Abstracts
Background - Bleeding ulcers are a major problem in public health and represent approximately half of all the cases of upper gastrointestinal hemorrhage in the United States. This study aims to determine the prognostic value of factors such as clinical history, laboratory and endoscopic findings in the occurrence of new episodes of bleeding in patients who have upper gastrointestinal hemorrhage caused by gastric or duodenal peptic ulcer. Methods - A cohort study with 94 patients was designed to investigate prognostic factors to the occurrence of new episodes of bleeding. Results - From the 94 patients studied, 88 did not present a new bleeding episode in the 7 days following hospital admission. The incidence of rebleeding was significantly higher in those patients with hemoglobin <6 g/dL at the admission (P = 0.03, RR = 6.2). The localization of the ulcers in bulb was positively associated to rebleeding (P = 0.003).The rebleeding group needed a greater number of units transfunded (P = 0.03) and the time of hospitalization was longer than the time of the hemostasia group (P = 0.0349). Conclusions - The identification of patients with risk of death by bleeding peptic ulcer remains as a challenge, once few factors are capable of predicting the severity of the evolution. The identification of such factors will allow the choice of the better therapeutic conduct improving the diagnosis and decreasing the rate of rebleeding and the mortality.
Peptic ulcer hemorrhage; Gastrointestinal hemorrhage; Risk factors
Introdução - Úlceras sangrantes constituem um grave problema de saúde pública e representam cerca da metade de todos os casos de hemorragia gastrointestinal nos Estados Unidos. O objetivo deste estudo foi determinar o valor prognóstico dos achados clínicos, laboratoriais e endoscópicos no ressangramento de pacientes com úlceras gástricas e duodenais sangrantes. Pacientes e Métodos - Foi realizado um estudo de coorte com 94 pacientes para avaliar fatores prognósticos no ressangramento por úlceras pépticas. Resultados - Dos 94 pacientes estudados, 88 não apresentaram novo episódio de sangramento durante os 7 dias de internação. A incidência de ressangramento foi significativamente maior nos pacientes com hemoglobina <6 g/dL na admissão (P = 0.03, RR=6.2). O grupo com ressangramento necessitou mais transfusão de sangue (P = 0,03) e o tempo de internação foi significativamente maior (P = 0,0349) que no grupo com hemostasia. Nas variáveis endoscópicas, a localização da úlcera no duodeno esteve associada positivamente com o ressangramento (P = 0,03). Conclusão - A identificação de paciente de risco de mortalidade por úlcera sangrante permanece um desafio, sendo que apenas alguns fatores são capazes de prever a gravidade da evolução. A identificação destes fatores auxiliou na escolha da melhor conduta terapêutica, melhorando o diagnóstico e diminuindo a taxa de ressangramento e de mortalidade.
Úlcera péptica hemorrágica; Hemorragia gastrointestinal; Fatores de risco
ARTIGO ORIGINAL / ORIGINAL ARTICLE
CLINICAL AND ENDOSCOPIC ASPECTS IN THE EVOLUTION OF PATIENTS WITH BLEEDING PEPTIC ULCER - a cohort study
Fábio SEGAL* + Clinical Hospital of Porto Alegre, Department of Internal Medicine, Post Graduation Course in Medicine: Gastroenterology, Federal University of Rio Grande do Sul - UFRGS, RS, Brazil. , João Carlos PROLLA** + Clinical Hospital of Porto Alegre, Department of Internal Medicine, Post Graduation Course in Medicine: Gastroenterology, Federal University of Rio Grande do Sul - UFRGS, RS, Brazil. , Ismael MAGUILNIK*** + Clinical Hospital of Porto Alegre, Department of Internal Medicine, Post Graduation Course in Medicine: Gastroenterology, Federal University of Rio Grande do Sul - UFRGS, RS, Brazil. and Fernando H. WOLFF**** + Clinical Hospital of Porto Alegre, Department of Internal Medicine, Post Graduation Course in Medicine: Gastroenterology, Federal University of Rio Grande do Sul - UFRGS, RS, Brazil.
