Abstracts
OBJECTIVE: To describe the neurological outcome of newborns with seizures. METHOD: Cohort study with newborns prospectively followed. Perinatal characteristics and etiological screening were related to outcome in a regression model. RESULTS: During the study 3659 newborns were admitted and 2.7% were diagnosed as having seizures. Hypoxic ischemic encephalopathy (51%) was the etiology more frequently associated to seizures and also to postneonatal epilepsy (53%). In the follow up 25 died during the acute neonatal illness and 9 during the first years of life, 19 were diagnosed as having post neonatal epilepsy, 35 had developmental delay and 11 an association among this two comorbidities. A significant association between abnormal postnatal EEG and neuroimaging to developmental delay (p=0.014, p=0.026) was observed. The group of newborns that had seizures presented an increased risk of developing epilepsy compared to newborns from the same cohort without seizures (19.3/100 vs. 1.8/100, p<0.001). CONCLUSION: In this study neonatal seizures predominated in term newborns with perinatal asphyxia an elevated perinatal mortality and post neonatal morbidity was observed.The follow up showed an increased risk for developing postnatal epilepsy and developmental delay.
neonatal seizures; follow up; epilepsy; neonatal EEG; preterm newborn
OBJETIVO: Avaliar o prognóstico neurológico de neonatos com crises convulsivas. MÉTODO: Estudo prospectivo, realizado em coorte de neonatos provenientes de hospital terciário. As características clínicas perinatais e os resultados de exames complementares foram correlacionados com prognóstico através de modelo de regressão logística. RESULTADOS: Durante o estudo 3659 neonatos foram internados, sendo que 101 apresentaram crises convulsivas (2,7%). A encefalopatia hipóxico-isquêmica foi a etiologia mais frequentemente associada às crises (51%). O seguimento evidenciou 25 óbitos no período neonatal e 9 durante os primeiros anos de vida, 19 lactentes desenvolveram epilepsia, 35 atraso no desenvolvimento e 11 associação entre os dois desfechos. O modelo de regressão logística aplicado mostrou associação significativa entre EEG pós neonatal anormal e neuroimagem anormal com atraso no desenvolvimento (p=0,014, p=0,026). Os neonatos em estudo, quando comparados aos demais da mesma coorte, que não apresentaram crises convulsivas tiveram maior probabilidade de desenvolver epilepsia (19,3/100 vs. 1,8/100, p<0,001). CONCLUSÃO: Neste estudo em que ocorreu predomínio de crises neonatais em neonatos a termo com asfixia perinatal, foi observada alta mortalidade perinatal e morbidade. O seguimento neurológico evidenciou elevado risco de desenvolvimento de epilepsia e/ou atraso no desenvolvimento neuropsicomotor.
convulsões neonatais; seguimento; epilepsia; EEG neonatal EEG; prematuridade
ARTICLES
Neurological outcome of newborns with neonatal seizures: a cohort study in a tertiary university hospital
Prognóstico neurológico de recém nascidos com crises convulsivas: estudo de coorte em hospital terciário
Magda Lahorgue NunesI; Maurer Pereira MartinsII; Bianca Menke BareaIII; Ricardo C. WainbergIII; Jaderson Costa da CostaIV
IMD, PhD, Associate Professor of Neurology and Pediatrics, PUCRS School of Medicine Neurodevelopment Outpatient Clinic and Laboratory of Clinical Neurophysiology, Hospital São Lucas, Pontifícia Universidade Católica do Rio Grande do Sul (PUCRS), School of Medicine, Porto Alegre RS, Brazil
IIMD, Fellow Pediatric Neurology, Division of Neurology Hospital São Lucas - PUCRS Neurodevelopment Outpatient Clinic and Laboratory of Clinical Neurophysiology, Hospital São Lucas, Pontifícia Universidade Católica do Rio Grande do Sul (PUCRS), School of Medicine, Porto Alegre RS, Brazil
IIIMD, Research Fellow Division of Neurology - PUCRS Neurodevelopment Outpatient Clinic and Laboratory of Clinical Neurophysiology, Hospital São Lucas, Pontifícia Universidade Católica do Rio Grande do Sul (PUCRS), School of Medicine, Porto Alegre RS, Brazil
IVMD, PhD, Professor of Neurology, PUCRS School of Medicine Neurodevelopment Outpatient Clinic and Laboratory of Clinical Neurophysiology, Hospital São Lucas, Pontifícia Universidade Católica do Rio Grande do Sul (PUCRS), School of Medicine, Porto Alegre RS, Brazil
ABSTRACT
OBJECTIVE: To describe the neurological outcome of newborns with seizures.
