Abstract
An evaluation was made of the effect of anthelmintic treatments on the performance of Simmental X Nellore crossbred calves before and after weaning. To this end, the calves were divided into three groups: (1) treated monthly with a low efficacy anthelmintic drug, ivermectin; (2) treated monthly with a highly effective anthelmintic drug, albendazole; and (3) untreated control group. All the groups in this experiment showed an average fecal egg count of less than 400 eggs per gram (EPG), and no clinical signs of parasitic gastroenteritis. The blood variables were within the normal range and no calf presented anemia. In most of the samplings, mean EPGs were significantly lower (P<0.05) in the group treated with albendazole. The calves received dietary supplementation before and after weaning, which enabled them to gain weight in every month of the experiment and reach a body weight of about 250 kg on the last sampling date, before turning one year old. The anthelmintic treatments did not affect body weight gain, leading to the conclusion that, when fed with suitable dietary supplements, Simmental X Nellore crossbred calves are not affected by gastrointestinal nematode parasites acquired by grazing.
Keywords:
Haemonchus; nutrition; ivermectin; albendazole; cattle
Resumo
O objetivo do experimento foi avaliar o efeito de tratamentos anti-helmínticos no desempenho de bezerros Simental x Nelore antes e após o desmame. Os bezerros foram alocados em três grupos: (1) tratado mensalmente com anti-helmínticos de baixa eficácia, ivermectina; (2) tratado mensalmente com anti-helmíntico de alta eficácia, albendazol e (3) controle não tratado. A média das contagens de ovos de nematoides durante o experimento foi inferior a 400 ovos por grama (OPG) em todos os grupos sem manifestação clínica de gastroenterite parasitária. As variáveis sanguíneas mantiveram-se dentro dos limites de normalidade e nenhum bezerro apresentou anemia. Na maioria das coletas, as médias de OPG foram significativamente inferiores (P<0,05) no grupo tratado com albendazol. Os bezerros receberam suplementação antes e depois do desmame, o que lhes permitiu ganhar peso em todos os meses do experimento, atingindo peso corporal em torno de 250 kg, ao final do experimento, antes de completarem um ano de idade. Não houve efeito dos tratamentos anti-helmínticos no ganho em peso, o que permitiu concluir que bezerros Nelore x Simental não são afetados pelo parasitismo por nematoides gastrintestinais sob condições de pastejo, quando devidamente suplementados com concentrado.
Palavras-chave:
Haemonchus; nutrição; ivermectina; albendazol; bovinos
Introduction
Brazil has more than 214 million heads of cattle ( ABIEC, 2019Associação Brasileira das Indústrias Exportadoras de Carne – ABIEC [online]. São Paulo: ABIEC; 2019 [cited 2019 Jul 9]. Available from: http://www.abiec.com.br/Sumario2019.aspx
http://www.abiec.com.br/Sumario2019.aspx...
), placing the country among the leaders in the sector, with the world’s second largest cattle inventory and the world’s largest beef exporter. However, cattle productivity presents losses due to parasitism caused by flies, ticks and endoparasites, with annual losses of more than 13 billion US dollars ( Grisi et al., 2014Grisi L, Leite RC, Martins JRS, Barros ATM, Andreotti R, Cançado PHD, et al. Reassessment of the potential economic impact of cattle parasites in Brazil. Rev Bras Parasitol Vet 2014; 23(2): 150-156. http://dx.doi.org/10.1590/S1984-29612014042. PMid:25054492.
http://dx.doi.org/10.1590/S1984-29612014...
). The body weight gain of young cattle may be affected by the interaction of several factors, such as genetic background, sanitary conditions and environmental factors, especially those pertaining to nutritional quality ( Pathak, 2017Pathak AK. Nutritional Bases to Control Gastrointestinal Parasites of Livestock. J Dairy Vet Sci 2017; 4(1): 555632. ; Fachiolli et al., 2017Fachiolli DF, Saes IL, Dellaqua JVT, Souza OA, Pinto LD, Santi PF, et al. Anthelmintic treatment and supplementation in Nellore calves performance in the post-weaning period. Semina: Ciênc Agrár 2017; 38(3): 1551-1560. http://dx.doi.org/10.5433/1679-0359.2017v38n3p1541.
http://dx.doi.org/10.5433/1679-0359.2017...
).
Gastrointestinal nematode (GIN) infections play an important role in the losses attributed to parasitic diseases in cattle. In Central Brazil, pioneering investigations into losses due to GIN infections in young Zebu or Zebu X European crossbred cattle were conducted in the 1970s. In Mato Grosso, Melo & Bianchin (1977)Melo HJH, Bianchin I. Estudos epidemiológicos de infecções por nematódeos gastrintestinais de bovinos de corte em zona de cerrado de Mato Grosso. Pesq Agropec Bras 1977; 12: 205-216. showed that Zebu calves strategically treated four times per year (mid-May, mid-July, mid-September and mid-December) with broad-spectrum anthelmintics (tetramisole) gained significantly more weight (43.3±11 kg) than untreated animals. In contrast, in Minas Gerais, Costa et al. (1977)Costa JO, Costa HMA, Guimarães MP, Freitas MG. Efeito de tratamentos anti-helmínticos sobre o OPG e o desenvolvimento ponderal de bezerros. Arq Esc Vet UFMG 1977; 29(2): 171-178. found no difference between the body weight gain of groups of 10-month-old calves treated with tetramisole twice (December and June) or four times a year (December, March, June and September) and that of a untreated control group.
Later, in Central Brazil, young treated cattle showed higher gains in body weight than untreated controls in the states of Mato Grosso do Sul ( Bianchin et al., 1995Bianchin I, Honer MR, Nunes S, Nascimento YA. Effect of stocking rates and anthelminthic treatments on weight gains in weaned Nellore cattle on improved pasture in the Brazilian cerrado. Trop Anim Health Prod 1995; 27(1): 1-8. http://dx.doi.org/10.1007/BF02236326. PMid:7770946.
http://dx.doi.org/10.1007/BF02236326...
, 2007Bianchin I, Catto BJ, Kichel NA, Torres AA, Honer MR. The effect of the control of endo- and ectoparasites on weight gains in crossbred cattle ( Bos taurus taurus × Bos taurus indicus ) in the central region of Brazil. Trop Anim Health Prod 2007; 39(4): 287-296. http://dx.doi.org/10.1007/s11250-007-9017-1. PMid:17847824.
http://dx.doi.org/10.1007/s11250-007-901...
; Borges et al., 2013Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Anthelmintic resistance impact on tropical beef cattle productivity: Effect on weight gain of weaned calves. Trop Anim Health Prod 2013; 45(3): 723-727. http://dx.doi.org/10.1007/s11250-012-0280-4. PMid:23076819.
http://dx.doi.org/10.1007/s11250-012-028...
; Catto et al., 2013Catto JB, Bianchin I, Feijó GLD, Araújo FR, Ramos CAN, Castelão ABC. Weight gain and control of endo- and ectoparasites of beef heifers treated with allopathic, herbal and homeopathic drugs. Rev Bras Parasitol Vet 2013; 22(4): 502-510. http://dx.doi.org/10.1590/S1984-29612013000400009. PMid:24473874.
http://dx.doi.org/10.1590/S1984-29612013...
; Heckler et al., 2016Heckler RP, Borges DGL, Vieira MC, Conde MH, Green M, Amorim ML, et al. New approach for the strategic control of gastrointestinal nematodes in grazed beef cattle during the growing phase in central Brazil. Vet Parasitol 2016; 221: 123-129. http://dx.doi.org/10.1016/j.vetpar.2016.03.010. PMid:27084483.
http://dx.doi.org/10.1016/j.vetpar.2016....
