Abstracts
RATIONALE: Metabolic acidosis induced by topiramate is a well documented but infrequent adverse event. The objective was to demonstrate the lowering of carbon dioxide serum levels, which is usually asymptomatic but may facilitate the occurrence of metabolic acidosis in patients using topiramate. METHODS: We evaluated, prospectively, the carbon dioxide serum levels of 18 patients seen at the epilepsy clinic of our university hospital, before and 3 months after introducing topiramate. RESULTS: Five patients were female and 13 were male, age ranging from 2 to 16 years old (mean=9.3). Carbon dioxide mean serum levels were 25 and 21.2 mmol/L (normal = 22 to 30), before and 3 months after introducing topiramate, respectively. Dose ranged from 2.08 to 11.76 mg/kg/day (mean=6.7mg/kg/day). Adverse events were anorexia, nausea and somnolence. CONCLUSION: We conclude that the lowering of carbon dioxide serum levels induced by topiramate is mostly asymptomatic, but may facilitate the occurrence of metabolic acidosis. Since patients in use of topiramate have refractory epilepsy, they may need epilepsy surgery, and must be carefully monitored for the risk of metabolic acidosis during surgery.
topiramate; metabolic acidosis; nephrolitiasis
INTRODUÇÃO: Acidose metabólica induzida por topiramato é evento adverso pouco frequente, mas bem documentado. Nosso objetivo foi demonstrar a diminuição dos níveis de dióxido de carbono, muitas vezes assintomática, mas que pode predispor ao aparecimento de acidose metabólica. MATERIAL E MÉTODO: Avaliamos prospectivamente os níveis de dióxido de carbono de 18 pacientes acompanhados no ambulatório de epilepsia infantil da UNICAMP, antes e 3 meses após o início do uso de topiramato. RESULTADOS: Foram avaliados 18 pacientes com idade entre 2 e 16 anos (média = 9,3 anos). Cinco pacientes eram do sexo feminino e 13, do sexo masculino. Os níveis médios de dióxido de carbono antes e após o uso de topiramato foram 25 e 21,2 mmol/L (normal = 22 a 30), respectivamente. A dose de topiramato utilizada foi 2,08 a 11,76 mg/kg/dia (média = 6,7 mg/kg/dia). Os eventos adversos mais frequentes foram anorexia, nausea e sonolência. CONCLUSÃO: Concluímos que a diminuição dos níveis de dióxido de carbono induzida por topiramato é na maioria das vezes assintomática entretanto, pode predispor à ocorrência de acidose metabólica. Como o topiramato é utilizado frequentemente no tratamento de epilepsia de difícil controle, os pacientes em uso desta medicação quando submetidos à cirurgia de epilepsia devem ser monitorizados, principalmente durante o intraoperatório, quanto à possibilidade de apresentarem acidose metabólica.
topiramato; acidose metabólica; nefrolitíase
PREDISPOSITION TO METABOLIC ACIDOSIS INDUCED BY TOPIRAMATE
MARIA AUGUSTA MONTENEGRO* * Departamento de Neurologia, Faculdade de Ciências Médicas (FCM) da Universidade Estadual de Campinas (UNICAMP). Research supported by Janssen-Cilag. Aceite: 18-julho-2000. , MARILISA M. GUERREIRO* * Departamento de Neurologia, Faculdade de Ciências Médicas (FCM) da Universidade Estadual de Campinas (UNICAMP). Research supported by Janssen-Cilag. Aceite: 18-julho-2000. , ANNA ELISA SCOTONI* * Departamento de Neurologia, Faculdade de Ciências Médicas (FCM) da Universidade Estadual de Campinas (UNICAMP). Research supported by Janssen-Cilag. Aceite: 18-julho-2000. , CARLOS A.M. GUERREIRO* * Departamento de Neurologia, Faculdade de Ciências Médicas (FCM) da Universidade Estadual de Campinas (UNICAMP). Research supported by Janssen-Cilag. Aceite: 18-julho-2000.
