Abstracts
We present here the clinical and molecular data of two patients with acromegaly treated with octreotide LAR after non-curative surgery, and who presented different responses to therapy. Somatostatin receptor type 2 and 5 (SSTR2 and SSTR5), and aryl hydrocarbon receptor-interacting protein (AIP) expression levels were analyzed by qPCR. In both cases, high SSTR2 and low SSTR5 expression levels were detected; however, only one of the patients achieved disease control after octreotide LAR therapy. When we analyzed AIP expression levels of both cases, the patient whose disease was controlled after therapy exhibited AIP expression levels that were two times higher than the patient whose disease was still active. These two cases illustrate that, although the currently available somatostatin analogs bind preferentially to SSTR2, some patients are not responsive to therapy despite high expression of this receptor. This difference could be explained by differences in post-receptor signaling pathways, including the recently described involvement of AIP. Arq Bras Endocrinol Metab. 2012;56(8):501-6
Apresentamos os dados clínicos e moleculares de dois pacientes com acromegalia tratados com octreotide LAR após cirurgia não curativa, com diferentes respostas a essa terapia medicamentosa. As expressões do receptor de somatostatina tipo 2 e 5 (SSTR2 e SSTR5) e da proteína de interação com o receptor aril hidrocarbono (AIP) foram analisadas por qPCR. Em ambos os casos, foi encontrada uma expressão elevada de SSTR2 e baixa do SSTR5. No entanto, o controle da doença foi obtido após tratamento com octreotide LAR em apenas um dos pacientes. Quando analisamos a expressão do AIP em ambos os casos, o paciente cuja doença foi controlada após a terapia medicamentosa apresentou uma expressão duas vezes maior do que a do paciente não controlado com o tratamento. Conclui-se que esses dois casos ilustram que, embora os análogos de somatostatina atualmente disponíveis se liguem preferencialmente ao SSTR2, alguns pacientes não respondem ao tratamento, apesar de uma elevada expressão desse receptor. Isso poderia ser explicado por alterações nas vias de sinalização pós-receptor, incluindo o envolvimento recentemente descrito da AIP. Arq Bras Endocrinol Metab. 2012;56(8):501-6
CASE REPORT
Resistance to octreotide LAR in acromegalic patients with high SSTR2 expression: analysis of AIP expression
Resistência ao octreotide LAR em pacientes acromegálicos com alta expressão do SSTR2: avaliação da expressão do AIP
Leandro KasukiI,II; Leandro M. ColliIII; Paula C. L. EliasIII; Margaret de CastroIII; Mônica R. GadelhaI
IEndocrinology Division, Clementino Fraga Filho University Hospital, Universidade Federal do Rio de Janeiro (HUCFF-UFRJ), Rio de Janeiro, RJ, Brazil
IIEndocrinology Unit, Bonsucesso Federal Hospital, Rio de Janeiro, RJ, Brazil
IIIEndocrinology Laboratory, Ribeirão Preto Medical School, Universidade de São Paulo (FMRP-USP), Ribeirão Preto, SP, Brazil
Correspondence Correspondence to: Mônica R. Gadelha Centro de Pesquisa em Neuroendocrinologia, Divisão de Endocrinologia, Hospital Universitário Clementino Fraga Filho, Universidade Federal do Rio de Janeiro Rua Professor Rodolpho Paulo Rocco, 255, sala 9F 21941-913 Rio de Janeiro, RJ, Brazil mgadelha@hucff.ufrj.br
SUMMARY
We present here the clinical and molecular data of two patients with acromegaly treated with octreotide LAR after non-curative surgery, and who presented different responses to therapy. Somatostatin receptor type 2 and 5 (SSTR2 and SSTR5), and aryl hydrocarbon receptor-interacting protein (AIP) expression levels were analyzed by qPCR. In both cases, high SSTR2 and low SSTR5 expression levels were detected; however, only one of the patients achieved disease control after octreotide LAR therapy. When we analyzed AIP expression levels of both cases, the patient whose disease was controlled after therapy exhibited AIP expression levels that were two times higher than the patient whose disease was still active. These two cases illustrate that, although the currently available somatostatin analogs bind preferentially to SSTR2, some patients are not responsive to therapy despite high expression of this receptor. This difference could be explained by differences in post-receptor signaling pathways, including the recently described involvement of AIP. Arq Bras Endocrinol Metab. 2012;56(8):501-6
