ABSTRACT:
An epidemiological survey was carried out by performing an Enzyme Linked Immuno Sorbent Assay (ELISA) test to determine the seroprevalence of Pythium insidiosum infection in equine in Rio Grande do Sul State (RS), Brazil. The serological study covered seven geographical regions of RS, classified according to the Instituto Brasileiro de Geografia e Estatística (IBGE). The samples were obtained from official veterinary service (Serviço Veterinário Oficial, SVO) linked to the Secretaria da Agricultura, Pecuária e Agronegócio of RS (SEAPA-RS) to proceed the investigation of equine infectious anemia in 2014. Samples were collected during the months of September and October of 2013, covering the seven geographical regions of RS, and totalized 1,002 serum samples. The seroprevalence for P. insidiosum in RS was 11.1% (CI95% 9.23 to 13.22). The relative risk (RR) of the presence of antibodies anti-P. insidiosum was in the regions Southeast 11.17 (CI95%, 4.65 to 26.8), Porto Alegre 4.62 (CI95%, 1.70 to 12.55), Southwest 11.17 (CI95%, 4.65 to 26.8) and Northwestern 3.72 (CI95%, 1.52 to 9.09). The highest prevalence (69.1%) was observed in females with RR of 1.59 (CI95%, 1.11 to 2.27). When the presence of dams was evaluated, the seropositivity was evident in 74.4%, presenting an association of 2.13 (CI95%, 1.16 to 3.91) compared to farms without dams. In properties with veterinary assistance, the frequency of 72.7% and RR of 3.04 (CI95%,, 1,85 to 4,98) of seropositivity were observed. Due to the importance of pythiosis in horse herds, this study highlights the presence of anti-P. insidiosum antibodies in horses in RS, Brazil.
Key words:
P. insidiosum; antibody; occurrence; horses; ELISA
RESUMO:
Um levantamento soroepidemiológico foi realizado através do teste de ELISA indireto para determinar a soroprevalência da infecção por Pythium insidiosum em equinos no estado do Rio Grande do Sul (RS), classificadas de acordo com o Instituto Brasileiro de Geografia e Estatística (IBGE). As amostras utilizadas eram provenientes do cadastro das propriedades do Serviço Veterinário Oficial (SVO), da Secretaria da Agricultura, Pecuária e Agronegócio do RS (SEAPA-RS), coletadas para o inquérito da anemia infecciosa equina de 2014. As coletas foram realizadas durante os meses de setembro e outubro de 2013, abrangendo as sete mesorregiões geográficas do RS, e totalizaram 1.002 amostras de soro. Do total das amostras testadas, 11.1% (CI95% 9.23 to 13.22) foram soropositivas para P. insidiosum. Constatou-se o risco relativo (RR) da presença de anticorpos anti-P. insidiosum nas regiões Sudeste 11,17(IC95%, 4,65-26,8), Porto Alegre 4,62 (IC95%, 1,70-12,55), Sudoeste 11,17 (IC95%, 4,65-26,8) e Noroeste 3,72 (IC95%, 1,52-9,09). Observou-se a maior soroprevalência (69,1%) em fêmeas com RR de 1,59 (IC95%, 1,11-2,27). Quanto à presença de açudes, evidenciou-se soropositividade em 74,4% das propriedades, apresentando associação de 2,13 (IC95%,1,16-3,91) em comparação com propriedades sem açude. Em propriedades com assistência veterinária, foi verificada a frequência de 72,7% e RR de 3,04 (IC95%,1,85-4,98). Diante da relevância da pitiose em rebanhos equinos, destaca-se a presença de anticorpos anti-P. insidiosum em equinos no estado do RS.
Palavras-chave:
P. insidiosum; anticorpo; ocorrência; equinos
INTRODUCTION:
Brazil has the third largest herd of horses in the world and the highest in Latin America (MAPA, 2014MAPA, 2014. Ministério da Agricultura Ministério da Agricultura, Pecuária e Abastecimento. Available from: <Available from: <http://www.agricultura.gov.br/animal/especies/equideos
>. Accessed: nov. 29, 2014.
http://www.agricultura.gov.br/animal/esp...
). The equine population in Rio Grande do Sul State (RS) is estimated at 468,691 heads (IBGE, 2014IBGE (INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA). 2014. Available from: <Available from: <http://www.ibge.gov.br/home/estatistica/economia/agropecuaria/censoagro/default.shtm
>. online. Accessed: nov. 29, 2014.
http://www.ibge.gov.br/home/estatistica/...