ABSTRACT ¾ RATIONALE: Metabolic acidosis induced by topiramate is a well documented but infrequent adverse event. The objective was to demonstrate the lowering of carbon dioxide serum levels, which is usually asymptomatic but may facilitate the occurrence of metabolic acidosis in patients using topiramate. METHODS: We evaluated, prospectively, the carbon dioxide serum levels of 18 patients seen at the epilepsy clinic of our university hospital, before and 3 months after introducing topiramate. RESULTS: Five patients were female and 13 were male, age ranging from 2 to 16 years old (mean=9.3). Carbon dioxide mean serum levels were 25 and 21.2 mmol/L (normal = 22 to 30), before and 3 months after introducing topiramate, respectively. Dose ranged from 2.08 to 11.76 mg/kg/day (mean=6.7mg/kg/day). Adverse events were anorexia, nausea and somnolence. CONCLUSION: We conclude that the lowering of carbon dioxide serum levels induced by topiramate is mostly asymptomatic, but may facilitate the occurrence of metabolic acidosis. Since patients in use of topiramate have refractory epilepsy, they may need epilepsy surgery, and must be carefully monitored for the risk of metabolic acidosis during surgery.
KEY WORDS: topiramate, metabolic acidosis, nephrolitiasis.
Predisposição a acidose metabólica induzida por topiramato
RESUMO ¾ INTRODUÇÃO: Acidose metabólica induzida por topiramato é evento adverso pouco frequente, mas bem documentado. Nosso objetivo foi demonstrar a diminuição dos níveis de dióxido de carbono, muitas vezes assintomática, mas que pode predispor ao aparecimento de acidose metabólica. MATERIAL E MÉTODO: Avaliamos prospectivamente os níveis de dióxido de carbono de 18 pacientes acompanhados no ambulatório de epilepsia infantil da UNICAMP, antes e 3 meses após o início do uso de topiramato. RESULTADOS: Foram avaliados 18 pacientes com idade entre 2 e 16 anos (média = 9,3 anos). Cinco pacientes eram do sexo feminino e 13, do sexo masculino. Os níveis médios de dióxido de carbono antes e após o uso de topiramato foram 25 e 21,2 mmol/L (normal = 22 a 30), respectivamente. A dose de topiramato utilizada foi 2,08 a 11,76 mg/kg/dia (média = 6,7 mg/kg/dia). Os eventos adversos mais frequentes foram anorexia, nausea e sonolência. CONCLUSÃO: Concluímos que a diminuição dos níveis de dióxido de carbono induzida por topiramato é na maioria das vezes assintomática entretanto, pode predispor à ocorrência de acidose metabólica. Como o topiramato é utilizado frequentemente no tratamento de epilepsia de difícil controle, os pacientes em uso desta medicação quando submetidos à cirurgia de epilepsia devem ser monitorizados, principalmente durante o intraoperatório, quanto à possibilidade de apresentarem acidose metabólica.
PALAVRAS-CHAVE: topiramato, acidose metabólica, nefrolitíase.
Topiramate (TPM) is a new antiepileptic drug (AED) used mainly as adjunctive therapy in partial and secondary generalized epilepsy. Important side effects include dizziness, ataxia, headache, paresthesia, tremor, somnolence, cognitive dysfunction, confusion, agitation, depression, amnesia, emotional lability, diarrhoea, diplopia and weight loss 1. Despite being a safe drug, it may seldom be associated with serious adverse events such as kidney stones and metabolic acidosis 2.
The objective of this study was to analyze the carbon dioxide (CO2) serum levels of 18 patients, before and during the use of TPM, since this drug may predispose the lowering of CO2 serum levels, which is usually asymptomatic but may facilitate the occurrence of metabolic acidosis.
METHOD
We evaluated, prospectively, the CO2 serum levels of patients seen at the epilepsy clinic of our University Hospital. All patients received TPM as adjunctive therapy for the treatment of severe epileptic encephalopaties and uncontrolled seizures.
Patients had to be receiving of at least one AED, had no history of kidney stones and could not be using of any carbonic anhydrase inhibitor either. Blood samples were drawn 15 days before and 3 months after TPM was introduced. Patients were seen monthly, and adverse events were assessed by physical examination and parent and/or children interview.
Statistical analysis was done using the Student "t" test for paired mean samples, with a significance level of 0.05.