ABSTRACT - Background - Bleeding ulcers are a major problem in public health and represent approximately half of all the cases of upper gastrointestinal hemorrhage in the United States. This study aims to determine the prognostic value of factors such as clinical history, laboratory and endoscopic findings in the occurrence of new episodes of bleeding in patients who have upper gastrointestinal hemorrhage caused by gastric or duodenal peptic ulcer. Methods - A cohort study with 94 patients was designed to investigate prognostic factors to the occurrence of new episodes of bleeding. Results - From the 94 patients studied, 88 did not present a new bleeding episode in the 7 days following hospital admission. The incidence of rebleeding was significantly higher in those patients with hemoglobin <6 g/dL at the admission (P = 0.03, RR = 6.2). The localization of the ulcers in bulb was positively associated to rebleeding (P = 0.003).The rebleeding group needed a greater number of units transfunded (P = 0.03) and the time of hospitalization was longer than the time of the hemostasia group (P = 0.0349). Conclusions - The identification of patients with risk of death by bleeding peptic ulcer remains as a challenge, once few factors are capable of predicting the severity of the evolution. The identification of such factors will allow the choice of the better therapeutic conduct improving the diagnosis and decreasing the rate of rebleeding and the mortality.
HEADINGS - Peptic ulcer hemorrhage. Gastrointestinal hemorrhage. Risk factors.
INTRODUÇÃO
Bleeding ulcers represent approximately half of all the cases of upper gastrointestinal hemorrhage in the United States(1, 17). The most frequently cited incidence data in reviews of upper gastrointestinal bleeding derives from several European series of bleeding patients. Overall, an incidence of approximately 50 to 150 hospital admissions per 100.000 individuals per year were noted(3). Although the majority of bleeding episodes (80%) ceases spontaneously, this is still a major problem of public health. In the non stopping bleeding lesions the mortality rate remained unaltered in the last decades varying widely due to non homogeneity of the groups, being estimated to be among 5% to 29%(2, 8, 15). However, this statistic does not take into account the increasing number of elderly patients with multiple medical problems that increase their risk for morbidity and mortality(11). Various hypothesis have been proposed to explain the maintenance of this mortality rate along the years, and the search for determining factors of the prognosis of these patients has the objective of identifying a subgroup whereby the more rigorous health care could allow for a more favorable prognostic.
The risk factors for morbidity and mortality by bleeding peptic ulcer could be classified in two groups:
related to the clinical presentation
related to the endoscopic aspect of the lesion
The clinical predictors of poor outcome have been: new onset of upper gastrointestinal hemorrhage while hospitalized for an unrelated clinical problem, presentation with shock or persistent hypotension; transfusion of more than five units of red blue cells (RBCs) within 24 hours of hospital admission; severe co-morbid medical-surgical conditions which increase surgical risk; advanced age; low levels of hemoglobin at hospital admission; and significant coagulopathies(2, 5, 11, 14, 26).
Regarding the specific endoscopic signs, known as "endoscopic stigmata of ulcer bleeding", they have been well defined by several investigators and predict the chance of continued bleeding or rebleeding from a particular ulcer, while helping in the choice of therapeutic decision, either endoscopic or surgical. Various endoscopic stigmata of ulcer hemorrhage can predict different outcomes. In general, the ulcer which presents a clean base or contains flat pigment spots presents a small risk of rebleeding (5% to 10%); in the cases of adhered red clot the rate of rebleeding is around 20% to 30% and non-bleeding visible vessel varies from 40% to 50%, in the oozing ulcers it varies from 75%-85% while in the spurting bleeding artery it varies from 80%-100%(5, 6, 12, 28, 29)..Poor prognostic features are large ulcers (larger than 15 mm) deep ulcers (more than 3 to 4 mm deep) , duodenal ulcers in the posterior and/or inferior position on the duodenal bulb and ulcer with large (> 5 mm) pulsatile visible vessels(11).
This study aims to determine the prognostic value of various factors such as clinical history, laboratory and endoscopic findings in the occurrence of new episodes of bleeding in patients who have upper gastrointestinal hemorrhage caused by gastric or duodenal peptic ulcer. Secondly, it aims to describe the evolution of patients submitted or not to endoscopic sclerotherapy.
