Abstracts
Temporal and spatial variations and environmental factors influencing the structure of tidepool fish assemblages were quantitatively investigated at Iparana beach, northeast Brazilian coast. The majority of the tidepool fishes sampled were suprabenthic juvenile individuals of great mobility. We recorded during monthly diurnal underwater visual censuses a total of 4,750 fish from 26 species, represented mainly by partial residents from the families Scaridae, Haemulidae, Gerreidae and Pomacentridae. The number of species and individuals showed significant variability among tidepools as a response to variations in their volume and type of substrate cover. The greatest species abundance and richness associated with rocks covered with algae suggested that substrate complexity is one of the main factors defining the spatial structure of the tidepool ichthyofauna. Temporal variability in species abundance and richness was associated with changes in salinity levels due to seasonal rainfalls. A higher number of juvenile fishes from December to May in our samples corroborate the hypothesis that the tidepools act as nursery sites. Therefore, the species associations found in this study and their relation to seasonal and spatial discontinuities may be partly explained according to their habitat requirements and reproductive cycles.
tidepool fishes; environmental factors; seasonality; diversity; abundance
Investigamos quantitativamente variações espaciais e temporais, assim como os fatores ambientais que podem influenciar a estrutura da fauna de peixes em poças de maré da praia de Iparana, nordeste do Brasil. A maioria dos peixes registrados eram indivíduos suprabênticos jovens de grande mobilidade. Registramos, através de censos visuais subaquáticos diurnos mensais, um total de 4.750 peixes pertencentes a 26 espécies, representados principalmente por residentes parciais das famílias Scaridae, Haemulidae, Gerreidae e Pomacentridae. Os números de espécies e indivíduos mostraram variação significativa entre as poças amostradas com relação ao seu volume e tipo de cobertura do substrato. As maiores abundância e riqueza de espécies foram associadas à presença de rochas cobertas com algas, sugerindo que a complexidade estrutural do substrato é um dos principais fatores que define a estrutura espacial da ictiofauna em poças de marés. A variabilidade temporal em abundância e riqueza de espécies foi associada com variações de salinidade devido à sazonalidade das chuvas. Um maior número de peixes jovens registrados entre dezembro e maio corrobora a hipótese de que as poças de maré funcionam como berçários. Portanto, as associações de espécies encontradas neste estudo, assim como suas relações de descontinuidades sazonais e espaciais podem ser explicadas parcialmente de acordo com as exigências de hábitat e ciclos reprodutivos.
peixes de poças de maré; fatores ambientais; sazonalidade; diversidade; abundância
ARTICLES
Temporal and spatial variations in tidepool fish assemblages of the northeast coast of Brazil
Variação espacial e temporal na fauna de peixes em poças de maré do nordeste do Brasil
Francisca Edna de Andrade CunhaI, IV, 1 1 Corresponding author: Francisca Edna de Andrade Cunha, e-mail: f_edna@yahoo.com.br ; Cassiano Monteiro-NetoII; Mara Carvalho NottinghamIII, IV
IDepartamento de Sistemática e Ecologia, Universidade Federal da Paraíba, Campus Universitário I, CEP 58059-900, João Pessoa, PB, Brasil
IIDepartamento de Biologia Marinha, Universidade Federal Fluminense, CP 100644, CEP 24001-970, Niterói, RJ, Brasil, e-mail: monteiro@vm.uff.br
IIIInstituto Brasileiro do Meio Ambiente e dos Recursos Naturais Renováveis IBAMA
IVGrupo de Ictiologia Marinha Tropical IMAT UFPE, Departamento de Oceanografia, Av. Arquitetura, s/n, CEP 50740-550, Recife, PE, Brasil, e-mail: mattin@secrel.com.br
ABSTRACT
Temporal and spatial variations and environmental factors influencing the structure of tidepool fish assemblages were quantitatively investigated at Iparana beach, northeast Brazilian coast. The majority of the tidepool fishes sampled were suprabenthic juvenile individuals of great mobility. We recorded during monthly diurnal underwater visual censuses a total of 4,750 fish from 26 species, represented mainly by partial residents from the families Scaridae, Haemulidae, Gerreidae and Pomacentridae. The number of species and individuals showed significant variability among tidepools as a response to variations in their volume and type of substrate cover. The greatest species abundance and richness associated with rocks covered with algae suggested that substrate complexity is one of the main factors defining the spatial structure of the tidepool ichthyofauna. Temporal variability in species abundance and richness was associated with changes in salinity levels due to seasonal rainfalls. A higher number of juvenile fishes from December to May in our samples corroborate the hypothesis that the tidepools act as nursery sites. Therefore, the species associations found in this study and their relation to seasonal and spatial discontinuities may be partly explained according to their habitat requirements and reproductive cycles.
