Abstracts
The aim of this study was to determine the trophic structure of the fish community in the Bananal stream subbasin, which belongs to a well-preserved Cerrado area (Brazilian Savanna) in Brasília National Park, Brazil. We also evaluated the influence of environmental variations in the diet of fish species. Four samples were taken in each 30 m long established transect, two in the rainy season and two in the dry season. A total of 1,050 stomachs of the 13 most abundant species were analyzed. A total of 36 food items were consumed, where 24 were autochthonous, 8 allochthonous, and 4 of undetermined origin. Non-metric multidimensional scaling (NMDS) analysis, in addition to the results of frequency of occurrence and abundance charts, was used to determine four groups of feeding guilds: detritivores, omnivores (tending toward herbivory and invertivory), invertivores and piscivores. Around 69% of the volume of resources consumed was allochthonous, which proves the importance of the resources provided by riparian vegetation. The contribution of autochthonous and allochthonous items in the diet differed due to seasonality for Aspidoras fuscoguttatus, Astyanax sp., Characidium xanthopterum, Hyphessobrycon balbus, Kolpotocheirodon theloura, Moenkhausia sp., Phalloceros harpagos, and Rivulus pictus. Despite the Cerrado climate characteristics, there was no significant influence of season on the fish diet. The absence of seasonal variation and the predominance of allochthonous items in the fish diet are probably associated with the presence of riparian vegetation, which acts as a transition area in the Cerrado biome and provides resources for the aquatic fauna. This work shows the importance of studies in non-disturbed areas considered here as a source of information concerning the biology of fish species and as a guide for direct conservation policies on the management of aquatic resources, recovery of damaged areas and determination of priority areas for conservation.
Feeding guilds; Seasonal variation; Stream fish; Trophic ecology
O presente estudo foi desenvolvido com o objetivo de determinar a estrutura trófica da comunidade de peixes da sub-bacia do ribeirão Bananal pertencente a uma área bem preservada de Cerrado no Parque Nacional de Brasília. Além disso, buscou-se verificar a influência das variações ambientais na dieta das espécies. Em cada trecho de 30 m de extensão foram realizadas quatro amostragens, duas no período seco e duas no período chuvoso. Foram analisados 1050 estômagos pertencentes às 13 espécies mais abundantes encontradas. No total, foram consumidos 36 itens alimentares sendo 24 autóctones, oito alóctones e quatro de origem indeterminada. A análise de escalonamento multidimensional não-métrico (NMDS), juntamente com os resultados dos gráficos de freqüência de ocorrência e abundância, agrupou as espécies em quatro guildas: detritívoros, onívoros (com tendência à herbivoria e invertivoria), invertívoros e piscívoros. Cerca de 69% dos recursos consumidos pelos indivíduos foram de origem alóctone, o que evidencia a importância dos recursos advindos das matas de galeria. A contribuição de presas autóctones e alóctones na dieta diferiu, de acordo com a estação para Aspidoras fuscoguttatus, Astyanax sp., Characidium xanthopterum, Hyphessobrycon balbus, Kolpotocheirodon theloura, Moenkhausia sp., Phalloceros harpagos e Rivulus pictus. Apesar das características climáticas do bioma Cerrado, não houve influência significativa da variação sazonal na dieta. A ausência de variação sazonal e a predominância de itens alóctones na dieta provavelmente estão associadas à presença de matas ciliares, que atuam como zonas de amortecimento e zonas provedoras de recursos para a biota aquática. O presente estudo demonstra a importância de estudos em regiões íntegras para fornecer conhecimentos sobre a biologia das espécies e permitir o direcionamento de ações públicas para o manejo dos recursos aquáticos, para a recuperação de áreas degradadas e para a determinação de áreas prioritárias para a conservação.
ILaboratório de Bentos, Departamento de Zoologia, Instituto de Ciências Biológicas, Universidade de Brasília, Campus Darcy Ribeiro, ICC norte, Asa Norte. 70.910-900 Brasília, DF, Brazil. marischneider224@gmail.com; pedropua@unb.br; mjsilva@unb.br
IIDepartamento de Ecologia, Instituto de Ciências Biológicas, Universidade de Brasília, Campus Darcy Ribeiro, ICC norte, Asa Norte. . 70.910-900 Brasília, DF, Brazil. padovesi@unb.br
ABSTRACT
The aim of this study was to determine the trophic structure of the fish community in the Bananal stream subbasin, which belongs to a well-preserved Cerrado area (Brazilian Savanna) in Brasília National Park, Brazil. We also evaluated the influence of environmental variations in the diet of fish species. Four samples were taken in each 30 m long established transect, two in the rainy season and two in the dry season. A total of 1,050 stomachs of the 13 most abundant species were analyzed. A total of 36 food items were consumed, where 24 were autochthonous, 8 allochthonous, and 4 of undetermined origin. Non-metric multidimensional scaling (NMDS) analysis, in addition to the results of frequency of occurrence and abundance charts, was used to determine four groups of feeding guilds: detritivores, omnivores (tending toward herbivory and invertivory), invertivores and piscivores. Around 69% of the volume of resources consumed was allochthonous, which proves the importance of the resources provided by riparian vegetation. The contribution of autochthonous and allochthonous items in the diet differed due to seasonality for Aspidoras fuscoguttatus, Astyanax sp., Characidium xanthopterum, Hyphessobrycon balbus, Kolpotocheirodon theloura, Moenkhausia sp., Phalloceros harpagos, and Rivulus pictus. Despite the Cerrado climate characteristics, there was no significant influence of season on the fish diet. The absence of seasonal variation and the predominance of allochthonous items in the fish diet are probably associated with the presence of riparian vegetation, which acts as a transition area in the Cerrado biome and provides resources for the aquatic fauna. This work shows the importance of studies in non-disturbed areas considered here as a source of information concerning the biology of fish species and as a guide for direct conservation policies on the management of aquatic resources, recovery of damaged areas and determination of priority areas for conservation.
Key words: Feeding guilds, Seasonal variation, Stream fish, Trophic ecology.
RESUMO
O presente estudo foi desenvolvido com o objetivo de determinar a estrutura trófica da comunidade de peixes da sub-bacia do ribeirão Bananal pertencente a uma área bem preservada de Cerrado no Parque Nacional de Brasília. Além disso, buscou-se verificar a influência das variações ambientais na dieta das espécies. Em cada trecho de 30 m de extensão foram realizadas quatro amostragens, duas no período seco e duas no período chuvoso. Foram analisados 1050 estômagos pertencentes às 13 espécies mais abundantes encontradas. No total, foram consumidos 36 itens alimentares sendo 24 autóctones, oito alóctones e quatro de origem indeterminada. A análise de escalonamento multidimensional não-métrico (NMDS), juntamente com os resultados dos gráficos de freqüência de ocorrência e abundância, agrupou as espécies em quatro guildas: detritívoros, onívoros (com tendência à herbivoria e invertivoria), invertívoros e piscívoros. Cerca de 69% dos recursos consumidos pelos indivíduos foram de origem alóctone, o que evidencia a importância dos recursos advindos das matas de galeria. A contribuição de presas autóctones e alóctones na dieta diferiu, de acordo com a estação para Aspidoras fuscoguttatus, Astyanax sp., Characidium xanthopterum, Hyphessobrycon balbus, Kolpotocheirodon theloura, Moenkhausia sp., Phalloceros harpagos e Rivulus pictus. Apesar das características climáticas do bioma Cerrado, não houve influência significativa da variação sazonal na dieta. A ausência de variação sazonal e a predominância de itens alóctones na dieta provavelmente estão associadas à presença de matas ciliares, que atuam como zonas de amortecimento e zonas provedoras de recursos para a biota aquática. O presente estudo demonstra a importância de estudos em regiões íntegras para fornecer conhecimentos sobre a biologia das espécies e permitir o direcionamento de ações públicas para o manejo dos recursos aquáticos, para a recuperação de áreas degradadas e para a determinação de áreas prioritárias para a conservação.
