Abstracts
Starch gel electrophoresis with L-leucyl-beta-naphthylamide as substrate revealed five aminopeptidases in extracts of Polistes versicolor. These enzymes are presumably products of five structural gene loci. All but Lap¹ aminopeptidases exhibited differential distribution in the developmental stages and in the tissues. Five dipeptidases were revealed with different dipeptides. These enzymes exhibited significant differences in their substrate preferences, but a more homogeneous distribution throughout ontogenetic developmental stages than did aminopeptidases. Electrophoretic variants of Lap4 and PepA² were detected and although a low intralocus heterozygosity was found due to the low frequency of these variants, phenotypical segregation observed at these loci in pupae extracts of some colonies points to the occurrence of more than one egg-laying female. Otherwise, the detection of Lap4 allozyme restricted to nests of one area suggests low dispersion ability of the adults of Polistes versicolor.
peptidases; substrate preference; developmental distribution; genetic variation; Polistes versicolor
Cinco aminopeptidases foram detectadas com o substrato L-leucil-beta-naftilamida em extratos de Polistes versicolor, presumidamente produtos de cinco locos gênicos distintos. Todas as aminopeptidases, exceto Lap¹, apresentaram distribuição diferencial nos estágios do desenvolvimento e nos tecidos. Cinco dipeptidases foram reveladas com a utilização de diferentes dipeptídeos. Essas enzimas exibiram marcantes diferenças na preferência aos substratos testados, mas distribuição mais homogênea no desenvolvimento ontogenético que as aminopeptidases. Variantes eletroforéticas de Lap4 e PepA² foram detectadas em colônias de P. versicolor e, embora heterozigosidades intralocos relativamente baixas tenham sido observadas em razão da baixa freqüência das variantes detectadas, a segregação dos fenótipos de Lap4 e PepA² em pupas de alguns ninhos indica a ocorrência de mais de uma fêmea ovipositora. Além disso, a detecção de aloenzimas restritas aos ninhos de uma área sugere baixa capacidade de dispersão dos adultos dessa espécie.
peptidases; preferência por substratos; distribuição no desenvolvimento; variação genética; Polistes versicolor
Genetic characterization of the peptidases of Polistes versicolor (Hymenoptera: Vespidae)
Caracterização genética das peptidases de Polistes versicolor (Hymenoptera: Vespidae)
Del Lama, m. A.; Ferreira, K. M.
Departamento de Genética e Evolução, Universidade Federal de São Carlos, Rodovia Washington Luís, km 235, CEP 13565-905, São Carlos, SP, Brazil
Correspondence Correspondence to Marco Antonio Del Lama Departamento de Genética e Evolução, Universidade Federal de São Carlos Rodovia Washington Luís, km 235 CEP 13565-905, São Carlos, SP, Brazil E-mail: dmdl@power.ufscar.br
ABSTRACT
Starch gel electrophoresis with L-leucyl-b-naphthylamide as substrate revealed five aminopeptidases in extracts of Polistes versicolor. These enzymes are presumably products of five structural gene loci. All but Lap1 aminopeptidases exhibited differential distribution in the developmental stages and in the tissues. Five dipeptidases were revealed with different dipeptides. These enzymes exhibited significant differences in their substrate preferences, but a more homogeneous distribution throughout ontogenetic developmental stages than did aminopeptidases. Electrophoretic variants of Lap4 and PepA2 were detected and although a low intralocus heterozygosity was found due to the low frequency of these variants, phenotypical segregation observed at these loci in pupae extracts of some colonies points to the occurrence of more than one egg-laying female. Otherwise, the detection of Lap4 allozyme restricted to nests of one area suggests low dispersion ability of the adults of Polistes versicolor.
Key words: peptidases, substrate preference, developmental distribution, genetic variation, Polistes versicolor.
