Abstracts
We redescribe Iracema caiana, a monotypic genus of the gymnotiform electric fish family Rhamphichthyidae. Iracema is known only from the type series (holotype and three paratypes) collected from the rio Jauaperi, affluent to the rio Negro in the Amazon basin, and was never collected again. Previous morphological studies were limited to features of external morphology. To study the osteology of Iracema we examined two specimens of different sizes using high-resolution x-ray computed tomography, a non-invasive and non-destructive technique to visualize internal anatomical structures. We describe and illustrate the osteology of Iracema caiana, and present data on morphometrics and external morphology. Contrary to previous hypotheses we propose that Iracema is the sister group to Rhamphichthys based on four synapomorphies: intermuscular bones present in the adductor mandibulae, reticulated texture of opercles, fully ossified Baudelot's ligaments, and elongate scales above the lateral line in the posterior portion of the body.
Amazon; Biodiversity; Guiana Shield; Osteology; Rare fish; Rhamphichthys
Iracema caiana, um genêro monotípico de peixes elétricos Gymnotiformes da família Rhamphichthyidae, é aqui redescrito. Iracema é conhecido apenas da série-tipo (holótipo e três parátipos) coletado no rio Jauaperi, afluente do rio Negro na bacia amazônica, nunca sendo coletado novamente. Estudos morfológicos prévios de Iracema foram limitados somente à morfologia externa. Neste trabalho, a osteologia do gênero foi examinada com base em dois espécimes de diferentes tamanhos usando-se tomografia computadorizada de alta resolução, uma técnica não invasiva para a visualização de estruturas anatômicas internas. Caracteres externos e morfométricos são apresentados e a osteologia é ilustrada e descrita. Contrariamente a hipóteses prévias, Iracema é proposto como grupo irmão de Rhamphichthys com base em quatro sinapomorfias: presença de ossos intermusculares no adductor mandibulae, textura reticulada do opérculo, ligamento de Baudelot completamente ossificado e escamas alongadas na região posterior do corpo acima da linha lateral.
University of Louisiana at Lafayette, Department of Biology, P.O. Box 42451, Lafayette, LA 70504, USA. tiagobio2002@yahoo.com.br (TPC), jalbert@louisiana.edu (JSA)
ABSTRACT
We redescribe Iracema caiana, a monotypic genus of the gymnotiform electric fish family Rhamphichthyidae. Iracema is known only from the type series (holotype and three paratypes) collected from the rio Jauaperi, affluent to the rio Negro in the Amazon basin, and was never collected again. Previous morphological studies were limited to features of external morphology. To study the osteology of Iracema we examined two specimens of different sizes using high-resolution x-ray computed tomography, a non-invasive and non-destructive technique to visualize internal anatomical structures. We describe and illustrate the osteology of Iracema caiana, and present data on morphometrics and external morphology. Contrary to previous hypotheses we propose that Iracema is the sister group to Rhamphichthys based on four synapomorphies: intermuscular bones present in the adductor mandibulae, reticulated texture of opercles, fully ossified Baudelot's ligaments, and elongate scales above the lateral line in the posterior portion of the body.
Key words: Amazon, Biodiversity, Guiana Shield, Osteology, Rare fish, Rhamphichthys.
RESUMO
Iracema caiana, um genêro monotípico de peixes elétricos Gymnotiformes da família Rhamphichthyidae, é aqui redescrito. Iracema é conhecido apenas da série-tipo (holótipo e três parátipos) coletado no rio Jauaperi, afluente do rio Negro na bacia amazônica, nunca sendo coletado novamente. Estudos morfológicos prévios de Iracema foram limitados somente à morfologia externa. Neste trabalho, a osteologia do gênero foi examinada com base em dois espécimes de diferentes tamanhos usando-se tomografia computadorizada de alta resolução, uma técnica não invasiva para a visualização de estruturas anatômicas internas. Caracteres externos e morfométricos são apresentados e a osteologia é ilustrada e descrita. Contrariamente a hipóteses prévias, Iracema é proposto como grupo irmão de Rhamphichthys com base em quatro sinapomorfias: presença de ossos intermusculares no adductor mandibulae, textura reticulada do opérculo, ligamento de Baudelot completamente ossificado e escamas alongadas na região posterior do corpo acima da linha lateral.
Introduction
Iracema caiana Triques is a member of the Rhamphichthyidae, a clade of Gymnotiformes with 15 valid species distributed in three genera (Ferraris, 2003; Lundberg, 2005; Carvalho et al., 2011). Rhamphichthyidae and Hypopomidae constitute the Rhamphichthyoidea, a well supported clade within Gymnotiformes (Mago-Leccia, 1978; Alves-Gomes et al., 1995; Albert, 2001). Iracema Triques was originally based on a unique combination of characters of external morphology. According to Triques (1996a) Iracema can be differentiated from other rhamphichthyids by the presence of one series of irregular to roundish blotches on the sides of the body; a broad and uniformly dark pigmented stripe along the dorsal margin of the body; and an intermediate number of anal-fin rays between Rhamphichthys Muller and Troschel, and Gymnorhamphichthys Ellis. Triques (1996a) briefly discussed putative relationships of Iracema within other rhamphichthyids, hypothesizing it as the sister group to Gymnorhamphichthys based on the shared loss of scales on the anterior portion of the body.
Iracema was first included in a formal phylogenetic analysis by Albert & Campos-da-Paz (1998), who were able to code only 46 characters-states in a data matrix of 170 characters due to absence of osteological information. Most osteological data on Iracema were not available due to a lack of specimens available for clearing and staining; the genus remains known only from four specimens in the type series. To avoid dissection and staining procedures with a limited type series we used an alternative method of visualizing skeletal morphology, high-resolution x-ray computed tomography (HRXCT). HRXCT is a non-destructive technique that allows reconstruction of a virtual skeleton in digital form. Similar approaches to recover osteological data from rare or extinct species have been previously adopted by ichthyologists (Schaefer, 2003; Schaefer & Fernández, 2009; Fink & Humphries, 2010).