). Equine breedings generate more than US$ 7.3 billion in economic activity annually. The equine industry in Brazil, especially in RS, is an important agricultural activity that combines business, sport and leisure in an economic impact program involving millions of people (ALMEIDA & SILVA, 2010ALMEIDA, F.Q.; SILVA, V.P. Scientific progress in equine production at 1st decade of the XXI century. Revista Brasileirade Zootecnia, v.39, p.119-129, 2010. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-35982010001300014
>. Accessed: nov. 29, 2014. doi: 10.1590/S1516-35982010001300014.
http://www.scielo.br/scielo.php?script=s...
). Knowledge of diseases that occur in the equine species is crucial for determining its economic importance, as well as the establishment of control procedures (MARCOLONGO-PEREIRA et al., 2014MARCOLONGO-PEREIRA, C. et al. Equine diseases in Southern Brazil., Pesquisa Veterinária Brasileira v.34, n.3, p.205-210, 2014. Available from: <Available from: <http://www.scielo.br/scielo.php?pid=S0100736X2014000300002&script=sci_arttext
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2014000300002.
http://www.scielo.br/scielo.php?pid=S010...
).
Pythiosis is a chronic granulomatous disease caused by the acquatic oomycete P. insidiosum that affects humans, domestic and wild animals. The most affected species is equine. The disease has a worldwide distribution, occurring in tropical and subtropical regions (MENDOZA et al., 1996MENDOZA, L. et al. Infections caused by the oomycetous pathogen Pythium insidiosumJournal Medical Mycology, v.6, n.4, p.151-164, 1996.). In Brazil, swamp areas such as the Pantanal Mato-Grossense (LEAL et al., 2001LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf
>. Accessed: nov. 29, 2014.
http://www.scielo.br/pdf/pvb/v21n4/7481....
;. SANTOS et al., 2014SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
http://www.j-evs.com/article/S0737-0806(...
) and RS (MARCOLONGO-PEREIRA et al., 2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
) are considered endemic for equine pythiosis.
The cases of clinical pythiosis in farm animals, especially in equine species, have been increasing in Brazil (LEAL et al., 2001LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf
>. Accessed: nov. 29, 2014.
http://www.scielo.br/pdf/pvb/v21n4/7481....
; SALLIS et al., 2003SALLIS, E.S.V. et al. Cutaneous pythiosis in horses: 14 cases., Ciência Rural v.33, n.5, p.899-903, 2003. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782003000500017
>. Accessed: nov. 29, 2014. doi: 10.1590/S0103-84782003000500017.
http://www.scielo.br/scielo.php?script=s...
; MARCOLONGO-PEREIRA et al., 2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
; SANTOS et al., 2014SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
http://www.j-evs.com/article/S0737-0806(...
). However, studies on the prevalence and incidence of equine pythiosis are limited (LEAL et al., 2001LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf
>. Accessed: nov. 29, 2014.
http://www.scielo.br/pdf/pvb/v21n4/7481....
; PEREIRA et al, 2008PEREIRA, D.I.B. et al. In vitro zoosporogenesis among oomycetes Pythium insidiosum isolates. Ciência Rural, v.38, n.1, p.143-147, 2008. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782008000100023
>. Accessed: nov. 29, 2014. doi: 10.1590/S0103-84782008000100023.
http://www.scielo.br/scielo.php?script=s...
; MARCOLONGO-PEREIRA et al., 2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
.). The available studies are based on the epidemiological aspects from retrospective cases (PIEREZAN et al., 2009PIEREZAN, F. et al. Necropsy findings related to the cause of death in 335 horses: 1968-2007., Pesquisa Veterinária Brasileira v.29, n.3, p.275-280, 2009. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2009000300015
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2009000300015.
http://www.scielo.br/scielo.php?script=s...
; SOUZA et al., 2011SOUZA, T.M. et al. Prevalence of equine skin tumors diagnosed at the Laboratory of Veterinary Pathology of the Federal University Santa Maria, Rio Grande do Sul, Brazil., Pesquisa Veterinária Brasileira v.31, n.5, p.379-382, 2011. Available from: <Available from: <http://www.scielo.br/scielo.php?pid=S0100-736X2011000500003&script=sci_arttext
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2011000500003.
http://www.scielo.br/scielo.php?pid=S010...
; MARCOLONGO-PEREIRA et al., 2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
) as well as analyses of clinical diseases diagnosed in endemic regions (LEAL et al., 2001LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf
>. Accessed: nov. 29, 2014.
http://www.scielo.br/pdf/pvb/v21n4/7481....
; SANTOS et al., 2014SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
http://www.j-evs.com/article/S0737-0806(...
). The objective of this study was to evaluate the prevalence of P. insidiosum infection in horses from RS State, southern of Brazil.
MATERIAL AND METHODS:
The seroepidemiological survey comprised the seven geographic mesoregions of RS, classified according to the IBGEIBGE (INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA). 2014. Available from: <Available from: <http://www.ibge.gov.br/home/estatistica/economia/agropecuaria/censoagro/default.shtm
>. online. Accessed: nov. 29, 2014.
http://www.ibge.gov.br/home/estatistica/...
