Abstract
Objective:
To evaluate the predictive value of epicardial fat thickness (EFT) in CHA2DS2-VASc (congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, previous stroke or transient ischemic attack, vascular disease, age 65-74 years, sex category) score risk groups.
Methods:
A total of 158 consecutive patients (75 females, 83 males, mean age 70.8±6.3 years) admitted routinely for cardiologic control were divided into two groups according to their CHA2DS2-VASc scores (scores 0 and 1 were regarded as low risk, and score ≥2 as high risk). One hundred twenty-five of 158 patients had a high-risk score.
Results:
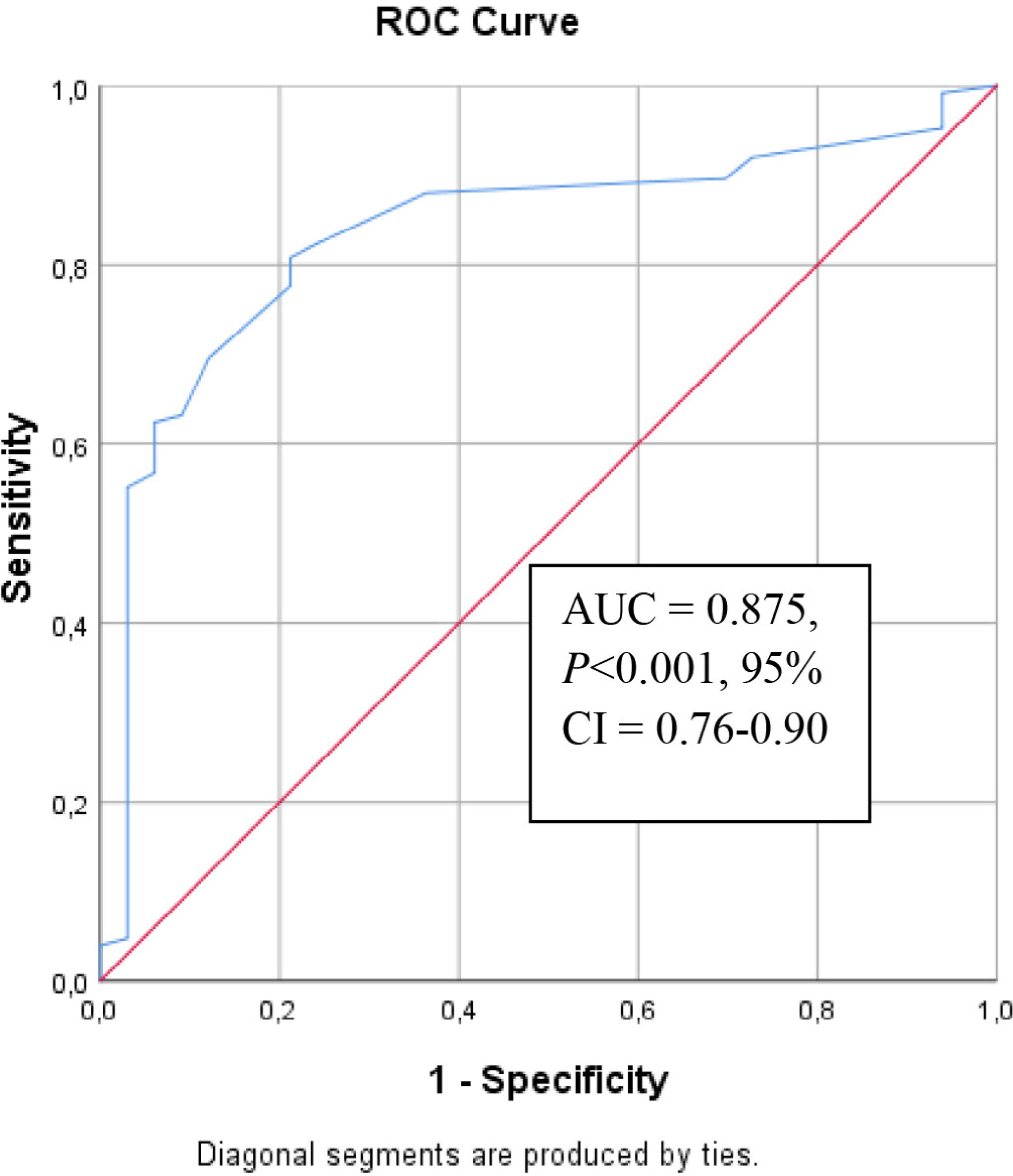
Mean EFT was significantly higher in the high-risk group than in the low-risk group (4.34±0.62 vs. 5.37±1.0; P<0.001). EFT was positively correlated with CHA2DS2-VASc score (r=0.577, P<0.001). According to receiver operating characteristics (ROC) analysis, EFT value of 4.4 mm was found to be predictive of high risk in CHA2DS2-VASc score with 80% of sensitivity and 79% of specificity (C-statistic = 0.875, P<0.001, 95% confidence interval [CI] = 0.76-0.90). And according to multivariate logistic regression analysis, EFT was an independent predictor of high thromboembolic risk in terms of CHA2DS2-VASc score.