SUMÁRIO
Apresentamos os dados clínicos e moleculares de dois pacientes com acromegalia tratados com octreotide LAR após cirurgia não curativa, com diferentes respostas a essa terapia medicamentosa. As expressões do receptor de somatostatina tipo 2 e 5 (SSTR2 e SSTR5) e da proteína de interação com o receptor aril hidrocarbono (AIP) foram analisadas por qPCR. Em ambos os casos, foi encontrada uma expressão elevada de SSTR2 e baixa do SSTR5. No entanto, o controle da doença foi obtido após tratamento com octreotide LAR em apenas um dos pacientes. Quando analisamos a expressão do AIP em ambos os casos, o paciente cuja doença foi controlada após a terapia medicamentosa apresentou uma expressão duas vezes maior do que a do paciente não controlado com o tratamento. Conclui-se que esses dois casos ilustram que, embora os análogos de somatostatina atualmente disponíveis se liguem preferencialmente ao SSTR2, alguns pacientes não respondem ao tratamento, apesar de uma elevada expressão desse receptor. Isso poderia ser explicado por alterações nas vias de sinalização pós-receptor, incluindo o envolvimento recentemente descrito da AIP. Arq Bras Endocrinol Metab. 2012;56(8):501-6
INTRODUCTION
The currently available somatostatin analogs (SSAs) octreotide and lanreotide are considered cornerstones of the medical treatment for acromegaly (1,2). SSAs act mainly by binding to somatostatin receptor type 2 (SSTR2) (3). Thus, as expected, SSTR2 expression is a predictor of the response to these drugs, and patients harboring tumors that present low SSTR2 expression are resistant to SSAs (4,5). However, in some cases, acromegaly is not controlled with SSAs therapy despite a high SSTR2 expression level in the somatotropinoma, indicating that although the presence of a high SSTR2 expression is essential for the action of SSAs, other factors, such as post-receptor signaling pathways, may be involved in the lack of response to SSAs in tumors presenting high expression of SSTR2 (5).
Recently, germline mutations in the aryl hydrocarbon receptor-interacting protein (AIP) gene have been described in the setting of familial isolated pituitary adenoma, and in seemingly sporadic young-onset pituitary adenoma patients (6,7). The patients harboring mutations in the AIP gene present worse response to SSAs therapy (8,9). Although somatic mutations have not been described, some sporadic tumors present low AIP expression, and patients harboring tumors with this feature have a lower probability of acromegaly control with SSAs treatment (10-12). Moreover, SSAs increase AIP expression, and, although the mechanism of action of SSAs is not completely understood, this feature appears to be important in the mechanism of action of this drug class (13).
To illustrate the importance of AIP expression in the response to SSAs therapy in acromegaly, we describe two patients whose somatotropinomas presented high SSTR2 expression levels, but exhibited different responses to octreotide LAR (OCT-LAR) treatment.
SUBJECTS AND METHODS
This study was approved by the Ethics Committees of the Clementino Fraga Filho University Hospital/Medical School, Universidade Federal do Rio de Janeiro, and Hospital das Clínicas/Ribeirão Preto Medical School, Universidade de São Paulo. All subjects signed an informed consent before entering the study.