(2006). RS was divided into seven strata (coinciding with the seven mesoregions) regarding the movement of horses (mostly intramesoregion), the horse skills (different in each mesoregion), animal stock (markedly different in each mesoregion), as well as edafoclimatic and socioeconomic characterisitcs in each mesoregion (Figure 1A).
A: Frequency of samples collected from horse herds in the mesoregions of Rio Grande do Sul State (RS). The percentages indicate the number of equine serum samples obtained from each mesoregion. B: Seroprevalence of P. insidiosum infection in horses from each mesoregion of RS. In this map, it is possible to identify the relative frequency of anti-P insidiosum antibodies in equine herds sampled in each mesoregion. Inside the parentheses are shown the absolute number of positive samples out of the total number of samples collected in each mesoregion.
The samples of equine serum used in this study were obtained from official veterinary service (Serviço Veterinário Oficial, SVO) linked to the Secretaria da Agricultura, Pecuária e Agronegócio of RS (SEAPA-RS). These samples were collected for the investigation of equine infectious anemia (EIA) in 2014. The sample type was defined in two stages. In the first stage, it was randomly drawn a predetermined number of farms that met the prerequisites of the study target population (presence of at least one equine above six months old). In the second stage, it was drawn systematically, a predetermined number of horses > six months of age. The calculation of sample size of farms was determined by THRUSFIELD (2007THRUSFIELD, M. Veterinary epidemiology. Oxford: Wiley-Blackwell, 2007.610p.) and performed in Ausvet software (SERGEANT, 2013SERGEANT, E.S.G. Epitools epidemiological calculators. AusVet Animal Health Services and Australian Biosecurity Cooperative Research Centre for Emerging Infectious Disease. 2013. Available from: <Available from: <http://epitools.ausvet.com.au
>. Accesed: nov. 29, 2014.
http://epitools.ausvet.com.au...
).The sampling parameters were defined based on the number of farms that had at least one equine with more than six months of age (n=103,180) enrolled in the equivalent to agricultural protection system (Sistema de Defesa Agropecuária, SDA) of RS. To calculate the sample, as the farm being a sampling unit, it was taken a confidence level of 95% and an absolute accuracy of 5%. Based on the described parameters, the number of samples was set 338 farms sampled in proportion to the number of registered properties in each mesoregion of RS. For the second stage, one prevalence intra herd of 25% was assumed a priori, and this simulated scenarios with different sensitivity and specificity until reaching 100%. As a result, in farms with less than 10 animals, all animals were sampled. In properties with more than 10 horses, samples of 10 animals were systematically collected. All of the samples were obtained during the months of September to October 2013 and totalized 1,002 sera, being 596 serum samples from females and 406 from males.
Indirect ELISA test was performed according to the methodology described by SANTURIO et al. (2006SANTURIO, J.M. et al. Indirect ELISA for the serodiagnostic of pythiosis., Pesquisa Veterinária Brasileira v.26, n.1, p.47-50, 2006. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2006000100010.
http://www.scielo.br/scielo.php?script=s...
). Each serum sample was tested in triplicate. Positive and negative control samples derived from seropositive and negative animals previously tested were used in all tests. The cut off point was based on the average value of the optical density (OD) added three times the standard deviation of the negative samples, with a confidence level of 99.8%. Samples with titre above the cut off were considered positive for P. insidiosum infection.
The data generated from the indirect ELISA were recorded and analyzed using the statistical software R, version 2.15.1. The cross-tabulation and descriptive statistics were used. All variables were screened first on the basis of the results of the response from ELISA test (positive or negative), and the variables with large amounts of missing data (>10%) and limited variability (<20%) were not included in these analysis. The other variables were inserted in a univariate logistic regression model when P<0.05, and it was built two models: one for animal level where the dependent variable was the presence or absence of anti-P insidiosum antibodies, in 1,002 horses, and one for herd level where the dependent variable was the presence of a positive animal by P. insidiosum infection (338 properties). The prevalence of anti-P. insidiosum antibodies at animal level was calculated with 95% confidence level using an exact binomial distribution (CLOPPER & PEARSON, 1934CLOPPER, C.J.; PEARSON, E.S. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika, v.26, p.404-413, 1934. Available from: <Available from: <http://www.jstor.org/discover/10.2307/2331986?uid=3737664&uid=2&uid=4&sid=21105329849283
>. Accessed: nov. 29, 2014.
http://www.jstor.org/discover/10.2307/23...
). Descriptive statistical analyzes were performed using Microsoft Office Excel(r) and R v.2.15.2 (R Development Core Team, 2012) programs. The spatial analyses were performed using ArcGis(r) 10 program.
