Abstracts
In children, vertical transmission is the main form of HIV infection. Our aim was to determine the prevalence of HIV-1 vertical transmission in mother-infant pairs in a public maternity ward in Presidente Prudente, SP. Additionally; we sought to identify characteristics associated with this form of transmission. The files of 86 HIV-1-infected mothers and their newborns referred to a Public Hospital from March 2002 to March 2007 were analyzed. The HIV-1-RNA viral load of the newborns was determined by bDNA. The HIV-1 vertical-transmission rate was 4.6%. Children that were born in the pre-term period and breastfed were at a higher risk of HIV-1 infection (p = 0.005 and p = 0.017 respectively) than children born at term and not breastfed. Prophylactic therapy with zidovudine after birth for newborns was associated with a lower risk of infection (p = 0.003). The number of newborns weighing < 2,500 g was significantly higher for infected children (p = 0.008) than for non-infected newborns. About 22.9% of mothers did not know the HIV-1 status of their newborns eight months after delivery. The study suggests that it is necessary to increase the identification of HIV-1 infection in pregnant women and their newborns as well as to offer and explain the benefits of ARV prophylaxis.
Brazil; HIV-1; MTCT; Characteristics
A transmissão vertical é a principal forma de infecção pelo HIV em crianças. Nosso objetivo foi determinar a prevalência da transmissão vertical do HIV em maternidade pública em Presidente Prudente, SP. Além disso, procuramos identificar características associadas a essa forma de transmissão. De março de 2002 a março de 2007, os prontuários de 86 mães HIV-1 positivo e seus bebês, nascidos na maternidade de Hospital Público foram analisadas. A carga viral do HIV-1-RNA dos recém-nascidos foi determinada por bDNA. A prevalência da transmissão vertical pelo HIV-1 foi de 4,6%. Crianças nascidas a pré-termo e amamentadas tiveram um risco maior de infecção (p = 0,005 e p = 0,017, respectivamente), comparado com crianças nascidas a termo e não amamentadas. A profilaxia com zidovudina depois do nascimento para os recém-nascidos foi associada ao menor risco de infecção (p = 0,003). O número de recém-nascidos com peso < 2.500 g foi maior entre as crianças infectadas (p = 0,008), comparado com crianças não infectadas. Oito meses depois do parto 22,9% das mães não tinham conhecimento da infecção de seus bebês pelo HIV. O presente estudo sugere a necessidade em aumentar a identificação do HIV em gestantes e seus bebês e explicar e oferecer os benefícios da profilaxia antiretroviral.
HIV
HIV-1-mother-to-child transmission and associated characteristics in a public maternity unit in Presidente Prudente, Brazil
Transmissão vertical do HIV e características associadas em maternidade pública em Presidente Prudente, Brasil
Luiz Euribel Prestes-CarneiroI; Patrícia Rodrigues Naufal SpirII,III, Armênio Alcântara RibeiroII; Vera Lúcia Maria Alves GonçalvesIV
IImmunology Department, Universidade do Oeste Paulista, Rua José Bongiovani 700, 19050-680 Presidente Prudente, São Paulo, Brazil
IIPediatrics Department, Universidade do Oeste Paulista, Rua José Bongiovani 1297, 19050-680 Presidente Prudente, São Paulo, Brazil
IIIPublic Regional Maternity Unit, Dr. Odilo Antunes Siqueira, Rua Doutor Albertino Sobrado 116, 19023-200 Presidente Prudente, São Paulo, Brazil
IVAdolfo Lutz Institute - Regional Laboratory of Presidente Prudente, Molecular Biology Section, Rua Coronel Marcondes 2357, 19013-050 Presidente Prudente, São Paulo, Brazil
Correspondence to Correspondence to: Luiz Euribel Prestes Carneiro Departamento de Imunologia Universidade do Oeste Paulista, Rua José Bongiovani 700, Cidade Universitária 19050-680 Presidente Prudente, São Paulo, Brasil. Phone/FAX: +55-18-229 1013. E-mail: luiz@unoeste.br
ABSTRACT
In children, vertical transmission is the main form of HIV infection. Our aim was to determine the prevalence of HIV-1 vertical transmission in mother-infant pairs in a public maternity ward in Presidente Prudente, SP. Additionally; we sought to identify characteristics associated with this form of transmission. The files of 86 HIV-1-infected mothers and their newborns referred to a Public Hospital from March 2002 to March 2007 were analyzed. The HIV-1-RNA viral load of the newborns was determined by bDNA. The HIV-1 vertical-transmission rate was 4.6%. Children that were born in the pre-term period and breastfed were at a higher risk of HIV-1 infection (p = 0.005 and p = 0.017 respectively) than children born at term and not breastfed. Prophylactic therapy with zidovudine after birth for newborns was associated with a lower risk of infection (p = 0.003). The number of newborns weighing < 2,500 g was significantly higher for infected children (p = 0.008) than for non-infected newborns. About 22.9% of mothers did not know the HIV-1 status of their newborns eight months after delivery. The study suggests that it is necessary to increase the identification of HIV-1 infection in pregnant women and their newborns as well as to offer and explain the benefits of ARV prophylaxis.