CASE REPORTS
Case 1
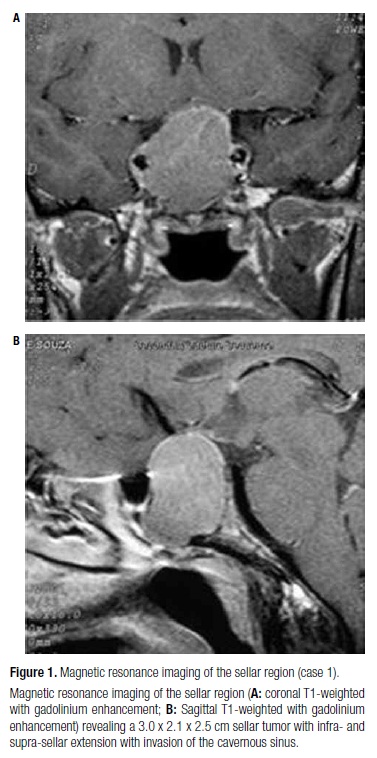
A 37 year-old female acromegalic patient was diagnosed due to a history of acral enlargement and amenorrhea. At diagnosis, she presented basal growth hormone (GH) level of 185.0 ng/mL and insulin-like growth factor type I (IGF-I) level of 1,470 ng/mL (age-adjusted normal range, 106-277). Anterior pituitary workup revealed normal basal serum cortisol (9.0 µg/dL); no hypothyroidism (TSH: 0.36 IU/mL; free T4:1.0 ng/dL), and slightly elevated prolactin of 41.0 ng/dL with LH and FSH levels in the normal range (1.0 and 2.2 mU/mL, respectively). Magnetic resonance imaging (MRI) of the sellar region revealed a 3.0 x 2.1 x 2.5 cm sellar tumor with supra-sellar extension and invasion of the cavernous sinus (Figure 1). Examination of her visual field was normal. The patient was submitted to transesphenoidal surgery, but remained with active disease after the surgical procedure. Anatomopathological study revealed a pituitary adenoma with positive immunostaining only for GH. Therefore, treatment with OCT-LAR was initiated at a dosage of 20 mg every four weeks, with control of the hormone levels after six months of medical therapy, with no further need for dose adjustment. A residual sellar lesion of 1.5 cm was present at MRI after transesphenoidal surgery and remained unchanged during follow-up. GH and IGF-I levels at diagnosis, post-surgery, and after OCT-LAR therapy are shown in table 2.
Case 2
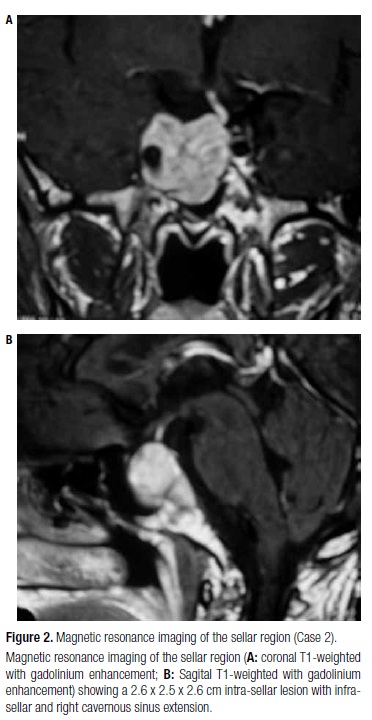
A 46-year-old male patient presented history of asthenia, hyperhidrosis, hand edema, and decreased libido that had started three years before. At initial laboratory evaluation, he presented hyperprolactinemia (prolactin, 91.4 ng/mL; normal range, 4.1-17.7), central hypothyroidism (free T4, 0.59 ng/dL; normal range, 0.8-1.9; TSH, 4.1 mcU/mL), GH level of 3.41 ng/mL, and IGF-I level of 224 ng/mL (age-adjusted normal range, 101-267). After treatment for central hypothyroidism, GH level was 15.6 ng/mL, and IGF-I level was 555 ng/mL. MRI of the sellar region showed a 2.6 x 2.5 x 2.6 cm intra-sellar lesion with infra-sellar and right cavernous sinus extension involving the right internal carotid artery (Figure 2). The patient was submitted to surgery; however, total macroscopic resection was not achieved, although he exhibited an improvement in GH and IGF-I levels (4.0 ng/mL and 411 ng/mL, respectively). The anatomopathological study revealed a pituitary adenoma with positive immunostaining for GH and prolactin. After surgery, treatment with OCT-LAR was initiated at a dosage of 20 mg every four weeks, with posterior up-titration to 30 mg every four weeks, without controlling the disease after one year of treatment. A residual lesion in the right cavernous sinus of 1.0 cm was present at MRI after transesphenoidal surgery and remained unchanged during follow-up. GH and IGF-I levels at diagnosis, three months post-surgery and after six months of OCT-LAR therapy are shown in table 3.