RESULTS:
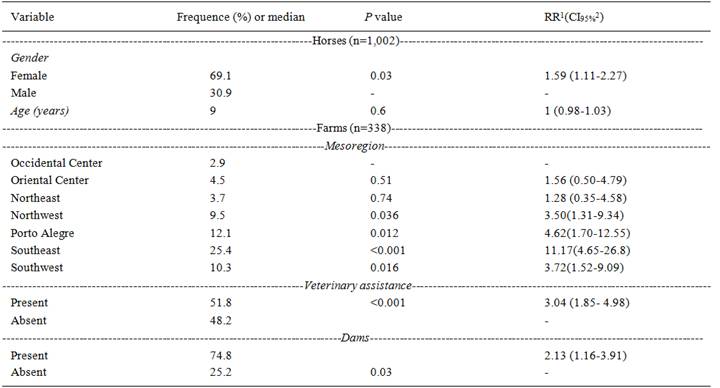
A total of 11.1% (CI95% 9.23 to 13.22) of the equine serum samples tested in RS, were seropositive for P. insidiosum infection, considering a cutoff in the indirect ELISA ≥0.1210. This study confirmed the presence of anti-P. insidiosum antibodies in the mesoregions of RS, specially Southeast, Metropolitan area of Porto Alegre, Southwest and Northwest (Table 1, Figure 1B). For logistic regression analysis in animal level it was indentified more probability for P. insidiosum infection in females with RR of 1.59 (CI95%, 1.11 to 2.27) compared to males (Table 1). In the analysis at the herd level, which evaluated the presence of water sources (dams, lakes, rivers and streams), it was also perceived as associated to the presence of dams with RR of 2.13 (CI95%, 1.16 to 3.91) compared to farms without dam. Furthermore, it was evidenced the relationship of anti-P. insidiosum antibodies in farms with veterinary assistance with RR of 3.04 (CI95%, 1.85 to 4.98).
In regard to the animal and farm levels, the following data were evaluated: age, breed, coat color, total area of each farm and purpose of horse breeding (work or recreational). Nevertheless, these variables were not significant (P≥0.05) (data not shown).
DISCUSSION:
In this study, it was verified that the seroprevalence for P. insidiosum infection in horse herds in RS was of 11.1%. This research was based on a sample design for EIA from the number of horses registered at SVO (SEAPA-RS), being representative of the number of animals presented in the horse herd of RS, in each mesoregion evaluated. For this reason, epidemiological information regarding to pythiosis, such as the presence of cutaneous lesions in animals, treatment alternatives and cure rates of this equine infection were not recorded.
Pythiosis is a very serious disease that causes important sanitary and economical problems in equine herds. In Brazil, RS is considered an endemic region for equine pythiosis, and its diagnosis is reported mainly in the Center and Southern regions of RS. MARCOLONGO-PEREIRA et al (2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
) verified that in samples of equine skin lesions from Southern RS, 14.5% were diagnosed as pythiosis. Previously, PIEREZAN et al (2009PIEREZAN, F. et al. Necropsy findings related to the cause of death in 335 horses: 1968-2007., Pesquisa Veterinária Brasileira v.29, n.3, p.275-280, 2009. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2009000300015
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2009000300015.
http://www.scielo.br/scielo.php?script=s...
) performed a retrospective study of necropsies in equine, during the years 1968 and 2007, in the Center region of RS, and the authors evinced that 3.58% of the cases were due to P. insidiosum infection. In the same region, during the period of 1999 and 2009, equine pythiosis represented 8.3% of the skin tumors diagnosed in horses (SOUZA et al., 2011SOUZA, T.M. et al. Prevalence of equine skin tumors diagnosed at the Laboratory of Veterinary Pathology of the Federal University Santa Maria, Rio Grande do Sul, Brazil., Pesquisa Veterinária Brasileira v.31, n.5, p.379-382, 2011. Available from: <Available from: <http://www.scielo.br/scielo.php?pid=S0100-736X2011000500003&script=sci_arttext
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2011000500003.
http://www.scielo.br/scielo.php?pid=S010...
). In the current study, it was evident the presence of anti-P. insidiosum antibodies in equine in the mesoregions of RS, mainly in the Southest, Porto Alegre, Southwest and Northwest (Figure 1B).
The serological technique used in this research was the indirect ELISA, considered a sensitive and specific method to detect pythiosis in humans and various animal species (MENDOZA et al., 1997MENDOZA, L. et al. Serodiagnosis of human and animal pythiosis using an enzyme-linked immunosorbent assay. Clinical and Diagnostic Laboratory Immunology, v.4, n.6, p.715-718, 1997. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC170646/
>. Accessed: nov. 29, 2014.
http://www.ncbi.nlm.nih.gov/pmc/articles...
