Abstracts
In homologous transplantation or allotranplantation of limbs, the great tissue diversity causes variability in the rejection process and, consequently, its immunology is very complex. Thus, limb transplantation is the most used prototype of compound tissue transplantation among the protocols of experimental studies. Composite tissue allotransplantation represents the experimental model to study the homologous transplantation (from an individual to another) of vascularized, innervated musclecutaneous units, joints, bone or even the whole member. Groups of rats were undergone allogeneic hindlimb transplantation. The receptors were randomized and control groups were established as: Control Group A: Autograft controls (F344 rats had its limbs reimplanted) and no immunosuppressive therapy. Control Group B: Allograft controls (BN rats limbs were transplanted to F344). Composite tissue homotransplantation allows the inclusion of innervated muscle-cutaneous units, joint and bone or even the hole limb, is considerably applicable in cases of congenital absence or deformity, trauma or greater resection due to malignant tumor. For many complex deformities, these transplantations would allow a more precise reconstruction than the current reconstruction techniques.
Tissue transplantation; Limb homotransplantation; Allotransplantation; Microsurgery, method; Rejection
Nos transplantes alógenos de membro a grande variabilidade de tecidos (pele, subcutâneo, músculo, osso, medula óssea, gânglios linfáticos, cartilagem, nervo, vasos, tendão, articulação) leva a grande variação dentro do processo de rejeição e consequentemente a sua imunologia é bastante complexa. Os transplantes alógenos de tecido composto representam o modelo experimental para se estudar o transplante homólogo (de um indivíduo para outro) de unidades músculo cutâneas inervadas, vascularizadas, articulações, osso ou mesmo de todo o membro. Os receptores foram randomizados e os grupos controle foram estabelecidos como: grupo controle A: transplante autógeno de membro em que ratos F344 tiveram o seu membro reimplantado e nenhuma medicação foi administrada e grupo controle B: transplante alógeno de membro (TAM) em que o membro dos ratos BN foi transplantado para os ratos F344 cujo membro havia sido amputado. Os transplantes homólogos de tecido composto (THTC) que possibilitam a inclusão de unidades músculo-cutâneas inervadas, articulação e osso ou mesmo de todo o membro, tem considerável aplicação nos casos de ausência ou deformidade congênita, trauma ou ressecção maior devido a tumor maligno. Para muitas deformidades complexas, estes THTC permitiriam uma reconstrução mais precisa do que as atuais técnicas reconstrutoras correntes.
Transplante de tecidos; Transplante homólogo de membro; Transplante alógeno; Microcirurgia; Rejeição
EXPERIMENTAL MODELS
Experimental model for composite tissue allotransplantations1 1 Plastic Surgery Division, Surgery Department, UNIFESP
Modelo experimental para alotransplantes de tecido composto
Lydia Masako FerreiraI; Luiz Roberto Kobuti FerreiraII
IHead and Chairwoman Plastic Surgery Division, Surgery Department, UNIFESP
IIMedical Graduate Student University of São Paulo
Correspondence Correspondence to Lydia Masako Ferreira MD, PhD Rua Napoleão de Barros, 715, 4º andar CEP: 04024-900 São Paulo SP Tel: (11)557604118 FAX: (11) 55716579 sandra.dcir@epm.br, lydia.dcir@epm.br
ABSTRACT
In homologous transplantation or allotranplantation of limbs, the great tissue diversity causes variability in the rejection process and, consequently, its immunology is very complex. Thus, limb transplantation is the most used prototype of compound tissue transplantation among the protocols of experimental studies. Composite tissue allotransplantation represents the experimental model to study the homologous transplantation (from an individual to another) of vascularized, innervated musclecutaneous units, joints, bone or even the whole member. Groups of rats were undergone allogeneic hindlimb transplantation. The receptors were randomized and control groups were established as: Control Group A: Autograft controls (F344 rats had its limbs reimplanted) and no immunosuppressive therapy. Control Group B: Allograft controls (BN rats limbs were transplanted to F344). Composite tissue homotransplantation allows the inclusion of innervated muscle-cutaneous units, joint and bone or even the hole limb, is considerably applicable in cases of congenital absence or deformity, trauma or greater resection due to malignant tumor. For many complex deformities, these transplantations would allow a more precise reconstruction than the current reconstruction techniques.
