Abstracts
OBJECTIVE: central neurocytoma is a low grade tumor of neuroglial origin and a relatively new histological entity. Only a few cases have been reported and its biological behavior is still uncertain. Some cases have shown an aggressive behavior (local recurrence, malignant dedifferentiation or CSF dissemination) and challenged the initial view of its relative benignity. A case of central neurocytoma with peritoneal dissemination is presented. CASE: a six years-old boy with recurrent neurocytoma of III ventricle and left thalamus showed fast growth of tumor rest and ascites three and a half years after subtotal removal of the lesion. Tumor cells were identified in the ascitic fluid and implanted in the peritoneum. Chemotherapy was initiated immediately after diagnosis of peritoneal dissemination (etoposide, carboplatin, doxorubicin and cyclophosphamide). The patient developed metabolic imbalance and respiratory failure due to rapid formation of ascitic fluid and died 3 days after the diagnosis of peritoneal dissemination was established. CONCLUSION: central neurocytoma is a low grade tumor with low values of the proliferative index in the majority of cases. In spite of that, some tumors may present a very aggressive behavior and extraneural dissemination. Evaluation of proliferative index may be a guideline parameter for planning adjuvant therapies after surgical treatment in selected cases. Extraneural dissemination may occur in some cases specially in patients with ventriculoperitoneal shunt.
central neurocytoma; intraventricular tumors; neuroglial tumor; peritoneal dissemination
OBJETIVO: Neurocitoma central é tumor de baixo grau, de origem neuroglial e uma entidade neurológica relativamente nova. Poucos casos foram relatados na literatura e seu comportamento biológico é ainda incerto. Alguns casos apresentam comportamento agressivo (recorrência local, diferenciação maligna ou disseminação liquórica) e desafiam a visão inicial de sua relativa benignidade. Apresentamos um caso de neurocitoma central com disseminação peritonial. CASO: Um menino de seis anos de idade com neurocitoma de III ventrículo e tálamo esquerdo recorrente apresentou rápido crescimento do resto tumoral e ascite três anos e meio após ressecção parcial da lesão. Células tumorais foram identificadas no fluido ascítico e implantadas no peritônio. Quimioterapia foi iniciada imediatamente após o diagnóstico de disseminação peritonial (etoposide, carboplatina, doxorubicina e ciclofosfamida). O paciente desenvolveu instabilidade metabólica e insuficiência respitatória devido à rápida formação de fluido ascítico e foi a óbito 3 dias após o estabelecimento do diagnóstico de disseminação peritonial. CONCLUSÃO: Neurocitoma central é tumor de baixo grau com baixos índices de proliferação na maioria dos casos. Apesar disto, alguns tumores podem apresentar comportamento muito agressivo e disseminação extraneural. A avaliação do índice proliferativo pode ser parâmetro indicativo para o planejamento de terapias adjuvantes pós tratamento cirúrgico em casos selecionados. Dissseminação extraneural pode ocorrer em alguns casos, especialmente em pacientes com derivação ventriculoperitonial.
neurocitoma central; tumores intraventriculares; tumor neuroglial; disseminação peritonial
Peritoneal dissemination from central neurocytoma: case report
Disseminação peritonial de neurocitoma central: relato de caso
Maurício Coelho NetoI; Ricardo RaminaI; Murilo Sousa de MenesesI; Walter Oleschko ArrudaII; Jerônimo Buzetti MilanoI
Instituto de Neurologia de Curitiba, Curitiba PR, Brazil
INeurosurgical Department
IINeurologist
ABSTRACT
OBJECTIVE: central neurocytoma is a low grade tumor of neuroglial origin and a relatively new histological entity. Only a few cases have been reported and its biological behavior is still uncertain. Some cases have shown an aggressive behavior (local recurrence, malignant dedifferentiation or CSF dissemination) and challenged the initial view of its relative benignity. A case of central neurocytoma with peritoneal dissemination is presented.
CASE: a six years-old boy with recurrent neurocytoma of III ventricle and left thalamus showed fast growth of tumor rest and ascites three and a half years after subtotal removal of the lesion. Tumor cells were identified in the ascitic fluid and implanted in the peritoneum. Chemotherapy was initiated immediately after diagnosis of peritoneal dissemination (etoposide, carboplatin, doxorubicin and cyclophosphamide). The patient developed metabolic imbalance and respiratory failure due to rapid formation of ascitic fluid and died 3 days after the diagnosis of peritoneal dissemination was established.