RESUMO
Cinco aminopeptidases foram detectadas com o substrato L-leucil-b-naftilamida em extratos de Polistes versicolor, presumidamente produtos de cinco locos gênicos distintos. Todas as aminopeptidases, exceto Lap1, apresentaram distribuição diferencial nos estágios do desenvolvimento e nos tecidos. Cinco dipeptidases foram reveladas com a utilização de diferentes dipeptídeos. Essas enzimas exibiram marcantes diferenças na preferência aos substratos testados, mas distribuição mais homogênea no desenvolvimento ontogenético que as aminopeptidases. Variantes eletroforéticas de Lap4 e PepA2 foram detectadas em colônias de P. versicolor e, embora heterozigosidades intralocos relativamente baixas tenham sido observadas em razão da baixa freqüência das variantes detectadas, a segregação dos fenótipos de Lap4 e PepA2 em pupas de alguns ninhos indica a ocorrência de mais de uma fêmea ovipositora. Além disso, a detecção de aloenzimas restritas aos ninhos de uma área sugere baixa capacidade de dispersão dos adultos dessa espécie.
Palavras-chave: peptidases, preferência por substratos, distribuição no desenvolvimento, variação genética, Polistes versicolor.
INTRODUCTION
Peptidases (EC 3.4) are enzymes that act in a peptide bond near the N- or C- termini portion or inside a polypeptide chain. In this structurally and functionally heterogeneous group, aminopeptidases (EC 3.4.11) are the most electrophoretically studied enzymes in plant and animal species. Among insects, aminopeptidases of Drosophila have been well characterized (Beckman & Johnson, 1964; Beckman et al., 1964; Sakai et al., 1969; Walker & Williamson, 1980; Walker et al., 1981; Hall, 1986, 1988), but aminopeptidases from other insects such as the coleopteran Attagenus megatoma (Baker & Woo, 1981), the moth Tineola (Ward, 1975), the dipteran tsétsé fly Glossina (Gooding & Rolselth, 1976), and the lepidopteran Sesamia nonagrioides Lef. (Ortego et al., 1996) have also been studied. Reports about complete cDNA sequences of different aminopeptidases as well as data indicating developmental and differential regulation of aminopeptidase expression are now available (Hua et al., 1998; Zhu et al., 2000; Emmerling et al., 2001).
Dipeptidases (EC 3.4.13) are enzymes that hydrolyze single amino acids from small peptides. These enzymes are less known (Del Lama et al., 1992) but dipeptidases of insect species such as Drosophila melanogaster (Voelker & Langley, 1978; Laurie-Ahlberg, 1982) and Calliphora erythrocephala (Collett, 1989) have been electrophoretically studied.
Despite their presumptive physiological significance, little data on the genetic properties of hymenopteran peptidases are available. Del Lama & Mestriner (1984) described comparative electrophoretic profiles of peptidases from fourteen bee species according to their substrate preference, developmental distribution, and to the occurrence of electrophoretic variants. Aminopeptidases of Apis mellifera have recently been better characterized based on their substrate preferences, the differential inhibitory effects of 1,10-phenanthroline and EDTA, and differential developmental and tissue distributions of these enzymes in adult drones, workers, and queens (Del Lama et al., 2001).
Polistes (Aphanilopterus) versicolor (Olivier), a very common wasp in urban areas, is neotropically distributed from Costa Rica to southern Brazil and Argentina (Richards, 1978) and much of its biology has been studied (Gobbi & Zucchi, 1980, 1985; Rodrigues, 1982; Itô, 1984; Valadão, 1986; Carneiro, 1991). It has been considered primitively eusocial, with independent-type nesting behaviour (Gadagkar, 1990), with the nest being founded by an association of females and with reproductive dominance characterized by physical aggression of the dominant female directed at the associated females. According to Giannotti & Mansur (1993), these associated females tend to be sisters.
This paper describes the electrophoretic patterns of the aminopeptidases and dipeptidases of Polistes versicolor, their substrate preferences, developmental and tissue distributions, and presence of genetic variants. A preliminary general picture will then be presented comparing the properties of these peptidases with those of other insect groups.
