Resumos
Recentes estudos demonstraram que os radicais livres de oxigênio parecem desempenhar papel importante nas lesões de isquemia-reperfusão. O alfa-tocoferol(AT) age in vivo como um anti-oxidante. Nosso objetivo foi o de verificar se o AT atenuava as lesões de isquemia-reperfusão em pata direita de ratos. Os animais, randomizados, foram divididos nos seguintes grupos(G): G1-controle sem isquemia; G2 e G3 - quatro horas isquemia e duas reperfusão. Os animais do G2 foram tratados com salina e os do G3 com AT, 50mg/kg. Como parâmetros examinamos as alterações de volume e circunferência do pé direito ao longo do experimento, assim como a captação de 99mTc-pirofosfato e a microscopia eletrônica de transmissão do músculo soleus direito. Os aumentos de volume e circunferência do pé dos animais do G2 foram estatisticamente maiores quando comparados com o G1. As medidas do G3 não diferiram do G1. A captação de 99mTc-pirofosfato foi bem aumentada nos G2 e G3, quando comparados ao G1, porém sem diferenças entre os grupos isquêmicos. As lesões ao MET foram menos intensas em três animais do G3, quando comparados ao G2. Conclusão: O tratamento com AT diminuiu a formação do edema mas só protegeu parcialmente as células musculares contra as lesões de isquemia-reperfusão.
Alfa tocoferol; Isquemia; Reperfusão; Ratos
Studies indicate that oxygen-derived free radicals may play a major role in ischemia-reperfusion injury. Alpha-tocopherol acts in vivo as a free radical scavenger. Our purpose was to elucidate whether alpha-tocopherol could change the evolution of ischemia/reperfusion in the right hindlimb of rats. The animals were randomly allocated in the following groups: Group 1(G1)- control without ischemia. Groups 2 and 3(G2,3): four hours ischemia and two hours reperfusion. The animals of group 2 were treated with saline and those of the group 3, treated with alpha-tocopherol, 50 mg/Kg. Parameters were the foot volume and circumference, 99mTc-Pyrophosfate uptake in the soleus muscle, plasmatic creatine-phosphokinase (CPK), and transmition eletronic microscopy (TEM)of muscle cells. The foot volume and circumference of the animals of G2 were significantly greater than in G1. In the G3 these measurements did not differ from the G1. Pyrophosphate uptake and CPK measurements were increased in G2,G3 when compared to the animals of G1, but there were no differences between the ischemic groups. Cell injury, when examined by TEM, was less severe in two of three animals of G3, when compared to the G2. Conclusion: The treatment with alpha-tocopherol diminuished edema formation, but only partially protected muscle cells from injury.
Alpha-tocopherol; Ischemia; Reperfusion; Rats
2 ORIGINAL ARTICLE
EFFECT OF ALPHA-TOCOPHEROL ON THE ISCHEMIA/REPERFUSION LESIONS INDUCED IN THE HINDLIMB OF RATS1 1 . From Faculty of Medicine of Botucatu - State University of São Paulo, Brazil. 2. Assistent Professor, PhD - Department of Surgery Faculty of Medicine - Marília, São Paulo, Brazil. 3. Full Professor Department Morphology, Biosciences Institute, Botucatu - State University of São Paulo, Brazil. 4. Full Professor Department of Surgery Faculty of Medicine, Botucatu - State University of São Paulo, Brazil.
José Bitu-Moreno2 1 . From Faculty of Medicine of Botucatu - State University of São Paulo, Brazil. 2. Assistent Professor, PhD - Department of Surgery Faculty of Medicine - Marília, São Paulo, Brazil. 3. Full Professor Department Morphology, Biosciences Institute, Botucatu - State University of São Paulo, Brazil. 4. Full Professor Department of Surgery Faculty of Medicine, Botucatu - State University of São Paulo, Brazil.