METHOD: Cohort study with newborns prospectively followed. Perinatal characteristics and etiological screening were related to outcome in a regression model.
RESULTS: During the study 3659 newborns were admitted and 2.7% were diagnosed as having seizures. Hypoxic ischemic encephalopathy (51%) was the etiology more frequently associated to seizures and also to postneonatal epilepsy (53%). In the follow up 25 died during the acute neonatal illness and 9 during the first years of life, 19 were diagnosed as having post neonatal epilepsy, 35 had developmental delay and 11 an association among this two comorbidities. A significant association between abnormal postnatal EEG and neuroimaging to developmental delay (p=0.014, p=0.026) was observed. The group of newborns that had seizures presented an increased risk of developing epilepsy compared to newborns from the same cohort without seizures (19.3/100 vs. 1.8/100, p<0.001).
CONCLUSION: In this study neonatal seizures predominated in term newborns with perinatal asphyxia an elevated perinatal mortality and post neonatal morbidity was observed.The follow up showed an increased risk for developing postnatal epilepsy and developmental delay.
Key words: neonatal seizures, follow up, epilepsy, neonatal EEG, preterm newborn.
RESUMO
OBJETIVO: Avaliar o prognóstico neurológico de neonatos com crises convulsivas.
MÉTODO: Estudo prospectivo, realizado em coorte de neonatos provenientes de hospital terciário. As características clínicas perinatais e os resultados de exames complementares foram correlacionados com prognóstico através de modelo de regressão logística.
RESULTADOS: Durante o estudo 3659 neonatos foram internados, sendo que 101 apresentaram crises convulsivas (2,7%). A encefalopatia hipóxico-isquêmica foi a etiologia mais frequentemente associada às crises (51%). O seguimento evidenciou 25 óbitos no período neonatal e 9 durante os primeiros anos de vida, 19 lactentes desenvolveram epilepsia, 35 atraso no desenvolvimento e 11 associação entre os dois desfechos. O modelo de regressão logística aplicado mostrou associação significativa entre EEG pós neonatal anormal e neuroimagem anormal com atraso no desenvolvimento (p=0,014, p=0,026). Os neonatos em estudo, quando comparados aos demais da mesma coorte, que não apresentaram crises convulsivas tiveram maior probabilidade de desenvolver epilepsia (19,3/100 vs. 1,8/100, p<0,001).
CONCLUSÃO: Neste estudo em que ocorreu predomínio de crises neonatais em neonatos a termo com asfixia perinatal, foi observada alta mortalidade perinatal e morbidade. O seguimento neurológico evidenciou elevado risco de desenvolvimento de epilepsia e/ou atraso no desenvolvimento neuropsicomotor.
Palavras-chave: convulsões neonatais, seguimento, epilepsia, EEG neonatal EEG, prematuridade.
Neonatal seizures are usually an acute manifestation of disturbance of the developing brain and are very common in the first weeks of life. Its incidence varies from 1-5 per 1000 live births1-5. This variability results, among other factors, from methodological differences (population based studies X high risk newborns selected at Neonatal Intensive Care Units) and the degree of sophistication utilized in their diagnoses4-9. The outcome of neonates with neonatal seizures has changed in recent years due to improved prenatal care, better obstetrical care and intensive neonatal care4,10-12. However, neonatal seizures remain an important predictor of future neurological complications5,13. In most instances, they result from severe diseases occurring in the neonatal period, among them, hypoxic-ischemic encephalopathy, intracranial hemorrhage, congenital infection and cerebral malformations5,13. Although mortality rates have been reduced, there remains a high morbidity rate, with epilepsy a frequent complication of neonatal seizures10-12. In addition it may be associated with other permanent neurological disorders such as mental retardation and cerebral palsy14-16. The occurrence of epilepsy after neonatal seizures varies in frequency as shown in previous studies from 3.5 to 56% according to sample selection9,14-17.