Introduction
The occurrence of fish species following patterns along environmental gradients as a result of the availability of resources may be determined starting with studies that focus on knowledge of fish feeding habits. Such studies provide a fair basis for understanding the ecological dynamics of fish populations and other aquatic organisms, besides the chance of making inferences on habitat usage, resources availability and behavioral characteristics (Hahn et al., 2004). In addition, knowledge of feeding habits also allows us to identify factors that affect the distribution and abundance of fish species (Deus & Petrere-Junior, 2003).
Stream fish live together with temporal and spatial variation in their food availability (Matthews, 1998). Lowe-McConnell (1987) states that the ichthyofauna of tropical streams is affected by seasonal changes due to the expansion and contraction of their habitat. Because of local changes in water speed, bottom type, depth and vegetation, these environments assume mosaic-like patterns, greatly increasing the availability of habitats (Luiz et al., 1998). According to Melo et al. (2003), seasonal rainfall variations create and/or eliminate micro-habitats. Heavy rains cause an increase in water flow, greatly augmenting a stream's discharge, carrying organic and inorganic materials and leading to changes in the stream's bed configuration and in the washing out of the biota (Luiz et al., 1998).
Stream bed configuration is preserved by adjacent vegetation, which is denser in the stream head region (Ribeiro & Walter, 1998). This vegetation partially blocks light penetration, hindering autotrophic production, contributing to plant and particulate organic matter (Vannote et al., 1980). As the stream grows, the bed-derived material becomes less important, significantly magnifying autochthonous primary production (Vannote et al., 1980).
These changes along the water course are important for determining the community's functional organization. At the stream heads, fish primarily depend on allochthonous material which derives from the surrounding vegetation (Lowe-McConnell, 1987). This material (such as leaves, branches and trunks) also affect autochthonous resources (which comes from inside the stream) creating microhabitats that bear young insect forms and other organisms as constituents of the basic autochthonous fish feeding resources (Russo et al., 2002).
Fish feeding habits represent an integration between feeding preferences and food availability and accessibility (Angermeier & Karr, 1984). Existing information on fish feeding preferences in tropical streams indicates that there is a lack of specialization or that specialization is rare and reversible (Lowe-McConnell, 1987).
The Cerrado biome (Brazilian Savanna) is home to the country's headwaters of the main hydrological regions, such as the upper Paraná, where knowledge of the ichthyofauna of its small rivers and streams is increasing (Ferrante et al., 2001; Novaes-Pinto, 1993; Langeani et al., 2007; Langeani et al., 2009). In the last decade, intense environmental degradation of this biome, due to the introduction of exotic species, has affected the biotic and abiotic characteristics of the region's rivers and streams (Novaes-Pinto, 1993).
The upper Paraná hydrographic region is completely within Brazilian territory, including the states of Paraná, São Paulo, Mato Grosso do Sul, Goiás, and Distrito Federal. One of the headwater regions of the upper Paraná basin is located in Brasília National Park, a subbasin of the Bananal stream. This subbasin is completely within the Park, and is a preserved area representative of a Cerrado biome.
The study of the characteristics of fish communities in well-preserved regions (streams, for example) is essential to determine the natural conditions to which each species is submitted, with the least possible anthropic interference, besides the important role such knowledge plays in the conservation of its biodiversity, management of aquatic resources, recovery of degraded areas and the determination of conservation priority areas (Agostinho et al., 2005).
Taking into account the lack of information on fish feeding habits in Cerrado streams and the influence of this biome's peculiar environmental variations on the fish community, the present study aimed at investigating the fish trophic structure in the Bananal stream subbasin by studying their feeding habits.
Material and Methods
Study area
Distrito Federal is located in the Central Brazilian Plateau, in the center of the Cerrado biome. Its high altitude and relief characteristics favor surface water drainage, giving home to the headwaters of three main Brazilian hydrographic regions: Tocantins-Araguaia, São Francisco, and Paraná (Novaes-Pinto, 1993; Ferrante et al., 2001).
This research was conducted in the Bananal stream subbasin belonging to the upper Paraná hydrographic region and fully encompassed by Brasília National Park (BNP). BNP embraces the Cerrado biome and corresponds to the Distrito Federal Integral Protection Conservation Unit (created by Law Act No. 241, passed on September 11, 1961, enhanced by Law No. 11,285, passed on August 3, 2006).
The Cerrado is characterized by dry winters and rainy summers (Ribeiro & Walter, 1998). In the Distrito Federal, there is greater rainfall during the summer (December to April), with some changes over the years (Nimer, 1989).
Sampling design
Fish were collected every three months at seven sites distributed along the Bananal stream subbasin, totaling four collections at each site during ten months (from October 2006 to July 2007). Rainfall corresponding to the sampling period indicates that the months of October 2006 (526.4 mm) and January 2007 (261.5 mm) had the most precipitation (rainy season), while the months of April (50.1 mm) and July (0 mm) 2007 the least precipitation (dry season).
At each site, 30 m long stretches were sampled along the headwaters and principal course of the stream. Sampling sites localization was performed according to the Strahler's hierarchical system (1957) (Table 1 and Figs. 1-2), aiming at including the whole Bananal stream subbasin. Field campaigns were sought for evaluating streams in the two seasons, with collections performed during the rainy (October 2006 and January 2007) and dry (April and July 2007) seasons.
At each sampling site, the stream's width, depth, current speed and vegetation cover upstream, in the middle and downstream, were measured. The width and depth were measured with a tape measure (30 m) and the current speed using flow meter for liquids. The canopy cover was measured with a spherical densiometer. More details can be found in Aquino et al. (2009).
Fish collection
Fish collection was performed in the daytime (from 8:00 am to 18:00 pm) by applying four methods: drag net, sieve, gill net, and fish hook. The combination of different fishing techniques was necessary due to the different habitat characteristics at each site sampled. Thus, we attempted to obtain representative samples of the total ichthyofauna present at each site. Sampling methods were all used until the number of individuals at each site tended toward zero (Malabarba & Reis, 1987).
Collected fish were preserved in the field in 10% formalin solution, and after 48 h were transferred in containers with 70% alcohol (Uieda & Castro, 1999). Each sample was identified, measured and weighed.
Species identification was done using keys for specific groups, aided by specialists in each group. Voucher material was sent to the Coleção Ictiológica da Universidade de Brasília (CIUnB). Some samples were addressed to the Coleção de Peixes do Departamento de Zoologia e Botânica da Universidade Estadual Paulista, São José do Rio Preto (DZSJRP), to confirm identification.
Diet analysis
In the laboratory, the stomach of each sampled fish was removed and preserved in 70% ethanol. Only species with more than five full stomachs were analyzed and their contents identified by means of a stereomicroscope.
To study the diet, the stomach contents were analyzed using the volumetric method proposed by Hyslop (1980). Stomach contents were separated into food item groups, and were identified with the aid of identification keys for aquatic insect groups (Merrit & Cummins, 1996) and terrestrial invertebrates (Borror & Delong, 1969). Each separated group was evenly compressed until reaching a height between 0.5 and 1 mm, and the volume was estimated according to the height and the area occupied on a strip of millimeter paper.