PATIENTS AND METHODS
From January to October 1996, 94 patients who were hospitalized in the Clinical Hospital of Porto Alegre, RS, Brazil (HCPA) due to upper gastrointestinal bleeding and who have endoscopic diagnosis of gastric or duodenal peptic ulcer as the cause of bleeding were included in this study. Another criteria used for inclusion of the patients was being older than 18 years. The subjects studied agreed in participating through a written informed consent signed by the patient or relative. Twenty seven patients with variceal bleeding and 18 patients with bleeding due to acute lesions of the gastroduodenal mucosa (without ulcer) were excluded from the study. None of the patients were excluded due to the presence of upper gastrointestinal bleeding which was treated in the last 30 days.
A cohort study was designed whereby the prognostic factors described below were investigated. These patients were evaluated clinically and at the laboratory in the first 24 hours following admission. Endoscopic injection therapy was used for the cases with active bleeding (oozing and spurting ulcers) and ulcers with stigmata of recent bleeding (visible vessel or adherent red clot). We have standardized our injection technique for ulcers. For actively bleeding ulcers, a 1:10.000 solution of epinephrine in saline is injected in 0.5-1.0 mL aliquots in four quadrants within 1-2 mm of the bleeding point. No limit is imposed on the total volume of the epinephrine solution injected. After achieved significant hemostasis, we injected a second solution ¾ ethanol 98% in 0.2 mL aliquots for a total volume of 2.0 mL or less in four quadrants immediately around the point which was bleeding.
For the patients presenting ulcer with an adhered dark clot, flat spot or gray slough, or a lesion with a clean base only medical management was adopted (histamine H2 receptor antagonist). The patients were followed daily, observing the presence of indicative signs of a new bleeding. In the cases where rebleeding occurred, a new diagnostic endoscopy was performed with the choice of a better therapeutic option, according to the criteria already described.
Rebleeding was defined as patients who, during hospitalization, presented one or more of the following signs: hematemesis, persistent melena (during reevaluation in 12 and 24 h), enterorrhagia, hypotension (fall of the sistolic pressure ³ 25 mm Hg or diastolic pressure ³ 15 mm Hg), tachycardia (CF ³ 100 bpm) or fall in the hemoglobin >2 g/dL in 12 hours.
Patients without these manifestations were considered as having obtained hemostasia, e.g., absence of the outcome. The information was obtained through a standardized questionnaire and tested in a previous pilot study. The interviewers, who were responsible for the collection of data at hospitalization, were previously trained medical students. The clinical data were reviewed by the main researcher who was also responsible for the collection of endoscopic data and evolution during hospitalization.
The variables studied were: age, sex, color, presence of hematemesis, melena, enterorrhagia, blood pressure, cardiac frequency, smoking habits, daily intake of alcohol in the last 12 months, use of aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs), use of corticosteroids, previous history of upper gastrointestinal bleeding, presence of associated diseases (systemic arterial hypertension, ischemic cardiopathy, diabetes mellitus, chronic obstructive pulmonary disease, neoplasia and coagulopathies), hematocrit and hemoglobin at admission and each 12 hours, platelets counting, prothrombin time, KTTP and presence of Helicobacter pylori in the gastric biopsy.
The evaluation of the prevalence of Helicobacter pylori was performed by the histopathologic exam of the endoscopic biopsy obtained from samples of two fragments of the gastric antrum. In all cases of duodenal ulcer and in the patients with gastric ulcer without active bleeding site, the research on the bacteria was performed simultaneously with the first endoscopy. In the cases of active bleeding of gastric lesions, the collection was made 30 days after, when the patient performed control endoscopy for confirmation of the benign nature of the lesion and for evaluation of the ulcer healing. This could have originated false-negative results once these patients were using antiulcer drugs. Besides, some of the patients did not come back for the follow-up, thus they did not perform control endoscopy being considered as lost cases in the statistical analysis.
RESULTS
In this period, 55% of the patients hospitalized due to gastric or duodenal bleeding peptic ulcer were submitted to endoscopic injection therapy and 45% performed conservative medical therapy. The general characteristics of the population studied can be seen in Table 1.
From the 94 patients studied, 88 did not present a new bleeding episode in the 7 days following hospital admission. Among the 52 patients who performed endoscopic injection therapy, hemostasia was obtained in 47 (90%). Among the 42 patients who did not perform endoscopic therapy, 98% did not rebleed. The six patients who presented new episode of bleeding were submitted to a new sclerotherapy and definitive hemostasia was obtained in five of them. For the sixth patient, a failure in the endoscopic treatment was detected and he underwent surgical treatment.