Analysis of AIP mutations
Deoxyribonucleic acid (DNA) was extracted using the QIAamp DNA MiniKit (Qiagen, Valencia, CA, USA) from pituitary adenoma tissue according to the manufacturer's protocol. The entire coding sequence of AIP (NM_003977.2), the conserved splice sites (from the conserved A of the upstream branch site to +10 downstream of each exon) and 1200 base pairs of the promoter region were directly sequenced, as described elsewhere (8).
Assessment of SSTR2, SSTR5 and AIP expression levels
SSTR2, SSTR5 and AIP expression levels were analyzed by quantitative polymerase chain reaction (qPCR) using Taqman® methodology. Tumoral ribonucleic acid (RNA) was extracted using the RNeasy® Mini kit (Qiagen) according to the manufacturer's protocol. The amount and quality of the extracted RNA were measured using NanoDrop 2000® (Thermo Fischer, Wilmington, DE, USA). The MultiScribeTM and High Capacity cDNA Reverse Transcription kit (Applied Biosystems, Carlsbad, CA, USA) were used for cDNA synthesis.
Thermocycling and fluorescence detection were performed using Applied Biosystems 7500 Real-Time PCR System®. The thermal cycling profile consisted of a preincubation step of 50°C for 2 min and denaturation at 95°C for 10 min followed by 40 cycles of 95°C for 15 seconds and 60°C for 1 min. In addition to SSTR2, SSTR5 and AIP, three housekeeping genes were analyzed [β-glucuronidase (GUSβ), TATA box-binding protein (TBP) and phosphoglycerate kinase 1 (PGK1)] as internal controls. Taqman® assays for each gene are depicted in table 1. Gene expression levels were calculated by QPCR software (14) using the efficiency of reaction (calculated for each reaction), and were compared with five normal pituitary tissues obtained from autopsies of subjects who had died of acute cardiovascular disease without evidence of previous endocrine disease.
RESULTS
Analysis of AIP mutations
No somatic mutations in the AIP gene were identified in either patient.
Analysis of SSTR2, SSTR5 and AIP expression levels
The expression level of SSTR2 was high in both tumors. The somatotropinoma of the patient 1 presented SSTR2 expression that was 15.51 times higher than that of normal pituitary tissue (NPT). The somatotropinoma of the patient 2 presented SSTR2 expression that was 22.95 times higher than that of NPT. SSTR2 expression level was 1.5 times higher in the adenoma of patient 2 compared with the adenoma of patient 1.
SSTR5 expression was very low in both tumors in comparison with NPT. The somatotropinoma of the patient 2 presented expression 0.34 times that of NPT. SSTR5 expression level in the somatotropinoma of patient 1 was 0.11 times that of NPT.
AIP expression of the somatotropinoma of patient 2 was similar to that of NPT (1.07x), while in patient 1, AIP expression level was 2.19 times higher than that of NPT. Comparing the two tumors, AIP expression was 2.05 times higher in the tumor of patient 1.