Material and Methods
Two specimens were scanned at the High-Resolution X-ray Computed Tomography Facility, The University of Texas, Austin. The head and cleithral region of a preserved paratype of Iracema caiana (MZUSP 49205, 345 mm SL) was scanned using the following parameters. FeinFocus microfocal X-ray source operating at 200 kV and 0.16 mA with no X-ray prefilter was employed. An empty container wedge was used. Slice thickness corresponded to two lines in a CCD image intensifier imaging system and equaled 0.0641 mm, with a source-to object distance of 92 mm. For each slice, 1,400 views were taken with three samples per view. The field of image reconstruction was 28 mm, and an image reconstruction offset of 4600 was used with a reconstruction scale of 7000. The dataset consists of 761 1024x1024-pixel HRXCT slices. A second specimen (MZUSP 49205, 235 mm SL) was scanned using the following parameters. An Xradia microXCT scanner operating at 40 kV and 7 W with a 0.15 mm glass X-ray prefilter was employed. An air wedge was used. Source-object distance was 125.3 mm and detector-object distance was 37.1 mm. A total of 521 views were gathered over a rotation of ±104 degrees. The acquisition time per view was 4 seconds. The dataset consists of 909 1024x1024-pixel HRXCT slices, with cubic voxels measuring 43.84 microns. The scan was taken along the long axis of the specimen from the tip of the snout to about the 8-9 vertebrae. Visualizations were produced in the commercial software package VGStudio Max®, at the Ichthyology department of the Academy of Natural Sciences of Philadelphia. Although the renderings appear similar to photographs, they represent the density differences of the biological materials as reflected in their X-ray opacity. Figures were based on still frames captured from digital animations of HRXCT and were prepared using Adobe Photoshop®CS. Homologous bone depicted in figures 3A and 3B were represented by the same spectral colors (hues) in VG Studio Max. These colors differ only in terms of tint (shading density). The shadowing option in VG Studio, used to help visualize objects in 3D produced different effects in these images due to shape differences. Some bones of Iracema caiana are very thin and overlap each other, and therefore it was impossible in the reconstructions to separate them into different color schemes (e.g. preopercle and interopercle). Differing from other fish with published Computed Tomography (CT) scan reconstructions, Iracema has thin and weakly ossified bones (e.g. those from the branchial basket), which are not clearly identified in the CT scans. In addition to the images reconstructed using the software we use the raw X-ray slices data to describe the morphology (e.g. contact between retroarticular and quadrate), however we did not include illustrations, because they have little value compared to the 3d images. The branchial basket was illustrated in the midsagittal plane instead of the usual coronal plane because there is a better resolution and less overlap of bones in these views.
Measurements were made to the nearest 0.1 mm with digital calipers. Standard length (SL), head length (HL) and snout length (SN) follow Nijssen et al. (1976: fig. 1). Standard length as used here is the same measurement as length to end of anal fin (LEA; Mago-Leccia, 1994). Caudal-appendage length (CL) and pectoral-fin length (PL) follow Schwassmann (1989: fig. 1). Anal-fin length (AL) is the distance from the anterior origin of the anal fin to the posterior end. Body depth (BD) is the greatest depth of the body, which is usually at about half the length of the standard length posterior to end of precaudal vertebrae. Caudal-filament depth (CD) is the depth of caudal appendage at the vertical of the last anal fin ray. Interorbital distance (IO) is the distance between the bony orbits, in the middle part of the orbit. Postorbital length (PO) is measured from the posterior limit of the eyeball to the posterior edge of bony opercle. Distance to posterior nares (PN) is the distance from the tip of snout to the anterior margin of the posterior nares (= measurement Prenasal 2 of Triques, 1996b; fig. 1). Opercular opening (BO) is the distance between the dorsal and the ventral corners of the branchial aperture (measurement 11 of Triques, 1996b; fig. 2). Morphometric data are expressed as percentages of standard length, except subunits of the head, which are expressed as percentages of head length. Osteological terminology is that of Fink & Fink (1981) and Fink & Fink (1996). Cleared and stained specimens (cs) were prepared according to the method of Taylor & Van Dyke (1985). Myological nomenclature follows Winterbottom (1974) and Datovo & Bockmann (2010). Electrocyte rows at the end of anal fin were observed under the transmitted light source in the stereomicroscope. One scale, at about the second third of body (above the lateral line) was removed from MZUSP 49205 (345 mm SL) and stained in alizarin red. Photographs of fish scales were taken using a digital camera attached to an Olympus SZX 12 microscope. Drawings were made using a camera lucida attached to an Olympus SZX 12 microscope. The number of vertebrae to the end of anal fin includes the four of the Weberian apparatus and end with the last vertebra with its hemal spine associated with the last anal-fin pterygiophore (Lundberg, 2005). Institutional abbreviations are as listed at http://research.calacademy.org/research/ichthyology/catalog/collections.asp.
Results
Iracema Triques, 1996
Type species.Iracema caiana Triques, 1996a: 91 by original designation and monotypy.
Diagnosis.Iracema is distinguished from other rhamphichthyids by the following characteristics: 240-257 anal-fin rays (vs. 139-211 in Gymnorhamphichthys and 304-470 in Rhamphichthys); 15 or 16 pectoral-fin rays (vs. 10-13 in Gymnorhamphichthys and 17-22 in Rhamphichthys); a series of dark, rounded pigment blotches on the side of the body along the lateral line (vs. no pigmentation in Gymnorhamphichthys and dark oblique transverse bands or no pigmentation in Rhamphichthys); caudal filament ovoid in cross section, its depth slightly greater than its width (vs. caudal filament laterally compressed in cross section, its depth about three times its width in Gymnorhamphichthys and Rhamphichthys).
Iracema caiana Triques, 1996
Iracema caiana Triques, 1996a: 91 [original description]. -Albert & Campos-da-Paz, 1998: 423, 425 [phylogeny and diagnosis]. -Albert, 2001: 63, 68 [phylogeny and diagnosis]. -Ferraris, 2003: 495 [listed]. -Albert & Crampton, 2005: 363 [listed]. -Crampton & Albert, 2006: 650 [listed and habitat description]. -Crampton 2011: 175 [listed]. -Winemiller & Willis, 2011: 231 [listed].
Diagnosis. The same as for genus.
Description. Morphometrics and meristics in Table 1. Adult body size moderate for Rhamphichthyidae (maximum size 360 mm SL). Dorsal profile of snout strongly concave anterior to eye, head profile slightly convex post-orbitally. Interorbital region slightly convex. Dorsal margin of body almost straight, slightly ascending at end of body cavity. Ventral body margin slightly concave from anal-fin origin to end of anal fin. Greatest body depth at about middle of standard length. Body tapering posterior to mid body. Caudal appendage long, somewhat ovoid in cross section, its depth slightly greater than its width. Mouth subterminal, lower jaw shorter than upper jaw (Fig. 2). Lower jaw included in upper jaw, rictus short and slightly upturned. Anterior and posterior nares present, without tubes. Anterior nares positioned terminally close to oral aperture, posterior nares positioned dorsally closer to tip of snout than eye. Branchial opening located posterior to opercle, its ventral margin extending to vertical with dorsal margin. No fleshy skin fold on body wall inside branchial opening. Eyes relatively large and positioned laterally. Urogenital papillae well developed, positioned more anteriorly in larger specimens. Anterior chamber of gas bladder not ossified. Posterior chamber of gas bladder apparently absent (MZUSP 49205, 345 mm SL; and Triques, 1996a: 92). Anal-fin origin at vertical with pectoral-fin insertion. Absence of thick fold of skin in proximal portion of anal-fin rays. Lateral line complete, with hourglass shape bony tubes. Lateral line with short ventral rami in anterior half of body.