Statistical analysis
Only the species that showed more than five individuals in total were used for statistical analysis. Comparison of the fish species with respect to diet was made using the technique known as non-metric multidimensional scaling (NMDS) analysis, based on the Bray-Curtis similarity matrix (Krebs, 1998), using the values of percentage composition of food items of all sampled individuals of each species (Hyslop, 1980). Percentage composition was based on the items' abundance values measured in volume (cm3). For matrix calculation, data were transformed into log(x+1) as to reduce the importance of more abundant items (Field et al. 1982). NMDS acts directly on the matrix ranking the similarity measurements between the species pairs, which are considered directly proportional to the distance in metric analysis. This analysis result does not assume linear relations (Clarke, 1993). Feeding preferences of the species found in the Bananal stream subbasin were determined using Costello's chart method (1990), modified by Amundsen et al. (1996).
To check whether or not there was variation in the species' diet according to food origin over the seasons, recorded food items were classified into autochthonous (coming from the stream itself) and allochthonous (coming from outside the stream). The relative participation of autochthonous and allochthonous items with respect to the seasons was calculated for each fish species. Analysis was performed with a Χ2 test with a 2 x 2 contingency table (Zar, 1999).
Similarity analysis (ANOSIM) was carried out to check for significant variations in fish diet between the dry and rainy seasons using the same NMDS similarity matrix. The ANOSIM is a multivariate randomization procedure analogous to variance analysis (ANOVA) of a factor (Chapman & Underwood, 1999).
Similarity analysis (ANOSIM) was performed aided by the statistical program R (R Development Core Team, 2007) using the vegan packs (Oksanen et al., 2007). The ordination (NMDS) was aided by the statistical program PC-ORD (McCune & Mefford, 1999). The significance level for all analyses was 0.05.
Results
For characterization of the sites, the environmental variables were collected at each study site and are shown in Table 2. Data refer to values obtained upstream, in the middle and downstream of each site for each variable. Measurements are presented with average ± pattern deviation/shift.
In general, all sites are narrow, well fitted with high vegetation cover and with little depth and width (Fig. 2 and Table 2). Exceptions to this pattern were site 2, which is the deepest one, and site 7, which is wider and deeper since it is of the fourth order. Vegetation cover of site 6 is low, with its surrounding vegetation composed of mid- and small-sized trees and shrubs.
Concerning seasons, the rainy season and the transition from rainy to dry weather showed the highest values in width and depth. Contrarily, the transition from dry to rainy season showed lower values for the same variables. Vegetation cover remained practically unchanged over the seasons at all sites (Table 2).
The highest current speed was found in the dry season, in which the smaller water volume increases current speed in the rapids. Nevertheless, as it can be observed in Table 2 that the standard deviation was high because in this season there were sites where the water flow was practically zero, forming pools along the stream course.
Feeding guilds
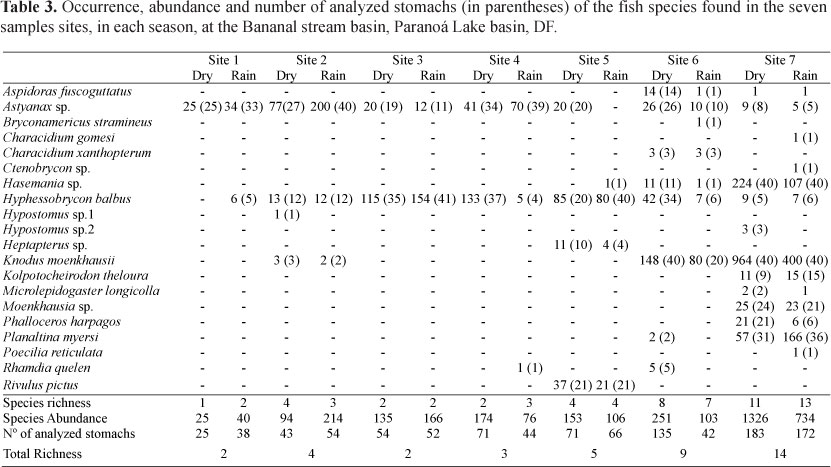
A thousand and fifty stomachs were analyzed, pertaining to the 20 species found in the Bananal stream subbasin. The species number and the number of analyzed stomachs, according to each species and season, are described in Table 3. Of the 20 recorded species, only 13 had stomach content studies, because they each had more than 5 stomachs available for analysis.
In accordance with the stomach analysis of the species found in the Bananal stream subbasin, 36 types of food were consumed, of which 24 were autochthonous, 8 were allochthonous, and 4 were of unknown origin (Table 4).
The non-metric multidimensional scaling analysis of the species' diet, irrespective of the season, showed a separation into four large groups (Fig. 3). The first two axes accounted for 86% of the data (R2 = 0.86). These groups reflect similarity in species' diet according to the dominant food items in the diet. The closer they are in the ordination, the greater the similarity is among species. Thus, the ordination revealed the separation of the species into four trophic guilds: detritivorous, omnivorous, invertivorous and piscivorous. The omnivorous could be separated into two groups: one with greater plant matter consumption (omnivorous tending toward herbivory), and the other with greater consumption of animal fragments (omnivorous tending toward invertivory).
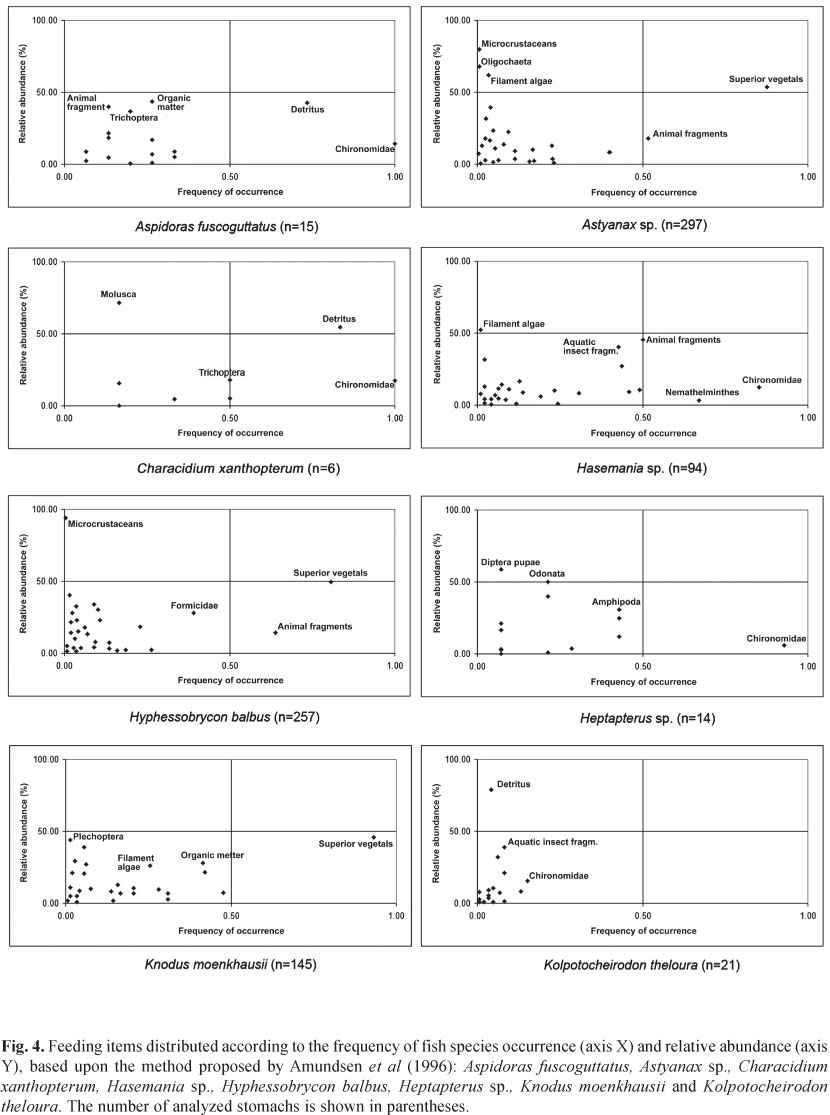
The gap found in the ordination is supported by the results of the food preference charts (Figs. 4-5), where group A represents the omnivorous species tending toward herbivory and the species in group B are the omnivorous tending toward invertivory, with various categories located at the bottom of the chart. The most representative food items of that group were animal fragments, belonging mainly to the families Formicidae and Chironomidae. The invertivorous group (C group) was formed by Rivulus pictus, Kolpotocheirodon theloura and Aspidoras fuscoguttatus, where aquatic invertebrates were the main food type. Group D is formed by Phalloceros harpagos and Characidium xanthopterum, which showed detritus as the main food item, followed by aquatic invertebrates. Heptapterus sp. and Rhamdia quelen species are separated from the others, and their diet displays different patterns. Heptapterus sp. consumed mostly aquatic invertebrates, while R. quelen ate fish, higher plant material, and Oligochaeta specimens. Even though Heptapterus sp. is classified as an invertivore, its location in the ordination remained separated from others of the same guild. This happened because Heptapterus sp. ingested Diptera, Odonata, and Amphipoda pupae, which did not occur in such high abundance and frequency with the other insectivorous species.
