Abstracts
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease that affects motor neurons in the cerebral cortex, brainstem, and spinal cord, brain regions in which conventional magnetic resonance imaging is often uninformative. Although the mean time from symptom onset to diagnosis is estimated to be about one year, the current criteria only prescribe magnetic resonance imaging to exclude "ALS mimic syndromes". Extensive application of non-conventional magnetic resonance imaging (MRI) to the study of ALS has improved our understanding of the in vivo pathological mechanisms involved in the disease. These modern imaging techniques have recently been added to the list of potential ALS biomarkers to aid in both diagnosis and monitoring of disease progression. This article provides a comprehensive review of the clinical applicability of the neuroimaging progress that has been made over the past two decades towards establishing suitable diagnostic tools for upper motor neuron (UMN) degeneration in ALS.
amyotrophic lateral sclerosis; primary lateral sclerosis; diffusion tensor imaging; proton spectroscopy; magnetic resonance imaging
A esclerose lateral amiotrófica (ELA) é uma doença neurodegenerativa fatal que afeta os neurônios motores em regiões nas quais a ressonância magnética (RM) é frequentemente pouco informativa. Embora o tempo médio desde a manifestação inicial até o diagnóstico esteja em torno de um ano, os critérios atuais apenas recomendam o emprego da RM para excluir as "síndromes mimetizadoras da ELA". A maior aplicação da RM não convencional tem melhorado nossa compreensão sobre os mecanismos patológicos in vivo envolvidos na ELA. Estas modernas técnicas de imagem foram adicionadas à lista de potenciais biomarcadores da ELA, contribuindo para o diagnóstico e para a monitorização da progressão da doença. Esta é uma revisão detalhada da aplicabilidade clínica dos recentes avanços da neuroimagem, que visa apontar as ferramentas mais apropriadas para o diagnóstico da degeneração do neurônio motor superior (NMS).
esclerose lateral amiotrófica; esclerose lateral primária; imagem de tensor por difusão; espectroscopia de prótons; ressonância magnética
VIEWS AND REVIEWS
Is magnetic resonance imaging a plausible biomarker for upper motor neuron degeneration in amyotrophic lateral sclerosis/primary lateral sclerosis or merely a useful paraclinical tool to exclude mimic syndromes? A critical review of imaging applicability in clinical routine
A ressonância magnética é um biomarcador aceitável da degeneração do neurônio motor superior em esclerose lateral amiotrófica/esclerose lateral primária ou apenas um instrumento paraclínico útil para a exclusão das síndromes mimetizadoras? Uma revisão crítica da aplicabilidade da imagem na rotina clínica
Antonio José da RochaI,II; Antonio Carlos Martins Maia JúniorI,II
IMD PhD; Division of Neuroradiology, Fleury Medicina e Saúde, São Paulo SP, Brazil
IIMD PhD; Division of Neuroradiology, Santa Casa de Misericórdia de São Paulo, São Paulo SP, Brazil
Correspondence Correspondence: Antonio José da Rocha Rua Dr. Cesário Motta Junior 112 01221-020 São Paulo SP Brasil E-mail: a.rocha@uol.com.br
ABSTRACT
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease that affects motor neurons in the cerebral cortex, brainstem, and spinal cord, brain regions in which conventional magnetic resonance imaging is often uninformative. Although the mean time from symptom onset to diagnosis is estimated to be about one year, the current criteria only prescribe magnetic resonance imaging to exclude "ALS mimic syndromes". Extensive application of non-conventional magnetic resonance imaging (MRI) to the study of ALS has improved our understanding of the in vivo pathological mechanisms involved in the disease. These modern imaging techniques have recently been added to the list of potential ALS biomarkers to aid in both diagnosis and monitoring of disease progression. This article provides a comprehensive review of the clinical applicability of the neuroimaging progress that has been made over the past two decades towards establishing suitable diagnostic tools for upper motor neuron (UMN) degeneration in ALS.
Key words: amyotrophic lateral sclerosis, primary lateral sclerosis, diffusion tensor imaging, proton spectroscopy, magnetic resonance imaging.
RESUMO
A esclerose lateral amiotrófica (ELA) é uma doença neurodegenerativa fatal que afeta os neurônios motores em regiões nas quais a ressonância magnética (RM) é frequentemente pouco informativa. Embora o tempo médio desde a manifestação inicial até o diagnóstico esteja em torno de um ano, os critérios atuais apenas recomendam o emprego da RM para excluir as "síndromes mimetizadoras da ELA". A maior aplicação da RM não convencional tem melhorado nossa compreensão sobre os mecanismos patológicos in vivo envolvidos na ELA. Estas modernas técnicas de imagem foram adicionadas à lista de potenciais biomarcadores da ELA, contribuindo para o diagnóstico e para a monitorização da progressão da doença. Esta é uma revisão detalhada da aplicabilidade clínica dos recentes avanços da neuroimagem, que visa apontar as ferramentas mais apropriadas para o diagnóstico da degeneração do neurônio motor superior (NMS).