DISCUSSION
The commercially available SSAs enable the control of GH and IGF-I levels in approximately 30% of the acromegalic patients (15,16). Considering their affinity to the five SSTRs, it is expected that the expression of SSTR2 is mandatory for good response to these drugs (3). Indeed, it has been demonstrated by our group and others, that disease activity is not controlled in the presence of low SSTR2 expression (4,5,17,18). However, the presence of a high SSTR2 expression is not invariably associated with good response to SSAs therapy (5). In fact, a considerable proportion of patients with high SSTR2 expression do not achieve disease control with treatment, suggesting that other factors, such as SSTR5 expression levels, the presence of truncated isoforms of this receptor, and alterations in post-receptor signaling pathways may be involved (4,19). Our group has previously demonstrated, with another qPCR method (SYBR® Green), that the presence of a low SSTR2/SSTR5 ratio (lower than 1.3) was associated with worse response to SSAs therapy (4).
Recent data have highlighted the importance of AIP expression in the response to SSAs therapy in acromegaly (9). Since the description of AIP germline mutations in familial isolated pituitary adenomas, it has been observed that patients harboring these mutations have worse response to SSAs treatment (8,9,20). Indeed, a study that compared 96 acromegalic patients with germline AIP mutations and 232 matched controls (acromegalic patients without mutations) demonstrated that patients harboring AIP mutations had a poorer response to SSAs treatment (9). The AIP-mutated patients had smaller decreases in GH and IGF-I levels, as well as reduced tumor shrinkage, thereby indicating that AIP may play a role in the mechanism of response to SSAs (9).
In addition to the data regarding familial acromegaly, it has been demonstrated that although no somatic mutation in the AIP gene has been found to date, a subset of sporadic somatotropinomas present low AIP expression (mainly the more invasive cases) (10,12,21). Our group has recently demonstrated that patients harboring tumors with low AIP protein expression have low chance of disease control with OCT-LAR therapy, independent of SSTR2 expression (11). These data reinforce the importance of AIP expression for good response to SSAs treatment.
Direct evidence of AIP involvement in the mechanism(s) of action of SSAs was provided by a study of Chahal and cols. (13). These authors observed that tumors from patients treated with lanreotide before surgery exhibited higher AIP expression than tumors from patients treated primarily by surgery. Furthermore, the authors performed an in vitro study and observed that treatment of GH3 cell lines with octreotide increased AIP expression. When AIP was knocked-down, the effect of octreotide in reducing cell proliferation was attenuated, and this was likely mediated through modulation of the expression of the tumor suppressor zinc-finger protein ZAC1, whose expression is known to be essential for the anti-proliferative effect of SSAs (13,22).
To illustrate these recent findings in the literature, we presented two cases of acromegalic patients who were not cured by surgery, were treated with OCT-LAR, and showed different responses to treatment. In both cases, the tumors presented high SSTR2 expression levels; however, patient 2 did not achieve disease control with OCT-LAR therapy despite having higher SSTR2 expression than patient 1. In both cases, low expression of SSTR5 was observed but, as previously described (4), it is the high expression of this receptor that is associated with poor response to octreotide LAR. When we compared AIP expression levels of both tumors, we observed that AIP expression was two times higher in the somatotropinoma from the patient whose disease was controlled with medical treatment (case 1). As previously discussed, the presence of a high SSTR2 expression is not always associated with good response to SSAs treatment, as in patient 2. One of the possible explanations is lower AIP expression, as AIP appears to be important in the post-receptor signaling of SSAs drug therapy (13).
The mechanism(s) of action of SSAs is not completely understood, and involves the regulation of ion channels (primarily potassium and calcium channels), adenylyl cyclase-cAMP-protein kinase A (PKA), mitogen-activated protein kinase (MAPK), and phosphotyrosine phosphatase (PTP) pathways (3). The exact place of AIP in this complex signaling machinery is not completely understood, and additional studies are necessary to clarify this issue.
One of the therapeutic possibilities for patient 2 is to associate cabergoline to octreotide LAR, as this approach is effective in patients with mildly elevated IGF-I levels (23). There are no reports on the possible role of AIP expression as a predictor of response to cabergoline in acromegaly, or if AIP is important in the mechanism of action of dopamine agonists.