Occurrence frequency charts and relative abundance values of each species' prey are described in Figs. 4 and 5 and demonstrate the feeding preferences.
Characidium xanthopterum and Phalloceros harpagos were classified as detritivorous. Omnivory was attributed to six species that showed a great variety of food items with no apparent preference for any category, occupying more than one trophic level (Astyanax sp., Hasemania sp., Hyphessobrycon balbus, Knodus moenkhausii, Moenkhausia sp., and Planaltina myersi). The latter group could be separated into two subgroups: omnivorous tending toward herbivory (Astyanax sp., Hyphessobrycon balbus, and Knodus moenkhausii) and omnivorous tending toward invertivory (Hasemania sp., Moenkhausia sp., and Planaltina myersi).
The third guild identified was invertivorous that ate mainly invertebrates, especially aquatic ones. Aspidoras fucoguttatus, Heptapterus sp., Kolpotocheirodon theloura, Rhamdia quelen, and Rivulus pictus belong to this guild. Aspidoras fucoguttatus, even though having detritus as the most frequent and abundant food item, displayed a high frequency of Chironomidae. Heptapterus sp. consumed large amounts of Diptera pupae and aquatic invertebrate larvae. Kolpotocheirodon theloura consumed a large variety of low frequency prey. Large amounts of Chironomidae larvae and other aquatic insects were consumed by Rivulus pictus, a fact that qualifies this species as quasi specialist on aquatic invertebrates.
The only species that displayed piscivorous habits was Rhamdia quelen; however, fragments of terrestrial insects and oligochaetes were also present in its diet, representing 39.16% of the consumed total, while fish accounted for 40.24% of the composition (Table 4). Due to the fish representativeness in its diet as a unique category, against the ensemble of terrestrial insect fragments and oligochaetes, this species was classified as piscivorous.
Allochthonous and autochthonous contributions
Taking the fish community as a whole and considering the abundance of items, 69% of all resources consumed by the species were allochthonous. The contribution of allochthonous and autochthonous prey to the samples' diet, according to the season (Fig. 6), was significant for Aspidoras fuscoguttatus (Χ2ð ð= 26.3, p < 0.001), Astyanax sp. (Χ2 = 11.8, p< 0.001), Characidium xanthopterum (Χ2 = 8.53, p< 0.004), Hyphessobrycon balbus (Χ2= 8.77, p< 0.003), Kolpotocheirodon theloura (Χ2 = 17.9, p< 0.001), Moenkhausia sp. (Χ2 = 13.3, p < 0.001), Phalloceros harpagos (Χ2 = 16.9, p< 0.001), Rhamdia quelen (Χ2 = 139.1 and p<< 0.001), and Rivulus pictus (Χ2 = 7.2, p= 0.007). As expected, four of these species (Aspidoras fuscoguttatus, Kolpotocheirodon theloura, Moenkhausia sp., and Phalloceros harpagos) did not show an increase in the consumption of allochthonous material in the rainy season.
Temporal variation
Variation in the consumption of food items between seasons was not significant (ANOSIM, R = -0,043, and p = 0.717). No significant variation was observed in between sites (ANOSIM, R = 0.06, and p = 0.364).
Discussion
Feeding guilds
The four guilds found did not occur at all sites, and they grew when getting away from the headwater region. According to Angermeier & Karr (1984), patterns explain that increases are correlated with the increase in food availability.
Omnivorous species showed wider distributions, and those tending toward herbivory were present at all sites. Because this is the case of a well-preserved area, resources of plant origin are abundant everywhere. In this guild, a wide spectrum of food items was consumed, with low abundances and frequencies, making classification impossible in terms of the dominance of a unique food category. These characteristics indicate low selectivity and much feeding opportunism (Mazzoni & Rezende, 2003). According to Abelha et al. (2001), there are few tropical species with specialized feeding habits, the majority of which are generalists, with a certain degree of preference but using the resources available in the environment (Pereira et al., 2007).
Higher vegetation was consumed in considerable amounts, compared with other items consumed by omnivorous tending toward herbivory. Among them, the genus Astyanax includes some of the most common species, widely distributed and with less defined taxonomical limits (Abelha et al., 2006). In general, there is the consensus that this genus comprises generalists with very variable diet (Zaret & Rand, 1971; Costa, 1987; Castro & Casatti, 1997; Luiz et al., 1998; Castro et al., 2003; Castro et al., 2004; Motta & Uieda, 2004; Bennemann et al., 2005; Oliveira & Bennemann, 2005; Ferreira & Casatti, 2006; Luz-Agostinho et al., 2006). The diet composition can vary according to the environmental characteristics, as well as food availability, which may be different from one region to another.
Invertivorous and omnivorous tending toward invertivory were found at sites very far from the headwaters, growing according to the stream's order. The increase in the number of insects consumed by fish species in upstream-downstream direction is in accordance with the predictions of the Continuum River Theory (Vannote et al., 1980), since larger amounts of organic matter are accumulated in the lower stream areas, making it easier for the settling of insects in the substrate. Omnivorous species tending toward invertivory showed a large variety of consumed items, where animal fragments were the most frequent and abundant in their diet. Animal fragments included remains of semi-digested insects and exoskeleton pieces of terrestrial and aquatic insects that could not be identified (Vilella et al., 2002; Melo et al., 2004). Catching this sort of food is considered an advantage because these remains maintain the insect's nutritional value as a whole (Melo et al., 2004). In the case of the Bananal stream species, the presence and consumption of several terrestrial and aquatic invertebrate categories explains the high frequency and abundance values of animal fragments. Thus, these species continue to be classified as omnivorous, even tough, in general, they prefer both terrestrial and aquatic insects.