Skeletal Anatomy. Skeletal anatomy based on computed tomography (CT) scan data from two specimens of Iracema caiana (Figs. 3-9).
Skull roof and neurocranium. Skull roof and neurocranium illustrated in Figure 4. Bones on anterior portion of skull elongate, mesethmoid (met), ventral ethmoid+vomer (v), parasphenoid (pas) and frontals (fr). Mesethmoid narrow, its length greater than antorbital region of frontal; anterior tip small and rounded, flexed ventrally around ventral ethmoid+vomer; posteriorly contacting medial portion of frontal. Ventral ethmoid fused with vomer (=ventral ethmoid+vomer); somewhat cross-shaped in ventral view, not fused anteriorly with mesethmoid. Lateral ethmoid not ossified and thus not visible on scan. Frontals covering more than half skull length. Antorbital process of frontals absent, lateral margin almost straight in dorsal region of orbit. Posterolateral region of frontals extending medially to orbit, contacting pterosphenoid (pts); posterior portion with imbricate suture with parietal (pa). Cranial fontanels large, frontals forming bridge in middle portion. Parietals forming lateral margins of fontanels contacting each other posteriorly, supporting supratemporal laterosensory canal. Small bony ridge on parietal at point of contact with extrascapular. Parietal ridge with inflection at point of contact with thin intermuscular bones of epaxial musculature. Occipital region of braincase consisting of supraoccipital (soc), basioccipital (bo), exoccipital (exo) and epioccipital (epo) bones. Supraoccipital contacting parietal anteriorly and epioccipital ventrolaterally. Epioccipital forming posterodorsal corner of brain case, contacting exoccipitals ventrally and pterotics anteriorly. Exoccipital contacting basioccipital ventrally, pterotics anterodorsally and supraoccipital posterodorsally. Posterodorsal portion of exoccipital together with posteroventral portion of supraoccipital forming condyle articulating skull with supraneural. Exoccipital forming most of foramen magnum, formed ventrally by basioccipital. Basioccipital forming ventral wall of lagenar otolith chamber, covered dorsally by exoccipital. Anterior portion of braincase (sphenoid and otic regions) consisting of orbitosphenoid (obs), pterosphenoid, prootic (pro), sphenotic (spo) and pterotics (pto) bones. Pterotic forming lateral portion of skull supporting semicircular canals of otic capsule. Pterotic contacting prootic and exoccipital ventrally, epioccipital posterodorsally, and exoccipital posteroventrally. Sphenotic contacting frontal dorsally and prootic ventrally. Parasphenoid extending to about three quarters of total length of neurocranium; anterior margin positioned dorsal to ventral ethmoid+vomer medially; posterior margin contacting basioccipital and prootic posteriorly; lateral margin extending dorsally to horizontal line with trigeminal foramen of prootic; ventral margin almost straight in lateral profile. Orbitosphenoid tapering anteriorly, contacting frontals dorsally and parasphenoid ventrally. Orbitosphenoid not contacting pterosphenoid, probably connected by cartilage (as in Rhamphichthys). Orbitosphenoid and pterosphenoid with porous, reticulated surfaces. Pterosphenoid contacting sphenotic posteriorly and parasphenoid ventrally. Sphenotic somewhat rectangular with anterodorsal process contacting frontal. Prootic, sphenotic and anteroventral portion of pterotic articulating with hyomandibula. Prootic contacting basioccipital, exoccipital, pterotics, sphenotics, and parasphenoid. Dorsolateral margin of sphenotic straight, anterior margin ventral to frontal. Prootic ossifying around numerous foramina for nerves and blood vessels. Prootic encapsulating anteriormost pair of otoliths.
Cephalic sensory canals. Small and tubular ossifications surrounding sensory canals of cephalic sensory canal system. Weakly ossified canals of preopercular-mandibular sensory canals; posteriorly contacting preopercle. Infraorbital series represented by five or six long bony tubes; series curved dorsoposteriorly at about third infraorbital. Two or three ossifications of supraorbital sensory canal anteriorly, canal entering frontal posteriorly. One or two ossifications in supratemporal sensory canal overlying parietals, with variable and asymmetrical appearance. Antorbital (ant) laminar; without canal (Fig. 5).
Suspensorium and oral jaws. Suspensorium consisting of hyomandibula (h), symplectic (sym), quadrate (q), metapterygoid (mpt) and endopterygoid (enp) bones (Fig. 5). Long axis of hyomandibula highly oblique, almost parallel to main axis of neurocranium (Fig. 3). Dorsal (proximal) head of hyomandibula narrow and robustly ossified, with two articulating surfaces. Ventral (distal) limb of hyomandibula narrow and thinly ossified, with slender shelf on its anterior margin at middle of bone. Ventral limb of hyomandibula articulating with metapterygoid anterodorsally, and symplectic anteroventrally (almost certainly by cartilage as in other rhamphichthyoids). Hyomandibula with single large foramen on dorsomedial surface, two foramina on lateral surface, and one on posterodorsal margin, all through which pass cranial nerves. Posterior lateral line nerve with separate lateral foramen located on posterodorsal margin of hyomandibula. Large foramen on lateral surface of dorsal portion of hyomandibula (laterosensory, Trigeminal, and Facial nerve components). Preopercular-mandibular (also laterosensory, Trigeminal, and Facial nerve components) foramen located on lateral surface of ventral hyomandibula about 2/3 distance to its tip. Metapterygoid narrow and triangular, with anterior dorsal process contacting endopterygoid. Posterior margin of metapterygoid directly abutting hyomandibula, anteroventral margin contacting symplectic. Symplectic triangular and narrow, about same size as metapterygoid. Anterior portion of symplectic contacting medial surface of quadrate between ascending blade and posteroventral process of quadrate. Quadrate contacting anguloarticular (ang) anteriorly and endopterygoid dorsally, with interdigitated contact margin. Quadrate anterodorsal process somewhat squared, not rounded. Endopterygoid elongate anteriorly, edentulous. Ascending process of endopterygoid long and thin, directed obliquely backward towards frontal and orbitosphenoid, not contacting these bones. Ascending endopterygoid process located about midlength of bone. Autopalatine not ossified and thus not visible in scan. Several isolated filamentous bones in adductor mandibulae (ib), located lateral to endopterygoid, extending from posterodorsal portion of dentary (d) obliquely to articulation of hyomandibula with neurocranium. Intermuscular ossifications absent in levator operculi, dilator operculi and protractor hyoidei.