), Minas Gerais ( Furlong et al., 1993Furlong J, Silva AM, Verneque RS, Gardner AL, Brockington NR. Análise bio-econômica do uso de anti-helmíntico em bezerros na zona da mata de Minas Gerais. Rev Bras Parasitol Vet 1993; 2(2): 119-126. ; Guimarães et al., 2000Guimarães MP, Ribeiro MFB, Facuri-Filho EJ, Lima WS. Strategic control of gastrointestinal nematodes in dairy calves in Florestal, Minas Gerais, Brazil. Vet Res Commun 2000; 24(1): 31-38. http://dx.doi.org/10.1023/A:1006373221169. PMid:10703752.
http://dx.doi.org/10.1023/A:100637322116...
; Lima et al., 1985Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335. ; 1997Lima WS, Fakuri E, Guimarães MP, Malacco MA. Dinâmica das helmintoses de bovinos de leite na região Metalúrgica de Minas Gerais. Rev Bras Parasitol Vet 1997; 6(2): 97-103. ), and São Paulo ( Oliveira & Freitas, 1998Oliveira GP, Freitas AR. Doramectin e levamizole no controle dos helmintos de bovinos no início da estação seca. Cienc Rural 1998; 28(2): 277-282. http://dx.doi.org/10.1590/S0103-84781998000200016.
http://dx.doi.org/10.1590/S0103-84781998...
; Soutello, 2001Soutello RVG. Influência do parasitismo e da suplementação no desenvolvimento ponderal de novilhos mestiços Angus-Nelore e da raça Guzerá [dissertação]. Ilha Solteira: Faculdade de Engenharia, Universidade do Estado de São Paulo; 2001. ). In contrast, no difference was found between treated and untreated cattle in the Pantanal region of Mato Grosso do Sul ( Catto & Furlong, 1982Catto JB, Furlong J. Desenvolvimento de bovinos criados extensivamente, submetidos a vários esquemas de tratamentos anti-helmínticos, no Pantanal Matogrossense. Pesq Agropec Bras 1982; 17(1): 131-136. ; Catto et al., 1993Catto JB, Barros ATM, Costa CAF. Efeito de tratamentos anti-helmínticos no ganho de peso de bezerros desmamados, criados em pastagens nativas, no pantanal mato-grossense, Brasil. Rev Bras Parasitol Vet 1993; 2(2): 127-132. ; Sereno et al., 2000Sereno JRB, Catto JB, Silva MP, Sereno FTPS. Veda e vermifugação como alternativas de manejo para desmama de bezerros nelore em pastagem nativa do Pantanal. Pesq Agropec Bras 2000; 35(10): 1099-2105. http://dx.doi.org/10.1590/S0100-204X2000001000023.
http://dx.doi.org/10.1590/S0100-204X2000...
) and in São Paulo ( Santos et al., 1977Santos LE, Mendes MFM, Fontenello D, Miguel O, Paulo MS, Santos OL. Avaliação de tratamentos antihelmínticos no ganho de peso, no nível de hemoglobina e na infestação parasitária de novilhas “Guzerá” na região de Sertãozinho, S. Paulo, Brasil. Rev Fac Med Vet Zootec Univ S Paulo 1977; 14(1): 149-169. http://dx.doi.org/10.11606/issn.2318-3659.v14i1p149-169.
http://dx.doi.org/10.11606/issn.2318-365...
). According to the above cited authors, the anthelmintics used in the experiments published before 1985 were benzimidazoles and imidazothiazoles (tetramisole and levamisole). The findings of the first investigation into the effect of a macrocyclic lactone (ivermectin) in young cattle were then published in 1985 ( Lima et al., 1985Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335. ). The use of these endectocides has become customary in the last few decades, and macrocyclic lactones, particularly ivermectin, are among the most widely sold drugs for the control of cattle parasites ( Delgado et al., 2009Delgado FEF, Lima WS, Cunha AP, Bello ACPP, Domingues LN, Wanderley RPB, et al. Verminoses dos bovinos: percepção de pecuaristas em Minas Gerais, Brasil. Rev Bras Parasitol Vet 2009; 18(3): 29-33. http://dx.doi.org/10.4322/rbpv.01803005. PMid:19772773.
http://dx.doi.org/10.4322/rbpv.01803005...
; Neves et al., 2014Neves JH, Carvalho N, Rinaldi L, Cringoli G, Amarante AFT. Diagnosis of anthelmintic resistance in cattle in Brazil: A comparison of different methodologies. Vet Parasitol 2014; 206(3-4): 216-226. http://dx.doi.org/10.1016/j.vetpar.2014.10.015. PMid:25468021.
http://dx.doi.org/10.1016/j.vetpar.2014....
). However, the continuous use of ivermectin has resulted in anthelmintic resistance, a widespread problem reported in the states of Minas Gerais ( Rangel et al., 2005Rangel VB, Leite RC, Oliveira PR, Santos EJ Jr. Resistência de Cooperia spp. e Haemonchus spp. às avermectinas em bovinos de corte. Arq Bras Med Vet Zootec 2005; 57(2): 186-190. http://dx.doi.org/10.1590/S0102-09352005000200008.
http://dx.doi.org/10.1590/S0102-09352005...
), Rio de Janeiro ( Cardoso et al., 2008Cardoso JMS, Martins IVF, Sant’anna FB, Correia TR, Tancredi IP, Coumendouros K, et al. Identification of ivermectin and doramectin-resistant Cooperia punctata (Linstow, 1907) in a dairy herd in the State of Rio de Janeiro, Brazil. Arq Bras Med Vet Zootec 2008; 45(Suppl.): 75-81. http://dx.doi.org/10.11606/S1413-95962008000700010.
http://dx.doi.org/10.11606/S1413-9596200...
), Santa Catarina ( Souza et al., 2008Souza AP, Ramos CI, Bellato V, Sartor AA, Schelbauer CA. Resistência de helmintos gastrintestinais de bovinos a anti-helmínticos no Planalto Catarinense. Cienc Rural 2008; 38(5): 1363-1367. http://dx.doi.org/10.1590/S0103-84782008000500026.
http://dx.doi.org/10.1590/S0103-84782008...
), Mato Grosso do Sul ( Borges et al., 2013Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Anthelmintic resistance impact on tropical beef cattle productivity: Effect on weight gain of weaned calves. Trop Anim Health Prod 2013; 45(3): 723-727. http://dx.doi.org/10.1007/s11250-012-0280-4. PMid:23076819.
http://dx.doi.org/10.1007/s11250-012-028...
), São Paulo ( Neves et al., 2014Neves JH, Carvalho N, Rinaldi L, Cringoli G, Amarante AFT. Diagnosis of anthelmintic resistance in cattle in Brazil: A comparison of different methodologies. Vet Parasitol 2014; 206(3-4): 216-226. http://dx.doi.org/10.1016/j.vetpar.2014.10.015. PMid:25468021.
http://dx.doi.org/10.1016/j.vetpar.2014....
), Paraná ( Holsback et al., 2015Holsback L, Silva MA, Patelli THC, Jesus AP, Sanches JR. Resistance of Haemonchus, Cooperia, Trichostrongylus , and Oesophagostomum to ivermectin in dairy cattle in Paraná. Semina: Ciênc Agrár 2015; 36(3): 2031-2036. http://dx.doi.org/10.5433/1679-0359.2015v36n3Supl1p2031.
http://dx.doi.org/10.5433/1679-0359.2015...
) and Rio Grande do Sul ( Ramos et al., 2016Ramos F, Portella LP, Rodrigues FS, Reginato CZ, Potter L, Cezar AS, et al. Anthelmintic resistance in gastrointestinal nematodes of beef catlle in the state of Rio Grande do Sul, Brazil. Int J Parasitol Drugs Drug Resist 2016; 6(1): 93-101. http://dx.doi.org/10.1016/j.ijpddr.2016.02.002. PMid:27054068.
http://dx.doi.org/10.1016/j.ijpddr.2016....
).