Many studies have been published concerning risk factors, etiology, identification, treatment and outcome of newborns with seizures. However, an important question as to why the outcome is so diverse among neonates with seizures is still open to discussion. Establishing risk factors that might predict outcome of newborns with seizures would be helpful in planning long term follow up and health assistance to these children.
The objective of this study was to describe the neurological outcome of newborns with seizures prospectively followed in a tertiary university hospital.
METHOD
We have performed an observational prospective study in a cohort of newborns that had clinically proven neonatal seizures and were followed in our Neurodevelopment Outpatient Clinic. Newborns included in the study were previously admitted in the Neonatal Intensive Care Unit of Hospital São Lucas, the University Hospital from Pontificia Universidade Católica do Rio Grande do Sul - School of Medicine in Porto Alegre, Brazil, from January 1999 to December 2003. This Unit is a referral center in southern Brazil and receives social security patients that are generally from low socio-economic status.
The diagnosis of neonatal seizures was based on clinical observation and correct description of seizure type according to Volpe's classification18. Furthermore, to be included in the study the newborns should have had at least two clinically proven episodes of seizures seen by the Neonatologist or Child Neurologist after maneuvers to attempt to restrain spontaneous events and to provoke events with tactile stimulation were performed19,20. Hospital records were reviewed by a pediatric neurologist (MLN) to verify the diagnosis of seizures and determine their etiology. Newborns that had only one single seizure, tremors or were pharmacologically paralyzed were excluded from the study. Seizure etiology was based on positive clinical data, laboratory data and/or imaging studies (ultrasonography, computed tomographic scan or magnetic resonance).
The time of occurrence of seizures was related to birth date and was considered early if seizures have started in the first week of life and late if seizures started among the 8th and 28th day of life. The use of antiepileptic drugs as acute therapy or in maintenance dosage (for periods longer than 48 hours after the first seizure) was also evaluated.
Clinical aspects and gestational history were obtained from hospital records in every neonate. Gestational age was determined by the Neonatologist according to Capurro21 and Ballard22 scales and newborns were classified as preterm if it was below 37 weeks.
Data from the recordings of newborns that were submitted to neonatal polysomnography were also evaluated. The recordings were blinded reviewed (MLN, JCC) and were classified as normal or abnormal, concerning the following aspects: organization of sleep stages, basal rhythm, presence of ictal activity or EEG seizures23,24.
The diagnosis of hypoxic-ischemic encephalopathy was defined by the presence of clear-cut neurological signs of neonatal encephalopathy as depressed level of consciousness, hypotonia and hypoactivity (neurological examination compatible with profiles 2, 3 or 4) associated with documented birth asphyxia (depressed Apgar scores, need of immediate resuscitation in the delivery room and, when information available, cord blood acidosis) and / or neuroradiological evidence of hypoxic-ischemic lesions9,17,25.
Data regarding neonatal neuroimaging (ultrasonography, computerized tomography or magnetic resonance) were obtained from hospital records and were considered abnormal when evidence of hypoxic ischemic or hemorrhagic lesions, malformations, migrational disorders or alterations in myelination was evidenced.
Information related to the diagnosis of central nervous system infection and metabolic disorders was also retrospectively collected.
The diagnosis of postnatal epilepsy was based on records of the Neurodevelopment Outpatient Clinic including clinical history, EEG and neuroimaging studies. Epilepsy was defined as a condition characterized by unprovoked recurring epileptic seizures (two or more), after the neonatal period26. Type of seizures and epileptic syndromes were determined based in the proposed classification by the International League Against Epilepsy (ILAE)26.
The diagnosis of developmental delay was based on results of Denver II screening test performed at ages 12 and 24 months, in the outpatient clinic associated to our protocol of neurological examination27,28.
Post neonatal EEGs were classified as normal or abnormal in report to the presence of ictal or interictal discharges, hypsarrhythmia or basal rhythm abnormalities.
All clinical variables evaluated were related to presence/absence of post neonatal epilepsy, developmental delay, perinatal and post-perinatal mortality.