RESULTS
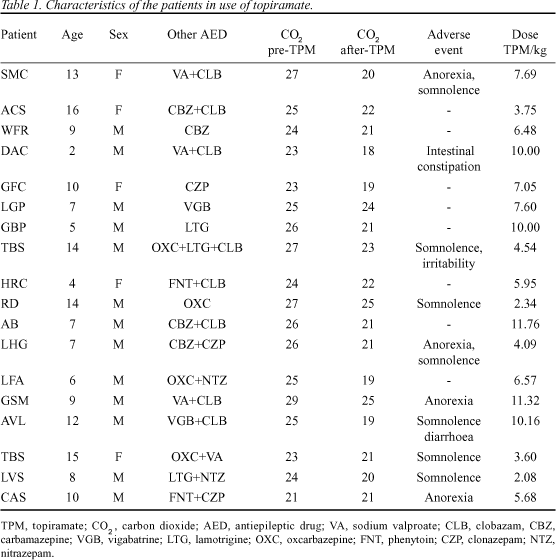
Eighteen patients were evaluated, 13 male and 5 were female. Ages ranged from 2 to 16 years-old (mean=9.3 years) and they were all using one or more AED when TPM was introduced (Table 1).
Carbon dioxide serum levels varied from 21 to 29 mmol/L (mean = 25) and 18 to 25mmol/L (mean = 21.2), 15 days before and 3 months after starting TPM, respectively, which was statistically significant (p=6.7 X10-8). The TPM dose varied from 2.08 mg/Kg to 11.6 mg/Kg (mean = 6.7).
Ten patients presented adverse events characterized by anorexia, somnolence, diarrhoea, irritability and intestinal constipation (Table 1).
DISCUSSION
Topiramate has three properties thought to be antiepileptic: state-dependent blocking of sodium channels, potentiation of GABA (g-aminobutyrate), and antagonistic effect on the kainate/AMPA (a-amino-3-hydroxy-5-methylisoxazole-4-propionic acid) subtype of receptor. It also inhibits isoenzimes of carbonic anhydrase (CA-II and CA-IV) 2.
Despite being a safe drug, and most of the patients do not present any sign of metabolic acidosis (hyperventilation, confusion, etc.), this is a well documented adverse event induced by TPM 2-5. Metabolic acidosis is probably due to the carbonic anhydrase inhibitory effect of TPM, which increases the bicarbonate lost in the urine, and consequently, decreases CO2 serum levels 2.
Takeoka et al. 4 reported a 10% lowering in the bicarbonate serum levels in 20 out of 29 children receiving TPM, showing a clear predisposition to metabolic acidosis. After TPM withdrawal, bicarbonate levels returned to baseline.
Despite our data showed that most of the patients had a decrease in CO2 serum levels, they presented only mild adverse events, and only one had to stop taking TPM due to anorexia. It is hard to know if there is any relationship between CO2 levels and mild adverse events. Although none of our patients presented metabolic acidosis, the decrease in CO2 serum levels may facilitate the occurrence of severe metabolic acidosis in situations where TPM is associated to the ketogenic diet, renal disease, dialysis, diarrhea or surgery. The combination of TPM and ketogenic diet or acetazolamide is contraindicated, not only because the increased risk of metabolic acidosis, but it may increase the occurrence of nephrolitiasis as well.
The occurrence of kidney stones induced by oral administration of carbonic anhydrase inhibitors in rats has been demonstrated by Molon-Noblot et al. 6 . The carbonic anhydrase inhibitory effect of TPM decreases the citrate excretion, with no effect on urinary calcium excretion. The hypocitraturia caused by TPM is probably the major risk factor for nephrolitiasis 7.
It is believed that nephrolitiasis is an idiosyncratic event, and it is not related to the TPM dose. In a study of 1200 patients, nephrolitiasis was seen in 18 patients (1.5%) treated with TPM. They were all male, and ages ranged from 21 to 54 years old. None needed open surgery and ¾ had spontaneous recovery 8. The incidence of 1.5% of nephrolitiasis associated with TPM is similar to those presented by other carbonic anhydrase drugs, such as acetazolamide 9,10.