Palavras-Chave: esclerose lateral amiotrófica, esclerose lateral primária, imagem de tensor por difusão, espectroscopia de prótons, ressonância magnética.
Amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig's disease, is a rare, chronic, fatal degenerative neurological disease that predominantly affects the motor system. Diagnosis of ALS is based on the presence of very characteristic clinical findings of upper motor neuron (UMN) compromise associated with lower motor neuron (LMN) affection in conjunction with the exclusion of "ALS mimic syndromes"1-3.
Clinical subtypes of motor neuron degeneration with only LMN degeneration, also called progressive muscular atrophy, or only UMN degeneration, called primary lateral sclerosis (PLS), and subtypes with predominant upper or LMN degeneration exist. Clinical UMN signs are initially absent in 7-10% of ALS cases. Therefore, delayed diagnosis (>18 months) is common in patients who present with isolated LMN signs. This pronounced delay between the onset of symptoms and diagnosis is possibly beyond the therapeutic window4.
Until recently, standard diagnostic criteria for ALS5,6, which were designed primarily for research purposes, have not been able to detect early disease. However, the Awaji criteria have recently been introduced to better define LMN degeneration, which has improved the sensitivity of early diagnostic methods for ALS7. In the Awaji criteria, needle electromyography is considered as an extension of the clinical examination, but the general principles of previous criteria are maintained. However, even if diagnosis can now be established earlier, clinicians need a reliable, objective biomarker that allows periodic assessment of the changes in disease progression or treatment efficacy.
Whereas the presence of LMN signs can be ascertained by electromyography, there is no widely accepted marker for UMN involvement. Paraclinical proof of UMN compromise is a challenging task, particularly in the early stages of the disease3,8. In the process of searching for UMN biomarkers, a broad spectrum of conventional and non-conventional magnetic resonance imaging (MRI) techniques, including proton magnetic resonance spectroscopy (1H-MRS), functional MRI (fMRI), diffusion tensor imaging (DTI), and magnetization transfer imaging (MTI) has been used with variable success in identifying neuropathological processes.
Previous reports have supported that MRI should be included in the workup of any patient with weakness and pyramidal signs9-11. Furthermore, some authors have suggested new MR techniques for the list of potential ALS biomarkers4,12-14. This article aimed to review the clinical applicability of the neuroimaging progress that has been made over the past two decades towards establishing suitable diagnostic tools of UMN disease.
CONVENTIONAL MRI
According to the current criteria, there are no neuroimaging tests that confirm the diagnosis of ALS6,7. However, in clinical practice, conventional MRI is performed in most ALS patients to identify a focal structural lesion that might account for a significant portion of the patient's symptoms. The vast list of brain disorders that mimic ALS may include skull base lesions, cervical myelopathies, conus lesions, and thoracolumbar sacral radiculopathy15,16.
Although degeneration of the entire corticospinal tract (CST) occurs in approximately 47% of autopsy specimens from ALS patients17, the demonstration of such abnormalities is limited on conventional MR acquisitions18,19. Areas of abnormal signal intensity in the CST documented on T1-weighted images (T1WI) and T2-weighted images (T2WI), including fluid-attenuated inversion recovery (FLAIR) images, have been previously reported in patients with ALS20-26. However, these abnormalities were inconsistent and unreliable because they were observed frequently in normal patients, and they do not correlate with clinical scores (Fig 1). T2-hyperintensity of the CST has low sensitivity (approximately ≤40%) and limited specificity (approximately ≤70%)12.
A thin line of cortical low signal intensity ("motor dark line" or "hypointense rim") of the precentral gyrus on T2WI or FLAIR images has been advocated to be a marker of UMN compromise in ALS, particularly in advanced disease20,27. However, this T2 shortening effect results from excessive iron deposition, fibrillary gliosis, and/or macrophage infiltration, and such a change is neither sensitive nor specific to the pathology of UMN degeneration in ALS and can be found in healthy people, as well as in those with other degenerative diseases10,28-30.
Precentral gyrus subcortical hyperintensity on T2WI or FLAIR images has been reported as an important finding that represents CST abnormalities in UMN degeneration20,27,29. This hyperintensity is probably due to ALS-related degenerative changes and contributes to the diagnosis of ALS in some studies, although with a low sensitivity20,28,31. Central sulcus enlargement and CST hyperintensity on FLAIR images have demonstrated low sensitivity and specificity for identifying ALS patients with pathological hyperreflexia or definite UMN signs and for confirming PLS32,33.