The project was approved by the Ethic Committee of Pontificia Universidade Católica do Rio Grande do Sul.
Statistics
All analyses were performed using SPSS 11.5. Fisher and chi square (c2) tests were used to compare qualitative variables. Mean value of numeric variables were compared using the Student's t test. The level of significance adopted was a=0.05. Multivariate analysis was performed with variables that reached statistical significance.
RESULTS
During the study period, 3659 newborns were admitted in the Neonatal Intensive Care Unit and 101 were diagnosed as having clinical seizures (2.7%). Of these, 25 died during the acute neonatal illness and 12 were lost to follow up after three negative attempts of search (by telephone, by mail, by domiciliary visit).
Considering the rate of perinatal mortality in the whole unit (8%), mortality among newborns with seizures is around 3 times more elevated (25%).
In this sample 57% of the newborns with seizures were male and 71.4% were term. The mean gestational age was 36.6±4 weeks, vaginal delivery occurred in 45.5% and 71% had birth weight adequate for gestational age. Mean Apgar score for the first minute was 5±0.3 and for the fifth, 7.1±2.5. The age of first seizure varied from <24 hours of life to 32 days (mean 4.8±7.2 days). Only one newborn had the first seizure after the 28th day of life, but this was a preterm newborn, with conceptional age below 40 weeks. 45% of the newborns had seizures during the first 24 hours of life.
The most common etiology was hypoxic ischemic encephalopathy (51%), followed by transient metabolic disturbances (14%), infection (congenital, septicemia, bacterial meningitis 9%) intraventricular hemorrhage (6%), venous infarct (3%), inborn error of metabolism and cerebral dysgenesis (2% each). It was not possible to determine etiology of seizures in 10 patients. No significant statistical relationship between etiology and the outcomes studied was observed. However, 10/19 patients that later developed epilepsy reached criteria to the diagnosis of hypoxic ischemic encephalopathy.
Results of the first EEG performed during the neonatal period in 71 newborns of the sample showed 3.5% with dysmaturity, 4% with ictal discharges, 21% with basal rhythm abnormalities, 53% with a combination of two or more alterations and 18.5% with normal results. From the newborns with seizures not submitted to an EEG during the neonatal period 21 (70%) were in a high risk situation and died in the Unit .The others had hypoglycemia or electrolyte disturbances, with seizures controlled after correction of the disorders.
Post neonatal EEGs were repeated in 42 patients during the first two years of life. In 21 patients the EEG was considered to be within normal limits, 17 had interictal epileptogenic activity (11 patients in this group developed epilepsy) and 4 had hypsarrhythmia (all of them were diagnosed as West syndrome). In the group with normal post neonatal EEGs, only 3 patients latter developed epilepsy.
Neuroimaging studies were performed in 67 neonates; in 18 it was considered normal and in 49 abnormal (63% with ischemic lesions, 28% with hemorrhagic lesions and 8% with malformations). Post neonatal neuroimaging studies were performed in 29 infants, in 7 it was considered normal. Among the abnormal results 68% had ischemic lesions, 18% had hydrocephalus and 14% hemorrhagic lesions.
During the neonatal period seizures were controlled in 63 patients (62%) with phenobarbital used in monotherapy (attack dosage varying from 20-40 mg/kg/day and maintenance 3-5 mg/kg/day). Five patients used only intravenous diazepam. In 26 newborns phenobarbital was used in association with one or more antiepileptic drugs. One patient had seizures controlled with pyridoxine. 45.5% of the newborns were discharged using antiepileptic drugs.
Concerning the outcome (mean follow up 33.2±21.3 months), 9 (8.9%) patients died after the neonatal period, 23 patients had repeated seizures during the follow up period, and 19 were latter diagnosed as having epilepsy (West syndrome=5, symptomatic epilepsy=12, undetermined epilepsy=1, benign idiopathic neonatal convulsions =1). The incidence of epilepsy at a mean age of 33 months was 30%. Developmental delay was observed in 35 patients, among them 11 had also epilepsy.