Upper jaw consisting of maxilla (m) and premaxilla (p). Maxilla elongate, sickle shape, its dorsal margin concave and ventral margin convex; anterior margin rounded; anterior maxillary process absent; descending process of maxilla narrow, tapering distally. Premaxilla gracile, with small ascending process. Bones of lower jaw consist in dentary, anguloarticular, retroarticular (rar), and coronomeckelian (cm) bones. Dorsal portion of dentary straight; posterior margin deeply forked; anteroventral portion with small hook-shaped process directed posteroventrally. Anguloarticular in close contact with dentary anteriorly and extensively contacting quadrate posteriorly. Anguloarticular condyle socket positioned along posterodorsal margin of lower jaw. Anguloarticular contributing more than retroarticular to joint with quadrate. Retroarticular triangular in shape, anterior margin pointed, dorsoposterior portion contacting quadrate (Fig. 5A). Coronomeckelian bone elongate and teardrop shaped, located at dorsoposterior portion of dentary posterior concavity. Teeth absent in both jaws.
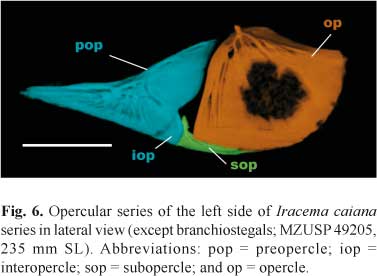
Opercular series. Opercular series consists of preopercle (pop), interopercle (iop), subopercle (sop), and opercle (op; Fig. 6). Preopercle long, anterior margin pointed; without concavity (as in Steatogenys Boulenger and Hypopygus Hoedeman). Dorsal margin concave, most concave in medial portion; anteroventral margin straight, posteroventral margin expanded. Posterodorsal portion of preopercle closely associated with posteroventral margin of hyomandibula. Preopercular-mandibular sensory canal fused with surface of preopercle, with five laterally oriented canal rami leading to pores. Subopercle thin, ventral surface convex and dorsal surface concave. Anterodorsal arm of subopercle long, reaching about same height of dorsoposterior portion of interopercle. Interopercle thin, weakly ossified, triangular in shape, anterior portion in close contact to preopercle. Posterior margin of interopercle convex in contact with subopercle. Opercle broad and fan shaped, dorsal margin concave posteriorly and with small crest anteriorly. Anterior margin of opercle almost straight, surface covered with series of vertical ridges and grooves. Central surface of opercle with reticulated texture. Opercle ventral margin rounded and convex, contacting subopercle anteroventrally.
Branchial basket. Gill arches illustrated in Figure 7. This description includes skeletal support of gills and associated elements, including basihyals (bh), urohyal (uh) and branchiostegals rays (br). Bones of gill arches poorly ossified (or demineralized during preservation), showing some degree of reticulated texture. Dorsal elements of branchial skeleton consisting of four epibranchials (bb), pharyngobranchials (poorly ossified?) and pharyngobranchial toothplate (tp). Epibranchial three with anterior uncinate process and epibranchial four forked posteriorly. Ventral elements of branchial skeleton consisting of five ceratobranchials and three basibranchials. Basihyal fused with basibranchial 1, forming long, rod shaped hollow bone (its core not ossified). Dorsal surface of basihyal rounded, not concave. Basibranchials two and three weakly ossified. Ventral hypohyal (vhh) located anterior to dorsal hypohyal (dhh), these located over anterior ceratohyal (ach). Anterior ceratohyal narrow medially. Posterior ceratohyal (pch) rounded, half ovoid, with distinct notch in dorsal margin. Hypobranchials apparently not ossified. Urohyal fusiform in ventral view; inverted T shape in cross section; with medial ventral slit along long axis, head large with lateral ridges, and anterior portion flat. Posterior portion of urohyal hyperossified forming elongate blade, reaching vertical with third ceratobranchial (cb). Interhyal small and located dorsoposteriorly to posterior ceratohyal. Four branchiostegal rays. First (anterior) branchiostegal ray filamentous, second thin, larger than first, both contacting anterior ceratohyal. Third and fourth branchiostegals lamellar, third contacting limit between anterior and posterior ceratohyal, fourth contacting posterior ceratohyal.
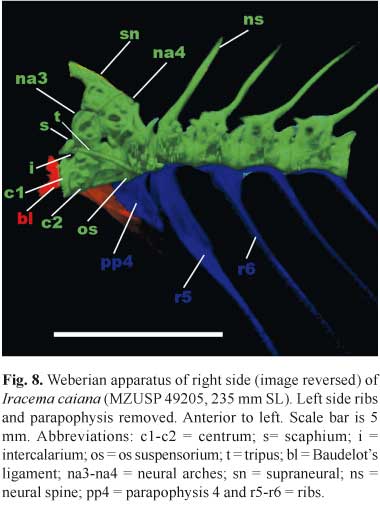
Weberian Apparatus. Anterior vertebrae and Weberian Apparatus illustrated in Figure 8. Neural arches (na) of third and fourth vertebrae and supraneural (sn) meet together forming roof of neural canal. Supraneural adjacent to supraoccipital and neural arch of third vertebra adjacent to exoccipital. Bones of Weberian complex forming socket for dorsal contact between occipital bones of skull and axial skeleton (in Gymnorhamphichthys only supraoccipital articulates with neural complex; Mago-Leccia, 1994: 40). Scaphium (s) barrel-shaped and located anterior to, and slightly ventral to third neural arch. Intercalarium (i) small and round, positioned posterior to scaphium and anterior to tripus (t). Tripus positioned obliquely and descending posteriorly between scaphium and ventral portion of fourth centrum. Tripus narrow anteriorly, broad in median region and tapering posteriorly. First centrum (c1) anteroposteriorly compressed and disk-like shape. Second centrum (c2) larger and with prominent parapophysis behind ossified Baudelot's ligament (bl), extending posterolaterally and fused with fourth parapophysis (pp4) and pleural rib complex. Third centrum located posterior to other components of Weberian complex. Fourth vertebra with fourth parapophysis and pleural rib complex fused to second parapophysis anteriorly. Os suspensorium (os) fused with ventral portion of fourth vertebrae, which extends anteriorly, tapering to reach ventral portion of second centrum. Fifth centrum bearing fifth rib (r5), slightly expanded along antero-posterior axis in midregion. Fifth vertebra bears first neural spine with spinous morphology, located in anterior portion of vertebra, and oriented obliquely posterodorsally. Subsequent ribs widest close to articulation with vertebrae; tapering distally. Anterior chamber of gas bladder not enclosed in bony capsule as in Rhamphichthys (Mago-Leccia, 1994: 40).