Crossbreeding between Zebu and European cattle breeds has become a common practice aimed at improving beef cattle weight gain and carcass grade. The age group most susceptible to GIN infections is young cattle. Therefore, in this study we evaluated Simmental X Nellore crossbred calves before and after weaning. Our aim was to determine whether the animals were more likely to acquire severe infections before or after weaning, and to evaluate the effect of such infections on their body weight gain (BWG). One group was not treated with anthelmintic drugs (control group), while the other was treated monthly with a highly effective anthelmintic (albendazole) to keep it as free as possible of GIN infections. Because BWG reportedly benefits from the use of macrocyclic lactones with low or moderate efficacy due to anthelmintic resistance ( Borges et al., 2013Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Anthelmintic resistance impact on tropical beef cattle productivity: Effect on weight gain of weaned calves. Trop Anim Health Prod 2013; 45(3): 723-727. http://dx.doi.org/10.1007/s11250-012-0280-4. PMid:23076819.
http://dx.doi.org/10.1007/s11250-012-028...
; Heckler et al., 2016Heckler RP, Borges DGL, Vieira MC, Conde MH, Green M, Amorim ML, et al. New approach for the strategic control of gastrointestinal nematodes in grazed beef cattle during the growing phase in central Brazil. Vet Parasitol 2016; 221: 123-129. http://dx.doi.org/10.1016/j.vetpar.2016.03.010. PMid:27084483.
http://dx.doi.org/10.1016/j.vetpar.2016....
), we also included a group treated monthly with an anthelmintic of low efficacy (ivermectin).
Materials and Methods
This study was carried out in accordance with the ethical guidelines of animal research and was approved by the local ethics committee on animal use (protocol 38/2014/CEUA-FMVZ).
Description of the experiment
We evaluated young beef cattle on a farm located in the municipality of Piracicaba, state of São Paulo, Brazil. The cattle on this farm presented an ivermectin resistant worm population, which was susceptible to albendazole ( Neves et al., 2014Neves JH, Carvalho N, Rinaldi L, Cringoli G, Amarante AFT. Diagnosis of anthelmintic resistance in cattle in Brazil: A comparison of different methodologies. Vet Parasitol 2014; 206(3-4): 216-226. http://dx.doi.org/10.1016/j.vetpar.2014.10.015. PMid:25468021.
http://dx.doi.org/10.1016/j.vetpar.2014....
). The calves we evaluated had not been treated with anthelmintics prior to this experiment.
The animals, identified by numbered ear tags, were born between January 09 and March 05, 2015. At the beginning of this study, on April 15, the youngest calf was 41 days old and the oldest was 96 days old. The average age (± standard deviation) of the ivermectin, albendazole and untreated control groups was 70±15.92, 72±10.67 and 69±13.96 days (P> 0.05), respectively.
The unweaned Simmental X Nellore crossbred calves initially grazed with their mothers. They were divided into three groups, based on to sex, paternal genealogy and previous stratification according to their nematode fecal egg counts (FEC) in individual fecal samples taken two days before administering the first anthelmintic treatment. The groups were as follows: G1 (8 females and 6 males) - treated monthly with an ineffective anthelmintic, ivermectin 1% (0.2 mg/kg, Ivomec ® , Merial), G2 (7 females and 10 males) - treated monthly with a highly effective anthelmintic, albendazole sulfoxide 10% (2.5 mg/kg; Albendathor ® , Fabiani Animal Health), and G3 (8 females and 9 males) - untreated Control group.
All the grazing animals were kept on pastures ( Urochloa spp.), at a stocking rate of approximately 1.4 livestock units per hectare, and had free access to a multimineral supplement (Fosbovi ® 15, DSM Animal Nutrition). From April to June, all the calves had access to creep feeding with 310 g of concentrate/animal/day. The amount of concentrate was increased to 630 grams/animal/day in July, and to 950 grams/animal/day from August to the end of the experiment. The concentrate was produced on the farm and consisted of soybean meal (25%), corn (70%) and millet enriched with minerals (5%). All the calves were weaned on 22 September 2015, when males and females were placed in separate paddocks of similar size and pasture grasses to prevent undesired pregnancy, since the bull calves were not castrated.
All the animals were vaccinated against foot-and-mouth disease, according to the mandatory vaccination schedule. Females were vaccinated against brucellosis on 10 June 2015, and all the animals were vaccinated against clostridiosis and rabies on 25 August 2015.
A single dose of Fipronil (1 mg/kg/BW, TopLine ® , Merial) was applied in September to control the ectoparasites Rhipicephalus microplus and Haematobia irritans .
Fecal testing
Fecal samples were collected from each animal every 28 days. The samples were processed individually using the Flotac dual technique with a sensitivity of two eggs/g, using saturated sodium chloride (NaCl, specific gravity 1.2) and zinc sulfate solution (ZnSO 4 , specific gravity 1.35) ( Cringoli et al., 2010Cringoli G, Rinaldi L, Maurelli MP, Utzinger J. FLOTAC: new multivalent techniques for qualitative and quantitative copromicroscopic diagnosis of parasites in animals and humans. Nat Protoc 2010; 5(3): 503-515. http://dx.doi.org/10.1038/nprot.2009.235. PMid:20203667.
http://dx.doi.org/10.1038/nprot.2009.235...
). The ZnSO 4 solution was used to detect Dictyocaulus viviparus larvae in the fecal tests carried out from 30 July up to the end of the experiment.
On the above-mentioned days, composite fecal cultures from each group were prepared to obtain third-stage larvae and identify them according to nematode genus ( Ueno & Gonçalves, 1998Ueno H, Gonçalves PC. Manual para diagnóstico das Helmintoses de ruminantes. Tokyo: Japan International Cooperation Agency; 1998. ; van Wyk & Mayhew, 2013van Wyk J, Mayhew E. Morphological identification of parasitic nematode infective larvae of small ruminants and cattle: A practical lab guide. Onderstepoort J Vet Res 2013; 80(1): 539. http://dx.doi.org/10.4102/ojvr.v80i1.539. PMid:23718204.
http://dx.doi.org/10.4102/ojvr.v80i1.539...
; Amarante, 2011Amarante AFT. Why is it important to correctly identify Haemonchus species? Rev Bras Parasitol Vet 2011; 20(4): 263-268. http://dx.doi.org/10.1590/S1984-29612011000400002. PMid:22166378.
http://dx.doi.org/10.1590/S1984-29612011...
). In the case of Haemonchus larvae, the distance between the tip of the larval tail and the end of the sheath tail was measured. Larvae with lengths of approximately 99.2 µm were identified as Haemonchus placei , while those with lengths of about 57.3 µm were identified as Haemonchus similis ( Amarante, 2011Amarante AFT. Why is it important to correctly identify Haemonchus species? Rev Bras Parasitol Vet 2011; 20(4): 263-268. http://dx.doi.org/10.1590/S1984-29612011000400002. PMid:22166378.
http://dx.doi.org/10.1590/S1984-29612011...
).
On two occasions during the experiment (April and October), additional individual fecal samples were taken 10 days post-treatment for a fecal egg count reduction (FECR) test to evaluate the effectiveness of albendazole sulfoxide and ivermectin. The FECR was calculated as described by Torgerson et al. (2014)Torgerson PR, Paul M, Furrer R. Evaluating faecal egg count reduction using a specifically designed package “eggCounts” in R and a user friendly web interface. Int J Parasitol 2014; 44(5): 299-303. http://dx.doi.org/10.1016/j.ijpara.2014.01.005. PMid:24556564.
http://dx.doi.org/10.1016/j.ijpara.2014....
.
Blood tests
Every four months, blood samples were drawn from the jugular vein into tubes containing EDTA (Vacutainer, BD ® , USA) in order to determine the packed cell volume (PCV) by microcentrifugation and total plasma protein (TPP) by refractometry. The eosinophil count in peripheral blood was performed in a Neubauer chamber after staining with Carpentier’s solution ( Dawkins et al., 1989Dawkins HJS, Windon RG, Eagleson GK. Eosinophil responses in sheep selected for high and low responsiveness to Trichostrongylus colubriformis.
Int J Parasitol 1989; 19(2): 199-205. http://dx.doi.org/10.1016/0020-7519(89)90008-8. PMid:2722393.
http://dx.doi.org/10.1016/0020-7519(89)9...