In conclusion, the cases reported here illustrate that, although the currently available SSAs bind preferentially to SSTR2, high expression of this receptor is not always associated with good response to therapy; high AIP expression may also be important for the mechanism of action of this drug class.
Grants: this study was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) (LK).
Disclosure: MRG received grant support from Novartis Biociências S.A. and Pfizer, and speaker fees from Novartis Biociências, Pfizer and Ipsen. LK, LMC, PCLE and MC have no conflict of interest to declare.
Received on July/31/2012
Accepted on Sept/10/2012
- 1. Vieira Neto L, Abucham J, Araujo LA, Boguszewski CL, Bronstein MD, Czepielewski M, et al. [Recommendations of Neuroendocrinology Department from Brazilian Society of Endocrinology and Metabolism for diagnosis and treatment of acromegaly in Brazil]. Arq Bras Endocrinol Metabol. 2011;55(2):91-105.
- 2. Barkan A, Bronstein MD, Bruno OD, Cob A, Espinosa-de-los-Monteros AL, Gadelha MR, et al. Management of acromegaly in Latin America: expert panel recommendations. Pituitary. 2010;13(2):168-75.
- 3. Ben-Shlomo A, Melmed S. Pituitary somatostatin receptor signaling. Trends Endocrinol Metab. 2010;21(3):123-33.
- 4. Taboada GF, Luque RM, Neto LV, Machado Ede O, Sbaffi BC, Domingues RC, et al. Quantitative analysis of somatostatin receptor subtypes (1-5) gene expression levels in somatotropinomas and correlation to in vivo hormonal and tumor volume responses to treatment with octreotide LAR. Eur J Endocrinol. 2008;158(3):295-303.
- 5. Wildemberg LE, Vieira Neto L, Costa DF, Nasciuti LE, Takiya CM, Alves LM, et al. Low somatostatin receptor subtype 2, but not dopamine receptor subtype 2, expression predicts the lack of biochemical response of somatotropinomas to treatment with somatostatin analogs. J Endocrinol Invest. 2012 Mar 26.
- 6. Vierimaa O, Georgitsi M, Lehtonen R, Vahteristo P, Kokko A, Raitila A, et al. Pituitary adenoma predisposition caused by germline mutations in the AIP gene. Science. 2006;312(5777):1228-30.
- 7. Cazabat L, Bouligand J, Salenave S, Bernier M, Gaillard S, Parker F, et al. Germline AIP mutations in apparently sporadic pituitary adenomas: prevalence in a prospective single-center cohort of 443 patients. J Clin Endocrinol Metab. 2012;97(4):E663-70.
- 8. Leontiou CA, Gueorguiev M, van der Spuy J, Quinton R, Lolli F, Hassan S, et al. The role of the aryl hydrocarbon receptor-interacting protein gene in familial and sporadic pituitary adenomas. J Clin Endocrinol Metab. 2008;93(6):2390-401.
- 9. Daly AF, Tichomirowa MA, Petrossians P, Heliovaara E, Jaffrain-Rea ML, Barlier A, et al. Clinical characteristics and therapeutic responses in patients with germ-line AIP mutations and pituitary adenomas: an international collaborative study. J Clin Endocrinol Metab. 2010;95(11):E373-83.
- 10. Kasuki Jomori de Pinho L, Vieira Neto L, Armondi Wildemberg LE, Gasparetto EL, Marcondes J, de Almeida Nunes B, et al. Low aryl hydrocarbon receptor-interacting protein expression is a better marker of invasiveness in somatotropinomas than Ki-67 and p53. Neuroendocrinology. 2011;94(1):39-48.
- 11. Kasuki L, Vieira Neto L, Wildemberg LE, Colli LM, de Castro M, Takiya CM, et al. AIP expression in sporadic somatotropinomas is a predictor of the response to octreotide LAR therapy independent of SSTR2 expression. Endocr Relat Cancer. 2012;19(3):L25-9.