Key words: Brazil; HIV-1; MTCT; Characteristics.
RESUMO
A transmissão vertical é a principal forma de infecção pelo HIV em crianças. Nosso objetivo foi determinar a prevalência da transmissão vertical do HIV em maternidade pública em Presidente Prudente, SP. Além disso, procuramos identificar características associadas a essa forma de transmissão. De março de 2002 a março de 2007, os prontuários de 86 mães HIV-1 positivo e seus bebês, nascidos na maternidade de Hospital Público foram analisadas. A carga viral do HIV-1-RNA dos recém-nascidos foi determinada por bDNA. A prevalência da transmissão vertical pelo HIV-1 foi de 4,6%. Crianças nascidas a pré-termo e amamentadas tiveram um risco maior de infecção (p = 0,005 e p = 0,017, respectivamente), comparado com crianças nascidas a termo e não amamentadas. A profilaxia com zidovudina depois do nascimento para os recém-nascidos foi associada ao menor risco de infecção (p = 0,003). O número de recém-nascidos com peso < 2.500 g foi maior entre as crianças infectadas (p = 0,008), comparado com crianças não infectadas. Oito meses depois do parto 22,9% das mães não tinham conhecimento da infecção de seus bebês pelo HIV. O presente estudo sugere a necessidade em aumentar a identificação do HIV em gestantes e seus bebês e explicar e oferecer os benefícios da profilaxia antiretroviral.
INTRODUCTION
About one third of all people living with HIV in Central and South America live in Brazil. In Latin America, although epidemiological data suggest that the HIV-1 epidemic has remained stable in the region, during the last decade there has been an increase in the incidence of HIV/AIDS among women of reproductive age (UNAIDS, 2010). In children, vertical transmission is the main form of infection and can occur during late pregnancy, during delivery or post-partum through breastfeeding (ORNE-GLIEMANN et al., 2008). Brazil was one of the first developing countries to implement measures to prevent mother-to-child transmission (MTCT) of HIV-1, resulting in a decrease in vertical transmission rates (PN-DST/AIDS, 2006; AMARAL et al., 2007; PRESTES-CARNEIRO et al., 2009). There are limited data addressing the prevalence and risk factors of MTCT in the western region of the São Paulo State (GONÇALVES et al., 2011). A recent study showed a growing AIDS dissemination among females in small-sized municipalities in the state (PRADO & CASTILHO, 2009). The objective of this study was to determine HIV-1 vertical transmission rates and some characteristics associated with MTCT in a public maternity ward located in the western region of the São Paulo State.
MATERIAL AND METHODS
Patients and samples: Located in the western part of Southeast Brazil, the Regional Health Department of Presidente Prudente (DRS XI) is composed of 45 municipalities, with approximately 750,000 inhabitants living mainly in small cities and rural communities. Located in Presidente Prudente, the Public Hospital Dr. Odilo Antunes Siqueira (PH) is a regional public maternity ward to which most pregnant women and women in the intrapartum period who are infected or at risk of HIV-1 infection are referred. The city also harbors the Adolfo Lutz Institute (ALI), the public reference laboratory responsible for confirming HIV-1 tests as well as measuring viral-load counts in HIV-1-positive pregnant women and infant-pairs of DRS XI.