Keywords: tidepool fishes, environmental factors, seasonality, diversity, abundance.
RESUMO
Investigamos quantitativamente variações espaciais e temporais, assim como os fatores ambientais que podem influenciar a estrutura da fauna de peixes em poças de maré da praia de Iparana, nordeste do Brasil. A maioria dos peixes registrados eram indivíduos suprabênticos jovens de grande mobilidade. Registramos, através de censos visuais subaquáticos diurnos mensais, um total de 4.750 peixes pertencentes a 26 espécies, representados principalmente por residentes parciais das famílias Scaridae, Haemulidae, Gerreidae e Pomacentridae. Os números de espécies e indivíduos mostraram variação significativa entre as poças amostradas com relação ao seu volume e tipo de cobertura do substrato. As maiores abundância e riqueza de espécies foram associadas à presença de rochas cobertas com algas, sugerindo que a complexidade estrutural do substrato é um dos principais fatores que define a estrutura espacial da ictiofauna em poças de marés. A variabilidade temporal em abundância e riqueza de espécies foi associada com variações de salinidade devido à sazonalidade das chuvas. Um maior número de peixes jovens registrados entre dezembro e maio corrobora a hipótese de que as poças de maré funcionam como berçários. Portanto, as associações de espécies encontradas neste estudo, assim como suas relações de descontinuidades sazonais e espaciais podem ser explicadas parcialmente de acordo com as exigências de hábitat e ciclos reprodutivos.
Palavras-chave: peixes de poças de maré, fatores ambientais; sazonalidade, diversidade, abundância.
Introduction
Coastal intertidal reefs and their tidepools are important habitats, especially for young-of-the-year of both coastal and oceanic fish species, many of which of special economic interest (Caddy & Sharp 1986, Griffiths et al. 2003). Coastal reefs in northeast Brazil often become uncovered at low tides, forming several tidepools, some of them entirely or partly isolated from the adjacent sea. Wind, solar radiation, and rainfall are factors that affect the water temperature, salinity, and dissolved oxygen, turning the tidepools into highly variable habitats both in time and space (Vasconcelos & Rocha 1986, Bridges 1994, Little & Kitching 1996).
Tidepools may serve as nursery habitats for larvae and juveniles of several marine organisms coming from the adjacent areas (Beckley 1985, Bennett 1987, Horn et al. 1999), also providing refuges for many other organisms during the low tides (Mahon & Mahon 1994). Fishes inhabiting tidepools can be categorized as: (1) true residents, represented by small-sized species, including benthic fishes of the families Gobiidae and Blenniidae (Horn et al. 1999); (2) partial residents, which are mainly juveniles from several families such as Scaridae, Pomacentridae, Acanthuridae, Pomacanthidae, and Chaetodontidae, also including some of economic interest as Lutjanidae, Serranidae, and Haemulidae (Gibson 1982, Mahon & Mahon 1994); and (3) transient or occasional species, which may be sporadically retained in tidepools at the low tides (e.g. Carangidae, Scianidae, Scombridae) (Mahon & Mahon 1994).
Most of the information on the composition, diversity, distribution, and abundance of tidepool fishes originated from studies conducted in North America (Thompson & Jones 1978, Yoshiyama 1981, Moring 1986, Yoshiyama et al. 1986), Caribbean (Mahon & Mahon 1994), Taiwan (Chang et al. 1977, Lee 1980), and South Africa (Christensen & Winterbottom 1981, Bennett & Griffiths 1984, Beckley 1985, Bennett 1987). In South America, Quijada & Cáceres (2000) studied the tidepool ichthyofauna in the temperate zone of Chile. In the northeastern tropical areas of Brazil Rosa et al. (1997, at the state of Paraíba) and Araújo et al. (2000, at the state of Ceará) studied species composition and biodiversity, but they did not investigate temporal and spatial patterns of the fish assemblages.
Much concern has been raised about the health of coastal ecosystems in Ceará, due to intense degradation of the coastal reefs by tourism, fisheries and coastal development (Campos et al. 2003). Nevertheless, the baseline studies to determine benchmarks for monitoring the health coastal reefs and tidepools are lacking. Following Meador & Matthews (1992), without the baseline spatial and temporal data on the inherent variability in sampling of aquatic biological communities, the detection of the effects of human-induced or natural perturbation becomes difficult. The present study aimed to investigate changes in tidepool fish assemblages structure on coastal reefs of Iparana, state of Ceará, NE Brazil, by (1) describing their taxonomic composition; (2) quantitatively assessing the patterns of variation across a range of spatial and temporal scales; (3) identifying which environmental variables may be important in influencing their structure; and (4) evaluating the importance of the tidepools as nursery areas for marine fishes.