Regarding the demographic or clinical variables studied we did not find a statistically significant increased risk of rebleeding for the groups studied, as can be seen in Table 2.
Regarding the laboratory findings we did not observe a statistically significant difference among the hemostasia and rebleeding groups taking into account the mean value of hematocrit and hemoglobin (Ht: 28,1 and 21,8; Hb: 9,1 and 7,1, respectively), nevertheless, when divided dicotomically amongst patients with hemoglobin level ³6 g/dL or <6 g/dL, an incidence of rebleeding was significantly higher in those patients with hemoglobin <6 g/dL (Table 3).
The interval between hospital admission and performance of endoscopy was in average 9.7 hours with a standard deviation of 7.4 hours, varying between 1 and 24 hours. There was no difference among the hemostasia and rebleeding group regarding the mean time until the performance of endoscopy (9.8 and 9.7 hours respectively, P = 0.816).
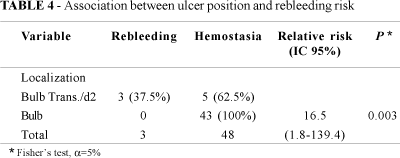
We observed the presence of a single ulcer in 71% of the cases, two lesions in 17% and three or more lesions in the remaining 12%. The number of ulcers was not associated to a high incidence of rebleeding (P = 0.7). There was 71 gastric lesions with three rebleeding ulcers (two patients in antrum and one case in incisura angularis) and 64 duodenal ulcers. There was no association between the ulcer position in the stomach and the occurrence of rebleeding, nevertheless, in the duodenum, the localization of the ulcers in the bulb transition to the second portion of the duodenum was positively associated to rebleeding, in three cases (Table 4).
The mean size of the duodenal ulcer was not associated to rebleeding (0.8 cm in the hemostasia group and 1.5 cm in the rebleeding group, P = 0.34), while the size of the gastric ulcer had nearly statistic significant results (1.4 cm in the hemostasia group and 2.5 cm in the rebleeding group, P = 0.056). The bigger ulcers had higher risk of rebleeding.
DISCUSSION
It is reported in the literature that clinical data associated to endoscopic findings allow for the identification of patients with a major risk of persistent hemorrhage or rebleeding, being considered to be prognostic indicators that allow the therapeutic planning(18). In the present study, regarding the clinical variables, only low levels of hemoglobin at admission to hospital were risk factors for rebleeding (P = 0.03, RR = 6.2), which is a similar finding to the one described by PETERSON(23), PETERSON et al.(22) and LAINE(14), where patients with low levels of hemoglobin at admission took more time to reach hemodinamic stabilization.
The need for transfusion and the amount of units of blood transfunded necessary for this stabilization, are also associated to the patient's prognostic(3). In our study the rebleeding group needed a greater number of units transfunded (P = 0.03) than the hemostasia group.
The association between NSAIDs and peptic ulcer is fairly known, being implicated as the etiologic of peptic disease and its complications, mainly the bleeding(7, 19, 20, 27). In this study a high prevalence of the use of these drugs was observed. Sixty nine patients (73,4%) used aspirin and/or NSAIDs previously to the episode of bleeding, nevertheless, such use did not constitute a prognostic factor for rebleeding.
Several studies have demonstrated the association between infection by Helicobacter pylori and the development of the gastric and duodenal peptic disease(4, 9, 13, 21, 24). In our study, Helicobacter pylori was detected in 55 out of the 72 patients whom we obtained biopsy from (77%). We did not collect material from 22 patients.
Rebleeding after endoscopic hemostasia occurs in up to 20% of the cases. A new sclerotherapy in the rebleeding cases results in permanent hemostasia in 50% of the cases, the other half, e.g., 10% of all originally treated patients, can still need surgical treatment(10). In our sample, from the six patients who rebled, the sclerotherapy had superior results with definitive hemostasia in 83% of the cases. Only one of the patients needed surgery.
In the rebleeding cases the time of hospitalization was very long, as it could be expected due to the severity of the clinical picture. The patients who presented rebleeding needed, in average, 16 days in the hospital while patients in whom hemostasia was obtained needed, in average, 5 days (P = 0.0349).