For this retrospective study, the files of mother-infant pairs born in the maternity ward of the PH from March 2002 to March 2007 - those whose blood samples were analyzed by the Molecular Biology Unit, in the Medical Biology Section of ALI, for HIV-1-RNA viral load determination, with the aim of assisting in the diagnosis of MTCT - were reviewed. The inclusion criteria were: children up to 18 months of age at the time of the first HIV-1-RNA viral load determination, and children that have undergone at least two blood samples for HIV-1-RNA viral load determination.
HIV-1-RNA analysis - Quantification of HIV-1-RNA was performed in plasma using the VERSANT® HIV-1 RNA 3.0 Assay (bDNA) (Bayer Corporation, Emervylle, California, USA) in accordance with the manufacturer's instructions. The assays were performed twice after a follow-up period of at least eight months between the first visit and the follow-up analysis. Newborns were considered uninfected if at least two independent blood samples were below the limit of detection of the HIV-1-RNA quantification test. Children were considered infected when at least two independent blood samples were positive in HIV-1-RNA quantification tests.
All of the women were hospitalized in the intrapartum period by the Unified National Health System (SUS).
The risk factors explored in the obstetric category were as follows: number of pregnancies, age groups, gestational age, mode of delivery and breastfeeding. The characteristics of infants analyzed were as follows: birth weight, birth width, use of zidovudine neonatal prophylaxis, use of trimethoprim/sulfamethoxazole (TMT/SMZ), and time of first analysis of the HIV-1 viral load of babies after birth. Antiretroviral (ARV) therapies were described as Zidovudine Monotherapy, dual Nucleoside Reverse Transcriptase Inhibitor therapy (NRTI, ZDV plus another NRTI), and Highly Active Antiretroviral Therapy (HAART; two NRTI plus one Non-Nucleoside Reverse Transcriptase Inhibitor - NNRTI; two NRTI plus one Protease Inhibitor - PI). Some women did not use any therapy during gestation or labor and delivery. This protocol was in accordance with the Brazilian STD/AIDS program3 and was approved by the Ethics Committee of ALI, SP, Brazil.
Statistical analysis - Fisher's exact test and the chi-square test were used to analyze the data using GraphPad Instat software (V4.0, San Diego, CA) and GraphPad Prism (V4.0, San Diego, CA). All p values are two-tailed; p values < 0.05 were considered to be statistically significant. Vertical HIV-1 transmission was calculated as the ratio of the number of newborns with a known HIV-1-positive diagnosis, divided by the total number of newborns.
RESULTS
From March 2002 to March 2007, 96 births of HIV-1-infected mothers occurred in the maternity ward of the PH, and blood samples of these newborns were analyzed by ALI for HIV-1-RNA determination. The analysis of the files of mother-infant pairs revealed that no twin births occurred. One child died during the neonatal period with two positive blood samples for HIV-1-RNA. Nine newborns were lost during the follow-up for different reasons such as transfer from the region of Presidente Prudente, lack of a second blood sample for HIV-1-RNA determination, children older than eighteen months for the first HIV-1-RNA analysis or few data in the records of mother-infant pairs. Data were available for 86 mother-infant pairs who completed the inclusion criteria established to prevent MTCT of HIV-1. The overall vertical HIV-1 transmission rate was 4.6% (4/86), (CI 1.2811.48), with no differences in infection relating to the sex of the infant. In this study, 12% of mothers brought their children for HIV-1 determination before they reached two months of age, 47.70% between two and four months, 10% between five and six months, 7.4% between seven and eight months and 22.9% over eight months of age. The distribution of births during the period was as follows: March 2002 to March 2003, 21% (18/86); April 2003 to March 2004, 27.9% (24/86); April 2004 to March 2005, 19.8% (17/86); April 2005 to March 2006, 13.9% (12/86) and from April 2006 to March 2007, 17.4% (15/86).
The risk factors were determined in 86 mothers referred to the Public Hospital (PH) during this period, although some data were missing in several of the files. Of the 86 files analyzed, 61.6% of the mothers lived in Presidente Prudente and 38.4% were from other cities of the DRS (Regional Health Directory) XI.