FLAIR images appear to have a higher sensitivity but lower specificity than fast spin-echo sequences (FSE) for detecting brain changes in ALS patients32,33. However, Hecht et al.28 have recommended caution in the evaluation of FLAIR images as they observed CST hyperintensity in both patients with ALS and control subjects. Gawne-Cain et al.34 have studied the range of white matter appearance on FLAIR images in healthy persons and showed increased signal intensity in the CST region at the level of the internal capsule in all subjects; this change extended up toward the centrum semiovale and down to the pons. In a previous report, we were not able to distinguish ALS patients based only on CST analysis in FLAIR images and found a large false positive occurrence10. Ngai et al.35 reported a statistically significant relationship between increasing age and the frequency of hyperintensity of the subcortical white matter of the precentral gyrus and hypointensity of the precentral gyrus gray matter on FLAIR images, which reinforces the idea that these signs may be present in the absence of UMN degeneration.
Compared to conventional T2-weighted sequences, proton density-weighted images (PDWI) definitely have greater specificity for the diagnosis of ALS (Fig 2). As PDWI is usually not performed in routine MRI, a radiologist must conduct the appropriate protocol to maximize the sensitivity of this method19,24,28. Cheung et al.23 noticed that a well-defined high signal in the CST on PDWI has specificity for ALS of 100% and a sensitivity of 4160%.
Hyperintensity on T1WI in the anterolateral column of the cervical cord has been observed in patients with ALS36, and intracranial anterolateral column hyperintensity has been observed in patients with predominant UMN signs, including spastic paraparesis26. However, spinal cord imaging and T1 analysis are not currently encouraged37.
Atrophy of the precentral gyrus may be identified indirectly as enlargement of the adjacent central sulcus in PLS and in ALS patients. The prominent atrophy in PLS patients has been explained by the long duration of the disease process in PLS as a result of longer survival. Although visualization of a clearly enlarged central sulcus may be helpful for the presence of motor cortex atrophy, it is not sensitive to identifying all phenotypes of UMN affection, and a reliable determination usually requires quantitative evaluation38 Furthermore, selective brain atrophy is difficult to recognize in early ALS patients and demands three-dimensional (3D) MR sequences and post-processed volumetric acquisitions.
Magnetic resonance voxel-based morphometry (VBM) involves automated segmentation and quantification of gray and white matter volumes to study regional differences. Application of VBM in ALS is not devoted to identifying UMN degeneration, but it is useful to demonstrate non-motor involvement with a widespread decrease in gray matter volume even without frank dementia12.
NON-CONVENTIONAL MRI
Diffusion tensor imaging
DTI provides quantitative information about the magnitude and directionality of water diffusion in 3D space. Diffusion is anisotropic in white matter tracts because axonal membranes and myelin sheaths present barriers to the motion of water molecules. Diffusivity is generally much higher in directions along fiber tracts than in the directions perpendicular to them. Mean diffusivity (MD) and fractional anisotropy (FA) are the most commonly used indices for estimating diffusion39. MD is a measure of the directionally averaged magnitude of diffusion and is related to the integrity of the local brain tissue. FA represents the degree of diffusion anisotropy and reflects the degree of alignment of cellular structures39. DTI techniques also allow interregional fiber tracking, known as diffusion tensor tractography, which allows tracking of the major WM tracts38.
DTI can provide important measures of UMN dysfunction. Changes in tissue structure including loss of pyramidal motor neurons in the primary motor cortex and axonal degeneration of the CST, proliferation of glial cells, extracellular matrix expansion, and intraneuron abnormalities may contribute to the observed CST DTI changes. DTI studies of patients with ALS have reported consistently decreased FA and increased MD values along the CST, using either region of interest-based approaches or tractography40-42. The posterior limb of the internal capsule is the place where the most pronounced decrease in FA and increase in MD have been demonstrated15,38.
Decreased FA in patients with ALS has been found to correlate with disease severity11,42,43, rate of disease progression44, and clinical38,45 and electrophysiological46 measures of UMN involvement.
The analysis of FA and MD support the view that ALS is a multisystem degenerative disease in which abnormalities of extra-motor areas play an important role in its pathophysiology, given that it has been possible to demonstrate that the abnormalities are not restricted to the motor tract (Fig 3) but also extend to extra-motor regions, including the corpus callosum, frontal and parietal WM, insula, and hippocampal formation14,38.