MATERIAL AND METHODS
Males and females of Polistes versicolor were analyzed in different ontogenetic developmental stages. Samples came from 27 nests collected in Franca/SP (Fra) and in eight areas of São Carlos/SP: Distrito de Jardim Paulista (Djp), Centro (Cen), Vila Carmem (Vca), Campus of the Universidade Federal de São Carlos (Ufs), Vila Lutfala (Vlu), Vila Prado (Vpr), Vila São Caetano (Vsc), and Vila São Gabriel (Vsg). Relative distances between these areas range from 300 m to 3.3 km. Sex identification was made by body color and number of abdominal segments.
The nests were taken in the early evening to assure that all adults of the colony were sampled. Each nest was placed in a plastic bag and removed from the support, usually a house wall, and put in a refrigerator for a few minutes to reduce adult activity, and allow separation and storage, done at 20°C, of eggs, larvae, pupae, and adults.
Horizontal electrophoresis was carried out in 14% corn starch gels (Penetrose 30Ô, Corn Brazil S/A) using tris-citrate-borate buffer (0.017M tris + 0.0023M citric acid, pH 8.0, in the gel; 0.3M borate, pH 8.3, in the electrodes). Samples of eggs, larvae (2-6 days), and young and old (pigmented) pupae and adults were prepared in a proportion of 100 mg to 0.2 ml of 0.2% 2-mercaptoethanol solution, centrifuged at room temperature for 15 minutes at 4000 g in an Eppendorf 541C centrifuge, and the supernatants were used for electrophoretic analysis. The electrophoretic run was developed for 4 hours at 10°C at a constant current of 30 mA.
Aminopeptidase activity was detected after incubating the gels for 2 hours at 37°C in 100 ml tris-maleate buffer (0.1M tris+ 0.1M maleic anhydride), pH 5.5, containing 30 mg substrate (aminoacyl derivatives of b-naphthylamine) and 40 mg Fast Garnet GBC salt (Beckman et al., 1964).
Dipeptidase activity was revealed when gels were incubated for 30-60 minutes at 37°C in a mixture of 15 ml 0.2M phosphate buffer, pH 7.5, 20 mg of different di and tripeptides (Table 1), 1 mg crude venom of Crotalus sp., 0.2 ml of a fresh solution of o-dianisidine dihydrochroride (25 mg/ml), 5 U peroxidase, 1 ml MgCl2 0.2M, and 15 ml 2% agar.
The zymograms, revealed using leucyl-b-naphthylamide (leu.NA) and leu-ala, were used as a control to compare the relative substrate preferences of the aminopeptidases and dipeptidases, respectively.
The presence of genetic aminopeptidase and dipeptidase variants was verified in whole extracts of male and female pupae. Allelle frequencies of Lap4 and PepA2 loci were estimated from the phenotypes of four randomly chosen females from 14 and 18 nests, respectively. The phenotypes were used to test genetic equilibrium agreement with the Hardy-Weinberg model.
RESULTS
Substrate preferences
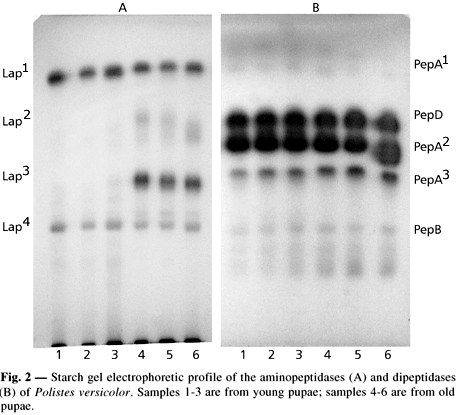
Five aminopeptidases were detected in Polistes versicolor and named Lap1 to Lap5 according to their decreasing anodic mobility. They are presumably products of five structural gene loci; their relative electrophoretic mobilities are illustrated in Figures 1 and 2. As can be seen, Lap2 and Lap3 showed a slightly diffuse banding pattern on the gel.