Material and Methods
The tidepools here studied are located at Iparana beach (3° 40' 18" S and 38° 36' 42" W), on the coast of Ceará (Figure 1). This area is located at the Brazilian northeastern coast, Western South Atlantic. Local Calcium Carbonate and Iron Oxide shore reefs are often elongated and discontinuous, extending from the beach face to nearly two hundred meters seaward (Morais 1969, Maida & Ferreira 1997, Leão & Dominguez 2000). These structures become partly exposed during low tides, isolating tidepools of several sizes and depths within the reef extension. The five tidepools monitored in this study were located and randomly distributed in the lower intertidal zone. Tidepool selection was based on three assumptions: a) to be exposed only at very low tides; b) to have the appropriate size and depth for conducting visual census; c) to become nearly isolated from neighboring tidepools.
Diurnal and monthly visual censuses were conducted at syzygy low tides lower than or equal to 0.1 m. Censuses consisted of an intensive species search, by snorkelling around the internal rim of the tidepools, using the methods suggested by Christensen & Winterbottom (1981) in respect to observation time. Each search lasted less than 25 minutes to avoid repetitive counts. A total of 55 samplings and approximately 23 hours of visual censuses were conducted between June 1998 and May 1999. Water visibility was always greater than the largest pool length.
Tidepools bottom cover was measured by using a square frame (0.5 x 0.5 m) placed over the substrate, and percent cover was categorized into 1) sand-rubble - SR; 2) bare rock - BR; and 3) rock covered with algae - RA. The surface area (A) of each tidepool was estimated by measuring and adding up triangular areas (m2) to match the approximate surface area of the tidepool. Average depth (D) in meters was estimated from three measurements taken at random locations along a longitudinal transect in order to estimate the pool volume (PV = A x D).
The salinity (SA), temperature (TE,°C), pH, and dissolved oxygen (DO, mg.L-1) of the seawater were recorded by using a multiparameter probe (Hydrolab). Data of rainfall (RF, mm yr-1) and wind velocity (WV, ms-1) during the studied period were obtained from the Fundação Cearense de Meteorologia (FUNCEME).
A full factorial two-way ANOVA was utilized for evaluating the tidepools (1 to 5) along the four trimesters (I June to August, II September to November, III December to February, and IV March to May), and also to analyse their interactions with the environmental variables, total species abundance, and species richness. Student-Newman-Keuls, a posteriori test, was applied for multiple comparisons. Variables were transformed as required by applying a logarithmic transformation of the type Y = log10 (x + 1) (Boesch 1977, Zar 1996).
Cluster analysis was used for organizing species (R-mode), trimesters or tidepools (Q-Mode), into nearly homogeneous groups based on a species vs. samples abundance matrix. Cluster analyses were performed by using the Euclidean Distance and Group Average (UPGMA) linkage methods (Boesch 1977, Valentin 1995). To minimize the effects of artificial groups and facilitate cluster interpretation, normal and inverse classifications were related to each other using nodal analysis, which determines species-collections coincidences. This was done using the concepts of Constancy and Fidelity as defined by Boesch (1977). Analyses were performed in SPSS for Windows (v.11) and Microsoft Excel 2000 Softwares.
Stepwise multiple linear regression was used to analyze the effects of the environmental variables recorded (i.e. tidepool volume, substrate type, salinity, temperature, pH, dissolved oxygen of the sea water, rainfall, and wind velocity) on total species abundance and richness (Draper & Smith 1981). When species determination was uncertain in the field, specimens were captured for laboratory identification. These specimens were not included in the statistical analysis. The collected material was identified, photographed, preserved and deposited at the Aquatic Biology Collection of the Department of Fisheries Engineering at the Federal University of Ceará.
Results
The 16 fish families recorded were represented by at least 26 species. Haemulidae was the richest family represented by six species. The most abundant families were Scaridae, Haemulidae, Gerreidae, and Pomacentridae, in a decreasing order. Of the total 4,750 fishes counted, 10 taxa amounted to about 98% of the total abundance, and the six most abundant species (Sparisoma cf. axillare, Haemulon parra, Eucinostomus spp., Abudefduf saxatilis, Lutjanus apodus, and Acanthurus chirurgus) represented about 94% of this total. With respect to the frequency of occurrence, individuals of Sparisoma cf. axillare were present in 96% of the censuses, followed by Eucinostomus spp. (95%); Abudefduf saxatilis and Lutjanus apodus (both in 87%), and Haemulon parra (85%) (Table 1).