One of the authors (CAMG) followed two patients with clear history of nephrolitiasis after TPM treatment. One of them, a 17 year-old boy using 400 mg/day of TPM for 4 months and the other a 32 year-old female using 200 mg/day of TPM for 8 months. She had temporal lobe epilepsy with hippocampal atrophy shown by MRI, and she was seizure free after TPM therapy, but when the drug was tapered down seizures relapsed. It was decided to restart TPM and she is seizure free again, with no new episode of nephrolitiasis in a follow up period of 1 year. Guerreiro et al. 11 reported a 2.5 year-old boy presenting "sand in the urine" after TPM therapy, which probably represented the early stages of nephrolitiasis. Symptoms disappeared after the withdrawal of the drug.
Patients receiving TPM may develop acute confusional state without changing TPM dosages. This condition may be difficult to diagnose, unless it is clinically suspected. The withdrawal of TPM (or even only a decrease in its dosage) causes total recovery 3. Severe metabolic acidosis was reported by Wilner et al. 2 during epilepsy surgery. Carbon dioxide serum levels were 28/29 mM and 24 mM before and after introducing TPM, respectively (both within normal limits). The authors believe that the metabolic acidosis was caused by an increase in the bicarbonate excretion by TPM, which became clinically significant during surgery. During the week prior to the surgery, the patient had presented diarrhea, which may have contributed to the clinical picture, by increasing the loss of alkali.
Since TPM is currently used mainly in patients with severe forms of refractory epilepsies, some might need epilepsy surgery. The decrease in CO2 serum levels, usually precedes the changes of serum pH, therefore, the monitoring of CO2 or bicarbonate is more precise in detecting subtle changes of acid-basic equilibrium. It is important to follow the bicarbonate and/or CO2 serum levels throughout visits and specially during surgery, for the risk of metabolic acidosis.
Acknowledgments - The authors thank José do Nascimento Oliveira from the Núcleo de Informática ¾ FCM/UNICAMP, for his valuable help with the statistical analysis.
Dra. Maria Augusta Montenegro ¾ Departamento de Neurologia, FCM / UNICAMP ¾ Caixa Postal 6111 ¾ 13083-970 Campinas SP ¾ Brasil.
- 1. Reife RA. Topiramate. In: Shorvon S, Dreifuss F, Fish D, Thomas D (eds). The treatment of epilepsy. London: Blackwell Science, 1996:471-481.
- 2. Wilner A, Raymond K, Pollard R. Topiramate and metabolic acidosis. Epilepsia 1999;40:792-795.
- 3. Stowe CD, Bollinger T, James LP, Haley TM, Griebel ML, Farrar HC 3rd. Acute mental status changes and hyperchloremic metabolic acidosis with long-term topiramate therapy. Pharmacotherapy 2000;20:105-109.
- 4. Takeoka M, Holmes GL, Thiele E, et al. Topiramate and metabolic acidosis in pediatric epilepsy. Epilepsia 1999; 40:Suppl 7.
- 5. Tartara A, Sartori I, Manni R, Galimberti CA, DiFazio M, Perucc E. Efficacy and safety of topiramate in refractory epilepsy: a long-term prospective study. Ital J Neurol Sci 1996;17:429-432.
- 6. Molon-Noblot S, Boussiguet-Leroux, Owen RA, et al. Rat urinary bladder hyperplasia induced by oral administration of carbonic nhydrase inhibitors. Toxicol Pathol 1992;20:93-102.
- 7. Wasserstein AG, Rak I, Reife RA. Investigation of the mechanistic basis of topiramate-associated nephrolitiasis : examination of urinary and serum constituents. Epilepsia 1995;36 (Suppl 3):S153.
- 8. Shorvon SD. Safety of topiramate : adverse events and relationship to dosing. Epilepsia 1996;37(Suppl 2):518-522.
- 9. Glauser TA. Topiramate. Sem Ped Neurol 1997;4:34-42.
- 10. Wasserstein AG, Rak I, Reife RA. Nephrolitiasis during treatment with topiramate. Epilepsia 1995;36(Suppl 3):S153.
- 11. Guerreiro MM, Manreza MLG, Guerreiro CAM, et al. A pilot study of topiramate in children with Lennox-Gastaut syndrome. Arq Neuropsiquiat 1999; 57:167-175.
Publication Dates
-
Publication in this collection
01 Dec 2000 -
Date of issue
Dec 2000
History
-
Accepted
18 July 2000