), and the counts were expressed as the number of cells per µL of blood.
Statistical analysis
Data were analyzed by one-way analysis of variance (one-way ANOVA) in the case of variables measured only once (BWG and daily BWG) and by ANOVA with repeated measures in the case of variables measured at several time points (FEC, body weight, PCV, TPP and eosinophils), using SAS ® version 9.4 software. Group and sex were the classes evaluated. Means were compared by Tukey’s test at a 5% level of significance, and only significant interactions at the 5% significance level are reported in the results. The EPG count and eosinophil-related data were analyzed under log transformation (log 10 (x +1)).
Results
The effectiveness of the anthelmintics was evaluated 10 days post treatment, in April and October. Albendazole proved to be highly effective, with a FECR of 99.2% (confidence interval: 100 – 99). In contrast, ivermectin effectiveness was only 45.3% (51; 40) in April and zero in October, with the following nematode larvae in cultures of this group: Cooperia spp. (96%) and H. placei (4%) in April; and Cooperia spp. (97%), H. placei (1%) and Oesophagostomum radiatum (2%) in October.
Throughout the experiment, the most prevalent parasite in the control group was Cooperia spp., followed, in decreasing order, by Haemonchus placei , Haemonchus similis , Oesophagostomum radiatum and Trichostrongylus spp. ( Table 1 ). The group treated with ivermectin showed similar percentages of genera, except for H. similis and Trichostrongylus , which were never identified in this group ( Table 1 ).
Percentage of third stage larvae (L 3 ) of Haemonchus placei (HP), Haemonchus similis (HS), Cooperia spp. (COO), Trichostrongylus sp. (TRI) and Oesophagostomum radiatum (OES) from fecal cultures of Simmental X Nellore crossbred calves treated every 28 days with ivermectin, albendazole sulfoxide, or not treated (Control).
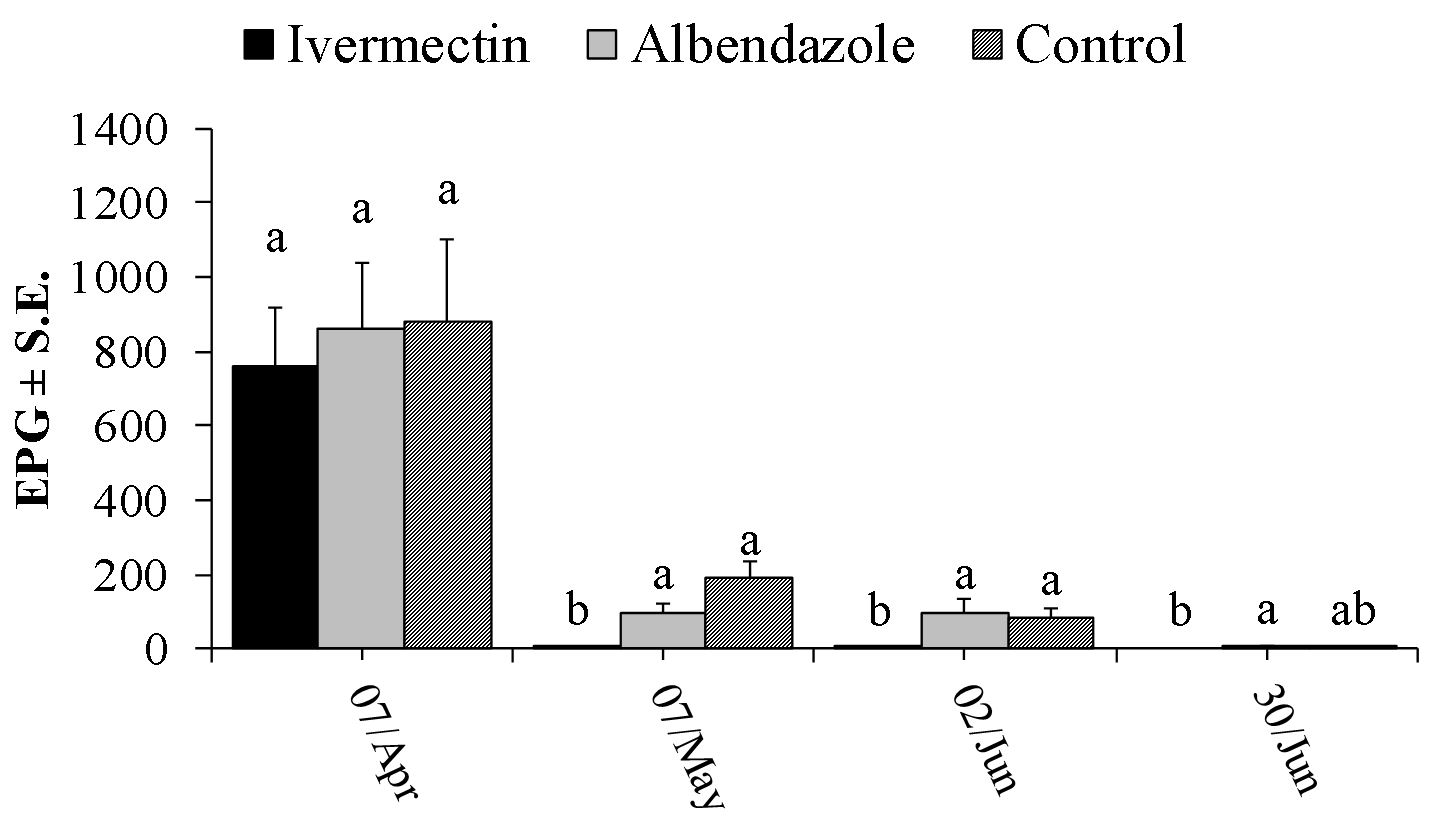
On the day of the first anthelmintic treatment (7 Apr), the mean EPG of strongylids were similar among groups: overall mean = 147.7 EPG with minimum and maximum values of zero and 518 EPG, respectively. The FECs of the control group remained relatively stable throughout the experiment, with means ranging from 117 to 289 EPG. On seven occasions, the average EPGs of the calves treated with albendazole were significantly lower than those of the ivermectin and control groups ( Figure 1 ). Nevertheless, calves in the albendazole treated group shed eggs in feces. The identification of infective larvae from fecal cultures indicated that these eggs were from Cooperia spp. ( Table 1 ). In only one of the 10 samplings (on 20 Oct), the mean FEC of ivermectin treated calves was significantly lower than that of the control. Sex was found to have an influence on FECs only in the last sampling, when females presented lower mean EPG (logEPG = 1.30) than males (logEPG = 2.07).
Mean number (± standard error) of strongyle eggs per gram of feces (EPG) from crossbred Simmental X Nellore calves treated every 28 days with ivermectin, albendazole sulfoxide or untreated (Control). On each sampling date, mean values with different letters differ significantly according to Tukey’s test (P<0.05). Bars represent standard error.
At the beginning of the experiment, the three groups showed high FECs of Strongyloides sp. ( Figure 2 ), with maximum individual values of 2,304 EPG and 3,504 EPG, respectively, in a female and in a male calf. Thereafter, the number of Strongyloides eggs gradually decreased until they were no longer detected after June ( Figure 2 ). In the samples collected on 07 May and 02 June, the mean EPG of the group treated with ivermectin was significantly lower than those of the other groups (P<0.05).
Mean number (± standard error) of Strongyloides spp. eggs per gram of feces (EPG) from crossbred Simmental X Nellore calves treated every 28 days with ivermectin, albendazole sulfoxide or untreated (Control). On each sampling date, mean values with different letters differ significantly according to Tukey’s test (P<0.05). Bars represent standard error.
Other parasites were found sporadically in some animals ( Table 2 ). In the first sampling, two animals of the ivermectin group presented Toxocara vitulorum eggs, which were absent after this anthelmintic was administered. Trichuris spp. and Moniezia spp. eggs were detected on a few occasions, while Eimeria spp. oocysts were found in most of the samples, albeit in small numbers. Dictyocaulus viviparus was found intermittently in some animals ( Table 2 ).