Two-way ANOVA did not show any significant results between the tidepools and the trimesters (F12.0 = 1.28, p > 0.05 and F12.0 = 0.78, p > 0.05), considering the total species abundance and richness.
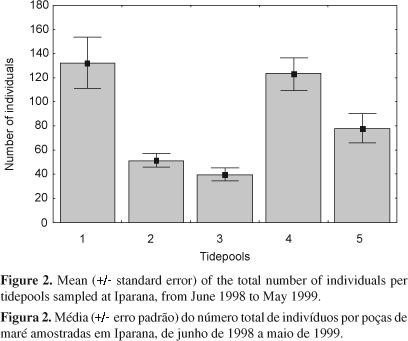
The isolated effect of tidepools was significant (F4.0 = 23.02, p < 0.01) on species abundance but not for species richness (F4.0 = 2.15, p = 0.88). Average total abundance was significantly higher on tidepools 1 and 4 in relation to tidepools 2, 3, and 5 (Newman-Keuls, p < 0.01, 1 = 4 > 2 = 3 = 5) (Figure 2).
Both species abundance (F3.0 = 16.15, p < 0.01) and richness (F3.0= 8.14, p < 0.01) varied significantly in relation to the sampling trimester. Total abundance was low on trimester I, high on III and IV, and intermediate on trimester II, showing a positive trend from the beginning to the end of the study period (Newman-Keuls, p < 0.05, I < II < III = IV). Average number of species was low on trimesters I and II and high on III and IV (Newman-Keuls, p < 0.05, I = II < III = IV) (Figure 3).
Multiple regression analysis significantly explained about 78% of the variation in abundance and 57% of the species richness as a function of the environmental variables considered. Species abundance was positively correlated with the tidepools volume and the presence of rock covered with algae, and negatively correlated with the wind velocity, bare rock and salinity. Species richness was also directly related to tidepools volume, but it was negatively related to the salinity (Table 2).
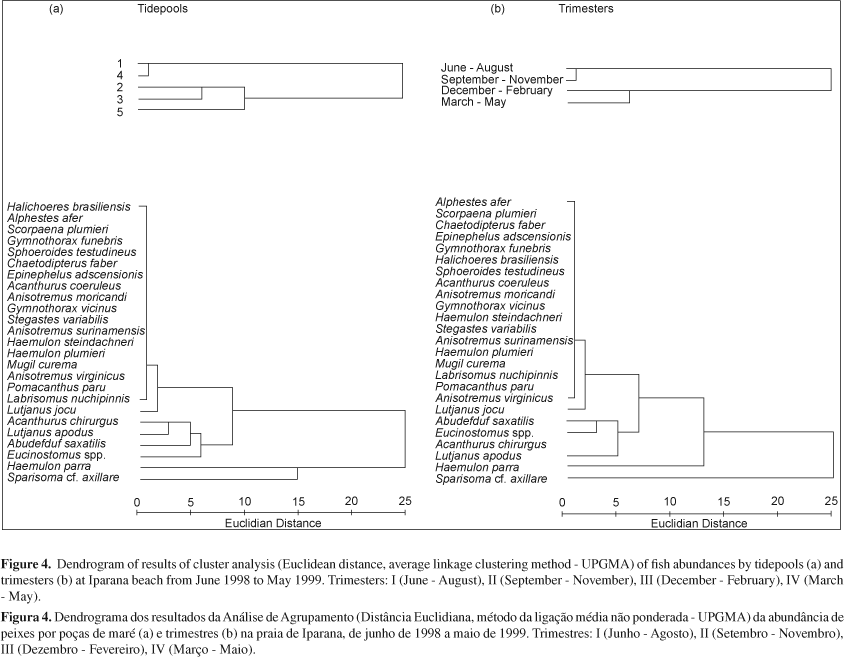
Q-mode cluster analysis resulted in groups which represented spatial and temporal discontinuities (Figure 4a). Tidepools 1 and 4 grouped together and represented the type 1 environment characterized by large volume (mean 187.4 m3) when compared with type 2 environment (average 49.3 m3 in volume; F(1) = 428.03, p < 0.01). The type 1 environment is also marked by a predominance of rock covered with algae (F(1) = 110.82, p < 0.01).