Vertebral column. Eighteen precaudal vertebrae (n=2; x-rayed specimens) including vertebrae of Weberian apparatus; 84 vertebrae to end of anal fin (n=1; x-rayed specimen). Neural and hemal spines positioned anteriorly on centrum in anterior vertebrae, transitioning to posterior position on centrum at about vertebra 62. Intermuscular bones (observed in CT scans) composed by dorsal series of myorhabdoi, epicentral bones and series of filamentous bones of anterior epaxial muscles. Epicentral bones in contact with dorsoposterior portion of ribs, rod shaped and somewhat sinusoidal, directed towards posterodorsal portion of body. First two epicentral bones not contacting ribs. Anterior portion of first epicentral bones located slightly anteriorly to vertical with scaphium; second slightly anterior to contact between neural arches three and four, third contacting fourth rib. Dorsal series of myorhabdoi obliquely positioned, anterior and posterior ends branched multiply filamentous bones in anterior dorsal muscles positioned posterior to epioccipital simple or sometimes branched.
We were not able to count precisely number of displaced hemal spines, but radiographs show at least more than six in MZUSP 49205, 345 mm SL. Displaced hemal spines all of similar size; absence of enlarged anterior displaced hemal spine (Albert, 2001: fig. 35c-d; posteroventral abdominal bone of Hilton et al., 2007: fig. 16b).
Anal fin and supports. Anal-fin rays range from 240-257 rays (n=4). All rays posterior to rays 23-42 branched, no rays branched more than once. Branched portions of rays always adnate from branching origin to distal end of fin. Three anal-fin pterygiophores between adjacent hemal spines.
Pectoral fin and girdle. Pectoral fin with 16-17 rays (n=4), anomalous specimen with 12 fin rays on left side (MZUSP 49205, 235 mm SL; as noted by Triques 1996: 92). Pectoral fin lanceolate, dorsal rays longer than ventral rays, distal fin margin slightly rounded. One or two dorsalmost rays unbranched.
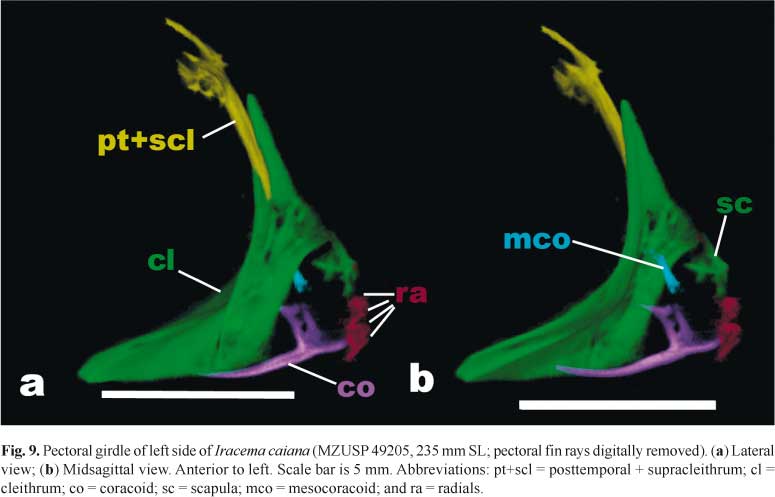
Dermal elements of pectoral girdle consisting of posttemporal (pt), supracleithrum (scl) and cleithrum (cl; Fig. 9); postcleithrae apparently absent. Posttemporal bone fused with supracleithrum (pt+scl), bearing large foramen for passage of posterior lateral line nerve. Dorsal portion of posttemporal + supracleithrum bones positioned over exoccipital and epioccipital. Cleithrum large, dorsal and ventral portions of similar size.
Chondral components of pectoral girdle consist of scapula (sc), coracoid (co) and mesocoracoid (mco; Fig. 9). Scapula not well ossified; axe-blade shape. Coracoid with long, thin and curved ventral process directed anteriorly. Dorsal process of coracoid hatchet-shaped with hook-shaped process on dorsal surface. Mesocoracoid present, rod-like hourglass shaped structure, obliquely oriented anterodorsally to posteroventrally. Support for pectoral fin rays composed of four radials (ra; MZUSP 49205, 345 mm SL). Baudelot's ligament ossified, branching anteriorly and posteriorly; extending from posteroventral portion of basioccipital and posterolateral portion of exoccipital to cleithrum; with "><" shape in lateral view.
Scales. Absence of scales on anterior two-thirds of body. Scales covering entire caudal appendage. Absence of scales in anterior portion of lateral line. Lateral line ossicles embedded in thick layer of skin. Scales above lateral line in posterior portion of body ovoid in shape, elongate on horizontal axis, about twice as long as deep (Fig. 10).
Electric organs. No information available regarding electric organ discharges (EOD) in Iracema caiana. Hypaxial electric organ extending anteriorly to mental region. Four rows of electrocytes present above posterior end of anal fin.
Color in Alcohol. Background color pale yellow, dorsolateral and lateral surfaces of head and body darker. Head almost unpigmented ventrally except for dark chromatophores on anterior portion of lower jaw. Dorsal portion of head and temporal region strongly pigmented, forming dark brown speckles from snout to nape region; speckles of about eye size; most concentrated dorsolaterally and absent on dorsal midline of head. Lateral region of body yellow with dark reddish and diffuse chromatophores. Anal-fin pterygiophore region lighter than other parts of body. Scattered dark brown speckles over anal-fin pterygiophores. About 20 to 30 dark brown diffuse blotches on lateral surface of body. Caudal appendage pale yellow, except for diffuse dark brown bands not extending to dorsal or ventral midline. Distal third of caudal appendage pale yellow, not darkly pigmented. Pectoral-fin membranes mostly hyaline with scattered chromatophores at distal end; anal-fin membrane hyaline except for dark brown band covering entire extent of distal two-thirds of rays. Iridescent coloration throughout entire lateral portion of body.
Distribution.Iracema caiana is known only from the type locality on the rio Jauaperi, a tributary of rio Negro in the Amazon basin. Despite a recent survey of several museum collections with large holdings of rhamphichthyid specimens, no additional material of Iracema caiana was identified. This makes Iracema the rarest species of Rhamphichthyidae and one of the rarest of Gymnotiformes (Nogueira et al., 2010). This limited distribution does not seem the result of a sample bias, since the rio Negro basin is relatively well surveyed (Goulding et al., 1988; Chao, 2001).
