - Sures B. Environmental parasitology: relevancy of parasites in monitoring environmental pollution. Trends Parasitol 2004; 20(4): 170-177. PMid:15099556. http://dx.doi.org/10.1016/j.pt.2004.01.014
» http://dx.doi.org/10.1016/j.pt.2004.01.014 - Takemoto RM, Pavanelli GC, Lizama MAP, Lacerda ACF, Yamada FH, Moreira LHA, et al. Diversity of parasites of fish from the Upper Paraná River floodplain, Brazil. Braz J Biol 2009; 69(2 Suppl): 691-705. PMid:19738975. http://dx.doi.org/10.1590/S1519-69842009000300023
» http://dx.doi.org/10.1590/S1519-69842009000300023 - Thatcher VE. Aquatic Biodiversity in Latin America Biodiversidade Acuática en América Latina – Amazon Fish Parasites. 2nd ed. Moscow: Pensoft; 2006.
- Vidal-Martínez VM, Pech D, Sures B, Purucker T, Poulin R. Can parasites really reveal environmental impact? Trends Parasitol 2010; 26(1): 44-51. PMid:19945346. http://dx.doi.org/10.1016/j.pt.2009.11.001
» http://dx.doi.org/10.1016/j.pt.2009.11.001 - Vital JF. Diversidade de Parasitas de Pygocentrus nattereri (Kner, 1858) (Characiformes: Characidae) durante o ciclo hidrológico em um lago de várzea e seu potencial como indicadora de qualidade ambiental [Dissertação]. Manaus: Universidade Federal do Amazonas, Instituto Nacional de Pesquisas da Amazônia; 2008.
- Zago AC, Franceschini L, Ramos IP, Zica EOP, Wunderlich AC, Carvalho ED, et al. Sphincterodiplostomum musculosum (Digenea, Diplostomidae) infecting Steindachnerina insculpta (Characiformes, Curimatidae) in the Chavantes Reservoir, Southeastern Brazil. Rev Bras Parasitol Vet 2013; 22(1): 98-103. PMid:24252954. http://dx.doi.org/10.1590/S1984-29612013000100018
» http://dx.doi.org/10.1590/S1984-29612013000100018
Publication Dates
-
Publication in this collection
Oct-Dec 2013
History
-
Received
5 June 2013 -
Accepted
5 Nov 2013