Table 1 shows that children who were born in the preterm period (< 37 weeks of gestational age) had a higher risk of HIV-1 infection than children who were delivered at term (≥ 37 weeks of gestational age) (p = 0.005). Not-breastfeeding was associated with a protection against infection in newborns (p = 0.017). Maternal age at the time of delivery varied from 15 to 40 years (mean 27.8; 95% CI 26.34 - 29.36 years), and only one mother was under 18 years old. No difference in MTCT related to maternal age was found. Elective cesareans and the number of pregnancies also did not correlate with HIV-vertical transmission (p > 0.05). Elective cesareans were conducted in 76.8% (53/69) of mothers. Zidovudine prophylaxis was administered to 33.3% (20/60) of mothers, and 13.3% (8/60) did not receive zidovudine.
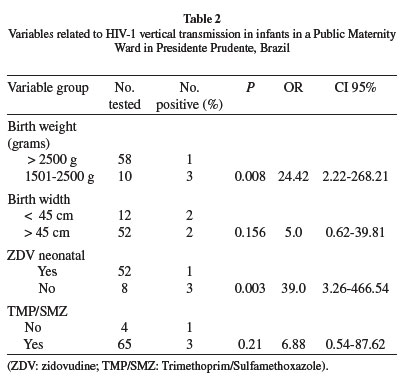
Table 2 shows the infant follow-up. Newborns < 2,500 g at birth had a significantly higher rate of HIV-1 transmission than those > 2,500 g (p = 0.008). In this study, prophylactic therapy with zidovudine in neonates was associated with a lower risk of infection (p = 0.003). Regarding timing of HIV-1 diagnosis, of 86 mothers analyzed, only 44 files showed this information, of which 50% (22/44) documented diagnosis before antenatal care, 40.9% (18/44) during antenatal care, and 9.1% (4/44) during or after delivery.
After delivery, mothers were instructed to take their babies to an infectious-disease specialist, and 70.4% (50/71) visited the doctor less than two months after delivery. The number of visits to the specialist during the first year of life was distributed as follows: 16.4% (11/67) visited fewer than two times, 53.7% (36/67) visited three to five times, and 29.8% (20/67) visited the clinic six to nine times.
Figure 1 shows the antiretroviral schema used among HIV-1-infected woman during pregnancy. Among 86 mothers analyzed during the period, only 60 files showed these data, from which 13.3% (8/60) did not use antiretroviral drugs, 33.3% (20/60), used the Nucleoside Analog Inhibitor (NRTI) zidovudine as a monotherapy and 8.3% (5/60) used a dual therapy with a combination of two drugs from the same group (NRTI). As a HAART regimen, with a combination of three or more drugs, 18.3% (11/60) used NRTI and Non-Nucleoside Analog Inhibitors (NNRTI). NRTI plus protease inhibitors (PI) were administered to 26.7% (16/60) of mothers.
DISCUSSION
Primary public healthcare centers and regional reference hospitals distributed throughout Brazilian municipalities represent a key role in the screening, diagnosis and management of populations considered to be at-risk of blood-borne diseases such as HIV-1 (PORTELINHA FILHO et al., 2009). The overall prevalence rate of vertical transmission of HIV-1 in this study was 4.6%. This rate is lower than that found in a nationwide multicenter study (7.1%), (CI 1.3-11.5 p > 0.05), conducted in 2001 (MENEZES SUCCI, 2007). In Brazil, the prevalence of HIV-1 vertical transmission has declined in the last decade due to global and regional measures implemented by the PN-DST/AIDS program (PN-DST/AIDS, 2006, UNAIDS, 2010). Limited by a reduced number of patients (86 pregnant and four positive mother-infant pairs) our data are in some aspects similar to the results obtained in other big cities and regions of Brazil (AMARAL et al., 2007; MENEZES SUCCI, 2007).
In this study, breastfeeding is found to be one of the main risk factors for maternal HIV-1 vertical transmission (Table 1). Infected mothers were advised not to breastfeed their infants and offered clinical or pharmacological support, as recommended by Government Protocol (UNAIDS, 2010). The government also provided specific nutritional formula. However, four mothers breastfed their babies, leading to a significant increase in HIV-1 vertical transmission in this group. There is specific evidence regarding the association between breastfeeding and MTCT. The occurrence of MTCT is 15-30% in non-breastfeeding populations. The risk increases considerably to 20-45% among populations who practice prolonged breastfeeding (AMARAL et al., 2007; ORNE-GLIEMANN et al., 2008).