Prematurity (p=0.008), low birth weight (p=0.01), abnormal neonatal neuroimaging (p=0.01), abnormal postnatal EEG (p=0.001) and postnatal neuroimaging (p<0.001), and earlier postnatal seizures were risk factors for developmental delay (Table 1A and 1B).
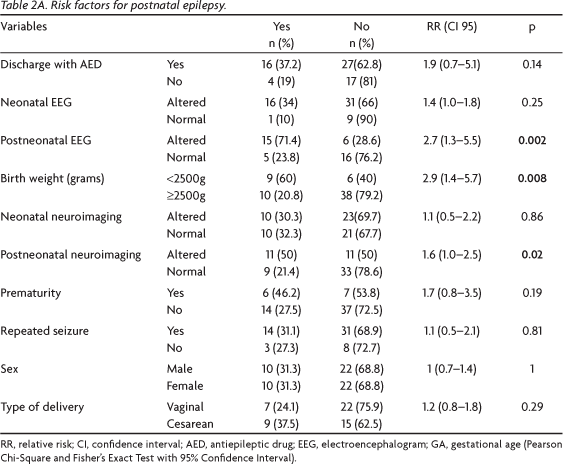
Low birth weight (p=0.008), abnormal postnatal EEG (0.002) and postnatal neuroimaging (0.02) were risk factors for the development of postnatal epilepsy (Table 2A and 2B).
The only variable significantly related to the outcome perinatal mortality was prematurity (Table 3). In the present study no variables were identified with significant association to the outcome post- perinatal mortality.
The logistic regression model applied to the outcomes studied showed significant association between abnormal postnatal EEG and neuroimaging to developmental delay (Table 4A and 4B).
Comparing newborns that had neonatal seizures (n=101) to newborns without neonatal seizures from the same cohort that were followed for a similar period in the Neurodevelopment Outpatient Clinic (n=480), the risk of developing epilepsy was 10.7 times greater in the former group (19.3/100 vs. 1.8/100) (p<0.001).
DISCUSSION
Seizures are still the most important clinical manifestation of neurological disorders in the neonatal period1,2. Previous studies have already shown close association between seizures and the development of permanent deficits, among them postnatal epilepsy3,4,9,10,12-14. Although the incidence of post neonatal epilepsy observed in this cohort could be considered elevate, it is similar to previous results published in the literature by different groups summarized in reference 32 .
The diagnosis of seizures in this study was based on clinical description. The variability inherent to the capacity of clinically identifying neonatal seizures has been evaluated previously; showing reliability in recognizing seizures based on the analysis of medical records28. It is widely known that the golden standard to recognize neonatal seizures is video-EEG. However, this equipment is not always available in many neonatal units around the world. Critics for over-diagnosing or misdiagnosing seizures using only clinical criteria have been pointed by many authors1,3,5,9,16,19,20,32,33. As described in methodology, we have used a very restrict criteria to diagnose clinical seizures and furthermore all the patients included in the study were prospectively followed in the neonatal unit since the suspicion of seizures. However, newborns that had exclusively electrographic seizures, without clinical manifestations were certainly misdiagnosed, and this is a limitation of this study.
In a recent publication, a systematic literature review of previous cohort studies of neonatal seizures, was performed: from the 36 selected studies, 24 (67%) were based only in clinical diagnosis of seizures, 7 used confirmed EEG seizures and only 2 of them were based on video-EEG. The interesting finding of this study is that the clinical profile of the newborns, risk factors and outcome were similar, independently of the methodology used to diagnose seizures32.
In studies using selected populations, including newborns treated in intensive care units, a higher incidence of epilepsy is expected than would usually be the case with population-based studies4,7-9,30. A higher rate of post neonatal epilepsy in this cohort was observed. However, our results are comparable to other cohort studies with similar sampling11,14-17. The diagnosis of severe forms of epilepsy and 58% of them in association with developmental delay is another important aspect of the study, which support the unfavorable evolution in patients with neonatal convulsions9,10,31 .
Neonatal seizures are usually reported to occur more frequently in premature infants32. However, in this study, we observed a predominance of seizures in term newborns with birth weight adequate to gestational age. These results agree with a previous cohort followed in our institution and with another study from the same country9,20. This finding might be related to regional peculiarities of maternal and birth care or perhaps due to the high incidence of perinatal asphyxia in our cohort. Anyway, both studies have observed that preterm and low birth weight infants, although minority in number, had higher probability of having an unfavorable outcome.