Material examined.Iracema caiana. Brazil. MZUSP 8952, holotype, 360 mm SL, Amazonas, rio Jauaperi, sand beach about 40-50 km above its mouth with the rio Negro, approximately 01º05'S 61º35'W, T. Roberts, 19 Nov 1968. MZUSP 49205, 3 paratypes, 208-345 mm SL, collected with the holotype.
Discussion
Triques (1996a) diagnosed Iracema caiana from other rhamphichthyids (Rhamphichthys and Gymnorhamphichthys) using a meristic character (anal-fin ray counts) and two color pattern characters. Iracema caiana has 240-257 anal-fin rays, contrasting with 139-211 in Gymnorhamphichthys and 304-470 in Rhamphichthys. Triques (1996a) also noted that Iracema caiana has a color pattern unique within the family, composed of irregularly shaped dark pigment blotches distributed along the sides of the body, and a uniform, broad and darkly pigmented stripe along the dorsal margin of the body. The first of these pigmentation characters is also present in some specimens of Rhamphichthys (an undescribed species; Rhamphichthys sp. INPA 17646).
Iracema has been consistently considered the sister group of Gymnorhamphichthys, based on derived features of squamation: 1) loss of scales from the anterior two thirds of the body (Triques, 1996a; Albert, 2001: character 16); 2) loss of scales on the anterior middorsum (Albert, 2001: character 15); and (3) possession of tubular lateral line scales (Triques, 1996a; Fig. 3). We observe that all rhamphichthyids have losses of scales to a certain degree. Juveniles Rhamphichthys have no scales on the anterior portion of body, these appearing later during development. In adult Rhamphichthys the squamation of the middorsum is not complete (Albert, 2001: character 16), but rather covered by scattered small round scales. This feature supports the hypothesis that Rhamphichthys is generally hypermorphic in Rhamphichthyidae, with further development in many traits as compared with the other genera. The presence of tubular lateral line scales in the anterior portion of the body is also observed in all juvenile rhamphichthyoids, and also in all adult Rhamphichthys (cf. Triques 1996a: 92) and Gymnorhamphichthys.
Albert (2001) diagnosed the clade formed by Iracema and Gymnorhamphichthys by the absence of an ascending process of the endopterygoid (his character 129) and all anal-fin rays unbranched (his character 197). Our examination of computed tomography images of Iracema caiana reveals the presence of an ascending process of the endopterygoid, which is long and very thin (Figs. 3 and 5), a condition shared also by Rhamphichthys but absent and derived in all species of Gymnorhamphichthys. In Iracema caiana, the anal-fin rays posterior to rays 23 to 42 are in fact bifurcated distally, although the rami are either adnate or very close together along most of the length of the fin. This appearance contrasts with the forked appearance of most anal-fin rays along the length of the fin in Rhamphichthys and other rhamphichthyoids.
An alternative sister group relationship between Iracema and Rhamphichthys is supported by the following synapomorphies: 1) filamentous intermuscular bones in the musculus adductor mandibulae; 2) central surface of opercle reticulated; 3) Baudelot's ligament ossified from its origin in the exoccipital and basioccipital to attachment with cleithrum; and 4) scales above lateral line in the posterior portion of body elongate in the horizontal axis, about twice as long as deep.
Intermuscular bones in the adductor mandibulae were noted by Albert (2001: character 44) in Rhamphichthys. We also observe a small number of intermuscular bones in the adductor mandibulae in Gymnotus carapo Linnaeus (USNM 225286), and a similar condition was described for Orthosternarchus tamandua Boulenger (Hilton et al., 2007). According to these authors O. tamandua also possesses intermuscular in the levator operculi and dilator operculi. The presence of intermuscular bones may be correlated with evolution of an elongate snout (although this does not explain their presence in the opercular muscles of Rhamphichthys). We observe intermuscular bones in the levator operculi, dilator operculi, and protractor hyoidei in all species of Rhamphichthys, but not Iracema or Gymnorhamphichthys.
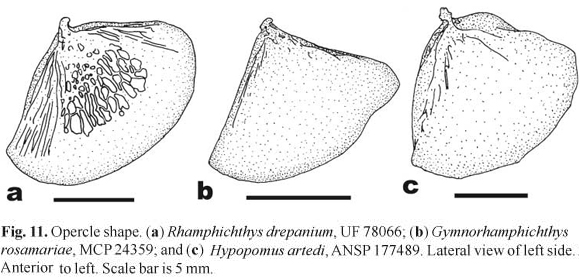
Rhamphichthys and Iracema share a reticulated texture to the central portion of the surface of the opercle, not seen in any other member of Rhamphichthyoidea (Fig. 11). In addition, Rhamphichthys presents a reticulated surface in other bones in the neurocranium, suspensorium and anterior vertebrae, which is not clearly seen in CT reconstructions of Iracema. The presence of reticulated texture to bones (including the opercle) is a derived trait in several other clades of gymnotiforms that inhabit the benthos of large river channels, especially Apteronotidae (except Apteronotus La Cepède s.s.; Albert, 2001: fig. 18: character 82; see also Hilton et al., 2007), and some species of the sternopygid genera Distocyclus Mago-Leccia, Eigenmannia Jordan & Evermann and Rhabdolichops Eigenmann & Allen. The function of the reticulated bone morphology is unknown, but as the pores and pits are usually filled with lipids and the trait may aid in reducing negative buoyancy.
Baudelot's ligament in Gymnotiformes has a single attachment to the neurocranium, separating it distally in two parts where it attaches to the fourth pleural rib and cleithrum (Fink & Fink, 1981). This ligament is partially ossified in some gymnotiforms (e.g. Sternopygus Müller & Troschel; Fink & Fink, 1981: fig. 19d). In Rhamphichthyidae the anterior portion of the ligament is connected to the exoccipital as well as the basioccipital. In the derived condition observed in Rhamphichthys and Iracema, the ligament is totally ossified (Figs. 3 and 8), whereas in Gymnorhamphichthys only the middle portion of the ligament is ossified.
Scales above the lateral line in the posterior portion of body are elongate in the horizontal axis, about twice as long as deep. Elongate scales evolved independently in several gymnotiform clades (Albert, 2001: character 18 and Albert et al., 2004: fig. 17: character 97). However, within rhamphichthyoids, horizontally elongate scales appear to have evolved only once in a clade formed by Iracema and Rhamphichthys.
The caudal appendage of Iracema caiana differs to a degree from all other rhamphichthyoids and can be considered an autapomorphy. The caudal appendage of Iracema is ovoid in cross section, and at its base (at the posterior end of the anal fin) it is about as wide as it is deep.