; GROOTERS et al., 2002GROOTERS, A.M. et al. Development and evaluation of an enzyme-linked immunosorbent assay for the serodiagnosis of pythiosis in dogs. Journal of Veterinary Internal Medicine, v.16, n.2, p.142-146, 2002. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/11899028
>. Accessed: nov. 29, 2014.
http://www.ncbi.nlm.nih.gov/pubmed/11899...
; VANITTANAKOM et al., 2004VANITTANAKOM, N. et al. Identification of emerging human-pathogenic Pythium insidiosum by serological and molecular assay-based methods. Journal of Clinical Microbiology, v.42, n.9, p.3970-3974, 2004. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC516349/
>. Accessed: nov. 29, 2014. doi: 10.1128/JCM.42.9.3970-3974.2004.
http://www.ncbi.nlm.nih.gov/pmc/articles...
; PEREZ et al., 2005PEREZ, R.C. et al. Epizootic cutaneous pythiosis in beef calves.Veterinary Microbiology, v.109, n.1-2, p.121-128, 2005. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/15961262
>. Accessed: Nov. 29, 2014. doi: 10.1016/j.vetmic.2005.04.020.
http://www.ncbi.nlm.nih.gov/pubmed/15961...
; SANTURIO et al., 2006SANTURIO, J.M. et al. Indirect ELISA for the serodiagnostic of pythiosis., Pesquisa Veterinária Brasileira v.26, n.1, p.47-50, 2006. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2006000100010.
http://www.scielo.br/scielo.php?script=s...
). CHAREONSIRISUTHIGUL et al. (2013CHAREONSIRISUTHIGUL, T. et al. Performance comparison of immunodiffusion, enzyme-linked immunosorbent assay, immunochromatography and hemagglutination for serodiagnosis of human pythiosis. Diagnostic Microbiology and Infectious Disease, v.76, n.1, p.42-45, 2013. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/23537786
>. Accessed: nov. 29, 2014. doi: 10.1016/j.diagmicrobio.2013.02.025.
http://www.ncbi.nlm.nih.gov/pubmed/23537...
) conducted a comparative study with the following techniques: ELISA, immunodiffusion, immunochromatography and hemagglutination. The authors concluded that regardless of the type of antigen used, ELISA is the technique with better accuracy for pythiosis diagnosis. In addition, the indirect ELISA for the diagnosis of pythiosis in horses developed by SANTURIO et al. (2006SANTURIO, J.M. et al. Indirect ELISA for the serodiagnostic of pythiosis., Pesquisa Veterinária Brasileira v.26, n.1, p.47-50, 2006. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2006000100010.
http://www.scielo.br/scielo.php?script=s...
) demonstrated a sensitivity of 97.72% and a specificity of 90.27%.
There was significant difference between the gender of horses, being observed that females had incresead serological response of anti-P insidiosum antibodies. Although the number of females was higher in the study, a higher prevalence in females was also verified by LEAL et al. (2001LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf
>. Accessed: nov. 29, 2014.
http://www.scielo.br/pdf/pvb/v21n4/7481....
) and SANTOS et al. (2014SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
http://www.j-evs.com/article/S0737-0806(...
) in the Brazilian Pantanal, as well as MOSBAH et al. (2012MOSBAH, E. et al. Diagnosis and surgical management of pythiosis in draft horses: report of 33 cases in Egypt. Journal of Equine Veterinary Science, v.32, n.3, p.164-169, 2012. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(11)00503-X/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2011.08.014.
http://www.j-evs.com/article/S0737-0806(...
) in Egypt.
P. insidiosum is an acquatic oomycete and its cycle is closely related to the presence of water and grass. In the farms with the presence of dams it was evidenced the highest frequency of horses with anti-P. insidiosum antibodies. Other authors have also associated the presence of water with P. insidiosum infection (CHAFFIN et al., 1995CHAFFIN, M.K. et al. Cutaneous pythiosis in the horse. Veterinary Clinics of North America: Equine Practice, v.11,n.1, p.91-103, 1995. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/7634168
>. Accessed: nov. 29, 2014.
http://www.ncbi.nlm.nih.gov/pubmed/76341...
; MARCOLONGO-PEREIRA et al., 2012MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
http://www.scielo.br/scielo.php?script=s...
; SANTOS et al., 2014SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract
>. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
http://www.j-evs.com/article/S0737-0806(...
).
Another important statement in this study was the association between the presence of anti-P.insidiosum antibodies with veterinary assistance to the farms. This may be related to immunotherapeutic treatment, whereas the indirect ELISA is not able to distinguish sick or P.insidiosum-inoculated animals. Regarding the humoral immunity, the ELISA is mentioned as highly sensitive and specific test (SANTURIO et al., 2006SANTURIO, J.M. et al. Indirect ELISA for the serodiagnostic of pythiosis., Pesquisa Veterinária Brasileira v.26, n.1, p.47-50, 2006. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010
>. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2006000100010.
http://www.scielo.br/scielo.php?script=s...