The invertivore guild comprises species that feed on both terrestrial and aquatic insects, as well as other invertebrates, such as molluscs. They form the largest trophic guild and possibly the most diverse (Goldstein & Simon, 1998). Species specifically invertivorous of the Bananal stream subbasin were Aspidoras fuscoguttatus, Heptapterus sp., Kolpotocheirodon theloura, and Rivulus pictus. The invertivorous feeding habit of Aspidoras fuscoguttatus is in agreement with that found in the literature, involving mostly aquatic insects (Melo et al., 2004; Ferreira & Casatti, 2006). Studies on the feeding habits of species the genus Heptapterus revealed that they were invertivorous (Sabino & Castro, 1990; Castro & Casatti, 1997; Uieda et al., 1997; Luiz et al., 1998; Esteves & Lobón-Cerviá, 2001; Castro et al., 2003; Ferreira & Casatti, 2006), with sporadic high consumption of aquatic invertebrates (Melo et al., 2004).
There are no studies on newer species concerning their biology and ecology. Little is known about the biology of Kolpotocheirodon theloura Malabarba & Weitzman, 2000, probably because it is a recently described species. The individuals sampled showed a diet composed of fragments of aquatic invertebrates, Chironomidae larvae and detritus. Rivulus pictus consumes mainly aquatic invertebrates, as found by Melo et al. (2004). Shibatta & Bennemann (2003), when studying the feeding plasticity of the same species in a small lagoon in Distrito Federal, recorded omnivory as its feeding habit tending toward planktivory. The large plankton abundance in lacustrine environments favors this feeding preference, unlike in riverine environments, which have little particulate organic matter leading to scarce primary production (Russo et al., 2002).
According to the theory proposed by Vannote et al. (1980), the detritivore guild is expected to become more diverse when far away from the stream's head, which is usually distant from the headwaters. In the case of the study area, detritivorous were present only at the sites very distant from the headwaters. When far from its headwaters, the decrease in current speed favors detritus deposition in the substrate, allowing the occurrence of detritivorous species (Vannote et al., 1980; Melo et al., 2004). Plant matter transport, originally from the riparian vegetation along the stream, restricts its use by detritivorous fish in the headwater area, since it has not yet transformed into detritus. The guild distribution pattern found in the Bananal stream follows the pattern proposed by Luiz et al. (1998) and Garutti (1988), who reported omnivores' predominance in the headwater region and detritivorous near the mouth in streams of the high Paraná basin.
In the case of Characidium xanthopterum, detritus was the main feeding item, followed by aquatic invertebrates. Studies on fish feeding habits of some species of the same genus (C. schubarti, C. lanei, C. zebra, C. gomesi, and C. pterostictum) in Brazilian streams demonstrated that their nurture is based almost exclusively on aquatic insect larvae (Uieda, 1984; Costa, 1987; Sabino & Castro, 1990; Castro & Casatti, 1997; Uieda et al., 1997; Aranha et al., 1998; Esteves & Lobón-Ceviá, 2001; Castro et al., 2004; Motta & Uieda, 2004; Barreto & Aranha, 2006; Luz-Agostinho et al., 2006). This variation in the results obtained is related to the characteristics of the environment in which specimens were collected. All Characidium xanthopterum samples belonging to the Bananal stream subbasin were collected at site 6, which had the smallest percentage of vegetation cover along with high current speed and turbidity. These characteristics can affect the abundance of aquatic insects barring their settling and permanence, because under such conditions they do not find substrate for attachment (leaves and branches). The species' feeding plasticity allows the exchange of preferential resources according to their abundance, taking detritus as an alternative food source as to ensure their survival at the site.
The only species considered piscivorous was Rhamdia quelen, which had almost 40% of its diet composed of fish or parts of fish. This guild eats mostly fish and it is not strictly piscivorous (Goldstein & Simon, 1998). High abundance of oligochaetes and fragments of terrestrial insects indicate some bias towards invertivory, moderately resembling the feeding habits of Heptapterus sp., which belong to the same family. Luz-Agostinho et al. (2006) found that this species is piscivorous, but occasionally and to a lesser degree complements its diet with plants and other invertebrates. Loureiro-Crippa & Hahn (2006) endorse that it is piscivorous. The classification of Rhamdia quelen into trophic guilds varies in the literature, and it can be found as an insectivore, consuming terrestrial insects followed by aquatic insect larvae (Castro & Casatti, 1997; Casatti, 2002; Ferreira & Casatti, 2006), and as a generalist, tending to consume animal items (Deus & Petrere-Junior, 2003; Gomiero et al., 2007).
Allochthonous and autochthonous contributions
In the current study, considering the nine species that showed significant feeding variations between seasons, there was higher consumption of autochthonous items. Nevertheless, when taking into account the totality of individuals of all sampled species, allochthonous items were more important. The higher representativeness of allochthonous items was due to the fact that the streams sampled showed headwater characteristics, with low primary productivity. Thus, there is more abundance of resources originating from riparian vegetation. Studies in a well-preserved stream belonging to the Mata Atlântica indicate that, despite that many species use autochthonous resources, when one considers its abundance, allochthonous resources grow in importance (Esteves & Lobón-Ceviá, 2001). Oliveira & Bennemann (2005) found that in streams with better preserved sites, the consumption of allochthonous items predominates.
It is worth highlighting that the environmental characteristics and the composition of fish communities are responsible for that variation in the consumption of items of different origin. However, there is a consensus that the production of autochthonous resources depends on the input of allochthonous resources (e.g., leaves, branches and seeds), which serve as a basis for the different aquatic invertebrate chains (Esteves & Aranha, 1999; Russo et al., 2002).
The season of the year affects the availability of autochthonous and allochthonous food resources. In the rainy season, the rain and the erosion wash out many terrestrial invertebrates toward the streams (Angermeier & Karr, 1984; Uieda & Kikuchi, 1995). In the case of the fish species found in the Bananal stream subbasin, the increase in the consumption of allochthonous material during the rainy period was observed only for five out of nine of the species that showed significant variations in the item's origin and the year's season (Astyanax sp., Characidium xanthopterum, Hyphessobrycon balbus, Rhamdia quelen, and Rivulus pictus). Of these, Rivulus pictus occurred only at site 5, where the stream is relatively narrow and has a dense vegetation cover. Its main diet was composed of aquatic insects. Thus, during the rainy season, the increase in water volume and the wash out of benthic communities yielded higher consumption of allochthonous material brought by rains.
The four other species (Aspidoras fuscoguttatus, Kolpotocheirodon theloura, Moenkhausia sp. and Phalloceros harpagos) showed significant variation in the consumption of items of different origin; but, as expected, they did not ingest high amounts of allochthonous items during the rainy season. This is because the species occurred at sites 6 (Aspidoras fuscoguttatus) and 7 (Kolpotocheirodon theloura, Moenkhausia sp., and Phalloceros harpagos), where the vegetation cover is less dense and the stream is wider. Site 7 is of the fourth order and represents the widest site with the smallest vegetation cover. In the dry season, the higher consumption of allochthonous material is due to the slow current speed, which allows the gathering of leaves and branches at the stream's banks, making them available for the fish community. In the rainy season the current is usually stronger and is prone to carry the inner habitat structures downstream, hindering their accumulation in the deposition areas. The relative importance of allochthonous food increases with the increase of the stream's size, while the vegetation cover diminishes (Angermeier & Karr, 1984). It is also worthwhile noting climatic effects, such as air turbulence which may be responsible for higher abundances of terrestrial organisms in open areas (Uieda & Kikuchi, 1995).
When evaluating a fish community´s diet, the preferential use of allochthonous resources varies according to the environment characteristics and the trophic niche occupied by the species under study. Variations found by different authors may be related to the sampling methods used, the stream's hydrological regime and the sampling period (Moyle & Senanayake, 1984).