Conclusion:
Our findings suggest that echocardiographic EFT measurement could provide additional information on assessing cardiovascular risks, such as thromboembolic events, and individuals with increased EFT should receive more attention to reduce unfavorable cardiovascular risk factors and the development of future cardiovascular events.
Keywords:
Thromboembolism/Prevention & Control; Pericardium; Adipose Tissue; Risk Assessment
INTRODUCTION
Ischemic stroke is a leading cause of death and long-term disability worldwide[11 GBD 2015 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1603-58.]. Control of risk factors is of particular importance for the prevention of cerebrovascular diseases. It is possible to stop progression or prevent these diseases by elimination or modification of modifiable risk factors in the light of treatment goals.
Epicardial adipose tissue (EAT), located between the myocardium and visceral pericardium, has emerged as an important cardiovascular risk predictor, in view of producing and releasing several adipocytokines[22 Iacobellis G, Malavazos AE, Corsi MM. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol. 2011;43(12):1651-4.,33 Sengül C, Özveren O. Epicardial adipose tissue: a review of physiology, pathophysiology, and clinical applications. Anadolu Kardiyol Derg. 2013;13(3):261-5.]. The importance of epicardial fat thickness (EFT) has been shown in recent years. Increased EFT is associated with hypertension, insulin resistance, and thromboembolic processes such as stroke and acute coronary syndrome[44 Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22(12):1311-9.
5 Gul I, Zungur M, Aykan AC, Gokdeniz T, Kalaycioglu E, Turan T, et al. The relationship between GRACE score and epicardial fat thickness in non-STEMI patients. Arq Bras Cardiol. 2016;106(3):194-200.-66 Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.].
The CHA2DS2-VASc risk score is a cheap and easy scoring system which is calculated by assigning 1 point for each: congestive heart failure (ejection fraction [EF] < 40%), hypertension, age between 65 and 74 years, diabetes mellitus, vascular disease (myocardial infarction or peripheral arterial disease), and female sex; and 2 points for: a history of stroke or transient ischemic attack (TIA) and age > 75 years. The CHA2DS2-VASc risk score is used to predict the thromboembolism risk in non-valvular atrial fibrillation (NVAF) patients[77 January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199-267.].
The present study aimed to determine whether EFT is more closely associated with high-risk patients according to the CHA2DS2-VASc risk score.
METHODS
The 158 consecutive patients (75 females, 83 males, mean age 70.8±6.3 years) admitted to the outpatient clinic of the Suleyman Demirel University Hospital, Department of Cardiology, and referred to our echocardiography laboratory due to suspicion of heart disease between June 2014 and May 2015 were enrolled in this prospective study. All patients underwent medical history assessment, physical examination, anthropometric measurements, electrocardiogram, and echocardiographic evaluation. The study was approved by the institutional ethics committee and all patients gave their informed consent. Exclusion criteria were pericardial effusion, poor echocardiographic window, history of chronic renal and liver disease, moderate to severe mitral and aortic regurgitation, moderate to severe mitral and aortic stenosis, malignancy, systemic or pulmonary embolism, chronic hematological diseases, acute or chronic inflammatory disease, autoimmune disease, hyperparathyroidism, hypercalcemia, hyperphosphatemia, and a prosthetic valve. According to CHA2DS2-VASc score, patients were divided into two groups: scores 0 and 1 were regarded as low risk, and score ≥2 as high risk.