The classic study conducted by the Pediatric AIDS Clinical Trials Group Protocol (PACTG 076), showed that MTCT can be reduced significantly through maternal antiretroviral treatment during pregnancy and delivery and infant antiretroviral prophylaxis after birth (CONNOR et al., 1994). The efficacy of a short-course peri-partum of the ARV-zidovudine prophylaxis regimen administered to HIV infected pregnant mothers and their newborns has been established in Brazil since 1996 (BRITO et al., 2005; PN-DST/AIDS, 2006). Regrettably, eight mothers (13.3%), and consequently their infants, did not receive zidovudine or ARV prophylaxis. Although the reasons for the lack of prophylaxis were not determined, one possibility is the fact that 9.1% of the analyzed mothers did not know their HIV-1 status before labor and delivery. The percentage is similar to recent data (6.6%) obtained from pregnant women at a Public Teaching Hospital in Presidente Prudente (PRESTES-CARNEIRO et al., 2009). Among the pregnant women who used ARV in the gestation or delivery period, ZDV monotherapy represented 33.3% and HAART (NRTI+PI) represented 23.3%. These data are in accordance with the protective effect of ZDV regarding the reduction of vertical transmission (CONNOR et al., 1994; AMARAL et al., 2007; ORNE-GLIEMANN et al., 2008).
To reduce the number of pregnant women not tested during antenatal care or whose HIV-1 status is undocumented during labor and delivery, a nationwide PN-STD/AIDS program of rapid HIV-1 testing was one of the main strategies introduced (PN-DST/AIDS, 2006; PRESTES-CARNEIRO et al., 2009). Despite all the planning and policy-making, the lack of decentralized services, the high number of women referred from small cities and rural areas and the continuous lack of rapid HIV-1 test kits can contribute to missed opportunities for preventing MTCT. In accordance with this hypothesis, 38.4% of women were referred from small cities of DRS XI. These results demonstrate the delicate nature of operating a public health system in the region during the period analyzed.
In these data, with regard to gestational age, premature babies had a higher risk of HIV-1 infection than full-term babies. Pre-term delivery and low birth weight were common among women on HAART during pregnancy, especially when therapy was started before or during the first trimester of pregnancy (ORNE-GLIEMAN et al., 2008). However, it was not possible to link prematurity and HIV-1 infection with ARV because we do not know in which period of pregnancy the ARV therapy occurred.
The growing number of pregnancies in adolescents and young women in Brazil in recent decades is alarming (ROCHA et al., 2010). Interestingly, only one of the mothers was under the age of 18, and the group's median age was 27.8 years. These findings suggest that, at least in this region, either HIV-1 infection is occurring after adolescence or young women are able to postpone or even avoid pregnancy, a theory supported in a Brazilian multicentric study of MTCT (MENEZES SUCCI, 2007). In contrast to the results reported herein, a study conducted in the Mato Grosso do Sul State, a surrounding DRS XI area, found that 19.8% of HIV-1 pregnant women were youths (DAL FABRO et al., 2005). In the data presented herein, the number of previous pregnancies, antenatal care visits and vaginal deliveries were not associated with HIV-1 maternal transmission. Vaginal delivery is strongly associated with an elevated risk of HIV-vertical transmission particularly in mothers with low levels of CD4+T lymphocytes and elevated HIV-viral load counts (ORNE-GLIEMANN et al., 2008; KAKEHASI et al., 2008). Regarding the mode of delivery, elective cesarean sections were performed in 76.8% of the pregnancies, independent of previous ARV regimen or viral-load counts, reflecting the high frequency of cesarean sections performed in Brazil (MENEZES SUCCI, 2007). It is currently accepted that for women with ARV treatment in gestation and elevated CD4+ levels, elective cesarean has no effect on HIV-1 transmission (ORNE-GLIEMANN et al., 2008).