PROTON MR SPECTROSCOPY
Proton MR spectroscopy (1H-MRS) is a method to detect and quantify tissue neurodegenerative changes by analyzing the levels of metabolites in vivo. The MR spectrum is a spectral graph in which the x-axis denotes the unique chemical composition of a metabolite in parts per million (ppm), and the y-axis reflects the concentration of that metabolite. The predominant brain metabolites detected by 1H-MRS are N-acetyl aspartate (NAA), a marker for neuronal integrity; choline (Cho), related to membrane turnover and cell proliferation; creatine (Cr), a marker of tissue energy state; and myoinositol, an astrocytic marker.
Proton MR spectroscopy can demonstrate the loss or dysfunction of motor neurons by reducing both the concentrations of NAA47 or ratios of NAA:Cr and NAA:Cho43. Pioro et al.48 reported that patients with motor neuron disease showed a decreased ratio of NAA:Cr in the primary motor cortex compared with controls and noted that two patients with spinal muscular atrophy without UMN signs showed no decrease in this ratio. Although regional analysis of these data showed more pronounced 1H-MRS changes in the precentral gyrus and corona radiata13, NAA reduction in the brainstem has also been reported in ALS patients who have prominent UMN or bulbar signs49.
A moderate correlation has been shown between the NAA concentration and its ratios in the motor cortex and clinical manifestations of ALS48-50. In addition to neuronal damage, increased glial cell activity, reflected by raised levels of myoinositol, has been demonstrated in the motor cortex of patients with ALS50.
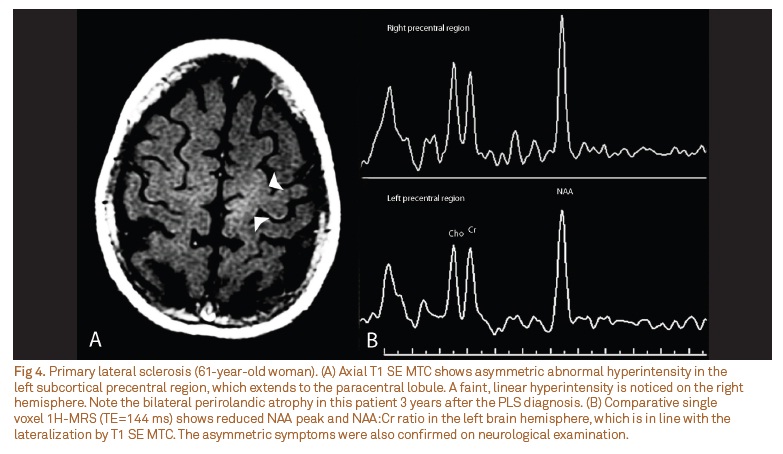
Increased myoinositol levels are probably associated with motor cortex hypointensity on T2WI51. Some authors have observed preeminent abnormalities on 1H-MRS that were associated with central sulcus enlargement and CST hyperintensity on FLAIR images32,33. The limits of the combined use of structural and metabolic MR techniques to define UMN degeneration are still not clearly established in the literature (Fig 4).
The application of 1H-MRS has been advocated as a surrogate marker for therapeutic efficacy. An increased NAA:Cr ratio in the motor cortex has been observed in some studies after a short course of treatment with riluzole52. It should be noted, however, that although the method can be used to monitor disease evolution, the diagnostic value of 1H-MRS is poor because of the considerable overlap of the metrics of patients with those of healthy controls15,16.
MAGNETIZATION TRANSFER IMAGING
MTI is based on the exchange of magnetization between spins in two proton pools: bound immobile protons associated with macromolecules (such as myelin) and free mobile protons associated with free water. Therefore, this technique can indicate the presence of structural changes in tissue associated with numerous diseases, even when they are not visible by other sequences, and allows qualitative and/or quantitative studies10.
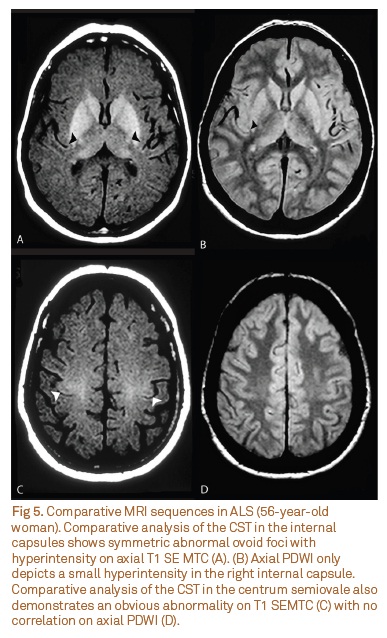
The magnitude of the effect depends on the relative water and macromolecular concentration, surface chemistry and biophysical dynamics of macromolecules and may be quantified by measuring the MT ratio (MTR). Typically, a low MTR indicates damage to myelin or to the axonal membrane, and therefore, MTI has been shown to be able to detect structural changes of the CST in ALS. We have reported CST hyperintensity, mainly in the supratentorial compartment, on T1-weighted spin-echo magnetization transfer contrast (T1 SE MTC), with great sensitivity (80%) and specificity (100%)10. This sequence is fast and simple to acquire, and it is particularly useful in early disease to demonstrate abnormal signal intensity in the CST in the centrum semiovale (Fig 5)9-11,53. This technique has recently been demonstrated to be highly specific to confirm a particular phenotype of imaging in ALS with UMN involvement11.