). In the present study, we observed a difference in the magnitude of the humoral response, since the antibody titers in ELISA were mostly low (data not shown). However, NEWTON & ROSS (1993NEWTON, J.C.; ROSS, P.S. Equine pythiosis: an overview of immunotherapy. Compendium on Continuing Education for the Practising Veterinarian-North American Edition, v.15, p.491-493, 1993.) and SANTOS et al. (2011SANTOS, C.E.P. et al. Does immunotherapy protect equines from reinfection by the oomycete Pythium insidiosum? Clinical and Vaccine Immunology, v.18, n.8, p.1397-1399, 2011. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3147340/
>. Accessed: nov. 29, 2014. doi: 10.1128/CVI.05150-11.
http://www.ncbi.nlm.nih.gov/pmc/articles...
) stated that high titers of anti-P insidiosum antibodies were detected in sick and recently vaccinated animals. In contrast, MENDOZA et al. (1992MENDOZA, L. et al. Evaluation of two vaccines for the treatment of pythiosis insidiosi in horses. Mycopathologia, v.119, p.89-95, 1992. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/1435952
>. Accessed: mar. 12, 2015.
http://www.ncbi.nlm.nih.gov/pubmed/14359...
) observed that antibody levels in treated horses did not increase even after treatment and disappeared after healing. Thus, it is suggested that greater part of the equine livestock evaluated in this study had antibody response to healing or even be due to the result of antigenic stimulus arising from the presence of the agent in the environment. It is believed that most mammals are resistant to infection by P. insidiosum and some eventually will develop the disease, possibly due to failure in the initial recognition of the antigen (VILELA &MENDOZA, 2013MENDOZA, E.; VILELA, R. The mammalian pathogenic oomycetes. Current Fungal Infection Reports, v.7, n.3, p.198-208, 2013. Available from: <Available from: <http://link.springer.com/article/10.1007%2Fs12281-013-0144-z
>. Accessed: nov. 29, 2014. doi: 10.1007/s12281-013-0144-z.
http://link.springer.com/article/10.1007...
; ZANETTE et al., 2013ZANETTE, R.A. et al. Toll deficient Drosophila is susceptible to Pythium insidiosum infection. Microbiology Immunology, v.57, n.10, p.732-735, 2013. Available from: <Available from: <http://onlinelibrary.wiley.com/doi/10.1111/1348-0421.12082/abstract
>. Accessed: Nov. 29, 2014. doi: 10.1111/1348-0421.12082.
http://onlinelibrary.wiley.com/doi/10.11...
). However, further studies are needed to prove these hypotheses.
CONCLUSION:
Due to the socioeconomic importance of equine breeding in Brazil, here we highlight the presence of anti-P insidiosum antibodies in horses in RS, indicating that the agent is present in different regions of the RS in Southern Brazil. It is also emphasized that there is need for further studies involving the epidemiology in a representative number of horse herds in Brazil.
ACKNOWLEDGMENTS
The authors thanks for the financial support from Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS) (Process 6386.284.15435.05062013) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (Process 473162/2013-0). Additional acknowledgedments to FAPERGS by the scholarship for the first author (Process 2443.269.15435.28022013).
REFERENCES:
- ALMEIDA, F.Q.; SILVA, V.P. Scientific progress in equine production at 1st decade of the XXI century. Revista Brasileirade Zootecnia, v.39, p.119-129, 2010. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-35982010001300014 >. Accessed: nov. 29, 2014. doi: 10.1590/S1516-35982010001300014.
» https://doi.org/10.1590/S1516-35982010001300014» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1516-35982010001300014 - CHAFFIN, M.K. et al. Cutaneous pythiosis in the horse. Veterinary Clinics of North America: Equine Practice, v.11,n.1, p.91-103, 1995. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/7634168 >. Accessed: nov. 29, 2014.
» http://www.ncbi.nlm.nih.gov/pubmed/7634168 - CHAREONSIRISUTHIGUL, T. et al. Performance comparison of immunodiffusion, enzyme-linked immunosorbent assay, immunochromatography and hemagglutination for serodiagnosis of human pythiosis. Diagnostic Microbiology and Infectious Disease, v.76, n.1, p.42-45, 2013. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/23537786 >. Accessed: nov. 29, 2014. doi: 10.1016/j.diagmicrobio.2013.02.025.
» https://doi.org/10.1016/j.diagmicrobio.2013.02.025» http://www.ncbi.nlm.nih.gov/pubmed/23537786 - CLOPPER, C.J.; PEARSON, E.S. The use of confidence or fiducial limits illustrated in the case of the binomial. Biometrika, v.26, p.404-413, 1934. Available from: <Available from: <http://www.jstor.org/discover/10.2307/2331986?uid=3737664&uid=2&uid=4&sid=21105329849283 >. Accessed: nov. 29, 2014.