CONCLUSION: central neurocytoma is a low grade tumor with low values of the proliferative index in the majority of cases. In spite of that, some tumors may present a very aggressive behavior and extraneural dissemination. Evaluation of proliferative index may be a guideline parameter for planning adjuvant therapies after surgical treatment in selected cases. Extraneural dissemination may occur in some cases specially in patients with ventriculoperitoneal shunt.
Key words: central neurocytoma, intraventricular tumors, neuroglial tumor, peritoneal dissemination.
RESUMO
OBJETIVO: Neurocitoma central é tumor de baixo grau, de origem neuroglial e uma entidade neurológica relativamente nova. Poucos casos foram relatados na literatura e seu comportamento biológico é ainda incerto. Alguns casos apresentam comportamento agressivo (recorrência local, diferenciação maligna ou disseminação liquórica) e desafiam a visão inicial de sua relativa benignidade. Apresentamos um caso de neurocitoma central com disseminação peritonial.
CASO: Um menino de seis anos de idade com neurocitoma de III ventrículo e tálamo esquerdo recorrente apresentou rápido crescimento do resto tumoral e ascite três anos e meio após ressecção parcial da lesão. Células tumorais foram identificadas no fluido ascítico e implantadas no peritônio. Quimioterapia foi iniciada imediatamente após o diagnóstico de disseminação peritonial (etoposide, carboplatina, doxorubicina e ciclofosfamida). O paciente desenvolveu instabilidade metabólica e insuficiência respitatória devido à rápida formação de fluido ascítico e foi a óbito 3 dias após o estabelecimento do diagnóstico de disseminação peritonial.
CONCLUSÃO: Neurocitoma central é tumor de baixo grau com baixos índices de proliferação na maioria dos casos. Apesar disto, alguns tumores podem apresentar comportamento muito agressivo e disseminação extraneural. A avaliação do índice proliferativo pode ser parâmetro indicativo para o planejamento de terapias adjuvantes pós tratamento cirúrgico em casos selecionados. Dissseminação extraneural pode ocorrer em alguns casos, especialmente em pacientes com derivação ventriculoperitonial.
Palavras-chave: neurocitoma central, tumores intraventriculares, tumor neuroglial, disseminação peritonial.
Since its first description by Hassoun in 19821, central neurocytomas have been reported as a low grade neuroglial tumor, and in the great majority of cases their biological behavior matches the histological findings. Due to its low incidence, the biological behavior of this tumor is still uncertain. Some cases of neurocytoma with aggressive behavior, changes in the histologic pattern (e.g. atypia), local recurrence and cerebrospinal fluid (CSF) dissemination have been described in the literature. These observations have changed the initial concept of ''benign lesion'' with indication for adjuvant treatment (radiotherapy) in aggressive tumors2-11.
This case is the first one reporting extraneural tumor dissemination of a central neurocytoma and emphasizes the clinical and histological malignant behavior that central neurocytomas may present.
CASE
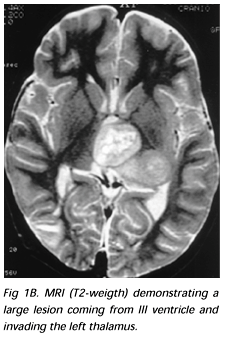
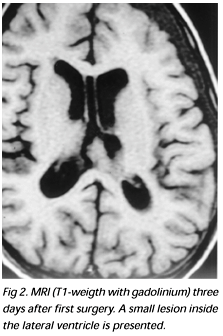
A 3-year-old boy presented right hand dystonia and signs of intracranial hypertension. MRI and CT scan examinations disclosed a very large lesion, with calcifications, inside the III ventricle, with extension to the left thalamus (Figs 1A, 1B). As the patient presented hydrocephalus, a ventriculoperitoneal shunt was inserted. The patient was referred to our clinic for treatment of the tumor. A stereotactic biopsy was performed and the initial neuropathological evaluation indicated a low grade glioma. He underwent stereotactic-guided surgery, and the tumor was subtotally resected, with a small lesion left inside the lateral ventricle (Fig 2). The definitive pathological diagnosis was central neurocytoma (Fig 3).