Echocardiography
The M-mode, two-dimensional, and Doppler echocardiographic examinations were obtained by an ultrasound machine (Philips iE 33 xMatrix) to assess left atrial (LA) diameter, interventricular septum (IVS) thickness, left ventricular posterior wall (LVPW) thickness, left ventricular end diastolic diameter (LVEDD), left ventricular end systolic diameter (LVESD), and left ventricular EF. LA and left ventricular dimensions and left ventricular EF were measured by M-mode echocardiography in the parasternal long axis view by using the American Echocardiography Society M-mode technique[88 Sahn DJ, DeMaria A, Kisslo J, Weyman A. Recommendations regarding quantitation in M-mode echocardiography: results of a survey of echocardiographic measurements. Circulation. 1978;58(6):1072-83.]. The presence of mitral and aortic insufficiency was evaluated by Doppler color flow mapping. EFT was identified echocardiographically as the echo-free space between the outer wall of the myocardium and the visceral layer of pericardium. EFT was measured at the point on the free wall of the right ventricle along the midline of the ultrasound beam, perpendicular to the aortic annulus at the end of systole[44 Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22(12):1311-9.] (Figure 1). As Iacobellis et al.[44 Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22(12):1311-9.] suggested, epicardial fat is best measured at end-systole, because it is compressed during diastole. The average value of three cardiac cycles was determined as EFT.
Blood Sampling
Blood samples were drawn from the antecubital vein by careful venipuncture in a 21 G sterile syringe without stasis at 08.00-10.00 AM after a fasting period of 12 h. Glucose, creatinine, and lipid profiles were determined by standard methods. Hemogram parameters were measured in a blood sample collected in dipotassium ethylenediaminetetraacetic acid (EDTA) tubes (Vacuette). An automatic blood counter (Beckman-Coulter Co, Miami, FL, USA) was used for whole blood counts.
Statistical Analysis
SPSS software package program, version 16.0, was used in this study's statistical analyses. Categorical variables were expressed as frequency (%) and compared using the χ2 test. A Kolmogorov-Smirnov test was used to test the distribution of numeric variables, and those with normal distribution were expressed as mean ± standard deviation and were compared using the Student's t-test. Data without normal distribution were expressed as median (inter-quartile range [IQR] of 25%-75% percentiles) and were compared using the Mann-Whitney U test. In all statistical analyses, P values <0.05 were considered as statistically significant. Univariate analysis of binary logistic regression was carried out to identify which factors were associated with high risk according to CHA2DS2-VASc risk score. After including each of these potential confounding factors, backward conditional binary logistic regression analysis was performed to estimate the odds ratio (OR) and 95% confidence interval (CI) for high risk according to CHA2DS2-VASc risk score. Receiver operating characteristics (ROC) curve analysis was used to analyze the prognostic value of EFT for high risk according to CHA2DS2-VASc risk score. C-Statistic (area under the curve [AUC]) was presented as a unified estimate of sensitivity and specificity. According to the cut-off value that was obtained by a ROC curve analysis, the study population could be segregated into two groups, as low risk and high risk. The correlations between CHA2DS2-VASc risk score, EFT, and other clinical, laboratory, and echocardiographic parameters were performed with Pearson and Spearman correlation analysis when appropriate.