Temporal variation
Despite the strong seasonality of the Cerrado biome, with one dry season and other rainy, the fish community in the Bananal stream subbasin did not show seasonal variation in its diet. This indicates that the hydrological variation over the year did not cause changes in prey availability along the year as to produce changes in the diet. Food items remained available throughout the year in sufficient amounts as not to induce variations in the feeding diet of the species inhabiting that environment. The literature shows that depending on the rain regime and on the characteristics of each study site, it is possible or not to detect diet variations according to seasonality (Esteves & Aranha, 1999). Esteves & Lobón-Cerviá (2001) found feeding regularity in the fish community of a Mata Atlântica stream over the year, with no significant differences between seasons. Seasonal variations on the fish diet are also found in the literature. Motta & Uieda (2004) determined those variations by studying the feeding habits of Astyanax scabripinnis and Characidium schubarti. The higher primary productivity and the greater abundance of Diptera larvae in the dry season assured its consumption in that year period. Mazzoni & Rezende (2003) found in the diet of Deuterodon sp. an alternation between items of plant and animal origin over the seasons. Diet differences between the seasons are due to changes in the proportions of the most important food items consumed by fish (Balcombe et al., 2005). Such changes can result in new feeding strategies, from generalist in the summer to specialists in the winter, according to the resources availability, as reported by Deus & Petrere-Júnior (2003).
Spatial and seasonal differences in the species' diet are related to temporal alterations, peculiar to each environment (Barreto & Aranha, 2006). The sites sampled showed headwater region characteristics, with high vegetation cover formed by well-preserved riparian vegetation. This guarantees the flow of allochthonous material for maintenance of the benthic community, supplying the organic matter necessary for primary production. Riparian vegetation floristic composition is characterized by perennial vegetation; i.e., non-deciduous vegetation cover that does not lose leaves in the dry season. Its proximity to the water courses guarantees its water supply throughout the whole year and its height and shape guarantee high relative humidity, even in the driest times of the year (Ribeiro & Walter, 1998). These characteristics of the riparian vegetation may mask the seasons' effect on the fish diet, since they keep the availability of resources practically constant throughout the year. Environmental changes concerning the rainy and dry seasons are more perceptible in the areas adjacent to the riparian vegetation. Besides, the region's high degree of preservation can also serve as contention means against hydric effects, hindering the perception of seasonal variations in the diet of the fish species present. Specialist species can become generalists whenever the availability of a specific resource diminishes (Abelha et al., 2001). However, some morphological adaptations can impose barriers to changes in their diet, halting the exploitation of diverse resources (Peres-Neto, 1999). These specificities and the dependence on the inflow of allochthonous resources, highlight the necessity of acquiring basic information concerning the species' biology (Esteves & Aranha, 1999). The dependence of some species on the resources originally from the riparian vegetation suggests that alterations in the structure and composition of the vegetation cover may cause serious impacts on the integrity of stream fish communities (Angermeier & Karr, 1984).
Ontogenetic, seasonal, spatial and individual changes in the diet make it difficult to classify a fish community into consistent trophic guilds, together with identifying patterns that allow reliable comparison between ecosystems. Long-term studies that seek to understand the current processes among fish species and their food source will significantly contribute to the management of aquatic environments and to the determination of priority conservation areas.
The ichthyofauna taxocenose of the Bananal stream subbasin is maintained by a great variety of resources where the ones originally from the riparian vegetation are essential for the maintenance of most of the fish biomass. The lack of seasonal variation and the predominance of allochthonous items in the diet are probably linked to the presence of riparian vegetation which acts as a buffer and resource supply zones for the aquatic biota. The preservation of the riparian vegetation guarantees the supply of resources for lotic environments, besides serving as a contention barrier for sudden environmental changes. Studies on preserved areas are essential for the identification of the natural conditions to which the communities are submitted. By doing so, it will be possible to evaluate anthropic actions on the fish communities and to formulate management plans aimed at the recovery of degraded areas.
List of voucher specimens: Voucher material of the 20 fish species collected in the Bananal stream subbasin, Paranoá Lake Basin, DF, registered in the Coleção Ictiológica da Universidade de Brasília (CIUnB) and in the Coleção de Peixes do Departamento de Zoologia e Botânica da Universidade Estadual Paulista, São José do Rio Preto (DZSJRP): Aspidoras fuscoguttatus CIUnB 14, 16, 173, 180, 220, DZSJRP 10813; Astyanax sp. CIUnB 1-3, 6, 7, 12, 50-52, 54, 55, 59, 60, 63, 161, 162, 165-168, 178, 183, 209-212, 218, 228, 312, 318, DZSJRP 10824; Bryconamericus stramineus CIUnB 61; Characidium gomesi CIUnB 15, Characidium xanthopterum CIUnB 13, 221, DZSJRP 10812; Ctenobrycon sp. CIUnB 64, Hasemania sp. CIUnB 22, 67, 93, 174, 185, 219, 223, 320, DZSJRP 10801, 01829; Hyphessobrycon balbus CIUnB 5, 9, 23, 53, 58, 65, 170, 215, 310, 311, 313, 316, 317, 319, 321, 326-330, 334-336, DZSJRP 10825; Hypostomus sp.1 CIUnB 164; Hypostomus sp.2 CIUnB 230; Heptapterus sp. CIUnB 11, 57, 171, 213, DZSJRP 10803, 01804, 01815; Knodus moenkhausii CIUnB 4, 20, 62, 68, 163, 177, 186, 187, 216, 222, DZSJRP 01808, 01828, 01830; Kolpotocheirodon theloura CIUnB 18, 179, 226, DZSJRP 10831; Microlepidogaster longicolla CIUnB 181, 229, DZSJRP 10827; Moenkhausia sp. CIUnB 21, 66, 188, 225, DZSJRP 10799, 10800, 10805; Phalloceros harpagos CIUnB 24, 182, 227, DZSJRP 10826; Planaltina myersi CIUnB 19, 69, 175, 184, 224, DZSJRP 10802; Poecilia reticulata CIUnB 17; Rhamdia quelen CIUnB 8, 172, 217; Rivulus pictus CIUnB 10, 56, 169, 214, DZSJRP 10814.
Acknowledgments
The authors thank the following people and agencies: Lilian Casatti for her assistance in the teaching methods indispensable for the realization of this work; Francisco Langeani, for helping with fish identification; Santos Balbino for his guidance in the field; Ricardo Pavan, José Braz, Thiago Couto, Bernardo Buta, Filipe Fortes, Juliana Fonseca, Carlos de Melo, Guthenberg Falcon, Paula Jota, and Flávio Cardoso for their assistance in collecting fish samples; the Post-Graduation Program in Ecology of the University of Brasília for financial support; the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), for granting a Master's scholarship to MS and PPUA; the Brasília National Park for logistic support; and finally, the Brazilian Environmental Institute (IBAMA), for providing a specimen collection license (Process No. 02001,003287/06-49 and license No. 187/2006).
Literature Cited
Submitted February 7, 2010
Accepted July 7, 2011
- Abelha, M. C. F., A. A. Agostinho & E. Gourlat. 2001. Plasticidade trófica em peixes de água doce. Acta Scientiarum, 23: 425-434.
- Abelha, M. C. F., E. Goulart, E. A. L. Kashiwaqui & M. R. Silva. 2006. Astyanax paranae Eigenmann, 1914 (Characiformes: Characidae) in the Alagados Reservoir, Paraná, Brazil: diet composition and variation. Neotropical Ichthyology, 4: 349-356.
- Agostinho, A. A., S. M. Thomaz & L. C. Gomes. 2005. Conservação da biodiversidade em águas continentais do Brasil. Megadiversidade, 1: 70-78.