The major factors governing MTCT can be broadly classified as maternal, obstetrical and neonatal (ORNE-GLIEMANN et al., 2008). Among the neonatal characteristics associated with HIV-1 vertical transmission (Table 2), the present study analysis of the data demonstrates the protective effect of starting AZT prophylaxis immediately after birth, as recommended by the PN-DST/AIDS program in reducing MTCT. Several studies have indicated that even incomplete AZT regimens were effective in protecting newborns from HIV infection (João et al., 2003; Mussi-Pinhata et al., 2007). In addition, the association between low birth weight and the HIV transmission observed has also been reported by others, linked mainly to poor nutritional status, anemia or low maternal body mass index. Low birth weight was found to be an important factor in predicting infant death (MEHTA et al., 2008).
Infectious diseases account for nearly 90% of early childhood deaths in developing countries (MUSSI-PINHATA et al., 2007). In this study, 93% of infants received trimethoprim/sulfamethoxazole in the neonatal period for prophylaxis against Pneumocystis jiroveci pneumonia. Only one death occurred in the neonatal period. According to the instructions of the Brazilian Ministry of Health HIV-AIDS program, the viral load of newborns should be determined at both two and four months following delivery. It should be noted that about 22.9% of mothers did not bring their babies for HIV-1-RNA determination eight months after delivery. The earliest possible screening for the HIV-1 viral status of newborns constitutes a key event in the follow-up of infants. One possibility is the distance between some counties or suburbs where many mothers live and the PH. However, an efficient public transport system for patients from other municipalities to referral centers such as PH is available in the region.
Because we only investigated mother-infant pairs referred to PH, the results drawn from this observational study may not be representative of HIV-infected women throughout the region. However, this limitation can be attenuated by the fact that PH is the only referral center for at-risk or HIV-1-infected pregnant women in the DRS XI area (CREMONEZI et al., 2005; ALVES et al., 2009). In this respect, the study involved 81% of all mother-infant pairs screened for MTCT who were referred to the Adolfo Lutz Institute (ALI), between 2002 and 2007. Another limitation of this study is the under-powered number of patients, precluding multivariate analysis. Nevertheless, the analyzed data could be used in the future to improve and compare the effectiveness of the measures adopted.
In conclusion, national and regional public-health policies were implemented in the last few years to prevent and control the vertical transmission of HIV. However, in the DRS XI region, the study suggests that it is necessary to increase the identification of HIV-1 infection in pregnant women and their infants as well as to offer and explain the benefits of ARV prophylaxis. Otherwise, in line with an observation made by Kallings (2008), we may win the battles, but we will lose the war.
ACKNOWLEDGMENTS
Supported by the Adolfo Lutz Institute and UNOESTE. Luiz Euribel Prestes Carneiro, MD, PhD, is an Institutional Researcher of UNOESTE.
Received: 27 March 2010
Accepted: 17 November 2011
- 1. Alves KCLRP, Fram DS, Diccini S, Belasco AGS, Barbosa DA. Prevalence and risk factors for human immunodeficiency virus infection in pregnant women. Acta Paul Enferm. 2009;22:307-12.
- 2. Amaral E, Assis-Gomes F, Milanez H , Cecatti JG, Vilela MM, Pinto e Silva JL. Implementaçăo oportuna de intervençőes para reduzir a transmissăo vertical do HIV: uma experięncia brasileira bem-sucedida. Rev Panam Salud Publica. 2007;21:357-64.
-
3Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Programa Nacional de DST e AIDS. Recomendações para profilaxia da transmissão vertical do HIV e terapia anti-retroviral em gestantes. Brasília: Ministério da Saúde, 2006. (Série Manuais No. 46).
- 4. Brito AM, Castilho EA, Szwarcwald CL. Regional patterns of the temporal evolution of the AIDS epidemic in Brazil following the introduction of antiretroviral therapy. Braz J Infect Dis. 2005;9:9-19.
- 5. Connor EM, Sperling RS, Gelber R, Kiselev P, Scott G, O'Sullivan MJ, et al. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. Pediatric AIDS Clinical Trials Group Protocol 076 Study Group. N Engl J Med. 1994;331:1173-80.