The postoperative period was uneventful and the patient showed no further deficit. Radiation therapy or chemotherapy were not indicated due to the benign pathologic characteristics (neither atypias nor necrosis) of the lesion. Inspite of revision of the shunt 18 months after surgery, he had a very good evolution for two years (Karnofsky performance scale 100). Two years after tumor removal the patient remained asymptomatic but a follow up MRI examination showed tumor growth of residual tumor. A new subtotal resection was carried out.
Three and a half years after diagnosis the patient presented normal social and school life. But on the 43rd month of follow up, hydrocephalus and tumor growth led to increased intracranial pressure syndrome. An additional shunt was inserted from the left lateral ventricle to the peritoneum. A week later, the patient presented increased abdominal volume and vomiting. Abdominal ultrasound showed ascites. Peritoneal shunt dysfunction was suspected and the peritoneal catheter was removed and a ventriculoatrial shunt was placed. To relieve the respiratory distress caused by ascites, 1.3 liters of ascitic fluid was drained. Fluid analysis did not show acute or chronic infection. In spite of peritoneal catheters removal, ascites kept increasing and a videolaparoscopy revealed the presence of small nodular lesions inside the peritoneal cavity. A biopsy of these tumors was taken.
The patient's condition deteriorated rapidly due to metabolic imbalance and respiratory distress. Pathological examination of the peritoneal lesions showed highly malignant cells, with characteristics of central neurocytoma (Figure 4a, 4b). Chemotherapy (etoposide, carboplatin, doxorrubicine and cyclophosphamide) was initiated immediately after peritoneal dissemination was diagnosed. The patient died three days later.
DISCUSSION
Central neurocytoma, described in 1982 by Hassoun et al.1, is a tumor with controversial histogenesis. Some authors suggest its origin from the germinal periventricular matrix, which has the capability to differentiate into neuronal or glial tissue12. Other authors attribute a neuronal origin of neurocytomas due to the unique imunohistochemical or ultrastructural findings suggesting neuronal differentiation13-14. On the other hand, glial component, when present, is more frequent in extraventricular tumors15. This tumor is typically located inside the lateral ventricles, usually next to the Monro's foramen and septum pellucidum, and may reach the third ventricle and periventricular tissue. Its frequency is extremely low. It represents 0.5% of all intracranial tumors15-16. It is more frequent from 20 to 40 years of age (about 70% of described cases). It is extremely rare in children15. Due to benign characteristics and good prognosis reported in the literature, this tumor was classified as benign (grade I) by the WHO Classification until 19932.
The description of cases with poor evolution due to aggressive behavior, recurrence or CSF dissemination, leaded to change of classification of neurocytomas to grade II by the WHO in 1999 (low or uncertain malignant potential or borderline malignancy)2-11,15,17-19. Thirty-two patients were described as having aggressive tumor based on clinical signs, radiological changes (progression or recurrence) or by histological findings (e.g. atypias, vascular proliferation or necrosis)2-5,9-11,17,19-21. Only 6 cases presented intraneural CSF dissemination of the tumor3-4,9,22.This is the first case reporting CSF dissemination outside the CNS.
This case presented an initial good clinical evolution, but showed a form of progression unknown for this type of tumor. The actual pattern of biological behavior of the central neurocytomas is poorly understood. Schild et al.16 described a series of central neurocytomas with benign histology and postoperative survival rate of 80% in 5 years of follow up. Different treatment schemes, follow up criteria and clinical evolution have been observed in other series2-3,16,18,23-24.
This kind of tumor is extremely rare in children15. Compared to other low grade tumors in children with benign behavior and a long survival (more than 5 years), central neurocytomas in childhood may eventually behave as a high grade tumor. Calcification, a common finding in this kind of tumors, may reflect slow tumor growth25. The presence of high proliferation index areas, measured through MIB-1, might be helpful to predict recurrence and dissemination22.
Surgery of deep-seated brain tumors has greatly improved in the last 10 years. Stereotactic-guided neurosurgery and neuronavigation have improved the rate of total tumoral resection26. Surgical treatment of these tumors should take into account the invasion of the periventricular tissue. Complete resection is easier in purely intraventricular tumors, when compared to tumors with thalamic involvement. Several authors report extension of resection as a key factor in prognosis. Subtotal resection would lead to higher rates of recurrence and lower survival11,16,25,27. A literature review of 127 cases has showed that only about half cases the central neurocytomas were totally resected25. Extraventricular extension has been considered by some authors as a factor of bad prognosis27,28. Some authors have tried to correlate findings of positron emission tomography (PET) with prognosis24. Spectroscopy may be a helpful diagnostic tool to define differentiation patterns23. Measurement of proliferative index through MIB-1 has been shown to be a reliable prognostic factor. Tumors with a proliferative index higher than 2 have a tendency to present a worse prognosis2,6,8. The small number of cases reported in the literature limited, however, the interpretation of the results.