Trimesters I and II clustered together and formed a winter-spring group (semester I), whereas the trimester III and IV formed a summer-fall group (semester II). Semester I was characterized by low rainfall (mean 37.33 mm), high salinity (mean 36.7) and wind velocity (mean 4.8 m.s-1), whereas semester II showed a higher precipitation (mean 198.1 mm), lower salinity (mean 35.2) and lower wind velocity (mean 3.5 m.s-1). All of these differences were significant.
R-mode cluster analysis formed three to four species groups, in which Sparisoma axillare and Haemulon parra were grouped together or considered as single species groups, depending on whether a spatial or temporal analysis was performed. The second group included Eucinostomus spp., Abudefduf saxatilis, Lutjanus apodus, and Acanthurus chirurgus; whereas the third group was formed by less abundant and frequent species (e.g. Lutjanus jocu, Labrisomus nuchipinnis and Pomacanthus paru). All 19 remaining species were clustered in this group. Either the groups two and three were consistent regardless if a spatial or temporal analysis was performed (Figure 4b).
Species and sample groups were reorganized into nodal contingency tables, aiming to maximize constancy and fidelity within each cell (nodes). Figure 5 shows the formation of four sampling dimensions (semesters 1 and 2 within environments 1 and 2) and seven species groups with the following characteristics:
Group I represented by the dominant and most abundant species, showed a very high constancy and low fidelity to all environmental dimensions analysed, and was composed by Sparisom cf. axillare, Haemulon parra, Lutjanus apodus, Eucinostomus spp., and Abudefduf saxatilis;
Group II had a moderate to very high constancy, especially in the Semester II, but low fidelity to all sampling dimensions, and included frequent species such as Acanthurus chirurgus, Lutjanus jocu, Anisotremus virginicus, Pomacanthus paru, and Labrisomus nuchipinnis;
Group III - this group presented low to moderate constancy and fidelity, particularly associated to Semester II and environment 1, and included Mugil curema, Haemulon plumieri, Stegastes variabilis, and Gymnothorax vicinus;
Groups IV VII these groups included occasional species, showed moderate to low constancy, but high to very high fidelity. The groups IV (Gymnothorax funebris, Epinephelus adscensionis) and V (Anisotremus moricandi, Acanthurus coeruleus, Sphoeroides testudineus, Chaetodipterus faber) were strongly associated with the environment 1, whereas the groups VI (Anisotremus surinamensis) and VII (Alphestes afer, Scorpaena plumieri, Halichoeres brasiliensis, Haemulon steindachneri) were strongly associated with the environment 2.
Discussion
The majority of the fishes in tidepools at Iparana were suprabenthic juvenile individuals of great mobility. According to Gibson & Yoshiyama (1999), the structure of tidepool fish communities depends mostly on the size of the sampled area, the height of the pool within the intertidal zone, and the sample size. Mahon & Mahon (1994) stated that large tidepools close to the lowest intertidal zone, such the ones sampled in this study contained more species, and a higher proportion of transient species than tidepools located at the highest intertidal zone. These findings may explain why the tidepools at Iparana showed a greater proportion of transient and occasional species. Another consideration was the fact that visual census sub-samples cryptic species such gobies and blennies, often regarded as true residents in tidepools (Christensen & Winterbottom 1981, Brock 1982, Ackerman & Bellwood 2000, Ackerman & Bellwood 2002). Methods that adequately sample cryptic fishes often include ichthyocides, anaesthetics or empty the tidepools. Whereas these methods may yield accurate estimates of species richness, they may not be adequate to sample less isolated lower intertidal pools. Furthermore, such methods may be incompatible with conservation objectives and assessment of highly impacted areas (F.E.A. Cunha, unpublished data).
The fish dwellers at Iparana tidepools were mainly species recruited from surrounding subtidal areas. Gibson & Yoshiyama (1999) considered that tidepools may contain large number of juveniles representing predominant subtidal species, suggesting that, within the intertidal zone, tide pools may act as nursery habitats for such species. Their suggestion is based on the assumption that tidepools are spatial and temporal refuges from large-sized and numerous predators of the subtidal zone. Mahon & Mahon (1994) also observed that intertidal pools at Barbados (Caribbean) have many partial residents which are juveniles of reef species, and concluded that the intertidal habitats are natural juvenile stocks for many reef fish populations. A higher number of juvenile fishes from December to May (both in number of individuals and species) further support the hypothesis that these tidepools act as nursery sites.