- Amundsen, P. A., H. M. Gabler & F. J. Staldvik. 1996. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. Journal of Fish Biology, 48: 607-614.
- Angermeier, P. L. & J. R. Karr. 1984. Fish communities along environmental gradients in a system of tropical streams. Enviromental Biology of Fishes, 9: 117-135.
- Aquino, P.P.U, M. Schneider, M. J. Martins-Silva, C. P. Fonseca, H. B. Arakawa & D. R. Cavalcante. 2009. Ictiofauna dos córregos do Parque Nacional de Brasília, bacia do Alto Rio Paraná, Distrito Federal, Brasil Central. Biota Neotropica, 9(1): 217-230.
- Aranha, J. M. R., D. F. Takeuti & T. M. Yoshimura. 1998. Habitat use and food partitioning of the fishes in a coastal stream of Atlantic Forest, Brazil. Revista de Biologia Tropical, 46(4): 951-959.
- Balcombe, S. R., S. E. Bunn, F. J. McKenzie-Smith & P. M. Davies. 2005. Variability of fish diets between dry and flood periods in an arid zone floodplain river. Journal of Fish Biology, 67: 1552-1567.
- Barreto, A. P. & L. M. R. Aranha. 2006. Alimentação de quatro espécies de Characiformes de um riacho da Floresta Atlântica, Guaraqueçaba, Paraná, Brasil. Revista Brasileira de Zoologia, 23: 779-788.
- Bennemann, S. T., A. M. Gealh, M. L. Orsi & M. L. Souza. 2005. Ocorrência e ecologia trófica de quatro espécies de Astyanax (Characidae) em diferentes rios da bacia do rio Tabagi, Paraná, Brasil. Iheringia, 95: 247-254.
- Borror, D. J. & D. M. Delong. 1969. Introdução ao Estudo dos Insetos. São Paulo, Edgard Blücher LTDA, 653p.
- Casatti, L. 2002. Alimentação dos peixes em um riacho do Parque Estadual Morro do Diabo, Bacia do Alto Rio Paraná, sudoeste do Brasil. Biota Neotropica, 2(2): 1-14.
- Castro, R. M. C. & L. Casatti. 1997. The fish fauna from a small Forest stream of the upper Paraná River basin, southeastern Brazil. Ichthyological Exploration of Freshwaters, 7: 337-352.
- Castro, R. M. C., L. Casatti, H. F. Santos, K. M. Ferreira, A. C. Ribeiro, R. C. Benine, G. Z. P. Dardis, A. L. A. Melo, R. Stopliglia, T. X. Abreu, F. A. Bockmann, M. Carvalho, F. Z. Gibran & F. C. T. Lima. 2003. Estrutura e composição da ictiofauna de riachos do Rio Paranapanema, Sudeste e Sul do Brasil. Biota Neotropica, 3(1): 1-31.
- Castro, R. M. C., L. Casatti, H. F. Santos, A. L. A. Melo, L. S. F. Martins, K. M. Ferreira, F. Z. Gibran, R. C. Benine, M. Carvalho, A. C. Ribeiro, T. X. Abreu, F. A. Bockmann, G. Z. Pelição, R. Stopliglia & F. Langeani. 2004. Estrutura e composição da ictiofauna de riachos da bacia do Rio Grande no estado de São Paulo, sudeste do Brasil. Biota Neotropica, 4(1) 1-39.
- Chapman, M. G. & A. J. Underwood. 1999. Ecological patterns in multivariate assemblages: information and interpretation of negative values in Anosim tests. Marine Ecology Progress Series, 180: 257-265.
- Clarke, K. R. 1993. Non-parametric multivariate analyses of changes in community structure. Australian Journal of Ecology, 18: 117-143.
- Costa, W. J. E. M. 1987. Feeding habits of a fish community in a tropical coastal stream, Rio Mato Grosso, Brazil. Studies of Neotropical Fauna and Environment, 22: 145-153.
- Costello, M. J. 1990. Predator feeding strategy and prey importance: a new graphical analysis. Journal of Fish Biology, 36: 261-263.
- Deus, C. P. & M. Petrere-Junior. 2003. Seasonal diet shift of seven fish species in an Atlantic rainforest stream in southeastern Brazil. Brazilian Journal of Biology, 63: 579-588.
- Esteves, K. E. & M. R. Aranha. 1999. Ecologia trófica de peixes de riachos. Pp. 157-182. In: Caramaschi, E. P., R. Mazzoni & P. R. Peres-Neto (Eds). Ecologia de peixes de riachos. Série Oecologia Brasiliensis, vol. VI. Rio de Janeiro, PPGE-UFRJ, Brasil, 260p.
- Esteves, K. E. & J. Lobón-Ceviá. 2001. Composition and trophic structure of a fish community of a clear water Atlantic rainforest stream in southeastern Brazil. Environmental Biology of Fishes, 62: 429-440.
- Ferrante, J. E. T., L. Rancan & P. B. Netto. 2001. Meio Físico. Pp. 45-55. In: Fonseca, F. O. (Org.). Olhares sobre o lago Paranoá. Brasília, Secretaria de Meio Ambiente e Recursos Hídricos (SEMARH), 425p.
- Ferreira, C. P. & L. Casatti. 2006. Integridade biótica de um córrego na bacia do Alto Rio Paraná avaliada por meio da comunidade de peixes. Biota Neotropica, 6: 1-25.
- Field, J. G., K. R. Clarke & R. M. Warwick. 1982. A practical strategy for analyzing multispecies distribution patterns. Marine Ecology Progress Series, 8: 37-52.
- Garutti, V. 1988. Distribuição longitudinal da ictiofauna em um córrego da Região Noroeste do Estado de São Paulo, Bacia do Rio Paraná. Revista Brasileira de Biologia, 48: 747-759.
- Goldstein, R. M. & T. P. Simon. 1998. Toward a united definition of guild structure for feeding ecology of north American freshwater fishes. Pp. 123-138. In: Simon, P.T. (Ed.). Assessing the sustainability and biological integrity of water resource using fish communities. Boca Raton, CRC Press LLC, 671p.
- Gomiero, L. M., U. P. Souza & F. M. S. Braga. 2007. Reprodução e alimentação de Rhamdia quelen (Quoy & Gaimard, 1824) em rios do Núcleo Santa Virgínia, Parque Estadual da Serra do Mar, São Paulo, SP. Biota Neotropica, 7(3): 1-8.
- Hahn, N. S., R. Fugi & F. Adrian. 2004. Trophic ecology of the fish assemblages. Pp. 247-269. In: Thomaz, S. M., A. A. Agostinho & N. S. Hahn (Eds). The upper Paraná River and its Floodplain: physical aspects, ecology and conservation. Leiden, The Netherlands, Backhuys Publishers, 393p.
- Hyslop, E. J. 1980. Stomach contents analysis - a review of methods and their application. Journal of Fish Biology, 17: 411-429.
- Krebs, C.J. 1998. Ecological Methodology. New York: Harper Collins. 654 p.
- Langeani, F., R. M. C. Castro, O. T. Oyakawa, O. A. Shibatta, C. S. Pavanelli & L. Casatti. 2007. Diversidade da ictiofauna do Alto Rio Paraná: composição atual e perspectivas futuras. Biota Neotropica, 7: 1-17.
- Langeani, F., P. A. Buckup, L. R. Malabarba, L. H. R. Py-Daniel, C. A. S. Lucena, R. S. Rosa, J. A. S Zuanon, Z. M. S. Lucena, M. R. Britto, O. T. Oyakawa & G. Gomes-Filho. 2009. Peixes de água doce. Pp. 211-230. In: Rocha, R.M. & W. A. Boeger (org). Estado da arte e perspectivas para a Zoologia no Brasil. Curitiba, UFPR, 296 p.