Magnetization transfer ratio studies applied to ALS have been inconsistent. In two of these studies, the authors found a decreased MTR in the CST, which ranged between 2.6 and 20% in patients with ALS compared to the control group54,55; however, in a recent publication by Charil et al.56, no significant differences were seen between patients and controls. It is assumed that reactive gliosis and axonal loss could have led to an MTR pseudonormalization in some cases56.
FUNCTIONAL MRI
fMRI allows noninvasive measurement of regional neuronal activity by determining the "blood oxygenation level dependent" or BOLD effect. This noninvasive technique is based on the increase in oxygen consumption and the secondary paramagnetic effect that alters the T2*-weighted MRI signal.
Functional MRI studies in patients with ALS have shown adaptive changes secondary to neuronal loss and demyelination of the CST, which were explained based on the phenomenon of neuronal plasticity and reorganization. fMRI might identify preserved, but non-executable, functions in ALS patients in the end stage and will set the direction for a new way of thinking about the functional capacities of these patients57. It is important to note that difficulty in controlling task performance in patients with ALS may be responsible for the variability of the results of fMRI studies16.
FRONTIERS AND PERSPECTIVES TO DEFINE IMAGING BIOMARKERS
Identification of imaging biomarkers that are sensitive and specific to ALS is not enough. A reliable imaging biomarker should recognize ALS phenotype particularities and identify those patients who are likely to have unusually fast or slow progression. In addition, imaging biomarkers should demonstrate specific involvement of the brainstem to ensure optimum and appropriate planning of care, including feeding and ventilation12.
Developing a tool that meets all of the requirements necessary to constitute an imaging biomarker of UMN degeneration in ALS patients is a great challenge. It demands comparable data in a large series of patients, including different stages and phenotypes of the disease4,16. In addition, it is important to be aware of how to transfer research results and their limitations to clinical practice, while addressing the needs of each patient. Individual patients must be interpreted with their particular clinical scenarios in mind, which demands imaging techniques that offer sensitive and specific results for variable phenotypes16.
Magnetization transfer imaging and DTI represent complementary, promising MR imaging techniques for diagnosing and studying early ALS. The T1 SE MTC technique can be easily implemented on many MR imagers to corroborate the clinical diagnosis of UMN involvement in ALS patients10,11,53. Quantitative measurement using DTI and 1H-MRS acquisitions on high-field magnets can be collected to identify UMN involvement and predict disease duration in patients with ALS43. DTI and MR VBM have the largest potential to become quantitative tools for evaluating anatomical integrity of the CST and motor and non-motor connectivity beyond the CST, respectively12,58.
The high sensitivity of MRI sequences to inherent cerebral motor and extra-motor pathology makes MRI a clear leader in the search for biomarkers. A reproducible MRI protocol was recently proposed with the aim of establishing a consensus on the various applications of MRI to the study of ALS and to explore the possibility of multicenter collaboration58.
FINAL REMARKS
Currently, MRI is a paraclinical tool that is useful in supporting ALS diagnosis. Due to the widespread availability of MRI and rapid evolution of the neuroimaging field, emerging imaging technologies might provide an opportunity for imaging in routine clinical practice with better sensitivity and specificity for confirming UMN degeneration.
Conventional MRI has shown low sensitivity and specificity in detecting UMN involvement in ALS/PLS, especially in the early stages of the disease. Our review encourages specialists to use T1 SE MTC, which has suitable sensitivity and high specificity for detecting early UMN degeneration. Advanced neuroimaging modalities such as 1H-MRS, DTI and MR VBM are emerging neuroimaging techniques that have a limited role in routine clinical practice but have shown promise in understanding the pathophysiology of motor neuron disease in vivo, depicting disease progression and identifying disease changes earlier, which will facilitate diagnosis in the future.