Key words: Tissue transplantation. Limb homotransplantation. Allotransplantation. Microsurgery, method. Rejection.
RESUMO
Nos transplantes alógenos de membro a grande variabilidade de tecidos (pele, subcutâneo, músculo, osso, medula óssea, gânglios linfáticos, cartilagem, nervo, vasos, tendão, articulação) leva a grande variação dentro do processo de rejeição e consequentemente a sua imunologia é bastante complexa. Os transplantes alógenos de tecido composto representam o modelo experimental para se estudar o transplante homólogo (de um indivíduo para outro) de unidades músculo cutâneas inervadas, vascularizadas, articulações, osso ou mesmo de todo o membro. Os receptores foram randomizados e os grupos controle foram estabelecidos como: grupo controle A: transplante autógeno de membro em que ratos F344 tiveram o seu membro reimplantado e nenhuma medicação foi administrada e grupo controle B: transplante alógeno de membro (TAM) em que o membro dos ratos BN foi transplantado para os ratos F344 cujo membro havia sido amputado. Os transplantes homólogos de tecido composto (THTC) que possibilitam a inclusão de unidades músculo-cutâneas inervadas, articulação e osso ou mesmo de todo o membro, tem considerável aplicação nos casos de ausência ou deformidade congênita, trauma ou ressecção maior devido a tumor maligno. Para muitas deformidades complexas, estes THTC permitiriam uma reconstrução mais precisa do que as atuais técnicas reconstrutoras correntes.
Descritores: Transplante de tecidos. Transplante homólogo de membro. Transplante alógeno. Microcirurgia. Rejeição
Introduction
The tissue transplantation are common practice in Plastic Surgery and the development of this specialty is based on breaking through biological obstacles that limit the accomplishment of transplantation from a human being to another.
During the last decades, organ transplantation has been object of intense research and there has been a continuous clinical and experimental progress in transplantation of many different organs.
Transplantation immunology of solid organs is relatively simple due to little variability in the process of rejection among the cell types of an organ and the rejection process is more homogeneous throughout the transplantation. In homologous transplantation or allotranplantation of limbs, the great tissue diversity causes variability in the rejection process and, consequently, its immunology is very complex. Thus, limb transplantation is the most used prototype of compound tissue transplantation among the protocols of experimental studies1,2.
Proposition
Composite tissue allotransplantation represents the experimental model to study the homologous transplantation (from an individual to another) of vascularized, innervated musclecutaneous units, joints, bone or even the whole member.
Method Description
Brown Norway (BN) rats with a RT-1n major histocompatibility (MHC) locus were donors and Fischer 344 (F344) rats with a RT-11 MHC locus were recipients2,3,4,5.
Groups of rats were undergone allogeneic hindlimb transplantation. The receptors were randomized and control groups were established as: Control Group A: Autograft controls (F344 rats had its limbs reimplanted) and no immunosuppressive therapy. Control Group B: Allograft controls (BN rats limbs were transplanted to F344).
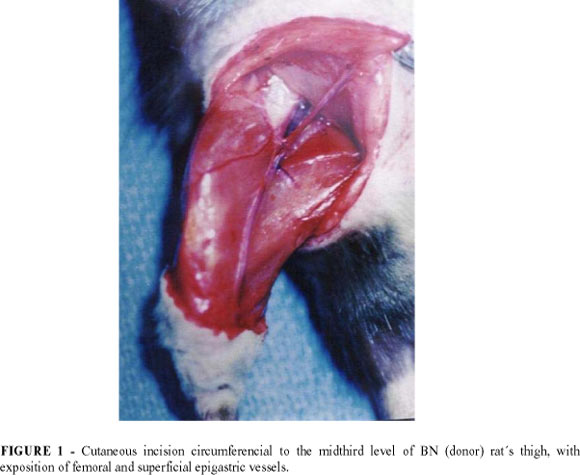
Sodium pentobarbital at a dose of 50mg/kg intraperitoneal was administred for anesthesia. Limbs were amputated at midfemur level followed by orthotopic transfer of the BN donor limb to the F-344 recipient site: a circumferencial cutaneous incision was made in the midthird level of the right thigh of the BN rats and the epigastric and femoral vessels were isolated (FIGURE 1).
Clamping, dissection and section of the femoral and superficial epigastric vessels were done at this level (FIGURE 2).