- Loureiro-Crippa, V. E. & N. S. Hahn. 2006. Use of food resources by the fish fauna of a small reservoir (rio Jordão, Brazil) before and shortly after its filling. Neotropical Ichthyology, 4: 357-362.
- Lowe-McConnell, R. H. 1987. Ecological studies in tropical fish communities. Cambridge, Cambridge Univ. Press, 382 p.
- Luiz, E. A., A. A. Agostinho, L. C. Gomes & N. S. Hahn. 1998. Ecologia trófica de peixes em dois riachos da Bacia do Rio Paraná. Revista Brasileira de Biologia, 58: 273-285.
- Luz-Agostinho, K. D. G., L. M. Bini, R. Fugi, A. A. Agostinho & H. F., Júlio Jr. 2006. Food spectrum and trophic structure of the ichthyofauna of Corumbá reservoir, Paraná River Basin, Brazil. Neotropical Ichthyology, 4:61-68.
- Malabarba, L. R. & R. E. Reis. 1987. Manual de técnicas para a preparação de coleções zoológicas. Sociedade Brasileira de Zoologia (Campinas), 36:1-14.
- Matthews, W. J. 1998. Patterns in freshwater fish ecology. Massachusetts, Chapman & Hall, Norwell, 756p.
- Mazzoni, R. & C. F. Rezende. 2003. Seasonal diet shift in a tetragonopterinae (Osteichthyes, Characidae) from the Ubatiba river, RJ, Brazil. Brazilian Journal of Biology, 63: 69-74.
- McCune, B. & M. J. Mefford. 1999. Multivariate Analysis of Ecological Data. Version 4.25. MjM Software, Gleneden Beach, Oregon, U.S.A.
- Melo, C. E., F. A. Machado & V. P. Silva. 2003. Diversidade de peixes em um córrego de cerrado no Brasil Central. Brazilian Journal of Ecology, 1-2: 17-23.
- Melo, C. E., F. A. Machado & V. Pinto-Silva. 2004. Feeding habits of fish from a stream in the savana of Central Brazil, Araguaia Basin. Neotropical Ichthyology, 2: 37-44.
- Merritt, R. W. & K. W. Cummins. 1996. An Introduction to the Aquatic Insects of North America. 3Ş edição. Dubuque, Iwoa, Kendall/Hunt Publishing Company, 862p.
- Motta, R. L. & V. S. Uieda. 2004. Dieta de duas espécies de peixes do Ribeirão do Atalho, Itatinga, SP. Revista Brasileira de Zoociências, 6: 191-205.
- Moyle, P. B. & F. R. Senanayake. 1984. Resource partitioning among the fishes of rainforest streams in Sri Lanka. Journal of Zoology, 202: 195-223.
- Nimer, E. 1989. Climatologia do Brasil., Rio de Janeiro Secretaria de Planejamento e Coordenação da Presidência da República e IBGE, 421p.
- Novaes-Pinto, M. 1993. Cerrado: caracterização, ocupação e perspectivas. Brasília, UnB, 681 p.
- Oksanen, J., R. Kindt, P. Legendre, B. O'Hara & M. H. H. Stevens. 2007. Vegan: Community Ecology Package. R package version 1.8-8, http://cran.r-project.org/, http://r-forge.r-project.org/projects/vegan/
- Oliveira, D. C & S. T. Bennemann. 2005. Ictiofauna, recursos alimentares e relações com as interferências antrópicas em um riacho urbano no Sul do Brasil. Biota Neotropica, 5 :1-13.
- Pereira, P. R., C. S. Agostinho, R. J. Oliveira & E. E. Marques. 2007. Trophic guilds of fishes in sandbank habitats of a Neotropical river. Neotropical Ichthyology, 5: 399-404.
- Peres-Neto, P. R. 1999. Alguns métodos e estudos de ecomorfologia de peixes de riachos. Pp. 209-236. In : Caramashi, E. P., R. Mazzoni & P. R. Peres-Neto (eds). Ecologia de Peixes de Riachos. Série Oecologia Brasiliensis, vol. VI. PPGE-UFRJ, Rio de Janeiro, Brasil, 260p.
- R Development Core Team. 2007. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, http://www.R-project.org
- Ribeiro, J. R. & B. M. T. Walter. 1998. Capitulo 3: Fitofisionomias do Bioma Cerrado. In: Sano, S. M. & S. P. Almeida (Eds). Cerrado: Ambiente e Flora. Embrapa: Empresa Brasileira de Pesquisa Agropecuária, Planaltina, DF, 556p.
- Russo, M. R., A. Ferreira & R. M. Dias. 2002. Disponibilidade de invertebrados aquáticos para peixes bentófagos de dois riachos da bacia do rio Iguaçu, Estado do Paraná, Brasil. Acta Scientiarum, 24 : 411-417.
- Sabino, J. & R. M. C. Castro. 1990. Alimentação, período de atividade e distribuição espacial dos peixes de um riacho da Floresta Atlântica (sudoeste do Brasil). Revista Brasileira de Biologia, 50 : 23-36.
- Shibatta, O. & S. T. Bennemann. 2003. Plasticidade alimentar em Rivulus pictus (Osteichthtyes, Cyprinodontiformes, Rivuliidae) de uma lagoa em Brasília, Distrito Federal, Brasil. Revista Brasileira de Zoologia, 20: 615-618.
- Strahler, A. N. 1957. Quantitative analysis of watershed geomorphology. American Geophysical Union Transactions, 38: 913-920.
- Uieda, V. S. 1984. Ocorrência e distribuição dos peixes em um riacho de água doce. Revista Brasileira de Biologia, 44: 203-213.
- Uieda, V. S. & R. Kikuchi. 1995. Entrada de material alóctone (detritos vegetais e invertebrados terrestres) num pequeno curso de água corrente na cuesta de Botucatu, São Paulo. Acta Limnológica Brasiliensia, 7: 103-114.
- Uieda, V. S., P. Buzzato & R. Kikuchi. 1997. Partilha de recursos alimentares em peixes de um riacho de serra do sudeste do Brasil. Academia Brasileira de Ciências, 69: 243-252.
- Uieda, V. S. & R. M. C. Castro. 1999. Coleta e fixação de peixes de riacho. Pp. 01-22. In: Caramaschi, E.P., R. Mazzoni & Peres-Neto, P.R. (Eds.). Ecologia de peixes de riachos. Série Oecologica Brasilenses, vol. VI. PPGE-UFRJ. Rio de Janeiro, Brasil, 260p.
- Vannote, R. L., G. W. Minshall, K. W. Cummins, J. R. Sedell & C. E. Cushing. 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences, 37: 130-137.
- Vilella, F. S., F. G. Becker & S. M. Hartz. 2002. Diet of Astyanax species (Teleostei, Characidae) in an Atlantic Forest river in southern Brazil. Brazilian Archives of Biology and Technology, 45(2): 223-232.
- Zar, J. R. 1999. Biostatistical Analysis. 4 edition. Prentice-Hall Inc. New Jersey, USA. 660 p.
- Zaret, T. M. & A. S. Rand. 1971. Competition in tropical streams fishes: support for the competitive exclusion principle. Ecology, 52: 336-342.
Trophic structure of a fish community in Bananal stream subbasin in Brasília National Park, Cerrado biome (Brazilian Savanna), DF
Publication Dates
-
Publication in this collection
02 Sept 2011 -
Date of issue
2011
History
-
Received
07 Feb 2010 -
Accepted
07 July 2011