Means (± standard error) of Trichuris spp. eggs per gram (EPG), Toxocara vitulorum EPG, and Dictyocaulus viviparus larvae per gram of feces; presence of Moniezia spp. eggs and Eimeria spp. oocysts in Simmental X Nellore calves of the groups treated with ivermectin (n=14), with albendazole (n=18), and in the untreated control group (n=17).
The anthelmintic treatments had no significant effect (P>0.05) on PCV, TPP and blood eosinophil counts ( Table 3 ). As for TPP, a significant group*sex interaction was identified on 28 Jul, when the albendazole-female group and male-control group showed mean values higher than 7 g/dL, while the other groups presented values below 6.7 g/dL, albeit without significant differences between group means (P>0.05). In the case of eosinophil counts, the influence of sex was detected on 7 Apr and 28 Jul, when females presented higher overall means than males.
Means (± standard error) of packed cell volume (PCV, %), total plasma protein (TPP, g/dL) and blood eosinophils (EOS, cells/µL) of crossbred Simmental X Nellore calves naturally infected with gastrointestinal nematodes. Groups were treated monthly with albendazole, ivermectin or untreated (Control).
The calves were all healthy during the experiment, with no clinical signs of parasitic diseases. Their development was satisfactory throughout the experiment and the anthelmintic treatments did not affect (P>0.05) their body weight gain ( Table 4 ).
Mean (± standard error) body weight (BW), BW gain and daily BW gain (kg) of crossbred Simmental X Nellore calves naturally infected with gastrointestinal nematodes, treated every 28 days with ivermectin, albendazole sulfoxide or untreated (Control).
Discussion
The mean FECs during the experiment were lower than 400 EPG in all the groups, which presented no clinical signs of parasitic gastroenteritis. The blood variables fell within the normal range and no calf showed signs of anemia. Mean EPGs were significantly higher in the control and ivermectin group than in the albendazole group. However, these differences in the degree of infection between groups did not have a statistically significant effect on body weight gain, which was similar in the three groups. This result was unexpected and differed from findings reported in other experiments conducted in Brazil, where anthelmintic treatments usually result in greater body weight gain among young cattle ( Ramos et al., 2018Ramos F, Marques CB, Reginato CZ, Rodrigues FS, Sangioni LA, Vogel FSF, et al. Economic viability of anthelmintic treatment in naturally infected beef cattle under different nutritional strategies after weaning. Parasitol Res 2018; 117(12): 3993-4002. http://dx.doi.org/10.1007/s00436-018-6108-z. PMid:30302586.
http://dx.doi.org/10.1007/s00436-018-610...
), sometimes even when they are treated with drugs that are ineffective due to anthelmintic resistance ( Borges et al., 2013Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Anthelmintic resistance impact on tropical beef cattle productivity: Effect on weight gain of weaned calves. Trop Anim Health Prod 2013; 45(3): 723-727. http://dx.doi.org/10.1007/s11250-012-0280-4. PMid:23076819.
http://dx.doi.org/10.1007/s11250-012-028...
; Heckler et al., 2016Heckler RP, Borges DGL, Vieira MC, Conde MH, Green M, Amorim ML, et al. New approach for the strategic control of gastrointestinal nematodes in grazed beef cattle during the growing phase in central Brazil. Vet Parasitol 2016; 221: 123-129. http://dx.doi.org/10.1016/j.vetpar.2016.03.010. PMid:27084483.
http://dx.doi.org/10.1016/j.vetpar.2016....
).
The calves involved in this experiment had been infected from an early age with gastrointestinal nematodes, beginning with Strongyloides spp., eliminated naturally through the development of immune response. Then, as expected, the parasitic fauna consisted predominantly of the strongylids ( Cooperia spp., Haemonchus spp. and Oesophagostomum spp.). The absence of clinical signs of parasitic gastroenteritis and the good performance of the calves in the control group suggest that the host-parasite interaction reached equilibrium under the continuous parasite challenge that occurred in this experiment.
The fecal cultures revealed the presence, in decreasing order of occurrence, of Cooperia spp., Haemonchus spp. and Oesophagostomum spp. This finding is consistent with other studies, which also found that the parasite with the highest infection intensity was Cooperia punctata , followed, in decreasing order, by Haemonchus ( H. placei and/or H. similis ) and Oesophagostomum radiatum ( Lima, 1998Lima WS. Seasonal infection pattern of gastrointestinal nematodes of beef cattle in Minas Gerais State- Brazil. Vet Parasitol 1998; 74(2-4): 203-214. http://dx.doi.org/10.1016/S0304-4017(97)00164-7. PMid:9561708.
http://dx.doi.org/10.1016/S0304-4017(97)...
; Pimentel Neto & Fonseca, 2002Pimentel Neto M, Fonseca AH. Epidemiologia das helmintoses pulmonares e gastrintestinais de bezerros em região de baixada do Estado do Rio de Janeiro. Pesq Vet Bras 2002; 22(4): 148-152. http://dx.doi.org/10.1590/S0100-736X2002000400004.
http://dx.doi.org/10.1590/S0100-736X2002...
; Bresciani et al., 2001Bresciani KDS, Nascimento AA, Costa AJ, Amarante AFT, Perri SHV, Lima LGF. Frequência e intensidade parasitária de helmintos gastrintestinais em bovinos abatidos em frigorífico da região noroeste do Estado de São Paulo, SP, Brasil. Semina: Ciênc Agrár 2001; 22(1): 93-97. http://dx.doi.org/10.5433/1679-0359.2001v22n1p93.
http://dx.doi.org/10.5433/1679-0359.2001...
). These three genera were detected in the group treated with ivermectin, which confirms their resistance to avermectins and is in agreement with a previous report of drug resistance on this farm ( Neves et al., 2014Neves JH, Carvalho N, Rinaldi L, Cringoli G, Amarante AFT. Diagnosis of anthelmintic resistance in cattle in Brazil: A comparison of different methodologies. Vet Parasitol 2014; 206(3-4): 216-226. http://dx.doi.org/10.1016/j.vetpar.2014.10.015. PMid:25468021.
http://dx.doi.org/10.1016/j.vetpar.2014....
). The nematode population was susceptible to albendazole, and only Cooperia was detected after treatment with this drug. This finding can be explained by the short prepatent period of this parasite (11-14 days), while H. placei and O. radiatum present a longer prepatent period of 26-28 and 35-41 days, respectively ( Wood et al., 1995Wood IB, Amaral NK, Bairden K, Duncan JL, Kassai T, Malone JB Jr, et al. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) second edition of guidelines for evaluating the efficacy of anthelmintics in ruminants (bovine, ovine, caprine). Vet Parasitol 1995; 58(3):181-213. https://doi.org/10.1016/0304-4017(95)00806-2
https://doi.org/10.1016/0304-4017(95)008...
). Therefore, it can be deduced that calves in the albendazole group become reinfected during the interval between drenches. In Minas Gerais, the FECs of treated calves were similar to those of the untreated control calves six weeks post-treatment with tetramisole or fenbendazole, indicating the rapid reestablishment of the parasite population ( Lima et al., 1983Lima WS, Costa HMA, Guimarães MP, Costa JO, Leite ACR. Reinfecção de bezerros após tratamentos anti-helmínticos. Arq Bras Med Vet Zootec 1983; 35(1): 101-112. ). Taken together, these results indicate that such treatments may not have a meaningful impact on the epidemiology of GIN infections.
In this experiment, we evaluated only the effect of GIN infections, since a single dose of fipronil prevented losses caused by ectoparasites. It is therefore difficult to compare our results with those of studies that used products also active against ectoparasites ( Bianchin et al., 2007Bianchin I, Catto BJ, Kichel NA, Torres AA, Honer MR. The effect of the control of endo- and ectoparasites on weight gains in crossbred cattle ( Bos taurus taurus × Bos taurus indicus ) in the central region of Brazil. Trop Anim Health Prod 2007; 39(4): 287-296. http://dx.doi.org/10.1007/s11250-007-9017-1. PMid:17847824.
http://dx.doi.org/10.1007/s11250-007-901...