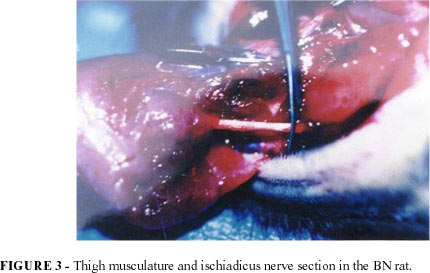
Muscle and sciatic nerve section (FIGURE 3).
Limb amputation at the midthird level of femur (FIGURE 4).
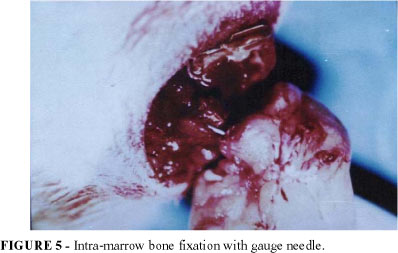
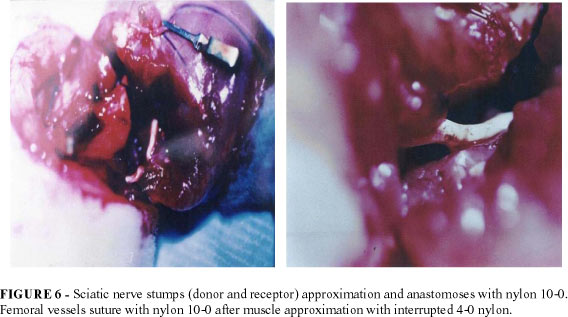
Donor limb was orthotopically transfered to the recipient area. Bone fixation was achieved using with a gauge needle as an intramedullary rod (FIGURE 5). The muscle was approximated using 4-0 nylon. The sciatic nerve, femoral artery and vein were repaired primarily with interrupted 10-0 nylon sutures using standart microsurgical technique (FIGURE 6). During dissection, the vessels were irrigated with physiological solution, heparin (100u/ml) and lidocaine 2% to lessen the vessels spasm, when needed.
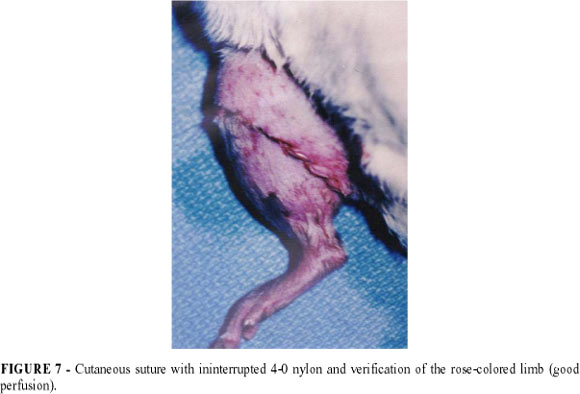
The skin was sutured with ininterrupted 5-0 nylon (FIGURE 7).
Femoral vessels suture with nylon 10-0 after muscle approximation with interrupted 4-0 nylon.
A protective wire collar was placed around the proximal thigh to prevent authophagia of the insensate limb (FIGURE 8).
No anticoagulation or antibiotic drugs were administered.
To compensate for intraoperative fluid loss and postoperative dehydration, subcutaneous saline (10cc) was injected for the first 5 days post-transplantation.
Daily, the limbs were evaluated on vascular permeability and rejection signals, including erythema, edema, cutaneous color, exfoliation, hair loss, nail growth, epidermolysis, ulceration, exudation and cutaneous necrosis (FIGURE 9).
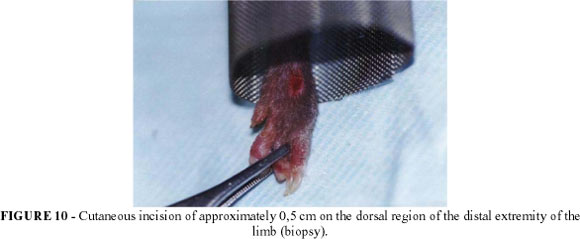
Skin biopsies were prepared (FIGURE 10): following remotion, the pieces were fixed in formol solution at 10%, histologically processed and posteriorly imbibed in paraffin. All pieces were stained with hematoxylin and eosin. The histological pattern scale system used by Saurat was used to quantify the rejection grade detected on the skin biopsy. All biopsies were blindly analyzed by a pathologist (FIGURE 11 and 12).