Table 1 presents the substrate preferences of these aminopeptidases which, as is evident, did not show absolute specificity for the b-naphthylamide derivatives employed. However, their highest relative activities were observed with leu.NA as substrate and, although some of these enzymes could be weakly detected with the few dipeptides used, they can be considered leucylaminopeptidases (EC 3.4.11.1). No activity was observed with L-benzoyl-argynyl-b-naphthylamide (BANA) as substrate.
Five dipeptidases were revealed on the gels (Figs. 1 and 2) and named PepA1, PepD, PepA2, PepA3, and PepB. The designation of PepA, PepB, and PepD followed the nomenclature employed by Harris & Hopkinson (1975) to characterize human dipeptidases.
PepA1 was detected with three of the substrates employed (leu-ala, leu-gly, and leu-tyr), while PepD (prolidase, EC 3.4.13.9) was revealed only with proline dipeptides (leu-pro and phe-pro). PepA2 was detected by using many dipeptides tested at different intensities. PepA3 exhibited electrophoretic mobility close to PepA2 and was detected with most of the dipeptides used. Although acting on some dipeptides, PepB was revealed with tripeptide substrates. This enzyme showed a slightly diffuse pattern on the gel.
Ontogenetic developmental distribution
Data on the distribution of aminopeptidase and dipeptidase activities throughout ontogenetic development are summarized in Fig. 3. Developmental stages of Polistes versicolor were defined according to Giannotti & Mansur (1993): egg 22.4 ± 6.2 days; larva 40.1 ± 7.7 days; and pupa 23.2 ± 9.1 days.
Lap1 was the single aminopeptidase observed in the egg extracts. In young larvae we detected Lap2 and Lap3 activity, while in older larvae Lap4 activity was detected. Only Lap1 and Lap4 are present in the young pupae and pos-defecation larvae, with Lap1 showing higher activity. Black-eyed pigmented body pupae exhibit all but Lap5 aminopeptidases, and in the newly-emerged adults Lap1, Lap3, and Lap4 activity is seen (Fig. 4).
Studies of distribution of dipeptidase activity during development showed PepA2 and PepD presence in egg extracts. PepA2, PepA3, PepB, and PepD were observed in young larvae homogenates, with the highest activity being that of PepD. From older larvae to newly-emerged adult extracts, all dipeptidases are present with different enzyme activity level, from low PepB activity in the older larva to high PepD activity in the newly-emerged adult.
In the head-thorax extracts of adult females were found Lap1, PepA1, PepA2, and PepD activity, while PepB, Lap2, Lap3, Lap4, and Lap5 were detected in abdomen extracts. Lap5 was visualized when abdomen extracts of the individuals presented an intense yellowish color, denoting the presence of food (pollen) inside the digestive tract. PepA3, Lap2, Lap3, and Lap4 activities were absent in head-thorax extracts of adult individuals.
Genetic variants
Lap4 phenotypes found may be explained by a locus with three codominant alleles. Due to the low frequency of the alleles that determine this polymorphism, some expected phenotypes were not observed. The variant S found in nests 12, 13, and 14, collected on the campus of UFSCar, represents a private polymorphism of colonies of this area (Table 2).
Allele frequencies estimated from four randomly chosen females from 14 nests (each mating was equally weighted) resulted in 0.982 ± 0.0125 and 0.018 ± 0.0125 for Lap4 M and Lap4 S alleles, respectively. Due to the very low frequency of the F variant, phenotypes determined by alleles F and S were clustered.
PepA2 phenotypes may be explained by a locus with two codominant alleles. An electrophoretic variant was observed in nests from different areas, in contrast to the private Lap4 polymorphism (Table 2). Considering the phenotypes of four females of 18 colonies, these allele frequencies were obtained: PepA2 F = 0.854 ± 0.029 and PepA2 S = 0.146 ± 0.029.
No significant c2 values were obtained when observed genotype frequencies were compared to the expected genotype frequencies according to the Hardy-Weinberg model. We can, therefore, conclude that Lap4 and PepA2 loci are in genetic equilibrium in this Polistes versicolor sample.