Eliza Aparecida Gregório3 1 . From Faculty of Medicine of Botucatu - State University of São Paulo, Brazil. 2. Assistent Professor, PhD - Department of Surgery Faculty of Medicine - Marília, São Paulo, Brazil. 3. Full Professor Department Morphology, Biosciences Institute, Botucatu - State University of São Paulo, Brazil. 4. Full Professor Department of Surgery Faculty of Medicine, Botucatu - State University of São Paulo, Brazil.
Francisco Humberto A. Maffei4 1 . From Faculty of Medicine of Botucatu - State University of São Paulo, Brazil. 2. Assistent Professor, PhD - Department of Surgery Faculty of Medicine - Marília, São Paulo, Brazil. 3. Full Professor Department Morphology, Biosciences Institute, Botucatu - State University of São Paulo, Brazil. 4. Full Professor Department of Surgery Faculty of Medicine, Botucatu - State University of São Paulo, Brazil.
Bitu-Moreno J, Gregório EA, Maffei FHA. Effect of alpha-tocopherol on the ischemia/reperfusion lesions induced in the hindlimb of rats. Acta Cir Bras [serial online] 2001 Apr-Jun;16(2). Available from URL: http://www.scielo.br/acb.
ABSTRACT: Studies indicate that oxygen-derived free radicals may play a major role in ischemia-reperfusion injury. Alpha-tocopherol acts in vivo as a free radical scavenger. Our purpose was to elucidate whether alpha-tocopherol could change the evolution of ischemia/reperfusion in the right hindlimb of rats. The animals were randomly allocated in the following groups: Group 1(G1)- control without ischemia. Groups 2 and 3(G2,3): four hours ischemia and two hours reperfusion. The animals of group 2 were treated with saline and those of the group 3, treated with alpha-tocopherol, 50 mg/Kg. Parameters were the foot volume and circumference, 99mTc-Pyrophosfate uptake in the soleus muscle, plasmatic creatine-phosphokinase (CPK), and transmition eletronic microscopy (TEM)of muscle cells. The foot volume and circumference of the animals of G2 were significantly greater than in G1. In the G3 these measurements did not differ from the G1. Pyrophosphate uptake and CPK measurements were increased in G2,G3 when compared to the animals of G1, but there were no differences between the ischemic groups. Cell injury, when examined by TEM, was less severe in two of three animals of G3, when compared to the G2. Conclusion: The treatment with alpha-tocopherol diminuished edema formation, but only partially protected muscle cells from injury.
KEY WORDS: 1. Alpha-tocopherol. 2. Ischemia. 3. Reperfusion. 4. Rats.
INTRODUCTION
Acute ischemia and reperfusion of a limb, specially if revascularization is delayed, can induce tissue metabolic and structural changes that can lead to significant morbidity, mortality and amputation rates.1-8 This syndrome was first well characterized by Haimovice 3 in 1960 and then by Legrain and Cormier 9 and is also known as Haimovice-Legrain-Cormier syndrome and consists of muscle pain, tenderness and edema associated with renal and systemic metabolic derangements.
Skeletal muscle is resistant to longer periods of ischemia, if compared to other tissues like cardiac muscle or brain. Severe long standing ischemia and reperfusion, however, often results in morphological and functional alterations in the muscle cells and microcirculation, including endothelial cell edema and denudation, increased vascular permeability, interstitial edema, accumulation, adhesion and infiltration of leucocytes, and finally tissue necrosis.10-19 Recent studies have shown that reactive oxygen metabolites may play an important role in this process, and that antioxidants and scavengers of free oxygen radicals may attenuate the ischemia/reperfusion injury in experimental models.16-21
Alpha-tocopherol (AT) acts "in vivo" as a free radical scavenger and antioxidant 22-24 and has been reported to protect several organs by decreasing acute pathological changes of ischemia/reperfusion injury.24-28 Little has been published, however, on the evaluation of the effect of alpha-tocopherol on the ischemia/reperfusion injury of the skeletal muscle.
The present study was therefore undertaken with the purpose of elucidating if alpha-tocopherol could change the evolution of tissue alterations in a model of ischemia/reperfusion injury in the hindlimb of rats.