The use of radiation therapy has been indicated for cases with subtotal resection16. Radiation therapy would be helpful for its indirect effect, with thrombosis of feeding vessels to tumor remnants5. Some authors have disagreed with this procedure due to its low effect in tumors with low proliferative index5,29. Radiosurgery has been mentioned as a very effective adjuvant treatment for recurrences, achieving control of the disease in the totality of the reported cases in the literature18,30-31. Radiosurgery also could avoid re-operations for small recurrences30-31. Radiation therapy of tumor bed with doses ranging from 48 to 60 Gy is recommended5,16. A few cases of CSF dissemination were reported3-4,9,22 and radiation therapy of the neuraxis and the use of chemotherapy for intrathecal implants were indicated. To our knowledge, there are no descriptions of cases with extraneural metastasis of central neurocytoma caused by ventriculoperitoneal shunt. Adjuvant therapies should probably be indicated in cases like ours. Radiation therapy was not used at first hand in our case due to patient's low age at the time of diagnosis and the benign histological findings. Radiation therapy might be performed in cases of subtotal resection of high proliferative index tumors (>2) and/or vascular proliferation, tumors with cellular atypias and necrosis, and in cases of recurrence and tumor rest growth even with low proliferative index (<2). In cases with intrathecal implants cerebrospinal irradiation would be indicated.
There are very few reports using chemotherapy as adjuvant treatment3-4,22,32. A good indication for this procedure would be cases with CSF tumor dissemination and small children.
In conclusion, central neurocytoma should be considered as a potentially aggressive tumor with uncertain behavior. Distant implant inside and outside the neuroaxis is reported in the literature and in our case. Early distant implants should be sought with MRI examination of the neuroaxis and cytological studies of CSF. Radiosurgery could be indicated for cases with recurrence and for cases when a subtotal resection was achieved and the proliferative index is high.
Acknowledgments - We thank Luiz Fernando Bleggi Torres, MD, Ph.D. and Mario Montemor, MD for providing the pathological studies and pictures.
Received 12 March 2003, received in final form 2 July 2003. Accepted 16 July 2003
Dr. Maurício Coelho Neto - Rua Jeremias Maciel Perreto 300 - 81210-310 Curitiba PR - Brazil. FAX: 55 41 264-1238 - E-mail: mcoelho29@yahoo.com
- 1. Hassoun J, Gambarelli D, Grisoli F, et al. Central neurocytoma. An electron microscopic study of two cases. Acta Neuropathol (Berl) 1982;56:151-156.
- 2. Ashkan K, Casey ATH, D'Arrigo C, et al. Benign central neurocytoma: a double misnomer ?. Cancer 2000;89:1111-1120.
- 3. Brandes AA, Amistà P, Gardiman M, et al. Chemotherapy in patients with recurrent and progressive central neurocytoma. Cancer 2000;88:169-174.
- 4. Eng DY, DeMonte F, Ginsberg L, et al. Craniospinal dissemination of central neurocytoma: report of two cases. J Neurosurg 1997;86:547-552.
- 5. Kim DG, Park SH, Kim IH, et al. Central neurocytoma: the role of radiation therapy and long term outcome. Cancer 1997;79:1995-2002.
- 6. Mackenzie IRA. Central neurocytoma: histologic atypia, proliferation potential, and clinical outcome. Cancer 1999;85:1606-1610.
- 7. Mrak RE. Malignant neurocytic tumor. Hum Pathol 1994;25:747-752.
- 8. Söylemezoglu F, Scheithauer BW, Esteve J, et al. Atypical central neurocytoma. J Neuropathol Exp Neurol 1997;56:551-556.
- 9. Tomura N, Hirano H, Watanabe O, et al. Central neurocytoma with clinically malignant behavior.AJNR Am J Neuroradiol, 1997:18:1175-1178.