DISCUSSION
The physiologic role of aminopeptidases and dipeptidases is poorly known. However, aminopeptidases are usually more active than carboxypeptidases in insects, strongly suggesting that these enzymes play an important role in protein metabolism (Terra & Ferreira, 1994).
Lap1, characterized by its high electrophoretic mobility towards the anode, is the most active aminopeptidase present in all ontogenetic developmental stages and tissues of males and females of Polistes versicolor. It exhibits no electrophoretic variants. An enzyme with similar properties was detected in each of thirteen Meliponini species (Del Lama & Mestriner, 1984), in Apis mellifera (Lap A; Del Lama et al., 2001), and in Drosophila melanogaster (Lap A; Beckman & Johnson, 1964). The nearly ubiquitous occurrence of this enzyme throughout most tissue, body parts, and developmental stages of these different insect groups points to a rather general metabolic role played by this enzyme, such as regulating aminoacid and small peptide concentrations in the hemolymph, and maintaining osmotic stability and a continuous supply of amino acids for protein anabolism.
A second group of aminopeptidases is characterized by smaller anodic mobility, lower activity compared to the aminopeptidase previously described, and higher activity in the pupal stage. Lap2, Lap3, and Lap4 aminopeptidases of P. versicolor are included in this group. Enzymes with similar properties were also observed in Apis mellifera (LapP, see Del Lama et al., 2001) and in D. melanogaster where, of the six aminopeptidases characterized, five are present exclusively in the pupal stage.
Lap2 and Lap3 activity is not detected from the final period of the larval stage (post-defecation larvae) until mid-pupal stage and their enzyme activity is concentrated in adult abdomen extracts. These findings suggest that such enzymes are probably involved in protein digestion occurring in the digestive tract.
Lap4 seems to be homologous to LapP of Apis mellifera because both show the same developmental distribution, higher activity in the pupal stage, and are detected in adult abdomen extracts.
Lap5 activity, observed in digestive tract homogenates of larvae and adults of P. versicolor, is related to the presence of food inside the digestive tract. A similar enzyme was described in Apis mellifera (LapG, see Del Lama et al., 2001) and Drosophila (Lap A , see Walker & Williamson, 1980).
Comparative analysis of dipeptidase activity among insect groups shows that Polistes versicolor and Drosophila do not exhibit tissue-specific dipeptidases. However, an enzyme, dipeptidase-B, restricted to head-thorax, and another, dipeptidase-C, concentrated in abdomen extracts, were reported in Apis mellifera (Bezerra, 1996). The preference demonstrated by PepD of P. versicolor for proline substrates suggests its homology to a prolidase of Apis mellifera (Del Lama, unpublished results) and to DipA of Drosophila melanogaster (Laurie-Ahlberg, 1982).
Similar electrophoretic profiles of aminopeptidases and dipeptidases were verified in males and females of Polistes. These results were like those described for Drosophila but different from those observed for Apis mellifera, where LapD shows very high activity in the reproductive tract of adult drones, particularly in mucus present either in the mucus gland or the ejaculatory bulb (Del Lama et al., 2001).
Segregational analysis of these polymorphic loci suggests that more than one female is responsible for the simultaneous oviposition. As shown in Table 2, Lap4 and PepA2 segregations observed in nest 13 in black-eyed pupae males presented a highly significant deviation from the 1:1 ratio expected in a heterozygous female for these markers.
The detection of Lap4 variants in adults from nests of a specific area (campus of UFSCar) characterizes a private polymorphism. A similar result was observed for a new alcohol dehydrogenase variant detected in extracts of young pupae of these same colonies (Gruber, unpublished results). According to Slatkin (1981), presence of rare private alelles indicates a low Nem and low gene flow. These observations suggest that adults of Polistes versicolor present low dispersion ability.