DTI is a promising candidate for an imaging marker in UMN disease, but it needs to be improved to offer confident diagnosis in individual patients. In the future, therefore, close cooperation among neurologists, neuroimaging researchers and neurophysiologists is mandatory to expand the knowledge and the comprehension of the natural history of ALS. An effective UMN imaging marker in ALS/PLS could emerge from the current knowledge of cross-sectional MRI combined with an approach using structural and functional techniques that will allow us to conduct comprehensive longitudinal studies.
Received 05 February 2012
Received in final form 23 February 2012
Accepted 02 March 2012
Conflict of interest: There is no conflict of interest to declare.
- 1. Bedlack RS. Amyotrophic lateral sclerosis: current practice and future treatments. Curr Opin Neurol 2010;23:524-529.
- 2. Orrell RW. Understanding the causes of amyotrophic lateral sclerosis. New Engl J Med 2007;357:822-823.
- 3. Rowland LP, Shneider NA. Amyotrophic lateral sclerosis. New Engl J Med 2001;344:1688-1700.
- 4. Pradat PF, Dib M. Biomarkers in amyotrophic lateral sclerosis: facts and future horizons. Mol Diagn Ther 2009;13:115-125.
- 5. Brooks BR. El Escorial World Federation of Neurology criteria for the diagnosis of amyotrophic lateral sclerosis. Subcommittee on Motor Neuron Diseases/Amyotrophic Lateral Sclerosis of the World Federation of Neurology Research Group on Neuromuscular Diseases and the El Escorial "clinical limits of amyotrophic lateral sclerosis" workshop contributors. J Neurol Sci 1994;124 Suppl:96-107.
- 6. Brooks BR, Miller RG, Swash M, Munsat TL, World Federation of Neurology Research Group on Motor Neuron D. El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 2000;1:293-299.
- 7. Carvalho M, Dengler R, Eisen A, et al. Electrodiagnostic criteria for diagnosis of ALS. Clinneurophysiol 2008;119:497-503.
- 8. Carvalho M, Swash M. Amyotrophic lateral sclerosis: an update. Curr Opin Neurol 2011;24:497-503.
- 9. Rocha AJ, Maia AC Jr., Nogueira RG, Lederman HM. Magnetic resonance findings in amyotrophic lateral sclerosis using a spin echo magnetization transfer sequence. Preliminary report. Arq Neuropsiquiatr 1999;57:912-915.
- 10. Rocha AJ, Oliveira AS, Fonseca RB, Maia AC, Jr., Buainain RP, Lederman HM. Detection of corticospinal tract compromise in amyotrophic lateral sclerosis with brain MR imaging: relevance of the T1-weighted spin-echo magnetization transfer contrast sequence. AJNR Am J Neuroradiol 2004;25:1509-1515.
- 11. Carrara G, Carapelli C, Venturi F, et al. A distinct MR imaging phenotype in amyotrophic lateral sclerosis: correlation between T1 magnetization transfer contrast hyperintensity along the corticospinal tract and diffusion tensor imaging analysis. AJNR Am J Neuroradiol 2012;33:733-739.
- 12. Turner MR, Kiernan MC, Leigh PN, Talbot K. Biomarkers in amyotrophic lateral sclerosis. Lancet Neurol 2009;8:94-109.
- 13. Mitsumoto H, Ulug AM, Pullman SL, et al. Quantitative objective markers for upper and lower motor neuron dysfunction in ALS. Neurology 2007;68:1402-1410.
- 14. Sage CA, Peeters RR, Gorner A, Robberecht W, Sunaert S. Quantitative diffusion tensor imaging in amyotrophic lateral sclerosis. Neuroimage 2007;34:486-499.
- 15. Agosta F, Chio A, Cosottini M, et al. The present and the future of neuroimaging in amyotrophic lateral sclerosis. AJNR Am J Neuroradiol 2010;31:1769-1777.
- 16. Chan S, Kaufmann P, Shungu DC, Mitsumoto H. Amyotrophic lateral sclerosis and primary lateral sclerosis: evidence-based diagnostic evaluation of the upper motor neuron. Neuroimaging Clin North Am 2003;13:307-326.
- 17. Brownell B, Oppenheimer DR, Hughes JT. The central nervous system in motor neurone disease. J Neurol Neurosurg Psychiatry 1970;33:338-357.
- 18. Guermazi A. Is high signal intensity in the corticospinal tract a sign of degeneration? AJNR Am J Neuroradiol 1996;17:801-802.
- 19. Comi G, Rovaris M, Leocani L. Review neuroimaging in amyotrophic lateral sclerosis. Eur J Neurol 1999;6:629-637.