The rejection day was established when the skin could be removed with a touch or was hard and scarified with hair loss2,3,4,5.
At the sacrifice moment, it was verified whether the vessels were permeable, by means of dissection and direct inspection of the femoral vessels anastomoses, in order to eliminate the possibility of ischemia, which could also lead to skin necrosis.
Perspectives
Composite tissue homotransplantation allows the inclusion of innervated muscle-cutaneous units, joint and bone or even the hole limb, is considerably applicable in cases of congenital absence or deformity, trauma or greater resection due to malignant tumor. For many complex deformities, these transplantations would allow a more precise reconstruction than the current reconstruction techniques8.
These compound tissue reconstruction would allow the surgeon to restore the lost contour, sensibility, movement and function making use of similar tissue reposition7.
The quest for safer and more effective immunosupressive treatment, in other words, the search for a less toxic imunotherapy, is essential because in these transplantations the skin is especially antigenic so that it is necessary a 2-3 times higher doses than in organ transplantation and the collateral effects are less acceptable since they are not vital surgeries as organ transplantation are.
So, there is still need for studies and this experimental model would help future experiments aiming at:
. Comprehension of the genetic mechanisms of composite tissue allotransplantation.
. Investigation of new immunosupressive drugs
. Other immunosupressive therapies combined in a manner to obtain a more efficient effect.
. Less toxicity by means of association of immunosupressive drugs in subtherapeutical doses.
. Better functional coverage of these transplantations with nerve regeneration studies.
. Decrease in tissue antigenicity, in determining the incidence and treatment of the graft versus host syndrome7.
. Prevention of late rejection3.
. Determine why GVHD occurs after bowel and not after limb allotransplantation9.
Conflito de interesse: nenhum
Fonte de financiamento: CNPq
Ferreira LM, Ferreira LRK. Modelo experimental para alotransplantes de tecido composto. Acta Cir Bras [serial online] 2004 Vol 19 Edição Especial. Disponível em URL: http://www.scielo.br/acb.
- 1. Ferereira LM, Ferreira LRK - Experimental model: historic and conceptual revision. Acta Cir Brás. 2003;18(sp):1-3.
- 2. Ferreira LM - Eficácia da associaçăo de doses subterapęuticas de CsA e RS-61443 em transplante alógeno microcirúrgico de membro. Estudo experimental em ratos. [Tese de Livre Docęncia]. Universidade Federal de Săo Paulo Escola Paulista de Medicina, 1994.
- 3. Benhaim P, Anthony JP, Ferreira LM, Borsanyi JP, Mathes SJ - Control of the rejection process treated with cyclosporine and RS-61443, in allogeneic microsurgery transplantation of a limb, in rats. Transplant. 1996; 61:527-31.
- 4. Ferreira LM, Borsanyi JP, Benhaim P, Anthony JP, Mathes S - Sinergic effect of subtherapeutic dose combination immunotherapy with cyclosporine and RS-61443 in rat limb allotransplantation. World J Plast Aesth and Reconstr Surg. 1999;3(1):108-11.
- 5. Saurat JH, Gluckman E, Bussel A - The lichen planuslike eruption after bone marrow transplantation. Br J Dermatol. 1975; 93:675-81.
- 6. Ferreira LM, Anthony JP, Mathes J, Andrews JM, Laredo Filho J - Complicaçőes em transplante alógeno microcirúrgico de membro (tecido composto), em ratos. Rev Assoc Med Brás. 1995; 41(3):213-8.
- 7. Ferreira LM, Andrews J, Laredo Filho J - Transplante homólogo de membro (tecido composto). Perspectivas para o futuro. Rev Assoc Med Bras. 1995; 41(2):151-7.
- 8. Ferreira, LM - Transplantes Homólogos de Membro In: Ferreira, LM Manual de Cirurgia Plástica, Editora Atheneu, Săo Paulo cap. 22, P 261-4, 1995.
- 9. Ferreira LM, Benhaim P, Anthony JP, Borsanyi JP, Mathes SJ, Andrews JM - Síndrome Enxerto versus Hospedeiro: uma possibilidade em transplante homólogo de membro Acta Orthop Bras. 1994;2(2):63-6.
Publication Dates
-
Publication in this collection
17 Mar 2005 -
Date of issue
Dec 2004