RESULTS
Baseline clinical features of the study population were summarized in Table 1. Age, female gender, hypertension, and diabetes mellitus were seen more often in high CHA2DS2-VASc score group than in low CHA2DS2-VASc score group. Only β-blocker and clopidogrel usages were significantly higher in the high CHA2DS2-VASc score group. Laboratory findings of the study population were summarized in Table 2. There was no statistically significant difference between the two groups except for fasting glucose (P=0.04). Cholesterol levels were similar between high- and low-risk groups according to CHA2DS2-VASc score. Echocardiographic findings of the study population were summarized in Table 3. LA diameter was significantly higher in patients with high-risk score than in low-risk score subjects in terms of CHA2DS2-VASc score (33±5.6 vs. 36±4.3 mm, respectively; P<0.001). IVS thickness was significantly higher in patients with high risk than in low-risk subjects (10±1.0 vs. 11±1.4 mm, respectively; P<0.001). LVEDD was significantly higher in patients with high risk than in low-risk subjects (44±1.7 vs. 45±3.7 mm, respectively; P<0.001). LVESD was significantly higher in patients with high risk than in low-risk subjects (27±1.7 vs. 28±3.3 mm, respectively; P<0.001). EFT was significantly higher in patients with high risk than in low-risk subjects (4.34±0.62 vs. 5.37±1.0 mm, respectively; P<0.001). Correlation analysis between EFT and CHA2DS2-VASc score with other clinical and echocardiographic parameters was shown in Table 4. EFT was positively correlated with CHA2DS2-VASc score (r=0.577, P<0.001). Also, EFT was positively correlated with age (r=0.520, P<0,001), LA (r=0.264, P<0.001), IVS (r=356, P<0.001), LVESD (r=0.262, P=0.011), and aorta diameter (r=0.22, P<0.001). Negative correlation was found between EFT and left ventricular EF (r=-0.199, P=0.012). CHA2DS2-VASc score was positively correlated with age (r=0.578, P<0,001), LA (r=0.235, P=0.003), IVS (r=386, P<0.001), LVESD (r=0.337, P<0.001), and aorta diameter (r=0.229, P=0.004). Negative correlation was found between CHA2DS2-VASc score and left ventricular EF (r=-0.154, P=0.05). Univariate and multivariate regression analyses results were shown in Table 5. Older age, LA diameter, aorta diameter, left ventricular EF, IVS diameter, and EFT achieved statistical significance in the univariate logistic analysis. Then, multivariate analysis was carried out with these variables; age and EFT were found to be independent predictors of high risk for CHA2DS2-VASc classification. According to ROC analysis, EFT value of 4.4 mm was predictive of high risk of CHA2DS2-VASc score with 80% of sensitivity and 79% of specificity (C-statistic = 0.875, P<0.001, 95 % CI= 0.76-0.90; Figure 2).
Clinical and echocardiographic parameters showing the significant correlation with EFT and CHA2DS2-VASc score.
Receiver operating characteristics (ROC) curve with calculated area under the curve (AUC) and optimal cut-off point for epicardial fat thickness to identify the presence of high risk of CHA2DS2-VASc (congestive heart failure, hypertension, age ≥75 years, diabetes mellitus, previous stroke or transient ischemic attack, vascular disease, age 65-74 years, sex category) score. CI=confidence interval
DISCUSSION
In the present study, we examined EFT in patients with high- and low-risk CHA2DS2-VASc scores. We found out that EFT was significantly higher in patients with high CHA2DS2-VASc score than in those with low CHA2DS2-VASc score and that EFT was positively correlated with CHA2DS2-VASc scores.
EAT is a true visceral fat tissue, deposited around the heart and particularly around the subepicardial coronary vessels. EAT is a complex organ, mainly composed of adipocytes, but it also includes a neuronal network, stromavascular, immune and inflammatory cells, all nourished by a rich microcirculation[22 Iacobellis G, Malavazos AE, Corsi MM. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol. 2011;43(12):1651-4.,33 Sengül C, Özveren O. Epicardial adipose tissue: a review of physiology, pathophysiology, and clinical applications. Anadolu Kardiyol Derg. 2013;13(3):261-5.,99 Mazurek T, Zhang L, Zalewski A, Mannion JD, Diehl JT, Arafat H, et al. Human epicardial adipose tissue is a source of inflammatory mediators. Circulation. 2003;108(20):2460-6.].