» http://www.jstor.org/discover/10.2307/2331986?uid=3737664&uid=2&uid=4&sid=21105329849283 - GROOTERS, A.M. et al. Development and evaluation of an enzyme-linked immunosorbent assay for the serodiagnosis of pythiosis in dogs. Journal of Veterinary Internal Medicine, v.16, n.2, p.142-146, 2002. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/11899028 >. Accessed: nov. 29, 2014.
» http://www.ncbi.nlm.nih.gov/pubmed/11899028 - IBGE (INSTITUTO BRASILEIRO DE GEOGRAFIA E ESTATÍSTICA). 2014. Available from: <Available from: <http://www.ibge.gov.br/home/estatistica/economia/agropecuaria/censoagro/default.shtm >. online. Accessed: nov. 29, 2014.
» http://www.ibge.gov.br/home/estatistica/economia/agropecuaria/censoagro/default.shtm - LEAL, A.B.M. et al. Equine pythiosis in the Brazilian Pantanal region: Clinical and pathological findings of typical and atypical cases. Pesquisa Veterinária Brasileira, v.21, n.4, p.151-156, 2001. Available from: <Available from: <http://www.scielo.br/pdf/pvb/v21n4/7481.pdf >. Accessed: nov. 29, 2014.
» http://www.scielo.br/pdf/pvb/v21n4/7481.pdf - MAPA, 2014. Ministério da Agricultura Ministério da Agricultura, Pecuária e Abastecimento. Available from: <Available from: <http://www.agricultura.gov.br/animal/especies/equideos >. Accessed: nov. 29, 2014.
» http://www.agricultura.gov.br/animal/especies/equideos - MARCOLONGO-PEREIRA, C. et al. Epidemiology of equine pythiosis in southern of Rio Grande do Sul State, Brazil., Pesquisa Veterinária Brasileira v.32, n.9, p.865-868, 2012. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009 >. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2012000900009.
» https://doi.org/10.1590/S0100-736X2012000900009» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2012000900009 - MARCOLONGO-PEREIRA, C. et al. Equine diseases in Southern Brazil., Pesquisa Veterinária Brasileira v.34, n.3, p.205-210, 2014. Available from: <Available from: <http://www.scielo.br/scielo.php?pid=S0100736X2014000300002&script=sci_arttext >. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2014000300002.
» https://doi.org/10.1590/S0100-736X2014000300002» http://www.scielo.br/scielo.php?pid=S0100736X2014000300002&script=sci_arttext - MENDOZA, L. et al. Evaluation of two vaccines for the treatment of pythiosis insidiosi in horses. Mycopathologia, v.119, p.89-95, 1992. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/1435952 >. Accessed: mar. 12, 2015.
» http://www.ncbi.nlm.nih.gov/pubmed/1435952 - MENDOZA, L. et al. Infections caused by the oomycetous pathogen Pythium insidiosumJournal Medical Mycology, v.6, n.4, p.151-164, 1996.
- MENDOZA, L. et al. Serodiagnosis of human and animal pythiosis using an enzyme-linked immunosorbent assay. Clinical and Diagnostic Laboratory Immunology, v.4, n.6, p.715-718, 1997. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC170646/ >. Accessed: nov. 29, 2014.
» http://www.ncbi.nlm.nih.gov/pmc/articles/PMC170646/ - MENDOZA, E.; VILELA, R. The mammalian pathogenic oomycetes. Current Fungal Infection Reports, v.7, n.3, p.198-208, 2013. Available from: <Available from: <http://link.springer.com/article/10.1007%2Fs12281-013-0144-z >. Accessed: nov. 29, 2014. doi: 10.1007/s12281-013-0144-z.
» https://doi.org/10.1007/s12281-013-0144-z» http://link.springer.com/article/10.1007%2Fs12281-013-0144-z - MOSBAH, E. et al. Diagnosis and surgical management of pythiosis in draft horses: report of 33 cases in Egypt. Journal of Equine Veterinary Science, v.32, n.3, p.164-169, 2012. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(11)00503-X/abstract >. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2011.08.014.
» https://doi.org/10.1016/j.jevs.2011.08.014» http://www.j-evs.com/article/S0737-0806(11)00503-X/abstract - NEWTON, J.C.; ROSS, P.S. Equine pythiosis: an overview of immunotherapy. Compendium on Continuing Education for the Practising Veterinarian-North American Edition, v.15, p.491-493, 1993.
- PEREIRA, D.I.B. et al. In vitro zoosporogenesis among oomycetes Pythium insidiosum isolates. Ciência Rural, v.38, n.1, p.143-147, 2008. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782008000100023 >. Accessed: nov. 29, 2014. doi: 10.1590/S0103-84782008000100023.