- 20. Zhang L, Ulug AM, Zimmerman RD, Lin MT, Rubin M, Beal MF. The diagnostic utility of FLAIR imaging in clinically verified amyotrophic lateral sclerosis. J Magn Reson Imaging 2003;17:521-527.
- 21. Hecht MJ, Fellner C, Schmid A, Neundorfer B, Fellner FA. Cortical T2 signal shortening in amyotrophic lateral sclerosis is not due to iron deposits. Neuroradiology 2005;47:805-808.
- 22. Mascalchi M, Salvi F, Valzania F, Marcacci G, Bartolozzi C, Tassinari CA. Corticospinal tract degeneration in motor neuron disease. AJNR Am J Neuroradiol 1995;16 4 Suppl:878-880.
- 23. Cheung G, Gawel MJ, Cooper PW, Farb RI, Ang LC, Gawal MJ. Amyotrophic lateral sclerosis: correlation of clinical and MR imaging findings. Radiology 1995;194:263-270.
- 24. Hofmann E, Ochs G, Pelzl A, Warmuth-Metz M. The corticospinal tract in amyotrophic lateral sclerosis: an MRI study. Neuroradiology 1998;40:71-75.
- 25. Oba H, Araki T, Ohtomo K, et al. Amyotrophic lateral sclerosis: T2 shortening in motor cortex at MR imaging. Radiology 1993;189: 843-846.
- 26. Waragai M. MRI and clinical features in amyotrophic lateral sclerosis. Neuroradiology 1997;39:847-851.
- 27. Hecht MJ, Fellner F, Fellner C, Hilz MJ, Neundorfer B, Heuss D. Hyperintense and hypointense MRI signals of the precentralgyrus and corticospinal tract in ALS: a follow-up examination including FLAIR images. J Neurol Sci 2002;199:59-65.
- 28. Hecht MJ, Fellner F, Fellner C, Hilz MJ, Heuss D, Neundorfer B. MRI-FLAIR images of the head show corticospinal tract alterations in ALS patients more frequently than T2-, T1- and proton-density-weighted images. J Neurol Sci 2001;186:37-44.
- 29. Iwasaki Y, Ikeda K, Shiojima T, Tagaya M, Kurihara T, Kinoshita M. Clinical significance of hypointensity in the motor cortex on T2-weighted images. Neurology 1994;44:1181.
- 30. Imon Y, Yamaguchi S, Katayama S, et al. A decrease in cerebral cortex intensity on T2-weighted with ageing images of normal subjects. Neuroradiology 1998;40:76-80.
- 31. Thorpe JW, Moseley IF, Hawkes CH, MacManus DG, McDonald WI, Miller DH. Brain and spinal cord MRI in motor neuron disease. J Neurol Neurosurg Psychiatry 1996;61:314-317.
- 32. Bowen BC, Pattany PM, Bradley WG, et al. MR imaging and localized proton spectroscopy of the precentralgyrus in amyotrophic lateral sclerosis. AJNR Am J Neuroradiol 2000;21:647-658.
- 33. Chan S, Shungu DC, Douglas-Akinwande A, Lange DJ, Rowland LP. Motor neuron diseases: comparison of single-voxel proton MR spectroscopy of the motor cortex with MR imaging of the brain. Radiology 1999;212:763-769.
- 34. Gawne-Cain ML, Silver NC, Moseley IF, Miller DH. Fast FLAIR of the brain: the range of appearances in normal subjects and its application to quantification of white-matter disease. Neuroradiology 1997;39:243-249.
- 35. Ngai S, Tang YM, Du L, Stuckey S. Hyperintensity of the precentralgyral subcortical white matter and hypointensity of the precentralgyrus on fluid-attenuated inversion recovery: variation with age and implications for the diagnosis of amyotrophic lateral sclerosis. AJNR Am J Neuroradiol 2007;28:250-254.
- 36. Waragai M, Shinotoh H, Hayashi M, Hattori T. High signal intensity on T1 weighted MRI of the anterolateral column of the spinal cord in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 1997;62:88-91.
- 37. Pradhan S, Yadav R, Mishra VN, Aurangabadkar K, Sawlani V. Amyotrophic lateral sclerosis with predominant pyramidal signs early diagnosis by magnetic resonance imaging. Magn Reson Imaging 2006;24:173-179.
- 38. Wang S, Melhem ER, Poptani H, Woo JH. Neuroimaging in amyotrophic lateral sclerosis. Neurotherapeutics 2011;8:63-71.
- 39. Basser PJ, Pierpaoli C. Microstructural and physiological features of tissues elucidated by quantitative-diffusion-tensor MRI. 1996. J Magn Reson 1996;111:209-219.