EFT is associated with thromboembolic diseases, including cardiovascular and neurovascular diseases[55 Gul I, Zungur M, Aykan AC, Gokdeniz T, Kalaycioglu E, Turan T, et al. The relationship between GRACE score and epicardial fat thickness in non-STEMI patients. Arq Bras Cardiol. 2016;106(3):194-200.,66 Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.]. Akil et al.[66 Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.] showed that EFT was significantly higher in patients with ischemic stroke than in healthy controls. Akdag et al.[1010 Akdag S, Simsek H, Sahin M, Akyol A, Duz R, Babat N. Association of epicardial adipose tissue thickness and inflammation parameters with CHA2DS2-VASASc score in patients with nonvalvular atrial fibrillation. Ther Clin Risk Manag. 2015;11:1675-81.] investigated the association of EFT, inflammatory, and thrombosis parameters with CHA2DS2-VASc score in NVAF patients. They determined that EFT, inflammatory, and thrombosis parameters were associated with the thromboembolic risk exhibited by CHA2DS2-VASc score in NVAF patients. In our study, we investigated the association of EFT with CHA2DS2-VASc score in patients with sinus rhythm. Our results were similar with those from that study. EFT was significantly higher in high CHA2DS2-VASc score than in low CHA2DS2-VASc score among patients with sinus rhythm. EAT is considered an endocrine and metabolically active organ. It is a source of several bioactive molecules that can influence the myocardium and coronary arteries[1111 Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nature Clin Pract Cardiovasc Med. 2005;2(10):536-43.]. Epicardial fat expresses and secretes a number of cytokines, pro- and anti-inflammatory adipokines, vasoactive factors, and growth factors[1111 Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nature Clin Pract Cardiovasc Med. 2005;2(10):536-43.,1212 Iacobellis G, Bianco AC. Epicardial adipose tissue: emerging physiological, pathophysiological and clinical features. Trends Endocrinol Metab. 2011;22(11):450-7.]. Accordingly, inflammation appears to play an important role in thromboembolic events, such as acute coronary syndrome and stroke[1313 Avanzas P, Arroyo-Espliguero R, Cosín-Sales J, Aldama G, Pizzi C, Quiles J, et al. Markers of inflammation and multiple complex stenoses (pancoronary plaque vulnerability) in patients with non-ST segment elevation acute coronary syndromes. Heart. 2004;90(8):847-52.,1414 Anrather J, Iadecola C. Inflammation and stroke: an overview. Neurotherapeutics. 2016;13(4):661-70.]. As a result, increased inflammatory mediators from EAT may have an important role in the pathogenesis of stroke and atherosclerosis. As also mentioned before, EAT is strongly associated with the pathogenesis of atherosclerosis due to sharing the same risk factors. In the present study, we have shown that high thromboembolic risk according to CHA2DS2-VASc score was positively correlated with EFT.
It has been shown that EAT is related to cardiovascular risk factors[1515 Vos AM, Prokop M, Roos CJ, Meijs MF, van der Schouw YT, Rutten A, et al. Peri-coronary epicardial adipose tissue is related to cardiovascular risk factors and coronary artery calcification in post-menopausal women. Eur Heart J. 2007;29(6):777-83.]. The studies using echocardiography to measure EFT on the right ventricle showed relations with waist circumference and left ventricular measurements[1515 Vos AM, Prokop M, Roos CJ, Meijs MF, van der Schouw YT, Rutten A, et al. Peri-coronary epicardial adipose tissue is related to cardiovascular risk factors and coronary artery calcification in post-menopausal women. Eur Heart J. 2007;29(6):777-83.
16 Iacobellis G, Assael F, Ribaudo MC, Zappaterreno A, Alessi G, Di Mario U, et al. Epicardial fat from echocardiography: a new method for visceral adipose tissue prediction. Obes Res. 2003;11(2):304-10.