- 10. von Deimling A, Janzer R, Kleihes P, et al. Patterns of differentiation in central neurocytoma. Acta Neuropathol 1990;79:473-479.
- 11. Yasargil MG, von Ammon K, von Deimling A, et al. Central neurocytoma: histopathological variants and therapeutic approaches. J Neurosurg 1992;76:32-37.
- 12. Valdueza JM, Westphal M, Vortmeyer A, et al. Central neurocytoma: clinical, immunohistologic, and biologic findings of a human neuroglial progenitor tumor. Surg Neurol 1996;45:49-56.
- 13. Lee MC, Nam JH, Choi C, et al. Ultrastructural characteristics of central neurocytoma in cell culture. Ultrastruct Pathol 1997;21:393-404.
- 14. Patt S, Schimdt H, Labrakakis C, et al. Human central neurocytoma cells show neuronal physiological properties in vitro. Acta Neuropathol 1996;91:209-214.
- 15. Figarella-Branger D, Söylemezoglu F, Kleihues P, et al. Central neurocytoma. In Kleihues P, Cavenee WK (eds) Pathology and genetics of tumors of the nervous system. Lyon: IARC Press, 2000:107-109.
- 16. Schild SE, Scheithauer BW, Haddock MG, et al. Central neurocytomas. Cancer 1997;79:790-795.
- 17. Kim DG, Ci JG, Park SH, et al. Intraventricular neurocytoma: clinicopathological analysis of seven cases. J Neurosurg 1992;76:759-765.
- 18. Kim DG, Kim JS, Chi JG, et al. Central neurocytoma : proliferative potential and biological behavior. J Neurosurg 1996;84:742-747.
- 19. Sgouros S, Carey M, Aluwihare N, et al. Central neurocytoma: a correlative clinicopathologic and radiologic analysis. Surg Neurol 1998;49:197-204.
- 20. Figarella-Branger D, Pellissier JF, Daumas-Duport C, et al. Central neurocytomas: critical evaluation of a small-cell neuronal tumor. Am J Surg Pathol 1992;16:97-109
- 21. Robbins P, Segal A, Narula S, et al. Central neurocytoma: a clinicopathological, immunohistochemical and ultrastructural study of 7 cases. Pathol Res Pract 1995;191:100-111.
- 22. Jay V, Edwards V, Hoving E, et al. Central neurocytoma: morphological, flow cytometric, polymerase chain reaction, fluorescence in situ hybridization, and karyotypic analyses. J Neurosurg 1999;90:348-354.
- 23. Kim DG, Choe WJC, Chang KH, et al. In vivo proton magnetic resonance spectroscopy of central neurocytoma. Neurosurgery 2000;46:329-334.
- 24. Mineura K, Sasajima, T, Itoh Y, et al. Blood flow and metabolism of Central neurocytoma. Cancer 1995;76:1224-1232.
- 25. Hassoun J, Soylemezoglu F, Gambarelli D, et al. Central neurocytoma: a synopsis of clinical and histological features. Brain Pathol 1993;3:297-306.
- 26. Ramina R, Neto MC, Meneses M, et al. Management of deep-seated gliomas. Crit Rev Neurosurg 1999;9:34-40.
- 27. Nishio S, Takeshita I, Kaneko Y, et al. Central neurocytoma: a new subset of benign neuronal tumors of the cerebrum. Cancer 1992;70:529-537.
- 28. Wichmann W, Schubiger O, von Deimling A, et al. Neuroradiology of central neurocytoma. Neuroradiology 1991;33:143-148.
- 29. Hara A, Araki Y, Shinoda J, et al. Central neurocytoma: proliferative assessment of nucleolar organizer region staining. Surg Neurol 1993;39:343-347.
- 30. Anderson RC, Elder JB, Parsa AT, et al. Radiosurgery for treatment of central neurocytomas. Neurosurgery 2001;48:1231-1238.
- 31. Pollock BE, Stafford SL. Stereotactic radiosurgery for recurrent central neurocytoma: case report. Neurosurgery 2001;48:441-443.
- 32. Odds D, Nonis J, Metha M, et al. Central neurocytoma: a clinical study of response to chemotherapy. J Neurooncol 1997;34:279-283.
Publication Dates
-
Publication in this collection
06 Jan 2004 -
Date of issue
Dec 2003
History
-
Reviewed
02 July 2003 -
Received
12 Mar 2003 -
Accepted
16 July 2003