; Catto et al., 2009Catto JB, Bianchin I, Santurio JM, Feijó GLD, Kichel AN, Silva JM. Grazing systems, rotenone and parasites control in crossbred calves: effect on live weight gain and on parasites burdens. Rev Bras Parasitol Vet 2009; 18(4): 37-43. http://dx.doi.org/10.4322/rbpv.01804007. PMid:20040207.
http://dx.doi.org/10.4322/rbpv.01804007...
; Soutello, 2001Soutello RVG. Influência do parasitismo e da suplementação no desenvolvimento ponderal de novilhos mestiços Angus-Nelore e da raça Guzerá [dissertação]. Ilha Solteira: Faculdade de Engenharia, Universidade do Estado de São Paulo; 2001. ). For instance, Catto et al. (2013)Catto JB, Bianchin I, Feijó GLD, Araújo FR, Ramos CAN, Castelão ABC. Weight gain and control of endo- and ectoparasites of beef heifers treated with allopathic, herbal and homeopathic drugs. Rev Bras Parasitol Vet 2013; 22(4): 502-510. http://dx.doi.org/10.1590/S1984-29612013000400009. PMid:24473874.
http://dx.doi.org/10.1590/S1984-29612013...
reported that Brangus heifers treated with moxidectin and an acaricide showed lower FECs and tick infestations than untreated animals or animals treated with alternative drugs, which resulted in heifers 22 to 30 kg heavier in the treated group. Hence, in those experiments, the benefits in body weight gain may have resulted from the control of both endo- and ectoparasites.
The calves in our study were fed dietary supplements before and after weaning, resulting in weight gains throughout the experiment and enabling them to reach a body weight of about 250 kg on the last sampling date, before turning one year old. This nutritional quality enabled the infected animals, especially those of the untreated control and ivermectin groups, to become resilient to GIN infections. Melo (1977)Melo HJH. Efeito de diferentes esquemas de tratamento anti-helmíntico no ganho de peso de bezerros nelore desmamados e criados extensivamente em pastagens de Jaraguá ( Hyparrhenia rufa (Nees Staft). Arq Esc Vet UFMG 1977; 29(3): 269-277. had already pointed out that the effect of anthelmintic drugs on BWG appeared to be related to environmental conditions, especially nutrition, confirming our findings. The good level of nutrition employed in this experiment may not correspond to the reality of cattle production in Brazil, especially in the past, when cattle normally failed to receive proper nutrition throughout the year, leading to low body weight gain in the rainy season and body weight loss in the dry season ( Melo, 1977Melo HJH. Efeito de diferentes esquemas de tratamento anti-helmíntico no ganho de peso de bezerros nelore desmamados e criados extensivamente em pastagens de Jaraguá ( Hyparrhenia rufa (Nees Staft). Arq Esc Vet UFMG 1977; 29(3): 269-277. ). It should be noted that tropical grasses have a high potential for mass production, but low crude protein content. The benefits of anthelmintic treatments may be greater under such stressful dietary conditions. To exemplify this statement, Lima et al. (1985)Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335. conducted a study of Holstein X Nellore and Chianina X Nellore crossbred cattle in Minas Gerais and reported that the animals’ performance improved in response to different ivermectin treatment strategies. In their study, the cattle grazed on Guinea grass ( Panicum maximum ) and their initial mean body weight (BW) varied from 104 kg to 134 kg at 6-8 months of age. In our experiment, the animals weaned at a similar age had a BW of about 200 kg, which indicates that the animals in our study were in much better nutritional condition than those of the experiment described by Lima et al. (1985)Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335. . This may explain the discrepant results of the two studies, i.e., Lima et al. (1985)Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335. reported a significant improvement in the BWG of treated animals over that of the controls, but this was not the case of the well-fed animals in our study. Therefore, the results of our experiment suggest that calves that are given proper dietary supplementation are more likely to develop normally, without significant losses due to GIN infections. Studies that include poorly managed nutrition may not reflect the current reality of the beef cattle industry, which is increasingly adopting techniques aimed at improving the animals’ nutritional status.
Grisi et al. (2014)Grisi L, Leite RC, Martins JRS, Barros ATM, Andreotti R, Cançado PHD, et al. Reassessment of the potential economic impact of cattle parasites in Brazil. Rev Bras Parasitol Vet 2014; 23(2): 150-156. http://dx.doi.org/10.1590/S1984-29612014042. PMid:25054492.
http://dx.doi.org/10.1590/S1984-29612014...
, who reassessed the potential economic impact of cattle parasites in Brazil, suggested that gastrointestinal nematode infections cause annual losses of US$ 7.11 billion. This estimate was based on two studies, which found that cattle up to two years of age – the age group most affected by gastrointestinal nematodes – present a reduction in weight gain ranging from 41 kg ( Bianchin et al., 1995Bianchin I, Honer MR, Nunes S, Nascimento YA. Effect of stocking rates and anthelminthic treatments on weight gains in weaned Nellore cattle on improved pasture in the Brazilian cerrado. Trop Anim Health Prod 1995; 27(1): 1-8. http://dx.doi.org/10.1007/BF02236326. PMid:7770946.
http://dx.doi.org/10.1007/BF02236326...
) to 67 kg ( Pinheiro, 1983Pinheiro AC. Programa integrado de controle de verminose de bovinos de corte. 2nd ed. Bagé: EMBRAPA-UEPAE; 1983. ) compared to animals treated with anthelmintics. These losses, which were recorded in studies conducted more than two decades ago, may be outdated and no longer represent the reality of today’s cattle industry, which has shown significant technological advances in recent years, especially in terms of nutritional management and crossbreeding strategies. Obviously, extrapolating our observations to other production systems would lead to inaccurate findings. However, considering the rapid development of the cattle industry, our results clearly indicate the need for further studies in different regions of Brazil in order to accurately estimate losses resulting from GIN infections.
There is also a need for the development of new control strategies less dependent on anthelmintics, given that the control methods recommended by Bianchin et al. (1995)Bianchin I, Honer MR, Nunes S, Nascimento YA. Effect of stocking rates and anthelminthic treatments on weight gains in weaned Nellore cattle on improved pasture in the Brazilian cerrado. Trop Anim Health Prod 1995; 27(1): 1-8. http://dx.doi.org/10.1007/BF02236326. PMid:7770946.
http://dx.doi.org/10.1007/BF02236326...
and Pinheiro (1983)Pinheiro AC. Programa integrado de controle de verminose de bovinos de corte. 2nd ed. Bagé: EMBRAPA-UEPAE; 1983. , which involve frequent drenches, are no longer sustainable due to the widespread problem of anthelmintic resistance. In addition, the excessive use of avermectins leaves behind residues that are harmful to the environment. The widespread use of ivermectin disrupted the ecosystem function by affecting species richness, abundance and biomass, resulting in significant changes in soil functionality ( Verdú et al., 2018Verdú JR, Lobo JM, Sánchez-Piñero F, Gallego B, Numa C, Lumaret JP, et al. Ivermectin residues disrupt dung beetle diversity, soil properties and ecosystem functioning: an interdisciplinary field study. Sci Total Environ 2018; 618: 219-228. http://dx.doi.org/10.1016/j.scitotenv.2017.10.331. PMid:29128770.
http://dx.doi.org/10.1016/j.scitotenv.20...
). Therefore, improved nutrition appears to be an excellent option to optimize prophylaxis, minimizing the need for anthelmintics.
In conclusion, our results indicate that Simmental X Nellore crossbred calves properly fed with suitable dietary supplementation were not affected by gastrointestinal nematode parasitism under grazing conditions.
-
Financial support: None.