17 Iacobellis G, Ribaudo MC, Assael F, Vecci E, Tiberti C, Zappaterreno A, et al. Echocardiographic epicardial adipose tissue is related to anthropometric and clinical parameters of metabolic syndrome: a new indicator of cardiovascular risk. J Clin Endocrinol Metab. 2003;88(11):5163-8.-1818 Iacobellis G, Ribaudo MC, Zappaterreno A, Iannucci CV, Leonetti F. Relation between epicardial adipose tissue and left ventricular mass. Am J Cardiol. 2004;94(8):1084-7.]. Our findings were similar to these studies. We reported a strong relationship between EAT and age, diabetes mellitus, and hypertension. CHA2DS2-VASc score includes these risk factors. Similarly, Cetin et al.[1919 Cetin M, Cakici M, Polat M, Suner A, Zencir C, Ardic I. Relation of epicardial fat thickness with carotid intima-media thickness in patients with type 2 diabetes mellitus. Int J Endocrinol. 2013;2013:769175.] reported a significant association between EFT and type 2 diabetic subjects with subclinical atherosclerosis. Dogan et al.[2020 Dogan M, Turak O, Akyel A, Grboviç E, Mendi MA, Öksüz F, et al. Increased epicardial adipose tissue thickness is linked to aortic stiffness in patients with primary hypertension. Blood Press. 2014;23(4):222-7.] showed that in patients with newly diagnosed hypertension, increased EFT was significantly linked to impaired aortic elastic properties. Iacobellis et al.[1717 Iacobellis G, Ribaudo MC, Assael F, Vecci E, Tiberti C, Zappaterreno A, et al. Echocardiographic epicardial adipose tissue is related to anthropometric and clinical parameters of metabolic syndrome: a new indicator of cardiovascular risk. J Clin Endocrinol Metab. 2003;88(11):5163-8.] showed a correlation between cholesterol levels and EFT. However, the relationship between EFT and CHA2DS2-VASc score was independent of cholesterol levels in our study.
In the echocardiographic evaluation, EFT, LA length, aorta diameter, and IVS length were correlated with CHA2DS2-VASc score and they are independent risk factors for high thromboembolic risk in CHA2DS2-VASc score, based on a multivariate analysis. Accordingly, Altun et al.[2121 Altun I, Unal Y, Basaran O, Akin F, Emir GK, Kutlu G, et al. Increased epicardial fat thickness correlates with aortic stiffness and N-terminal pro-brain natriuretic peptide levels in acute ischemic stroke patients. Texas Heart Inst J. 2016;43(3):220-6.] have shown a significant association between EFT, N-terminal pro b-type natriuretic peptide (NT-proBNP) levels, and arterial dysfunction in patients who had sustained acute ischemic stroke.
Stroke is one of the most important causes of death and long-term disability. Control of the risk factors can prevent the development of stroke[11 GBD 2015 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1603-58.]. Recent studies have shown a relation between EFT and stroke[66 Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.,2222 Tsao HM, Hu WC, Tsai PH, Lee CL, Liu FC, Wang HH, et al. The abundance of epicardial adipose tissue surrounding left atrium is associated with the occurrence of stroke in patients with atrial fibrillation. Medicine (Baltimore). 2016;95(14):e3260.]. Akil et al.[66 Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.] demonstrated for the first time the association between EFT and cerebral ischemic stroke.
As mentioned before, EAT has the same blood supply as the adjacent myocardium and also shown paracrine functions. This causes risk for cardiac structures due to local pathogenic inflammatory effects[22 Iacobellis G, Malavazos AE, Corsi MM. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol. 2011;43(12):1651-4.,1111 Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nature Clin Pract Cardiovasc Med. 2005;2(10):536-43.,1212 Iacobellis G, Bianco AC. Epicardial adipose tissue: emerging physiological, pathophysiological and clinical features. Trends Endocrinol Metab. 2011;22(11):450-7.]. A computed tomography (CT) evaluation from the Framingham Heart study showed that pericardial fat volume could predict atrial fibrillation (AF) risk independently of other measurements of adiposity[2323 Thanassoulis G, Massaro JM, O'Donnell CJ, Hoffmann U, Levy D, Ellinor PT, et al. Pericardial fat is associated with prevalent atrial fibrillation: the Framingham Heart Study. Cir Arrhythm Electrophysiol. 2010;3(4):345-50.]. Tsao et al.[2222 Tsao HM, Hu WC, Tsai PH, Lee CL, Liu FC, Wang HH, et al. The abundance of epicardial adipose tissue surrounding left atrium is associated with the occurrence of stroke in patients with atrial fibrillation. Medicine (Baltimore). 2016;95(14):e3260.] showed that periatrial EAT was increased and was correlated with atrial dysfunction in patients with AF-related stroke.