METHODS
EXPERIMENTAL PROCEDURES
Fifty eight male Wistar rats weighing 220-250g were used. The animals were anesthetized with intra-peritoneal pentobarbital sodium (30mg/Kg). Small, complementary doses of anesthetic drug were given throughout the procedure if necessary.
After anesthesia, skin, subcutaneous tissue and muscles around the right hip joint were sectioned and only the femoral vessels and femur were left intact. One external jugular vein was dissected free and cannulated for drug injection.
In the animals of the groups in which ischemia was induced, a microvascular occluding clamp was placed at the femoral artery. After 4 hours the vascular clamp was removed and blood was allowed to recirculate. Saline or AT were injected immediately before clamp removal and 99mTc-Pyrophosfate was injected 1 hour later in treatment groups. Reperfusion was allowed for 2 hours. After this time biopsies for transmition eletronic mycroscopy were taken from soleus muscles from both hindlimbs, through a posterior incision in the leg and the animals were killed with an averdose of pentobarbital.
EXPERIMENTAL GROUPS
The animals were divided in 3 experimental groups:
Group 1: Control group without ischemia/reperfusion. 15 animals were submitted to all operative procedures except arterial occlusion and reperfusion. Four hours after exposure of the femoral vessels, the animals were treated with saline injected by the jugular vein, in the same volume/kg used for the treatment with alpha-tocopherol (AT).
Group 2: Treatment control. 19 animals were submitted to all operative procedures including artery occlusion and reperfusion and were treated with saline as in group 1.
Group 3: Treatment group. 17 animals were submitted to all surgical procedures and treated with AT (30mg/KG) injected by the jugular vein just before reperfusion.
The animals were randomly allocated to each treatment group after muscle section and vessel exposure. Operative procedures and measurements were undertaken by the first author blindly, without the knowledge of the used drug.
EVALUATED PARAMETERS
Effects of artery clamping and withdrawn of the clamp was evaluated by the inspection of the color of the foot. During ischemia an intense pallor was observed, and immediately after reperfusion a reactive hyperhemia could be seen.
Edema was evaluated by the measurement of the circumference of the foot, 1 cm bellow the malleolus medialis, and by the determination of the foot volume, by measuring the water displacement in a plethismograph for small animals, constructed of glass. These measurements were performed after anesthesia (before artery clamping in groups 2 and 3), four hours after vessels dissection with or without artery clamping, and 2 hours after revascularization or the corresponding time in group 1.
Uptake of 99mTc-Pyrophosfate was undertaken to estimate muscle necrosis. One hour after vessels dissection, a bolus of 37 Kbq/g of 99mTc-Pyrophosfate was injected in the jugular vein of each animal. One hour later, with the animal still alive, the soleus muscle was removed from the treated leg and from the opposite leg, that was used as control. The muscles were then weighted and counted for radioactivity in a gamma well counter AUTO-LOGIC (ABBOT), after correction for radioactivity decay. Results were expressed in counts per minute. The same procedure was repeated 12 hours after, in the dried muscles.
Creatine phosphokinase (CPK) activity was determined, using CK-NAC-Creatine Kinase kits and a biochemical auto analyzer RA-1000 (TECHNICON), in blood samples, taken by intra-cardiac puncture 2 hours after reperfusion.
Muscle biopsies for transmition electron microscopy were cut and fixed in 2.5% buffered glutaraldehyde (pH 7.31), post fixed in 1.0% osmiumtetroxide, dehydrated in acetone and imbedded in araldite. Ultra-thin sections were stained in a saturated solution of uranyl acetate and lead citrate and examined in a Philips EM 301 electron microscope.
STATISTICS
Analysis of variance was used for the variables circumference and volume in the same group of animals in 3 moments. The central tendency was expressed as mean ± standard error of the mean (SEM) in each moment of each group. For the variables CPK and 99mTc-Pyrophosfate, evaluated at the end of the experiment, the central tendency was calculated with median (Md) in each group. The comparison between the 3 groups was performed with the use of the no parametric test of Kruskal-Wallis, with the statistical calculus H and the minimal difference for contrasts between pair of groups.