- 40. Ellis CM, Simmons A, Jones DK, et al. Diffusion tensor MRI assesses corticospinal tract damage in ALS. Neurology 1999;53:1051-1058.
- 41. Toosy AT, Werring DJ, Orrell RW, et al. Diffusion tensor imaging detects corticospinal tract involvement at multiple levels in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 2003;74:1250-1257.
- 42. Cosottini M, Giannelli M, Siciliano G, et al. Diffusion-tensor MR imaging of corticospinal tract in amyotrophic lateral sclerosis and progressive muscular atrophy. Radiology 2005;237:258-264.
- 43. Wang S, Poptani H, Woo JH, et al. Amyotrophic lateral sclerosis: diffusion-tensor and chemical shift MR imaging at 3.0 T. Radiology 2006;239:831-838.
- 44. Ciccarelli O, Behrens TE, Altmann DR, et al. Probabilistic diffusion tractography: a potential tool to assess the rate of disease progression in amyotrophic lateral sclerosis. Brain 2006;129:1859-1871.
- 45. Cosottini M, Giannelli M, Vannozzi F, et al. Evaluation of corticospinal tract impairment in the brain of patients with amyotrophic lateral sclerosis by using diffusion tensor imaging acquisition schemes with different numbers of diffusion-weighting directions. J Comp Assist Tomogr 2010;34:746-750.
- 46. Iwata NK, Aoki S, Okabe S, et al. Evaluation of corticospinal tracts in ALS with diffusion tensor MRI and brainstem stimulation. Neurology 2008;70:528-532.
- 47. Pohl C, Block W, Karitzky J, et al. Proton magnetic resonance spectroscopy of the motor cortex in 70 patients with amyotrophic lateral sclerosis. Arch Neurol 2001;58:729-735.
- 48. Pioro EP, Antel JP, Cashman NR, Arnold DL. Detection of cortical neuron loss in motor neuron disease by proton magnetic resonance spectroscopic imaging in vivo. Neurology 1994;44:1933-1938.
- 49. Cwik VA, Hanstock CC, Allen PS, Martin WR. Estimation of brainstem neuronal loss in amyotrophic lateral sclerosis with in vivo proton magnetic resonance spectroscopy. Neurology 1998;50:72-77.
- 50. Kaufmann P, Pullman SL, Shungu DC, et al. Objective tests for upper motor neuron involvement in amyotrophic lateral sclerosis (ALS). Neurology 2004;62:1753-1757.
- 51. Young K, Govind V, Sharma K, Studholme C, Maudsley AA, Schuff N. Multivariate statistical mapping of spectroscopic imaging data. Magn Reson Med 2010;63:20-24.
- 52. Kalra S, Tai P, Genge A, Arnold DL. Rapid improvement in cortical neuronal integrity in amyotrophic lateral sclerosis detected by proton magnetic resonance spectroscopic imaging. J Neurol 2006;253: 1060-1063.
- 53. Alvarez-UríaTejero MJ, Sáiz Ayala A, Fernández Rey C, SantamartaLiébana ME, Costilla García S. Diagnosis of amyotrophic lateral sclerosis: advances in magnetic resonance imaging. Radiologia 2011;53:146-155.
- 54. Kato Y, Matsumura K, Kinosada Y, Narita Y, Kuzuhara S, Nakagawa T. Detection of pyramidal tract lesions in amyotrophic lateral sclerosis with magnetization-transfer measurements. AJNR Am J Neuroradiol 1997;18:1541-1547.
- 55. Tanabe JL, Vermathen M, Miller R, Gelinas D, Weiner MW, Rooney WD. Reduced MTR in the corticospinal tract and normal T2 in amyotrophic lateral sclerosis. Magn Reson Imaging 1998;16:1163-1169.
- 56. Charil A, Corbo M, Filippi M, et al. Structural and metabolic changes in the brain of patients with upper motor neuron disorders: amultiparametric MRI study. Amyotroph Lateral Scler 2009;10: 269-279.
- 57. Konrad C, Henningsen H, Bremer J, et al. Pattern of cortical reorganization in amyotrophic lateral sclerosis: a functional magnetic resonance imaging study. Exp Brain Res 2002;143:51-56.
- 58. Turner MR, Grosskreutz J, Kassubek J, et al.Towards a neuroimaging biomarker for amyotrophic lateral sclerosis. Lancet Neurol 2011;10:400-403.
Correspondence:
Publication Dates
-
Publication in this collection
23 July 2012 -
Date of issue
July 2012
History
-
Received
05 Feb 2012 -
Accepted
02 Mar 2012 -
Reviewed
23 Feb 2012