Thus, the identification of patients with risk of death by bleeding peptic ulcer remains as a challenge, once few factors are capable of predicting the severity of the evolution. The identification of such factors will allow the choice of the better therapeutic conduct improving the diagnosis and decreasing the rate of rebleeding and mortality(16, 25).
ACKNOWLEDGMENT
To students Daniela Ferreira, Letícia H. Pitrez and Rafael Boeno for the dedication and aid in the conduction of this research.
To Dr. Sandra Leistner for the the version for the English language of this work.
Segal F, Prolla JC, Maguilnik I, Wolff FH. Aspectos clínicos e endoscópicos na evolução de pacientes com úlcera péptica sangrante ¾ um estudo de coorte. Arq Gastroenterol 2000;37(3):162-167.
RESUMO - Introdução - Úlceras sangrantes constituem um grave problema de saúde pública e representam cerca da metade de todos os casos de hemorragia gastrointestinal nos Estados Unidos. O objetivo deste estudo foi determinar o valor prognóstico dos achados clínicos, laboratoriais e endoscópicos no ressangramento de pacientes com úlceras gástricas e duodenais sangrantes. Pacientes e Métodos - Foi realizado um estudo de coorte com 94 pacientes para avaliar fatores prognósticos no ressangramento por úlceras pépticas. Resultados - Dos 94 pacientes estudados, 88 não apresentaram novo episódio de sangramento durante os 7 dias de internação. A incidência de ressangramento foi significativamente maior nos pacientes com hemoglobina <6 g/dL na admissão (P = 0.03, RR=6.2). O grupo com ressangramento necessitou mais transfusão de sangue (P = 0,03) e o tempo de internação foi significativamente maior (P = 0,0349) que no grupo com hemostasia. Nas variáveis endoscópicas, a localização da úlcera no duodeno esteve associada positivamente com o ressangramento (P = 0,03). Conclusão - A identificação de paciente de risco de mortalidade por úlcera sangrante permanece um desafio, sendo que apenas alguns fatores são capazes de prever a gravidade da evolução. A identificação destes fatores auxiliou na escolha da melhor conduta terapêutica, melhorando o diagnóstico e diminuindo a taxa de ressangramento e de mortalidade.
DESCRITORES - Úlcera péptica hemorrágica. Hemorragia gastrointestinal. Fatores de risco.
Recebido para publicação em 11/10/1999.
Aprovado para publicação em 18/2/2000.
* Gastroenterologist ¾ President of the Gastroenterologic Society of Rio Grande do Sul (SGG), Porto Alegre, RS, Brazil.
** Professor of Medicine, Chairman of the Internal Medicine Department, Clinical Hospital of Porto Alegre, UFRGS.
*** Professor of Medicine, Chairman of the Gastrointestinal Endoscopic Unit, Clinical Hospital of Porto Alegre, UFRGS.
**** Physician. UFRGS.
Address for correspondence: Dr. Fábio Segal - Rua Ramiro Barcelos 910/904 - 90035-001 - Porto Alegre, RS, Brazil.
-
1American Society for Gastrointestinal Endoscopy. The national ASGE survey on upper gastrointestinal bleeding: I. Study design and baseline data. Gastrointest Endosc 1981;(2):73-9.
- 2. Cook DJ, Guyatt GH, Salena BJ, Laine LA. Endoscopic therapy for acute nonvariceal upper gastrointestinal hemorrhage: a meta-analysis. Gastroenterology 1992;102:139-48.
- 3. Consensus Development Panel. Consensus statement on therapeutic endoscopy and bleeding ulcers. Gastrointest Endosc 1990;36:S62-S5.
- 4. Cullen DJE, Hawkey CJ. Role of non-steroidal anti-inflammatory drugs and Helicobacter pylori in bleeding peptic ulcer [abstract]. Paper presented at the Digestive Disease Week, 1994.
- 5. Dudnick R, Martin P, Friedman LS. Treatment of hemorrhagic ulcers. Med Clin North Am 1991;75:963-83.
- 6. Freeman ML. The endoscopic diagnosis and intensive care unit management of severe ulcer and other nonvariceal upper gastrointestinal hemorrhage. Gastrointest Endosc Clin North Am 1991;1:209-39.