RESULTS
Circumference of the foot of the rats (Figure 1). The foot circumference of the animals with ischemia-reperfusion treated with saline was greater than in the animals of the control group without ischemia. (p<.0.05) In the animals treated with alpha-tocopherol these meaurements did not differ from the control group without ischemia.
Volume of the foot of the rats (Figure 2). The foot volume of the animals with ischemia-reperfusion treated with saline was greater than in the animals of the control group without ischemia (P<0,01). In the animals treated with alpha-tocopherol, these measurements showed no statistical significant difference to the control animals.
CPK. (Table 1) The CPK measurements were more elevated in the animals with ischemia-reperfusion when compared to the animals of the control group (p<0,05). In the animals treated with alpha-tocopherol, these measurements did not differ from the other groups.
99m-Tc-Phyrofosfate (Table 2 and 3). The 99m-Tc-Phyrofosfate uptake measurements were more elevated in the animals with ischemia-reperfusion when compared to the animals of the control group without ischemia (P<0,001).
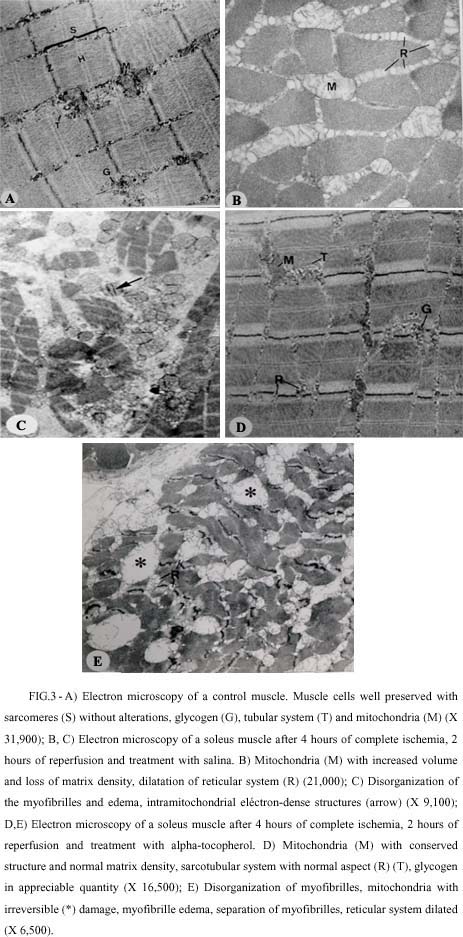
Electron microscopy (Figure 3). In animals of control group, without ischemia, the myofibrilles showed a sarcomere pattern well represented, with regular and not disrupted Z line, sarcotubular system with normal aspect, presence of glycogen granules and mitochondrias with preserved structure, normal matrix density and organization of cristas.
The changes in the fine structure of the myofibrils in specimens from the ischemia/reperfusion groups were: myofibrillar edema, separation of filaments in the myofibrils, widening of the myofibrillar spaces, swelling and dilatation of sarcotubular system, absence of glycogen granules, and mitochondrial abnormalities such as: swelling, loss of matrix density and appearance of intra-mitochondrial dense bodies. The sarcomere pattern was well preserved in many muscle fibers, but disrupture of this pattern was evident in other regions, characterizing focal lesions. The damaged areas appeared rarefied with partial loss of myofilaments, and more severe lesions consisting of smudged myofilaments which accumulated in dense clumps, with distorted or disrupted Z line, and mitochondria detected as dense-homogenous bodies. The effects were more intense in all specimens of the ischemia/reperfusion group treated with saline, when compared with the specimens of the animals treated with alpha-tocopherol, in which 2 of 3 animals presented less severe alterations.