- 12. Jaffrain-Rea ML, Angelini M, Gargano D, Tichomirowa MA, Daly AF, Vanbellinghen JF, et al. Expression of aryl hydrocarbon receptor (AHR) and AHR-interacting protein in pituitary adenomas: pathological and clinical implications. Endocr Relat Cancer. 2009;16(3):1029-43.
- 13. Chahal HS, Trivellin G, Leontiou CA, Alband N, Fowkes RC, Tahir A, et al. Somatostatin analogs modulate AIP in somatotroph adenomas: The Role of the ZAC1 Pathway. J Clin Endocrinol Metab. 2012;97(8):E1411-20.
- 14. Pabinger S, Thallinger GG, Snajder R, Eichhorn H, Rader R, Trajanoski Z. QPCR: application for real-time PCR data management and analysis. BMC Bioinformatics. 2009;10:268.
- 15. Mercado M, Borges F, Bouterfa H, Chang TC, Chervin A, Farrall AJ, et al. A prospective, multicentre study to investigate the efficacy, safety and tolerability of octreotide LAR (long-acting repeatable octreotide) in the primary therapy of patients with acromegaly. Clin Endocrinol (Oxf). 2007;66(6):859-68.
- 16. Colao A, Cappabianca P, Caron P, De Menis E, Farrall AJ, Gadelha MR, et al. Octreotide LAR vs. surgery in newly diagnosed patients with acromegaly: a randomized, open-label, multicentre study. Clin Endocrinol (Oxf). 2009;70(5):757-68.
- 17. Vieira Neto L, Taboada GF, Gadelha MR. Somatostatin receptors subtypes 2 and 5, dopamine receptor type 2 expression and gsp status as predictors of octreotide LAR responsiveness in acromegaly. Arq Bras Endocrinol Metabol. 2008;52(8):1288-95.
- 18. Ferone D, de Herder WW, Pivonello R, Kros JM, van Koetsveld PM, de Jong T, et al. Correlation of in vitro and in vivo somatotropic adenoma responsiveness to somatostatin analogs and dopamine agonists with immunohistochemical evaluation of somatostatin and dopamine receptors and electron microscopy. J Clin Endocrinol Metab. 2008;93(4):1412-7.
- 19. Duran-Prado M, Gahete MD, Martinez-Fuentes AJ, Luque RM, Quintero A, Webb SM, et al. Identification and characterization of two novel truncated but functional isoforms of the somatostatin receptor subtype 5 differentially present in pituitary tumors. J Clin Endocrinol Metab. 2009;94(7):2634-43.
- 20. Pinho LK, Vieira Neto L, Wildemberg LE, Moraes AB, Takiya CM, Frohman LA, et al. Familial isolated pituitary adenomas experience at a single center: clinical importance of AIP mutation screening. Arq Bras Endocrinol Metabol. 2010;54(8):698-704.
- 21. Raitila A, Georgitsi M, Karhu A, Tuppurainen K, Makinen MJ, Birkenkamp-Demtroder K, et al. No evidence of somatic aryl hydrocarbon receptor interacting protein mutations in sporadic endocrine neoplasia. Endocr Relat Cancer. 2007;14(3):901-6.
- 22. Theodoropoulou M, Zhang J, Laupheimer S, Paez-Pereda M, Erneux C, Florio T, et al. Octreotide, a somatostatin analogue, mediates its antiproliferative action in pituitary tumor cells by altering phosphatidylinositol 3-kinase signaling and inducing Zac1 expression. Cancer Res. 2006;66(3):1576-82.
- 23. Vilar L, Azevedo MF, Naves LA, Casulari LA, Albuquerque JL, Montenegro RM, et al. Role of the addition of cabergoline to the management of acromegalic patients resistant to longterm treatment with octreotide LAR. Pituitary. 2011;14(2):148-56.
Correspondence to:
Publication Dates
-
Publication in this collection
02 Jan 2013 -
Date of issue
Nov 2012
History
-
Received
31 July 2012 -
Accepted
10 Sept 2012