-
How to cite:Neves JH, Carvalho N, Amarante AFT. Gastrointestinal nematode infections do not hinder the development of Simmental X Nellore crossbred calves raised with a nutritionally enhanced diet. Braz J Vet Parasitol 2020; 29(1): e015819. http://dx.doi.org/10.1590/S1984-29612020006
References
- Amarante AFT. Why is it important to correctly identify Haemonchus species? Rev Bras Parasitol Vet 2011; 20(4): 263-268. http://dx.doi.org/10.1590/S1984-29612011000400002 PMid:22166378.
» http://dx.doi.org/10.1590/S1984-29612011000400002 - Associação Brasileira das Indústrias Exportadoras de Carne – ABIEC [online]. São Paulo: ABIEC; 2019 [cited 2019 Jul 9]. Available from: http://www.abiec.com.br/Sumario2019.aspx
» http://www.abiec.com.br/Sumario2019.aspx - Bianchin I, Catto BJ, Kichel NA, Torres AA, Honer MR. The effect of the control of endo- and ectoparasites on weight gains in crossbred cattle ( Bos taurus taurus × Bos taurus indicus ) in the central region of Brazil. Trop Anim Health Prod 2007; 39(4): 287-296. http://dx.doi.org/10.1007/s11250-007-9017-1 PMid:17847824.
» http://dx.doi.org/10.1007/s11250-007-9017-1 - Bianchin I, Honer MR, Nunes S, Nascimento YA. Effect of stocking rates and anthelminthic treatments on weight gains in weaned Nellore cattle on improved pasture in the Brazilian cerrado. Trop Anim Health Prod 1995; 27(1): 1-8. http://dx.doi.org/10.1007/BF02236326 PMid:7770946.
» http://dx.doi.org/10.1007/BF02236326 - Borges FA, Almeida GD, Heckler RP, Lemes RT, Onizuka MKV, Borges DGL. Anthelmintic resistance impact on tropical beef cattle productivity: Effect on weight gain of weaned calves. Trop Anim Health Prod 2013; 45(3): 723-727. http://dx.doi.org/10.1007/s11250-012-0280-4 PMid:23076819.
» http://dx.doi.org/10.1007/s11250-012-0280-4 - Bresciani KDS, Nascimento AA, Costa AJ, Amarante AFT, Perri SHV, Lima LGF. Frequência e intensidade parasitária de helmintos gastrintestinais em bovinos abatidos em frigorífico da região noroeste do Estado de São Paulo, SP, Brasil. Semina: Ciênc Agrár 2001; 22(1): 93-97. http://dx.doi.org/10.5433/1679-0359.2001v22n1p93
» http://dx.doi.org/10.5433/1679-0359.2001v22n1p93 - Cardoso JMS, Martins IVF, Sant’anna FB, Correia TR, Tancredi IP, Coumendouros K, et al. Identification of ivermectin and doramectin-resistant Cooperia punctata (Linstow, 1907) in a dairy herd in the State of Rio de Janeiro, Brazil. Arq Bras Med Vet Zootec 2008; 45(Suppl.): 75-81. http://dx.doi.org/10.11606/S1413-95962008000700010
» http://dx.doi.org/10.11606/S1413-95962008000700010 - Catto JB, Barros ATM, Costa CAF. Efeito de tratamentos anti-helmínticos no ganho de peso de bezerros desmamados, criados em pastagens nativas, no pantanal mato-grossense, Brasil. Rev Bras Parasitol Vet 1993; 2(2): 127-132.
- Catto JB, Bianchin I, Feijó GLD, Araújo FR, Ramos CAN, Castelão ABC. Weight gain and control of endo- and ectoparasites of beef heifers treated with allopathic, herbal and homeopathic drugs. Rev Bras Parasitol Vet 2013; 22(4): 502-510. http://dx.doi.org/10.1590/S1984-29612013000400009 PMid:24473874.
» http://dx.doi.org/10.1590/S1984-29612013000400009 - Catto JB, Bianchin I, Santurio JM, Feijó GLD, Kichel AN, Silva JM. Grazing systems, rotenone and parasites control in crossbred calves: effect on live weight gain and on parasites burdens. Rev Bras Parasitol Vet 2009; 18(4): 37-43. http://dx.doi.org/10.4322/rbpv.01804007 PMid:20040207.
» http://dx.doi.org/10.4322/rbpv.01804007 - Catto JB, Furlong J. Desenvolvimento de bovinos criados extensivamente, submetidos a vários esquemas de tratamentos anti-helmínticos, no Pantanal Matogrossense. Pesq Agropec Bras 1982; 17(1): 131-136.
- Costa JO, Costa HMA, Guimarães MP, Freitas MG. Efeito de tratamentos anti-helmínticos sobre o OPG e o desenvolvimento ponderal de bezerros. Arq Esc Vet UFMG 1977; 29(2): 171-178.
- Cringoli G, Rinaldi L, Maurelli MP, Utzinger J. FLOTAC: new multivalent techniques for qualitative and quantitative copromicroscopic diagnosis of parasites in animals and humans. Nat Protoc 2010; 5(3): 503-515. http://dx.doi.org/10.1038/nprot.2009.235 PMid:20203667.
» http://dx.doi.org/10.1038/nprot.2009.235 - Dawkins HJS, Windon RG, Eagleson GK. Eosinophil responses in sheep selected for high and low responsiveness to Trichostrongylus colubriformis. Int J Parasitol 1989; 19(2): 199-205. http://dx.doi.org/10.1016/0020-7519(89)90008-8 PMid:2722393.
» http://dx.doi.org/10.1016/0020-7519(89)90008-8 - Delgado FEF, Lima WS, Cunha AP, Bello ACPP, Domingues LN, Wanderley RPB, et al. Verminoses dos bovinos: percepção de pecuaristas em Minas Gerais, Brasil. Rev Bras Parasitol Vet 2009; 18(3): 29-33. http://dx.doi.org/10.4322/rbpv.01803005 PMid:19772773.
» http://dx.doi.org/10.4322/rbpv.01803005 - Fachiolli DF, Saes IL, Dellaqua JVT, Souza OA, Pinto LD, Santi PF, et al. Anthelmintic treatment and supplementation in Nellore calves performance in the post-weaning period. Semina: Ciênc Agrár 2017; 38(3): 1551-1560. http://dx.doi.org/10.5433/1679-0359.2017v38n3p1541
» http://dx.doi.org/10.5433/1679-0359.2017v38n3p1541 - Furlong J, Silva AM, Verneque RS, Gardner AL, Brockington NR. Análise bio-econômica do uso de anti-helmíntico em bezerros na zona da mata de Minas Gerais. Rev Bras Parasitol Vet 1993; 2(2): 119-126.
- Grisi L, Leite RC, Martins JRS, Barros ATM, Andreotti R, Cançado PHD, et al. Reassessment of the potential economic impact of cattle parasites in Brazil. Rev Bras Parasitol Vet 2014; 23(2): 150-156. http://dx.doi.org/10.1590/S1984-29612014042 PMid:25054492.
» http://dx.doi.org/10.1590/S1984-29612014042 - Guimarães MP, Ribeiro MFB, Facuri-Filho EJ, Lima WS. Strategic control of gastrointestinal nematodes in dairy calves in Florestal, Minas Gerais, Brazil. Vet Res Commun 2000; 24(1): 31-38. http://dx.doi.org/10.1023/A:1006373221169 PMid:10703752.
» http://dx.doi.org/10.1023/A:1006373221169 - Heckler RP, Borges DGL, Vieira MC, Conde MH, Green M, Amorim ML, et al. New approach for the strategic control of gastrointestinal nematodes in grazed beef cattle during the growing phase in central Brazil. Vet Parasitol 2016; 221: 123-129. http://dx.doi.org/10.1016/j.vetpar.2016.03.010 PMid:27084483.