DISCUSSION
In our experiment rats submitted to 4 hours of complete warm hindlimb ischemia developed during reperfusion considerable foot edema, traduced as an increase in foot volume and circumference, observed also in other investigations.10-12,14,20 A significant increase in 99m-Tc-Phyrofosfate uptake and high levels of CPK were also observed reflecting indirectly the extension of irreversible lesions and muscle necrosis in the ischemic-reperfunded hindlimb.29-32 Ultrastructural studies of the soleus muscle of these animals showed alterations such as widening of myofibrillar spaces, myofibrillar edema, separation of filaments in the myofibrils, dilatation of sarcotubular system, diminution in the amount of glycogen granules, and mitochondrial abnormalities such as swelling, loss of matrix density and appearance of intramitochondrial dense bodies. These ultrastructural abnormalities appeared in focal pattern, with less injured cells side to side with severely damaged cells, and were in accordance with the results of 99mTc-Pyrophosfate uptake and CPK levels.
These alterations were more intense in the group treated with saline, compared with the group treated with alpha-tocopherol. In this group the increase in foot volume and circumference was less severe and did not differ from the control without ischemia. A probable effect of alpha-tocopherol acting as a free radical scavenger and antioxidant, breaking the chain of lipidic peroxidation and protecting the endothelial membrane integrity 23 could explain this effect. Other referred properties of alpha-tocopherol such as inhibition of xanthine oxidase formation in endothelial cells 33 and interference with the participation of polymorphonuclear leukocytes in ischemia-reperfusion process 34 could also explain this protection against the increase in vascular permeability as a component of reperfusion injury, although we have not found leukocytic infiltration in the studied samples of muscle.
The attenuation of the reperfusion edema in skeletal muscle was obtained by others authors using different oxygen-free radicals scavengers, such as superoxide dismutase,14,35 catalase,14,32 mannitol,32 deferoxamine,16 apotransferrine,16 allopurinol and oxypurinol,14,18 dimethyl sulfoxide,14 and other methods, as control of the conditions of reperfusion, composition of the reperfusate,20,21 and use of calcium channel blocking drugs 20,21
The ultrastructural analysis of the soleus muscle of the animals treated with alpha-tocopherol, revealed a certain grade of cellular protection in 2 of 3 animals in which the alterations did not differ from the control group. This fact could justify the tendency for a smaller 99m-Tc-Phyrofosfate uptake and a lower levels of seric CPK in the treated animals. In one of the alpha-tocopherol treated animals, the ultrastructural alterations were particularly severe, with all examined cells uniformly damaged and a pattern of congested capillares, referred by Harman 36 Strock & Majno 10 and Quiñones-Baldrich 37 as characteristic of the no-reflow phenomenon.
CONCLUSION
the treatment with alpha-tocopherol of rats in which ischemia/reperfusion was induced in the hindlimb diminished edema formation, but only partially protected muscle cells from injury.
Bitu-Moreno J, Gregório EA, Maffei FHA. Efeitos do alfa-tocoferol nas lesões de isquemia-reperfusão induzidas em pata direita de ratos. Acta Cir Bras [serial online] 2001 Abr-Jun;16(2). Disponível em URL: http://www.scielo.br/acb.
RESUMO: Recentes estudos demonstraram que os radicais livres de oxigênio parecem desempenhar papel importante nas lesões de isquemia-reperfusão. O alfa-tocoferol(AT) age in vivo como um anti-oxidante. Nosso objetivo foi o de verificar se o AT atenuava as lesões de isquemia-reperfusão em pata direita de ratos. Os animais, randomizados, foram divididos nos seguintes grupos(G): G1-controle sem isquemia; G2 e G3 - quatro horas isquemia e duas reperfusão. Os animais do G2 foram tratados com salina e os do G3 com AT, 50mg/kg. Como parâmetros examinamos as alterações de volume e circunferência do pé direito ao longo do experimento, assim como a captação de 99mTc-pirofosfato e a microscopia eletrônica de transmissão do músculo soleus direito. Os aumentos de volume e circunferência do pé dos animais do G2 foram estatisticamente maiores quando comparados com o G1. As medidas do G3 não diferiram do G1. A captação de 99mTc-pirofosfato foi bem aumentada nos G2 e G3, quando comparados ao G1, porém sem diferenças entre os grupos isquêmicos. As lesões ao MET foram menos intensas em três animais do G3, quando comparados ao G2. Conclusão: O tratamento com AT diminuiu a formação do edema mas só protegeu parcialmente as células musculares contra as lesões de isquemia-reperfusão.