Tidepools on rocky reefs at Iparana may also serve as refuges for opportunistic estuarine species. Eucinostomus spp., Mugil curema, and the species of Engraulidae were frequent on censuses, indicating that the studied area, which is located near the mouth of the Ceará river estuary, within large stretches of sandy beaches, uses to be visited by these species, probably seeking shelter and/or food during migrating periods to and from estuarine nursery grounds. The temporal structure of species communities in these areas is often the result of seasonal settlement or consecutive waves of migration by the juvenile stages. However, it is generally accepted that the movement of the juvenile fishes into the intertidal zone is related with feeding (Amara & Paul 2003). Another hypothesis focuses on the predation avoidance provided by the shallow intertidal waters (Gibson 1994).
Reproductive patterns in tropical latitudes are often linked with seasonal changes on wind strength and velocity, and also with the rainfall (Lowe-McConnell 1987). Similar correlations were found for fishes (Costa et al. 1995, Cunha et al. 2000) and lobsters (Fonteles-Filho 1986) on the coast of Ceará, in which the greatest abundance was observed during the rainy season. In fact, Alves & Lima (1978) observed that most of the fishes found off the coast of Ceará showed rest reproductive cycles during the dry windy season.
Multiple linear regression analysis was partly consistent with the environmental discontinuities indicated by Q-Mode cluster analysis. The positive correlation of both species abundance and richness and the presence of rock-algae substrate in environment 1 suggested that substrate structural complexity was probably one of the main factors defining spatial community structure. Bennett & Griffiths (1984) and Gibson & Yoshiyama (1999) reported that the area, volume, and rugosity of the tidepools are factors of great importance affecting the fish assemblage structure. Furthermore, the presence of holes and crevices of varied sizes in the rocky walls, and multiple sized boulders at the bottom further enhanced habitat complexity and, therefore, the abundance and diversity of the fish assemblages (Bennett 1987, Mahon & Mahon 1994, Núñez Lara & Arias González 1998). Ferreira et al. (2001) studying the rocky coastline in southern Brazil also observed that not only biotic factors (like abundance of sessile benthic invertebrates), but also the physical structure (like the surface area), explains the highest diversity and abundance of fishes.
The fact that the tidepools volume was a significant factor in this study corroborates the literature, and support the hypothesis that tidepools size also plays an important role at Iparana, mostly by controlling the habitat variability. For instance, Rosa & Moura (1997) suggested that large volume tidepools are less affected by temperature and salinity variations, which can reach critical levels in such micro-ecosystems. The positive correlation of species richness and tidepools volume, and its negative correlation with salinity, may be an important evidence of the buffering effect of large tidepools vs. the small ones at Iparana.
The species associations found in this study and their relation to seasonal and spatial discontinuities may be partly explained according to their habitat requirements and reproductive cycles. Species belonging to group I in our classification were considered by Alves & Lima (1978) as ubiquitous, abundant and nearly year round spawners in the coast of Ceará, whereas those of groups II and III often show reproduction peaks matching the semester II of our classification. Species groups II and III also showed a greater affinity with larger and more complex tidepools. Groups I and III give a substantial contribution in number of species and individuals to the fish community in the tidepools at Iparana, being considered as true and partial residents, according to the classification of Mahon & Mahon (1994). The species Labrisomus nuchippinis and Gymnothorax vicinus are true cryptic residents. The other species of Groups I and III are partial residents in the tidepools, occurring mainly as juveniles. Species belonging to Groups from IV to VII were classified as transient or occasional species.
In summary, we conclude that the tidepool fish fauna at Iparana presents complex seasonal and spatial relationships, dependent upon cyclic changes on physical environment and reproduction of its fish species. Connectivity and interdependence between the tidepools and the adjacent ecosystems such as the subtidal reefs, sandy shores and estuaries was also apparent in our study. However, despite our findings that some environmental and habitat variables appeared to influence the structure of the tidepool fish assemblages, it is necessary to investigate other mechanisms related within community structure and population size such as recruitment, predation, and competition patterns, assessing theirs effects on such fish assemblage structures.
Acknowledgments
We thank the members of the Tropical Marine Ichthyology Group, Universidade Federal do Ceará - UFC for their help in the course of this study. This research was made possible through grants and fellowships provided by Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq (Research Fellow), Fundação Cearense de Apoio ao Desenvolvimento Científico e Tecnológico FUNCAP (MSc fellowship) and the Graduate program on Fisheries Engineering UFC.
Recebido em 17/01/06
Versão reformulada recebida em 17/10/06
Publicado em 31/03/07
ISSN 1676-0603
- ACKERMAN, J.L. & BELLWOOD, D.R. 2000. Reef fish assemblages: a re-evaluation using enclosed rotenone stations. Mar. Ecol. Prog. Ser. 206:227-237.