» https://doi.org/10.1590/S0103-84782008000100023» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782008000100023 - PEREZ, R.C. et al. Epizootic cutaneous pythiosis in beef calves.Veterinary Microbiology, v.109, n.1-2, p.121-128, 2005. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pubmed/15961262 >. Accessed: Nov. 29, 2014. doi: 10.1016/j.vetmic.2005.04.020.
» https://doi.org/10.1016/j.vetmic.2005.04.020» http://www.ncbi.nlm.nih.gov/pubmed/15961262 - PIEREZAN, F. et al. Necropsy findings related to the cause of death in 335 horses: 1968-2007., Pesquisa Veterinária Brasileira v.29, n.3, p.275-280, 2009. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2009000300015 >. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2009000300015.
» https://doi.org/10.1590/S0100-736X2009000300015» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2009000300015 - SALLIS, E.S.V. et al. Cutaneous pythiosis in horses: 14 cases., Ciência Rural v.33, n.5, p.899-903, 2003. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782003000500017 >. Accessed: nov. 29, 2014. doi: 10.1590/S0103-84782003000500017.
» https://doi.org/10.1590/S0103-84782003000500017» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-84782003000500017 - SANTOS, C.E.P. et al. Does immunotherapy protect equines from reinfection by the oomycete Pythium insidiosum? Clinical and Vaccine Immunology, v.18, n.8, p.1397-1399, 2011. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3147340/ >. Accessed: nov. 29, 2014. doi: 10.1128/CVI.05150-11.
» https://doi.org/10.1128/CVI.05150-11» http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3147340/ - SANTOS, C.E.P. et al. Epidemiological survey of equine pythiosis in the Brazilian Pantanal and nearby areas: results of 76 cases., Journal of Equine Veterinary Science v.34, n.2, p.270-274, 2014. Available from: <Available from: <http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract >. Accessed: nov. 29, 2014. doi: 10.1016/j.jevs.2013.06.003.
» https://doi.org/10.1016/j.jevs.2013.06.003» http://www.j-evs.com/article/S0737-0806(13)00415-2/abstract - SANTURIO, J.M. et al. Indirect ELISA for the serodiagnostic of pythiosis., Pesquisa Veterinária Brasileira v.26, n.1, p.47-50, 2006. Available from: <Available from: <http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010 >. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2006000100010.
» https://doi.org/10.1590/S0100-736X2006000100010» http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-736X2006000100010 - SERGEANT, E.S.G. Epitools epidemiological calculators. AusVet Animal Health Services and Australian Biosecurity Cooperative Research Centre for Emerging Infectious Disease. 2013. Available from: <Available from: <http://epitools.ausvet.com.au >. Accesed: nov. 29, 2014.
» http://epitools.ausvet.com.au - SOUZA, T.M. et al. Prevalence of equine skin tumors diagnosed at the Laboratory of Veterinary Pathology of the Federal University Santa Maria, Rio Grande do Sul, Brazil., Pesquisa Veterinária Brasileira v.31, n.5, p.379-382, 2011. Available from: <Available from: <http://www.scielo.br/scielo.php?pid=S0100-736X2011000500003&script=sci_arttext >. Accessed: nov. 29, 2014. doi: 10.1590/S0100-736X2011000500003.
» https://doi.org/10.1590/S0100-736X2011000500003» http://www.scielo.br/scielo.php?pid=S0100-736X2011000500003&script=sci_arttext - THRUSFIELD, M. Veterinary epidemiology. Oxford: Wiley-Blackwell, 2007.610p.
- VANITTANAKOM, N. et al. Identification of emerging human-pathogenic Pythium insidiosum by serological and molecular assay-based methods. Journal of Clinical Microbiology, v.42, n.9, p.3970-3974, 2004. Available from: <Available from: <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC516349/ >. Accessed: nov. 29, 2014. doi: 10.1128/JCM.42.9.3970-3974.2004.
» https://doi.org/10.1128/JCM.42.9.3970-3974.2004» http://www.ncbi.nlm.nih.gov/pmc/articles/PMC516349/ - ZANETTE, R.A. et al. Toll deficient Drosophila is susceptible to Pythium insidiosum infection. Microbiology Immunology, v.57, n.10, p.732-735, 2013. Available from: <Available from: <http://onlinelibrary.wiley.com/doi/10.1111/1348-0421.12082/abstract >. Accessed: Nov. 29, 2014. doi: 10.1111/1348-0421.12082.
» https://doi.org/10.1111/1348-0421.12082» http://onlinelibrary.wiley.com/doi/10.1111/1348-0421.12082/abstract
-
1
CR-2015-0056.R2
Publication Dates
-
Publication in this collection
17 Nov 2015 -
Date of issue
Jan 2016
History
-
Received
19 Jan 2015 -
Accepted
20 Apr 2015 -
Reviewed
22 July 2015