DESCRITORES: 1. Alfa tocoferol. 2. Isquemia. 3. Reperfusão. 4. Ratos.
Conflito de interesses: nenhum
Fontes de financiamento: nenhuma
Address for correspondence:
José Bitu Moreno
Pça. Athos Fragata 25, 502
Marília SP
17501-220
e-mail: jbmoreno@famema.br
Data do recebimento: 12/12/2000
Data da revisão: 03/02/2001
Data da aprovação: 15/04/2001
- 1.Blaisdell FW, Lim RC, Amberg JR. Pulmonary microembolism: a cause of morbidity and death afeter major vascular surgery. Arch Surg 1966;93:776-86.
- 2.Bonamigo T. Embolectomias tardias com o uso do catéter de Fogarty [Tese Doutorado]. Faculdade de Medicina da Universidade de Săo Paulo; 1972.
- 3.Haimovici H. Arterial embolism with acute massive ischemic myopathy and myoglobinuria. Surgery 1960;47:739-47.
- 4.Eriksson I, Holmberg JT. Analysis of factors affecting limb salvage and mortality after embolectomy. Acta Chir Scand 1977;143:237-40.
- 5.Burihan E, Ciscato JG, Miranda Jr F et al. Avaliaçăo crítica de alguns aspectos da embolia arterial: revisăo de 164 embolias, 1969-1978. Rev Col Bras Cir 1979;5:187.
- 6.Francisco Jr J, Miranda Jr F, Barros JR., N. et al. The Haimovici-Legrain-Cormier syndrome in arterial embolectomy. J Cardiovasc Surg 1983;424: 39.
- 7.Abbot WM, Mc Cabe C, Maloney RD et al. Embolism of the popliteal artery. Surg Gynecol Obstet 1984;159:533-6.
- 8.Lastória S, Maffei FHA. Oclusőes arteriais agudas. In: Maffei FHA, Lastória S, Yoshida WB et al. (ed). Doenças vasculares periféricas. Rio de Janeiro: MEDSI, 1987. P 299-321.
- 9.Cormier JM, Legrain M. L˘hyoerkaličmie, complication trčs grave des syndromes d˘ischémie des membres. J Chir 1962;83:473-88.
- 10.Strock PE, Majno GM. Vascular responses to experimental tourniquet ischemia. Surg Gynecol Obstet 1969a; 129: 309-318.
- 11.Strock PE, Majno GM. Microvascular changes in acutely ischemic rat muscle. Surg Gynecol Obstet 1969b;129:1213-24.
- 12.Diana JN, Laughlin MH. Effect of ischemia on capillary pressure and equivalent pore radius in capillaries of the isolated dog hind limb. Circ Res 1974;35:77-101.
- 13.Gidlöf A, Lewis DH, Hammersen F. The effect of prolonged total ischemia on the ultrastructure of human skeletal muscle capillaries: a morphometric analysis. Int J Microcirc Clin Exp 1988;7:67-70.
- 14.Korthuis RJ, Granger DN, Townsley MI et al. The role of oxygen-derived free radicals in ischemia-induced increases in canine skeletal muscle vascular permeability. Circ Res 1985;57:599-609.
- 15.Korthuis RJ, Grisham MB, Granger DN. Leukocyte depletion attenuates vascular injury in postischemic skeletal muscle. Am J Physiol 1988;254:H283-7.
- 16.Smith JK, Carden DL, Grisham MB et al. Role of iron in postischemic microvascular injury. Am J Physiol 1989a;256:H1472-7.
- 17.Smith JK, Carden DL, Korthuis RJ. Role of hydroxyl radicals in ishcemia/reperfusion-induced injury to skeletal muscle microvasculature. FASEB J 1989b;3: A1234.
- 18.Smith JK, Carden DL, Sadasivan KK et al. Role of xanthine oxidase in postischemic microvascular injury in skeletal muscle. Am J Physiol 1989c;257:H1782-9.
- 19.Oredsson S, Qvarfordt P, Plate G. Polymorphonuclear leucocytes increase reperfusion injury in skeletal muscle. Intern Angiol 1995;14:80-8.