The biochemical polymorphisms characterized in this work and others previously described in our laboratory (Simokomaki, 1996) can be powerful tools for estimating population size, intracolonial genetic relationships, and populational viscosity of this wasp species.
Acknowledgments The authors are grateful to Isabel C. Godoy and Caroline V. Gruber for valuable technical assistance. This work was supported by Fundação de Amparo à Pesquisa do Estado de Paulo (FAPESP).
Received April 1, 2002 Accepted July 16, 2002 Distributed May 31, 2003
- BAKER, J. E. & WOO, S. M., 1981, Properties and specificities of a digestive aminopeptidase from larvae of Attagenus megatoma (Coleoptera: Demestidae). Comp. Biochem. Physiol., 69B: 189-193.
- BECKMAN, L. & JOHNSON, F. M., 1964, Genetic control of aminopeptidases in Drosophila melanogaster. Hereditas, 51: 221-230.
- BECKMAN, L., BJORLING, G. & CHRISTODOULOU, C., 1964, Genetics of leucine aminopeptidase isozymes in maize. Genetics, 50: 899-904.
- BEZERRA, R. M., 1996, Caracterização genética das exopeptidases de Apis mellifera. Tese de Mestrado, Universidade Federal de São Carlos, 72p.
- CARNEIRO, A., 1991, Determinação do sistema de substituição de fêmeas dominantes em colônias de Polistes (Vespidae-Polistinae) Tese de Doutorado, UNESP, Rio Claro, 206p.
- COLLETT, J. I., 1989, Characterization of the peptidases of Calliphora: many features allow the utilization of small peptides as an amino acid reservoir. Insect Biochem., 19: 535-547.
- DEL LAMA, M. A. & MESTRINER, M. A., 1984, Starch gel electrophoretic patterns of exopeptidase phenotypes in 14 different species of bees. Braz. J. Genet., 2: 9-20.
- DEL LAMA, S. N.; DEL LAMA, M. A.; MESTRINER, M. A. & MORTARI, N., 1992, Peptidase B polymorphism in cattle erythrocytes. Biochem. Genet., 30: 247-255.
- DEL LAMA, M. A., BEZERRA, R. M., SOARES, A. E. E. & RÚVOLO-TAKASUSUKI, M. C. C., 2001, Genetic, ontogenetic, and tissue-specific variation of aminopeptidases in Apis mellifera Apidologie, 32: 25-35.
- EMMERLING, M., CHANDLER, D. & SANDEMAN, M., 2001, Molecular cloning of three cDNAs encoding aminopeptidases from the midgut of Helicoverpa punctigera, the Australian native budworm. Insect Biochem. Molec. Biol., 31: 899-907.
- GADAGKAR, R., 1990, Evolution of eusociality: the advantages of assured fitness returns. Phil. Trans. R. Soc. London B, 329: 17-25
- GIANNOTTI, E. & MANSUR, C. B., 1993, Dispersion and foundation of new colonies in Polistes versicolor (Oliver) (Hymenoptera: Vespidae). An. Soc. Entomol. Brasil, 22: 307-316.
- GOBBI, N. & ZUCCHI, R., 1980, On the ecology of Polistes versicolor versicolor (Olivier) in southern Brazil (Hymenoptera: Vespidae: Polistini) I. Phenological account. Naturalia, 6: 97-104.
- GOBBI, N. & ZUCCHI, R., 1985, On the ecology of Polistes versicolor versicolor (Olivier) in southern Brazil (Hymenoptera: Vespidae: Polistini) II. Colonial Productivity. Naturalia, 10: 21-25.
- GOODING, R. H. & ROLSELTH, B. M., 1976, Digestive processes of haematophagous insects. XI. Partial purification and some properties of six proteolytic enzymes from the tsetse fly Glossiva morsitans morsitans. Can. J. Zool., 54: 1950-1959.
- HALL, N. A., 1986, Peptidase in Drosophila melanogaster I. Characterization of dipeptidase and leucine aminopeptidase activities. Biochem. Genet., 24: 775-793.