- 6. Cremonezi D, Mesquita PE, Romăo MM, Prestes-Carneiro LE. Prevalence of indeterminate human immunodeficiency virus Western blot results in pregnant women attended at a public hospital in Presidente Prudente, Brazil. Braz J Infect Dis. 2005;9:506-9.
- 7. Dal Fabbro MM, Cunha RV, Paniago AM, Lindenberg AS, Freitas GM, Nogueira SA. Prospective study on the prevention of vertical transmission of HIV in Campo Grande, Mato Grosso do Sul, Brazil, from 1996 to 2001. Braz J Infect Dis. 2005;9:20-7.
- 8. Gonçalves VL, Troiani C, Ribeiro AA, Spir PR, Gushiken EK, Vieira RB, Prestes-Carneiro LE. Vertical transmission of HIV-1 in the western region of the State of Săo Paulo. Rev Soc Bras Med Trop. 2011;44:4-7.
- 9. Joăo EC, Cruz ML, Menezes JA, Matos HJ, Calvet GA, d'Ippolito MM, et al Vertical transmission of HIV in Rio de Janeiro, Brazil. AIDS. 2003;17:1853-5.
- 10. Kakehasi FM, Pinto JA, Romanelli RM, Carneiro M, Cardoso CS, Tavares MC, et al Determinants and trends in perinatal human immunodeficiency virus type 1 (HIV-1) transmission in the metropolitan area of Belo Horizonte, Brazil: 1998 - 2005. Mem Inst Oswaldo Cruz. 2008;103:351-7
- 11. Kallings LO. The first postmodern pandemic: 25 years of HIV/ AIDS. J Intern Med. 2008;263:218-43.
- 12. Menezes Succi RC. Mother-to-child transmission of HIV in Brazil during the years 2000 and 2001: results of a multi-centric study. Cad Saude Publica. 2007;23(Suppl 3):S379-89.
- 13. Mehta S, Manji KP, Young AM, Brown ER, Chasela C, Taha TE, et al. Nutritional indicators of adverse pregnancy outcomes and mother-to-child transmission of HIV among HIV-infected women. Am J Clin Nutr. 2008;87:1639-49.
- 14. Mussi-Pinhata MM, Freimanis L, Yamamoto AY, Korelitz J, Pinto JA, Cruz ML, et al Infectious disease morbidity among young HIV-1-exposed but uninfected infants in Latin American and Caribbean countries: the National Institute of Child Health and Human Development International Site Development Initiative Perinatal Study Group. Pediatrics. 2007;119:e694-704.
- 15. Orne-Gliemann J, Becquet R, Ekouevi DK, Leroy V, Perez F, Dabis F. Children and HIV/AIDS: from research to policy and action in resource-limited settings. AIDS. 2008;22:797-805.
- 16. Portelinha Filho AM, Nascimento CU, Tannouri TN, Troiani C, Ascęncio EL, Bonfim R, et al Seroprevalence of HBV, HCV and HIV co-infection in selected individuals from state of Săo Paulo, Brazil. Mem Inst Oswaldo Cruz. 2009;104:960-3.
- 17. Prado RR, Castilho EA. A epidemia de AIDS no estado de Săo Paulo: uma aplicaçăo do modelo espaço-temporal bayesiano completo. Rev Soc Bras Med Trop. 2009;42:537-42.
- 18. Prestes-Carneiro LE, Miguel NA, Ascęncio EL, Amâncio M, Alcântara VL, Portelinha-Filho JA. Rapid HIV diagnostic test in undocumented pregnant women applied at an inner-city teaching hospital. Rev Inst Med Trop Sao Paulo. 2009; 51:273-6
- 19. Rocha RC, de Souza E, Soares EP, Nogueira ES, Chambô Filho A, Guazzelli CA. Prematurity and low birth weight among Brazilian adolescents and young adults. J Pediatr Adolesc Gynecol. 2010;23:142-5.
-
20UNAIDS Report on the global AIDS epidemic 2010. [cited 2010 Dec 5] Available from: http://www.unaids.org/GlobalReport/Global_report.htm
Publication Dates
-
Publication in this collection
23 Feb 2012 -
Date of issue
Feb 2012
History
-
Received
27 Mar 2010 -
Accepted
17 Nov 2011