- ACKERMAN, J.L. & BELLWOOD, D.R. 2002. Comparative efficiency of clove oil and rotenone for sampling tropical reef fish assemblages. J. Fish Biol. 60:893-901.
- ALVES, M.I.M. & LIMA, H.H. 1978. Sobre a época de desova de alguns peixes marinhos do estado do Ceará, Brasil. Bol. Ciên. Mar 30:1-7.
- AMARA, R. & PAUL, C. 2003. Seasonal patterns in the fish and epibenthic crustaceans community of an intertidal zone with particular reference to the population dynamics of plaice and Brown shrimp. Estuar. Coast Shelf S. 56:807-818.
- ARAÚJO, M.E., CUNHA, F.E.A., CARVALHO, R.A.A., FREITAS, J.E.P., NOTTINGHAM, M.C. & BARROS, B.M.N. 2000. Ictiofauna Marinha do estado do Ceará, Brasil: II. Elasmobranchii e Actinopterygii de arrecifes de arenito da região entremarés. Arq. Ciên. Mar 33:133-138.
- BECKLEY, L.E. 1985. The fish community of East Cape tidal pools and an assessment of the nursery function of this habitat. S. Af. J. Zool. 20:21-27.
- BENNETT, B.A. 1987. The rock-pool fish community of Koople Alleen and an assessment of the importance of Cape rock-pools as nurseries for juvenile fish. S. Af. J. Zool. 22:25-32.
- BENNETT, B.A. & GRIFFITHS, C.L. 1984. Factors affecting the distribution, abundance and diversity of rock-pool fishes on the Cape Peninsula, South Africa. S. Af. J. Zool. 19:97-104.
- BOESCH, D.F. 1977. Application of numerical classification in ecological investigations of water pollution. Corvallis Environmental Research Laboratory, Oregon.
- BRIDGES, C.R. 1994. Ecophysiology of intertidal fish. In Fish ecophysiology (J.C. Rankin & F.B. Jensen eds.). Chapman & Hall, London, p.375-399.
- BROCK, R.E. 1982. A critique of the visual census method for assessing coral reef fish populations. B. Mar. Sci. 32:269-276.
- CADDY, J.F. & SHARP, G.D. 1986. An ecological framework for marine fishery investigations. FAO Fisheries Technical, Rome, Paper 283.
- CAMPOS, A.A., MONTEIRO, A.Q. & MONTEIRO-NETO, C. 2003. A Zona Costeira do Ceará: Diagnóstico para a Gestão Integrada. AQUASIS, Fortaleza-CE.
- CHANG, K.H., LEE, S.C. & WU, W.L. 1977. Fishes of reef limestone platform at Maopitou, Taiwan: diversity an abundance. B. I. Zool. Acad. Sinica 16:9-21.
- CHRISTENSEN, M.S. & WINTERBOTTOM, R.A. 1981. Correction factor for, and its application, visual censuses of littoral fish. S. Af. J. Zool. 16:73-79.
- COSTA, P.S.R., SANTOS, M.A.M., ESPÍNOLA, M.F.A. & MONTEIRO-NETO, C. 1995. Biologia e biometria do coró, Pomadasys corvinaeformis (Steindachner) (Teleostei: Pomadasidae), em Fortaleza, Ceará, Brasil. Arq. Ciên. Mar 29:20-27.
- CUNHA, F.E.A., FREITAS, J.E.P., FEITOSA, C.V. & MONTEIRO-NETO, C. 2000. Biologia e biometria da palombeta Chloroscombrus chrysurus (Linnaeus, 1766) (Teleostei: Carangidae) em Fortaleza, Ceará, Brasil. Arq. Ciên. Mar 33:143-148.
- DRAPER, N.R. & SMITH, H. 1981. Applied Regression Analysis. John Wiley & Sons, New York.
- FERREIRA, C.E.L., GONÇALVES, J.E.A. & COUTINHO, R. 2001. Community structure of fishes and habitat complexity on a tropical rocky shore. Environ. Biol. Fish. 61:353-369.
- FONTELES-FILHO, A.A. 1986. Influência do recrutamento e da pluviosidade sobre a abundância das lagostas Panulirus argus (Latreille) e Panulirus laevicauda (Latreille) (Crustacea: Palinuridae), no nordeste do Brasil. Arq. Ciên. Mar 25:13-31.
- GIBSON, R.N. 1982. Recent studies on the biology of intertidal fishes. Oceanogr. Mar. Biol. Annu. Rev. 20:363-414.