EAT, a metabolically active tissue can induce fibrotic changes on the atrial myocardium by releasing proinflammatory cytokines and adipo-fibrokines. EAT can be an infiltrated adipocyte on the atrial myocardium. This can cause blockage of local conduction and promote the micro-reentry circuit. As a result, the occurrence of AF increases. Two potential mechanisms can be proposed for this association: firstly, the actions of proinflammatory cytokines and adipo-fibrokines released from EAT, such as activin A, adiponectin, and resistin, which can induce fibrotic changes on the atrial myocardium[22 Iacobellis G, Malavazos AE, Corsi MM. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol. 2011;43(12):1651-4.,44 Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22(12):1311-9.,1111 Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nature Clin Pract Cardiovasc Med. 2005;2(10):536-43.]; and secondly, adipocyte infiltration on the atrial myocardium, which can cause blockage of local conduction and promote the micro-reentry circuit; and potential modulations of the autonomic nervous system by the ganglionic plexus within the EAT, which may influence the occurrence of AF. In this study, we comprehensively assessed the relationship between the CHA2DS2-VASc score with EFT around the right ventricle. They were independently associated with each other, based on a multivariate analysis. Consequently, we can say that EFT is a risk indicator for stroke.
Study Limitations
The relatively limited number of patients could limit the strength of the results and the conclusion obtained from this study. Echocardiographic EAT is a linear measurement, and thus it may not assess the total epicardial fat volume that varies at several myocardial locations. As a result of EAT being a metabolically active tissue, inflammatory cytokines and inflammatory markers could be investigated in future studies.
CONCLUSION
In conclusion, our findings suggest that echocardiographic EFT measurement could provide additional information on assessing cardiovascular risks, such as thromboembolic events, and individuals with increasing EFT should receive more attention to reduce unfavorable cardiovascular risk factors and the development of future cardiovascular events.
-
This study was carried out at the Department of Cardiology, Suleyman Demirel University, Medical School, Isparta, Turkey.
-
No financial support.
REFERENCES
-
1GBD 2015 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 315 diseases and injuries and healthy life expectancy (HALE), 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1603-58.
-
2Iacobellis G, Malavazos AE, Corsi MM. Epicardial fat: from the biomolecular aspects to the clinical practice. Int J Biochem Cell Biol. 2011;43(12):1651-4.
-
3Sengül C, Özveren O. Epicardial adipose tissue: a review of physiology, pathophysiology, and clinical applications. Anadolu Kardiyol Derg. 2013;13(3):261-5.
-
4Iacobellis G, Willens HJ. Echocardiographic epicardial fat: a review of research and clinical applications. J Am Soc Echocardiogr. 2009;22(12):1311-9.
-
5Gul I, Zungur M, Aykan AC, Gokdeniz T, Kalaycioglu E, Turan T, et al. The relationship between GRACE score and epicardial fat thickness in non-STEMI patients. Arq Bras Cardiol. 2016;106(3):194-200.
-
6Akil E, Akil MA, Varol S, Özdemir HH, Yücel Y, Arslan D, et al. Echocardiographic epicardial fat thickness and neutrophil to lymphocyte ratio are novel inflammatory predictors of cerebral ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23(9):2328-34.
-
7January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014;130(23):e199-267.
-
8Sahn DJ, DeMaria A, Kisslo J, Weyman A. Recommendations regarding quantitation in M-mode echocardiography: results of a survey of echocardiographic measurements. Circulation. 1978;58(6):1072-83.