» http://dx.doi.org/10.1016/j.vetpar.2016.03.010 - Holsback L, Silva MA, Patelli THC, Jesus AP, Sanches JR. Resistance of Haemonchus, Cooperia, Trichostrongylus , and Oesophagostomum to ivermectin in dairy cattle in Paraná. Semina: Ciênc Agrár 2015; 36(3): 2031-2036. http://dx.doi.org/10.5433/1679-0359.2015v36n3Supl1p2031
» http://dx.doi.org/10.5433/1679-0359.2015v36n3Supl1p2031 - Lima WS, Costa HMA, Guimarães MP, Costa JO, Leite ACR. Reinfecção de bezerros após tratamentos anti-helmínticos. Arq Bras Med Vet Zootec 1983; 35(1): 101-112.
- Lima WS, Fakuri E, Guimarães MP, Malacco MA. Dinâmica das helmintoses de bovinos de leite na região Metalúrgica de Minas Gerais. Rev Bras Parasitol Vet 1997; 6(2): 97-103.
- Lima WS, Guimarães MP, Leite ACR. Custo-benefício de diferentes dosificações anti-helmínticas em relação ao ganho de peso de bezerros de corte. Pesq Agropec Bras 1985; 20(11): 1333-1335.
- Lima WS. Seasonal infection pattern of gastrointestinal nematodes of beef cattle in Minas Gerais State- Brazil. Vet Parasitol 1998; 74(2-4): 203-214. http://dx.doi.org/10.1016/S0304-4017(97)00164-7 PMid:9561708.
» http://dx.doi.org/10.1016/S0304-4017(97)00164-7 - Melo HJH, Bianchin I. Estudos epidemiológicos de infecções por nematódeos gastrintestinais de bovinos de corte em zona de cerrado de Mato Grosso. Pesq Agropec Bras 1977; 12: 205-216.
- Melo HJH. Efeito de diferentes esquemas de tratamento anti-helmíntico no ganho de peso de bezerros nelore desmamados e criados extensivamente em pastagens de Jaraguá ( Hyparrhenia rufa (Nees Staft). Arq Esc Vet UFMG 1977; 29(3): 269-277.
- Neves JH, Carvalho N, Rinaldi L, Cringoli G, Amarante AFT. Diagnosis of anthelmintic resistance in cattle in Brazil: A comparison of different methodologies. Vet Parasitol 2014; 206(3-4): 216-226. http://dx.doi.org/10.1016/j.vetpar.2014.10.015 PMid:25468021.
» http://dx.doi.org/10.1016/j.vetpar.2014.10.015 - Oliveira GP, Freitas AR. Doramectin e levamizole no controle dos helmintos de bovinos no início da estação seca. Cienc Rural 1998; 28(2): 277-282. http://dx.doi.org/10.1590/S0103-84781998000200016
» http://dx.doi.org/10.1590/S0103-84781998000200016 - Pathak AK. Nutritional Bases to Control Gastrointestinal Parasites of Livestock. J Dairy Vet Sci 2017; 4(1): 555632.
- Pimentel Neto M, Fonseca AH. Epidemiologia das helmintoses pulmonares e gastrintestinais de bezerros em região de baixada do Estado do Rio de Janeiro. Pesq Vet Bras 2002; 22(4): 148-152. http://dx.doi.org/10.1590/S0100-736X2002000400004
» http://dx.doi.org/10.1590/S0100-736X2002000400004 - Pinheiro AC. Programa integrado de controle de verminose de bovinos de corte. 2nd ed. Bagé: EMBRAPA-UEPAE; 1983.
- Ramos F, Marques CB, Reginato CZ, Rodrigues FS, Sangioni LA, Vogel FSF, et al. Economic viability of anthelmintic treatment in naturally infected beef cattle under different nutritional strategies after weaning. Parasitol Res 2018; 117(12): 3993-4002. http://dx.doi.org/10.1007/s00436-018-6108-z PMid:30302586.
» http://dx.doi.org/10.1007/s00436-018-6108-z - Ramos F, Portella LP, Rodrigues FS, Reginato CZ, Potter L, Cezar AS, et al. Anthelmintic resistance in gastrointestinal nematodes of beef catlle in the state of Rio Grande do Sul, Brazil. Int J Parasitol Drugs Drug Resist 2016; 6(1): 93-101. http://dx.doi.org/10.1016/j.ijpddr.2016.02.002 PMid:27054068.
» http://dx.doi.org/10.1016/j.ijpddr.2016.02.002 - Rangel VB, Leite RC, Oliveira PR, Santos EJ Jr. Resistência de Cooperia spp. e Haemonchus spp. às avermectinas em bovinos de corte. Arq Bras Med Vet Zootec 2005; 57(2): 186-190. http://dx.doi.org/10.1590/S0102-09352005000200008
» http://dx.doi.org/10.1590/S0102-09352005000200008 - Santos LE, Mendes MFM, Fontenello D, Miguel O, Paulo MS, Santos OL. Avaliação de tratamentos antihelmínticos no ganho de peso, no nível de hemoglobina e na infestação parasitária de novilhas “Guzerá” na região de Sertãozinho, S. Paulo, Brasil. Rev Fac Med Vet Zootec Univ S Paulo 1977; 14(1): 149-169. http://dx.doi.org/10.11606/issn.2318-3659.v14i1p149-169
» http://dx.doi.org/10.11606/issn.2318-3659.v14i1p149-169 - Sereno JRB, Catto JB, Silva MP, Sereno FTPS. Veda e vermifugação como alternativas de manejo para desmama de bezerros nelore em pastagem nativa do Pantanal. Pesq Agropec Bras 2000; 35(10): 1099-2105. http://dx.doi.org/10.1590/S0100-204X2000001000023
» http://dx.doi.org/10.1590/S0100-204X2000001000023 - Soutello RVG. Influência do parasitismo e da suplementação no desenvolvimento ponderal de novilhos mestiços Angus-Nelore e da raça Guzerá [dissertação]. Ilha Solteira: Faculdade de Engenharia, Universidade do Estado de São Paulo; 2001.
- Souza AP, Ramos CI, Bellato V, Sartor AA, Schelbauer CA. Resistência de helmintos gastrintestinais de bovinos a anti-helmínticos no Planalto Catarinense. Cienc Rural 2008; 38(5): 1363-1367. http://dx.doi.org/10.1590/S0103-84782008000500026
» http://dx.doi.org/10.1590/S0103-84782008000500026 - Torgerson PR, Paul M, Furrer R. Evaluating faecal egg count reduction using a specifically designed package “eggCounts” in R and a user friendly web interface. Int J Parasitol 2014; 44(5): 299-303. http://dx.doi.org/10.1016/j.ijpara.2014.01.005 PMid:24556564.
» http://dx.doi.org/10.1016/j.ijpara.2014.01.005 - Ueno H, Gonçalves PC. Manual para diagnóstico das Helmintoses de ruminantes. Tokyo: Japan International Cooperation Agency; 1998.
- van Wyk J, Mayhew E. Morphological identification of parasitic nematode infective larvae of small ruminants and cattle: A practical lab guide. Onderstepoort J Vet Res 2013; 80(1): 539. http://dx.doi.org/10.4102/ojvr.v80i1.539 PMid:23718204.
» http://dx.doi.org/10.4102/ojvr.v80i1.539 - Verdú JR, Lobo JM, Sánchez-Piñero F, Gallego B, Numa C, Lumaret JP, et al. Ivermectin residues disrupt dung beetle diversity, soil properties and ecosystem functioning: an interdisciplinary field study. Sci Total Environ 2018; 618: 219-228. http://dx.doi.org/10.1016/j.scitotenv.2017.10.331 PMid:29128770.
» http://dx.doi.org/10.1016/j.scitotenv.2017.10.331 - Wood IB, Amaral NK, Bairden K, Duncan JL, Kassai T, Malone JB Jr, et al. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) second edition of guidelines for evaluating the efficacy of anthelmintics in ruminants (bovine, ovine, caprine). Vet Parasitol 1995; 58(3):181-213. https://doi.org/10.1016/0304-4017(95)00806-2
» https://doi.org/10.1016/0304-4017(95)00806-2
Publication Dates
-
Publication in this collection
30 Mar 2020 -
Date of issue
2020
History
-
Received
02 Sept 2019 -
Accepted
16 Dec 2019