- GIBSON, R.N. 1994. Impact of habitat quality and quantity on the recruitment of juvenile flatfishes. Neth. J. Sea Res. 32:191-206.
- GIBSON, R.N. & YOSHIYAMA, R.M. 1999. Intertidal fish communities. In Intertidal fishes: life in two worlds (M. H. Horn, K. L. Martin & M. A. Chotkowski eds.). Academic Press, London, p.264-296.
- GRIFFITHS, S.P., WEST, R.J. & DAVIS, A.R. 2003. Effects of intertidal elevation on the rockpool ichthyofaunas of temperate Australia. Environ. Biol. Fish. 68:197-204.
- HORN, M.H., MARTIN, K.L.M. & CHOTKOWSKI, M.A. 1999. Intertidal fishes: life in two worlds. Academic Press, London.
- LEÃO, M.A.N. & DOMINGUEZ, J.M.L. 2000. Tropical Coast Brazil. Mar. Pollut. Bull. 41:112-122.
- LEE, S.C. 1980. Intertidal fishes of the rocky pools at Lanyu (Botel Tobago), Taiwan. B. I. Zool. Acad. Sinica. 19:1-13.
- LITTLE, C. & KITCHING, J.A. 1996. The biology of rocky shores. Oxford University Press, Oxford.
- LOWE-McCONNELL, R.H. 1987. Ecological studies in tropical fish communities. Cambridge University Press, Cambridge.
- MAHON, R. & MAHON, S.D. 1994. Structure and resilience of a tidepool fish assemblages at Barbados. Environ. Biol. Fish. 41:171-190.
- MAIDA, M. & FERREIRA, B.P. 1997. Coral reefs of Brazil: an overview. Proc. 8th Int. Coral Reef Symp. 1:263-274.
- MEADOR, M.R. & MATTHEWS, W.J. 1992. Spatial and temporal patterns in fish assemblage structure of an intermittent Texas stream. Am. Mid. Nat. 127:106-114.
- MORAIS, J.O. 1969. Contribuição ao estudo do "Beach-Rocks" do Nordeste do Brasil. Trab. Oceanogr. UFPE 9(11):79-94.
- MORING, J.R. 1986. Seasonal presence of tidalpool fish species in a rocky intertidal zone of northern California, USA. Hydrobiol. 134:21-27.
- NÚÑEZ LARA, E. & ARIAS GONZÁLEZ, E. 1998. The relationship between reef fish community structure and environmental variables in the southern Mexican Caribbean. J. Fish Biol. 53(Suppl. A):209-221.
- QUIJADA, P.A. & CÁCERES, C.W. 2000. Patrones de abundancia, composición trófica Y distribución espacial del ensamble de peces intermareales de la zona centro-sur de Chile. Rev. Chil. Hist. Nat. 73:8-18.
- ROSA, R.S. & MOURA, R L. 1997. Visual assessment of reef fish community structure in the Atol das Rocas Biological Reserve, off Northeastern Brazil. Proc. 8th Int. Coral Reef Symp. 1:983-986.
- ROSA, R.S., ROSA, I.L. & ROCHA, L.A. 1997. Diversidade da ictiofauna de poças de maré da Praia do Cabo Branco, João Pessoa, Paraíba, Brasil. Revta bras. Zool. 14:201-212.
- THOMPSON, M.J. & JONES, R.S. 1978. Comparison of Florida reef fish assemblages using a rapid visual technique. B. Mar. Sci. 28:159-172.
- VASCONCELOS, F.P. & ROCHA, C.A.S. 1986. Análise da influência das marés sobre os parâmetros físico-químicos da água de microambientes em rochas-de-praia. Arq. Ciên. Mar 25:51-61.
- VALENTIN, J.L. 1995. Agrupamento e ordenação. Tópicos em tratamento de dados biológicos. Instituto de Biologia (UFRJ), Rio de Janeiro.
- YOSHIYAMA, R.M. 1981. Distribution and abundance patterns of rocky intertidal fishes in central California. Environ. Biol. Fish. 6:315-332.
- YOSHIYAMA, R.M., SASSAMAN, C. & LEA, R.N. 1986. Rocky intertidal fish communities of California: temporal and spatial variation. Environ. Biol. Fish. 17:23-40.
- ZAR, J. 1996. Biostatistical analysis. Prentice-Hall Inc, New Jersey.
Publication Dates
-
Publication in this collection
21 Aug 2007 -
Date of issue
2007
History
-
Received
17 Jan 2006 -
Accepted
31 Mar 2007