- 7. Gabriel SE, Jaakkimainen L, Bombardier C. Risk for serious gastrointestinal complications related to use of nonsteroidal anti-inflamatory drugs. A meta-analysis. Ann Intern Med 1991;115:787-96.
- 8. Gilbert DA. Epidemiology of upper gastrointestinal bleeding. Gastrointest Endosc 1990;36:S8-S13.
- 9. Graham DY, Heeps KS, Ramirez FC, Lew GM, Saeed ZA. Treatment of Helicobacter pylori reduces the rate of rebleeding in peptic ulcer disease. Scand J Gastroenterol 1993;28:939-42.
- 10. Jaspersen D, Koerner T, Schorr W, Brennenstuhl M, Raschka C, Hammar CH. Helicobacter pylori eradication reduces the rate of rebleeding in ulcer hemorrhage. Gastrointest Endosc 1995;41:5-7.
- 11. Jensen D. The bleeding ulcer ž stigmata, needles, probes and antibiotics? ASGE Postgraduate Course ž Digestive Disease Week (DDW) 1997, May 23-24.
- 12. Kohler B, Riemann JF. Upper GI bleeding-value and consequences of emergency endoscopy and endoscopic treatment. Hepatogastroenterology 1991;38:198-200.
- 13. Labenz J, Börsch G. Role of Helicobacter pylori eradication in the prevention of peptic ulcer bleeding relapse. Digestion 1994;55:19-23.
- 14. Laine L. Rolling review: upper gastrointestinal bleeding. Aliment Pharmacol Ther 1993;7:207-32.
- 15. Laine L, Peterson WL. Bleeding peptic ulcer. N Engl J Med 1994;331:717-27.
- 16. Laine L. Endoscopic therapy in bleeding ulcers. ASGE Clinical Symposium. Digestive Disease Week (DDW), San Diego, May 1995.
- 17. Laine L. The long-term management of patients with bleeding ulcers: Helicobacter pylori eradication instead of maintenance antisecretory therapy [editorial]. Gastrointest Endosc 1995;41:77-8.
- 18. Lin HJ, Lee FY, Lee SD, Lee CH. What kind of non-bleeding visible vessel in a peptic ulcer needs aggressive therapy? Endoscopy 1990;22:8-11.
- 19. Loeb DS, Ahlquist DA. Management of gastroduodenopathy associated with use of nonsteroidal anti-inflammatory drugs. Mayo Clin Proc 1992;67:354-64.
- 20. Mertz HR, Walsh JH. Physiopathology of peptic ulcer. Med Clin North Am 1991;75:807-23.
- 21. NIH Consensus Conference. Helicobacter pylori in peptic ulcer disease. JAMA 1994;272:65-9.
- 22. Peterson WL, Barnett CC, Smith HJ, Allen MH, Corbett DB. Routine early endoscopy in upper gastrointestinal tract bleeding. N Engl J Med 1981;304:925-9.
- 23. Peterson WL. Clinical risk factors. Gastrointest Endosc 1990;36:S14-5.
- 24. Rauws EAJ, Tytgat GNJ. Cure of duodenal ulcer associated with eradication of Helicobacter pylori Lancet 1990;335:1233-5.
- 25. Saaed ZA, Ramirez FC, Hepps KS, Cole RA, Graham DY. Prospective validation of the Baylor bleeding score for predicting the likelihood of rebleeding after endoscopic hemostasis of peptic ulcers. Gastrointest Endosc 1995;41:561-5.
- 26. Schiller KFR, Truelove SC, Williams G. Haematemesis and melaena, with special reference to factors influencing the outcome. Br Med J 1970;2:7-14.
- 27. Schubert TT, Bologna SD, Nensey Y, Schubert AB, Mascha EJ. Ulcer risk factors: interaction between Helicobacter pylori infection, nonsteroidal use, and age. Am J Med 1994;94:413-8.
- 28. Sugawa C, Joseph AL. Endoscopic interventional management of bleeding duodenal and gastric ulcers. Surg Clin North Am 1992;72:317-34.
- 29. Zuccaro G Jr. Bleeding peptic ulcer: pathogenesis and endoscopic therapy. Gastroenterol Clin North Am 1993;22:737-50.
Publication Dates
-
Publication in this collection
29 Nov 2002 -
Date of issue
July 2000
History
-
Received
11 Oct 1999 -
Accepted
18 Feb 2000