The data presented here contradicted all previously published hypotheses of interrelationships of Iracema caiana that regarded it as the sister group to Gymnorhamphichthys (Triques, 1996a; Albert & Campos-da-Paz, 1998; Albert, 2001). These data suggest that Iracema is the sister group to Rhamphichthys and we will not include it in this genus. Also, Rhamphichthys species share several synapomorphies not present in Iracema caiana, e.g. the anterior chamber of gas bladder enclosed in a bony capsule (Mago-Leccia, 1994), and the presence of a fleshy skin fold on the body wall, inside the opercular opening (Triques, 2005), which reinforce its monophyly.
Comparative material.Apteronotus albifrons: Venezuela. Monagas. UF 29921, 11 (1 cs), Laguna Grande. Brachyhypopomus occidentalis: Costa Rica. Limón. ANSP 163176, 9 (1 cs), creek close to Shiroles. Eigenmannia virescens: Venezuela. Guarico. UF 78117, 27 (2 cs), río Tiznados at Guardatinajas. Gymnorhamphichthys bogardusi: Venezuela. Delta Amacuro. ANSP 187349, 7 (2 cs), río Orinoco, downstream from caño Remolina. Gymnorhamphichthys hypostomus: Brazil. Amazonas. MCP 24292, 2 (1 cs), rio Negro, 18.5 miles above Manaus. Pará. MCP 26580, 3 (1 cs), rio Trombetas 0.8 miles upstream Vila Aracua. Gymnorhamphichthys petiti: Brazil. Mato Grosso. MCP 30373, 4 (1 cs), ribeirão Macuco on highway BR-163, about 74 km N of Sinop. MCP 40189, 5 (1 cs), creek tributary to rio Suiazinho, on highway BR-158 N of Ribeirão Cascalheira. Gymnorhamphichthys rondoni: Brazil. Pará. MCP 15161, 3 (1 cs), rio Tapajós, beach at island 5 km upstream Itaituba. MZUSP 97147, 9, (1cs), rio Curuá tributary of rio Iriri at Vila Castelo dos Sonhos. Gymnorhamphichthys rosamariae: Brazil. Amazonas. MCP 24359, 2 (1 cs), rio Negro 18.5 miles upstream Manaus. MCP 24872, 9 (2 cs), rio Negro upstream rio Branco mouth, between Carvoeiro and Vila Guajara. Gymnotus carapo: Suriname. Nickerie. USNM 225286, 15 (1 cs), Koekwie Kreek. Hypopomus artedi: Suriname. ANSP 177489, 2 (1 cs), Burro-Burro River. CAS 72233, 2 of 4, Parwapa Kreek. Hypopygus lepturus: Brazil. Amazonas. MCP 41121, 10 (2 cs), creek on highway BR-319. Rhamphichthys apurensis: Venezuela. Bolivar. ANSP 162300, 60 (1 cs), río Orinoco near mouth of río Caura. Rhamphichthys drepanium: Venezuela. Guarico. UF 78066, 2 (1 cs), Morichal 2.3 km N of San Fernando de Apure. Rhamphichthys hahni: Brazil. Mato Grosso do Sul. MZUSP 59297, 2 (1 cs), rio Novo tributary to rio Negro at brejo de Santa Sofia. Rhamphichthys lineatus: Brazil. Amazonas. MCP 33457, 2 (1 cs), paraná Maiana, lago Mamirauá. Rhamphichthys marmoratus: Brazil. Pará. MZUSP 44493, 2 (1 cs), rio Apeu, at Boa Vista de Apeu. Rhamphichthys rostratus: Brazil. Amazonas. MCP 27756, 23 (1 cs), rio Negro 15 miles downstream Moura. Rhamphichthys sp.: Brazil. Amazonas. INPA 17646, 7 (1 cs), Costa do Catalão. Steatogenys elegans: Brazil. Amazonas. MCP 24290, 8 (2 cs), rio Amazonas downstream rio Negro. Sternopygus macrurus: Venezuela. Apure. MCNG 3733, 4 cs, río Apure.
Acknowledgements
We thank J. Maisano and M. Colbert (UT Austin) for preparation of computed tomography x-rays, K. Luckenbill (ANSP) for help in learning image editing with VGStudio Max, and O. Oyakawa (MZUSP) for the loan of types specimens. We also thank C. Lucena (MCP), O. Oyakawa and J. Figueiredo (MZUSP), M. Sabaj-Pérez, K. Luckenbill and J. Lundberg (ANSP), L. Py-Daniel and M. Rocha (INPA), and R. Robbins (UF) for the loan of specimens or support in the fish collections. We acknowledge J. Lundberg and E. Maxime for discussions of gymnotiform morphology. This project was supported in part by the Student Exchange Program of DeepFin. TPC is partially supported by a research assistantship of NSF-0741450 to JSA.
Literature Cited
Submitted January 4, 2011
Accepted August 19, 2011
Published September 16, 2011
- Albert, J. S. 2001. Species diversity and phylogenetic systematics of American knifefishes (Gymnotiformes, Teleostei). Miscellaneous Publications, Museum of Zoology, University of Michigan, 190: 1-127.
- Albert, J. S. & R. Campos-da-Paz. 1998. Phylogenetic systematics of Gymnotiformes with diagnoses of 58 clades: a review of available data. Pp. 419-446. In: Malabarba, L. R., R. E. Reis, R. P. Vari, Z. M. S. Lucena & C. A. S. Lucena (Eds.). Phylogeny and classification of Neotropical fishes. Porto Alegre, Edipucrs, 603p.
- Albert J. S. & W. G. R. Crampton. 2005. Diversity and phylogeny of Neotropical electric fishes (Gymnotiformes). Pp. 360-409.In: Bullock, T. H., C. D. Hopkins, A. N. Popper & R. R. Fay (Eds.). Electroreception. New York, Springer Handbook of Auditory Research, 485p.
- Albert, J. S., W. G. R. Crampton, D. H. Thorsen & N. R. Lovejoy. 2004. Phylogenetic systematics and( historical biogeography of the Neotropical electric fish Gymnotus (Teleostei: Gymnotiformes). Systematic and Biodiversity, 2: 375-417.
- Alves-Gomes, J. A., G. Orti, M. Haygood, A. Meyer & W. Heiligenberg. 1995. Phylogenetic analysis of the South American electric fishes (Order Gymnotiformes) and the evolution of their electrogenic system: a synthesis based on morphology, electrophysiology, and mitochondrial sequence data. Molecular Biology and Evolution, 12: 298-318.
- Carvalho T. P., C. S. Ramos & J. S. Albert. 2011. A new species of Gymnorhamphichthys (Gymnotiformes: Rhamphichthyidae) from Paraná-Paraguay system. Copeia, 2011(3): 400-406.