- HALL, N. A., 1988, Peptidases in Drosophila melanogaster II. The variation of peptidase activities during development. Insect Biochem., 18: 145-155.
- HARRIS, H. & HOPKINSON, D. A., 1975, Handbook of enzyme electrophoresis in human genetics North-Holland, Amsterdan.
- HUA, G., TSUKAMOTO, K. & IKEZAWA, H., 1998, Cloning and sequence analysis of the aminopeptidase N isozyme (APN2) from Bombyx mori midgut. Comp. Biochem. Physiol. B. Biochem. Mol. Biol., 121: 213-222.
- ITÔ, Y., 1984, Shifts of females between adjacent nests of Polistes versicolor (Hymenoptera: Vespidae) in Panama. Insectes Soc., 31: 103-111.
- LAURIE-AHLBERG, C. C., 1982, Genetic, ontogenetic, and tissue-specific variation of dipeptidases in Drosophila melanogaster. Biochem. Genet., 20: 407-424.
- ORTEGO, F., NOVLLO, C. & CASTANERA, P., 1996, Characterization and distribution of digestive proteases of the stalk corn borer, Sesamia nonagrioides Lef. (Lepidoptera: Noctuidae). Arch. Insect Biochem. Physiol., 33: 163-180.
- RICHARDS, O. W., 1978, The social wasps of the Americas excluding the Vespinae British Museum (Natural History), London, 580p.
- RODRIGUES, V. M., 1982, Vespídeos sociais: a reabsorção de ovócitos e estrutura social de Polistes (Aphanilopterus) versicolor (Olivier) (Polistinae, Polistini). Dusenia, 13: 123-126.
- SAKAI, R. K., TUNG, D. A. & SCANDALIOS, J. G., 1969, Developmental genetic studies of aminopeptidases in Drosophila melanogaster Mol. Gen. Genet., 105: 24-29.
- SLATKIN, M., 1981, Estimating levels of gene flow in natural populations. Genetics, 99: 323-335.
- SIMOKOMAKI, K., 1996, Parentesco e estrutura genética em colônias de Polistes (Aphanilopterus) versicolor (Olivier, 1791) (Hymenoptera: Vespidae) Tese de Doutorado, Universidade Federal de São Carlos, 139p.
- TERRA, W. R. & FERREIRA, C., 1994, Insect digestive enzymes: properties, compartmentalization and function. Comp. Biochem. Physiol., 109B: 1-62.
- VALADÃO, R., 1986, Dados sobre a interação prole-adultos em Polistes versicolor, Olivier (Vespidae-Polistinae) Dissertação de Mestrado, UNESP, Rio Claro, 136p.
- VOELKER, R. A. & LANGLEY, C. H., 1978, Dipeptidase-A: a polymorphic locus in Drosophila melanogaster. Genetica, 49: 233-236.
- WALKER, V. & WILLIAMSON, J. H., 1980, Ontogeny and tissue distribution of leucine aminopeptidase in Drosophila melanogaster Insect Biochem., 10: 535-541.
- WALKER, V. K., WILLIAMSON, J. H. & CHURCH, R. B., 1981, Differential characterization of two leucine aminopeptidases in Drosophila melanogaster Biochem. Genet., 19: 47-60.
- WARD, C. W., 1975, Aminopeptidases in webbing clothes moth larvae. Properties and specificity of the major enzyme of low electrophoretic mobility. Int. J. Biochem., 6: 765-768.
- ZHU, Y. C., KRAMER, K. J., OPPERT, B. & DOWDY, A. K., 2000, cDNAs of aminopeptidase-like protein genes from Plodia interpunctella strains with different susceptibilities to Bacillus thuringiensis toxins. Insect Biochem. Molec. Biol., 30: 215-224.
Correspondence to
Publication Dates
-
Publication in this collection
15 Aug 2003 -
Date of issue
May 2003
History
-
Accepted
31 May 2003 -
Reviewed
16 July 2002 -
Received
01 Apr 2002