- 20.Beyersdorf F, Matheis G, Hanselmann A et al.Reducing reperfusion injury after acute ischemia in the hindlimb by controlled reperfusion. Thorac Cardiovasc Surg 1987;35:94-5.
- 21.Beyersdorf F, Unger A, Wildhirt A et al. Studies of reperfusion injury in skeletal muscle: preserved cellular viability after extended periods of warm ischemia. J Cardiovasc Surg 1991;32:664-76.
- 22.Halliwell B. Biochemical mechanisms accouting for the toxic action of oxygen on living organisms: the key role of superoxide dismutase. Cell Biol Int Rep 1978;2:113-28.
- 23.Burton GW, Ingold KU. Autoxidation of biological molecules: the antioxidant activity of vitamin E and related chain-breaking phenolic antioxidants in vitro. J Am Chem Soc 1981;103:6472-7.
- 24.Ikezawa T, Hishikimi N, Oba Y. Lipid peroxides in the mechanism of ischemia/reperfusion injury in skeletal muscle-experimental studies.Vasc Surg 1993;191-201.
- 25.Yamamoto M, Shima T, Uozumi T et al. A possible role of lipid peroxidation in cellular damages caused by cerebral ischemia and the protective effect of alpha-tocopherol administration. Stroke 1983;14:977-82.
- 26.Marubayashi S, Dohi K, Uchi K et al. Role of free radicals in ischemic rat liver cell injury: prevention of damage by alph-tocopherol administration. Surgery 1986;99:184-92.
- 27.Ferrari R, Curello S, Boffa, GM et al. Oxygen free radical-mediatec heart injury in animal models and during bypass surgery in humans: effects of alpha-tocopherol. Ann NY Acad Sci 1989;57:237-53.
- 28.Massey KD, Burton KP. Alpha-tocopherol attenuates nyocardial membrane-related alterations resulting from ischemia and reperfusion. Am J Physiol 1989; 256:H1192-9.
- 29.Buja LM, Tofe AJ, Kulkarni PV et al. Sites and mechanisms of localization of techenetium-99m phosphorous radiopharmaceuticals in actue myocardial infarcts and other tissues. J Clin Invest 1977;60:724-40.
- 30.Hargens AR, Schmidt DA, Evans CL et al. Quantitation of skeletal-muscle necrosis in a model compartment syndrome. J Bone Joint Surg 1981;63:631-6.
- 31.Hobson II RW, Neville R, Watanabe B. et al. Role of heparin in reducing skeletal muscle infarctions in ischemia-reperfusion. Microcirc Endothelium Lymphatics 1989;5:259-76.
- 32.Catignani GL, Dinning JS. Role of vitamin E in the regulation of rabbit liver xanthine dehydrogenase activity. J Nutr 1971;101:1327-30.
- 33.Ricci MA, Graham AM, Corbisiero R. et al. Are free radical scavengers beneficial in the treatment of compartment syndrome after acute arterial ischemia? J Vasc Surg 1989;9:244-9.
- 34.Engle WA, Yoder MC, Baurley JL. et al. Vitamin E decreases superoxide anion production by polymorphonuclear leukocytes. Pediatr Res 1988;23:245-8.
- 35.Lee KR, Cronenwett JR, Shlafer M. et al. Effect of superoxide dismutase plus catalse on Ca2+ transport in ischemic and reperfused skeletal muscle. J Surg Res 1987;42:24-32.
- 36.Harman JW. The significance of local vascular phenomena in the production of ischemic necrosis in skeletal muscle. Am J Pathol 1948;24:625-44.
- 37.Quińones-Baldrich WJ. The role of fibrinolysis during reperfusion of ischemic skeletal muscle. Microcirc. Endothelium Lymphatics 1989;5:299-31.
Datas de Publicação
-
Publicação nesta coleção
11 Set 2003 -
Data do Fascículo
Jun 2001
Histórico
-
Aceito
15 Abr 2001 -
Revisado
03 Fev 2001 -
Recebido
12 Dez 2000