- Chao, N. L. 2001. The fishery, diversity and conservation of ornamental fishes in the Rio Negro Basin, Brazil - a review of the Project Piaba (1989-99). Pp. 161-204. In: Chao N. L., P. Petry, G. Prang. L. Sonneschien & M. Tlusty (Eds.). Conservation and management of ornamental fish resources of the Rio Negro Basin, Amazonia, Brazil - Project Piaba. Manaus, EDUA, 310p.
- Crampton, W. G. R. 2011. An ecological perspective on diversity and distributions. Pp. 165-189. In: Albert, J. S. & R. E. Reis (Eds.). Historical Biogeography of Neotropical Freshwater Fishes. Berkeley, UCPress, 388p.
- Crampton, W. G. R. & J. S. Albert. 2006. Evolution of electric signal diversity in gymnotiform fishes. Pp. 647-731. In: Ladich, F., S. P. Collin, P. Moller & B. G. Kapoor (Eds.). Communication in fishes. Enfield, Science Publishers, 870p.
- Datovo, A. & F. A. Bockmann. 2010. Dorsolateral head muscles of the catfish families Nematogenyidae and Trichomycteridae (Siluriformes: Loricarioidei): comparative anatomy and phylogenetic analysis. Neotropical Ichthyology, 8: 193-246.
- Ferraris Jr., C. J. 2003. Rhamphichthyidae (sand knifefishes). Pp. 495-497. In: Reis, R. E., S. O. Kullander & C. J. Ferraris, Jr. (Eds.). Checklist of the Freshwater Fishes of the South and Central America. Porto Alegre, Edipucrs, 729p.
- Fink S. V. & W. L. Fink. 1981. Interrelationships of the Ostariophysan fishes (Teleostei). Zoological Journal of the Linnean Society, 72: 297-353.
- Fink S. V. & W. L. Fink. 1996. Interrelationships of Ostariophysan fishes (Teleostei). Pp. 209-249. In: Stiassny, M. L. J., L. R. Parenti & D. Johnson (Eds.). San Diego, Academic Press, 496p.
- Fink, W. L. & J. H. Humphries. 2010. Morphological description of the extinct North American sucker Moxostoma lacerum (Ostariophysi, Catostomidae), based on high-resolution x-ray computed tomography. Copeia, 2010: 5-13.
- Goulding, M., M. L. Carvalho & E. G. Ferreira, 1988. Rio Negro, rich life in poor water: Amazonian diversity and foodchain ecology as seen through fish communities. The Hague, SPB Academic Publishing, 200p.
- Hilton E. J., C. Cox Fernandes, J. P. Sullivan, J. G. Lundberg & R. Campos-da-Paz. 2007. Redescription of Orthosternachus tamandua (Boulenger, 1998) (Gymnotiformes, Apteronotidae), with reviews of its ecology, electric organ discharges, external morphology, osteology, and phylogenetic affinities. Proceedings of the Academy of Natural Sciences of Philadelphia, 156: 1-25.
- Lundberg, J. G. 2005. Gymnorhamphichthys bogardusi, a new species of sand knifefish (Gymnotiformes: Rhamphichthyidae) from the Rio Orinoco, South America. Notula Naturae, 479: 1-4.
- Mago-Leccia, F. 1978. Los Peces de la familia Sternopygidae de Venezuela, incluyendo una descripción de la osteología de Eigenmannia virescens y una nueva definición y classificación del Orden Gymnotiformes. Acta Cientifica Venezuelana, 29: 1-51.
- Mago-Leccia, F. 1994. Electric fishes of the continental waters of America. Caracas, Fundación para el Desarollo de las Ciencias Físicas, Matemáticas y Naturales, Electric Fishes: 1-206.
- Nijssen, H., I. J. H. Isbrucker & J. Géry. 1976. On the species of Gymnorhamphichthys Ellis, 1912, translucent sand-dwelling gymnotid fishes from South America (Pisces, Cypriniformes, Gymnotoidei). Studies on Neotropical Fauna and Environment, 11: 37-63.
- Nogueira C., P. A. Buckup, N. A. Menezes, O. T. Oyakawa, T. P. Kasecker, M. B. Ramos Neto & J. M. C. da Silva. 2010. Restricted-range fishes and the conservation of Brazilian freshwaters. PLOS One, 5: 1-10.
- Schaefer, S. A. 2003 Relationships of Lithogenes villosus Eigenmann, 1909 (Siluriformes, Loricariidae): evidence from high-resolution computed microtomography. American Museum Novitates, 3401: 1-55.
- Schaefer S. A. & L. Fernández. 2009. Redescription of the Pez Graso, Rhizomichthys totae (Trichomycteridae) of Lago Tota, Colombia, and aspects of cranial osteology revealed by microtomography. Copeia, 2009: 510-522.
- Schwassmann, H. O. 1989. Gymnorhamphichthys rosamariae, a new Species of knifefish (Rhamphichthyidae, Gymnotiformes) from the upper Rio Negro, Brazil. Studies on Neotropical Fauna and Environment, 24: 157-167.
- Taylor, W. R. & G. C. Van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium, 9: 107-119.
- Triques, M. L. 1996a. Iracema caiana, a genus and species of electrogenic Neotropical freshwater fish (Rhamphichthyidae: Gymnotiformes: Ostariophysi: Actionpterygii). Revue Française d'Aquariologie, 23: 91-92.
- Triques, M. L. 1996b. Eigenmannia vicentespelea, a new species of cave dwelling electrogenic neotropical fish (Ostariophisi: Gymnotiformes: Sternopygidae). Revue Française d'Aquariologie, 23: 1-4.
- Triques, M. L. 2005. Novas sinapomorfias para Rhamphichthys Muller & Troschel, 1848 (Teleostei: Rhamphichthyidae). Lundiana, 6: 35-39.
- Winemiller, K. O. & S. C. Willis. 2011. The Vaupes Arch and Casiquiare Canal: barriers and passages. Pp. 225-242. In: Albert, J. S. & R. E. Reis (Eds.). Historical Biogeography of Neotropical Freshwater Fishes. Berkeley, UCPress, 388p.
- Winterbottom, R. 1974. A descriptive synonymy of the striated muscles of the Teleostei. Proceedings of the Academy of Natural Sciences of Philadelphia, 125(12): 225-317.
Redescription and phylogenetic position of the enigmatic Neotropical electric fish Iracema caiana Triques (Gymnotiformes: Rhamphichthyidae) using x-ray computed tomography
Publication Dates
-
Publication in this collection
29 Sept 2011 -
Date of issue
2011
History
-
Received
04 Jan 2011 -
Accepted
19 Aug 2011