-
9Mazurek T, Zhang L, Zalewski A, Mannion JD, Diehl JT, Arafat H, et al. Human epicardial adipose tissue is a source of inflammatory mediators. Circulation. 2003;108(20):2460-6.
-
10Akdag S, Simsek H, Sahin M, Akyol A, Duz R, Babat N. Association of epicardial adipose tissue thickness and inflammation parameters with CHA2DS2-VASASc score in patients with nonvalvular atrial fibrillation. Ther Clin Risk Manag. 2015;11:1675-81.
-
11Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nature Clin Pract Cardiovasc Med. 2005;2(10):536-43.
-
12Iacobellis G, Bianco AC. Epicardial adipose tissue: emerging physiological, pathophysiological and clinical features. Trends Endocrinol Metab. 2011;22(11):450-7.
-
13Avanzas P, Arroyo-Espliguero R, Cosín-Sales J, Aldama G, Pizzi C, Quiles J, et al. Markers of inflammation and multiple complex stenoses (pancoronary plaque vulnerability) in patients with non-ST segment elevation acute coronary syndromes. Heart. 2004;90(8):847-52.
-
14Anrather J, Iadecola C. Inflammation and stroke: an overview. Neurotherapeutics. 2016;13(4):661-70.
-
15Vos AM, Prokop M, Roos CJ, Meijs MF, van der Schouw YT, Rutten A, et al. Peri-coronary epicardial adipose tissue is related to cardiovascular risk factors and coronary artery calcification in post-menopausal women. Eur Heart J. 2007;29(6):777-83.
-
16Iacobellis G, Assael F, Ribaudo MC, Zappaterreno A, Alessi G, Di Mario U, et al. Epicardial fat from echocardiography: a new method for visceral adipose tissue prediction. Obes Res. 2003;11(2):304-10.
-
17Iacobellis G, Ribaudo MC, Assael F, Vecci E, Tiberti C, Zappaterreno A, et al. Echocardiographic epicardial adipose tissue is related to anthropometric and clinical parameters of metabolic syndrome: a new indicator of cardiovascular risk. J Clin Endocrinol Metab. 2003;88(11):5163-8.
-
18Iacobellis G, Ribaudo MC, Zappaterreno A, Iannucci CV, Leonetti F. Relation between epicardial adipose tissue and left ventricular mass. Am J Cardiol. 2004;94(8):1084-7.
-
19Cetin M, Cakici M, Polat M, Suner A, Zencir C, Ardic I. Relation of epicardial fat thickness with carotid intima-media thickness in patients with type 2 diabetes mellitus. Int J Endocrinol. 2013;2013:769175.
-
20Dogan M, Turak O, Akyel A, Grboviç E, Mendi MA, Öksüz F, et al. Increased epicardial adipose tissue thickness is linked to aortic stiffness in patients with primary hypertension. Blood Press. 2014;23(4):222-7.
-
21Altun I, Unal Y, Basaran O, Akin F, Emir GK, Kutlu G, et al. Increased epicardial fat thickness correlates with aortic stiffness and N-terminal pro-brain natriuretic peptide levels in acute ischemic stroke patients. Texas Heart Inst J. 2016;43(3):220-6.
-
22Tsao HM, Hu WC, Tsai PH, Lee CL, Liu FC, Wang HH, et al. The abundance of epicardial adipose tissue surrounding left atrium is associated with the occurrence of stroke in patients with atrial fibrillation. Medicine (Baltimore). 2016;95(14):e3260.
-
23Thanassoulis G, Massaro JM, O'Donnell CJ, Hoffmann U, Levy D, Ellinor PT, et al. Pericardial fat is associated with prevalent atrial fibrillation: the Framingham Heart Study. Cir Arrhythm Electrophysiol. 2010;3(4):345-50.
Publication Dates
-
Publication in this collection
Jan-Feb 2019
History
-
Received
07 Aug 2018 -
Accepted
24 Sept 2018