Perinatal asphyxia, in this study, in agreement with other series, was the most frequently etiology related to neonatal seizures5,9,33,34.
Comparing two cohorts followed in our institution, in the former (1987-1997) the incidence of neonatal seizures was 24.2:1000 live birth, mortality 15%, postnatal epilepsy 28.3% at 36 months and 33.8% at 48 months of follow up9. In the present study we have observed a slight increase in the incidence of neonatal seizures (27.6:1000 live birth ), an increase in perinatal mortality (24.7%) and similar rates of postnatal epilepsy (29.6%). The main difference among both studies was the way data was collected, in the first from hospital records and in the second with prospective follow up. The percentage of preterm infants was slight higher in the first (34.6% x 28.6%) but mean gestational age was lower in the second (34±2.2 x 32±3.8 weeks).
In conclusion, we have observed in this study were neonatal seizures predominated in term newborns with perinatal asphyxia an elevated perinatal mortality, and post neonatal morbidity was observed. Furthermore, the follow up showed an increased risk for developing postnatal epilepsy and developmental delay. Abnormal post-neonatal EEG and neuroimaging were good predictors for the outcome developmental delay.
Received 11 September 2007, received in final form 28 January 2008. Accepted 1 February 2008.
BMB and RCW were supported by scholarships from FAPERGS and CNPq.
MLN was supported by CNPq (300364/2004-0).
Dra. Magda Lahorgue Nunes Division of Neurology / Hospital São Lucas / PUCRS - Avenida Ipiranga 6690 / room 220 - CEP 90610-000 Porto Alegre RS, Brasil. E-mail: nunes@pucrs.br
- 1. Scher MS, Painter MJ. Controversies concerning neonatal seizures. Pediatr Clin N Am 1989;36:281-310.
- 2. Lombroso CT. Prognosis in neonatal seizures. In Delgado-Escueta AV, Wasterlain CG, Treiman DM, Porter RJ (Eds). Status epilepticus: mechanisms of brain damage and treatment. Advances in Neurology. New York: Raven Press,1983:101-113.
- 3. Lombroso CT. Neonatal seizures. Historic notes and present controversies. Epilepsia 1996;37(Suppl):S5-S13.
- 4. Scher MS. Seizures in the newborn infant: diagnosis, treatment and outcome. Clin Perinatol 1997;24:735-772.
- 5. Lombroso CT. Neonatal seizures: a clinician's overview. Brain Develop 1996;18:1-28.
- 6. Ericksson M, Zetterstrom R. Neonatal convulsions: incidence and causes in the Stockholm area. Acta Pediatr Scand 1979;68:807-811.
- 7. Ronen GM, Penney S, Andrews W. The epidemiology of clinical neonatal seizures in Newfoundland: a population based study. J Pediatr 1999;134:71-75.
- 8. Lanska MJ, Lanska DJ, Baumann RJ, Kryscio RJ. A population-based study of neonatal seizures in Fayette County, Kentucky. Neurology 1995;45:724-732.
- 9. Garcias da Da Silva LF, Nunes ML, Da Costa JC. Risk factors for developing epilepsy after neonatal seizures. Pediatr Neurol 2004;30:271- 277.
- 10. Bernes SM, Kaplan AM. Evolution of neonatal seizures. Pediatr Clin North Am 1994;41:1069-1104.
- 11. Arpino C, Domizio S, Carrieri MP, et al. Prenatal and perinatal determinants of neonatal seizures occurring in the first week of life. J Child Neurol 2001;16:651-656.
- 12. Keen JH, Lee D. Sequelae of neonatal convulsions: study of 112 infants. Arch Dis Child 1973;48:542-546.
- 13. Dennis J. Neonatal convulsions: etiology, late neonatal status and long-term outcome. Dev Med Child Neurol 1978;20:143-58.
- 14. Curatolo P, Arpino C, Stazi MA, Medda E. Risk factors for the co-occurence of partial epilepsy, cerebral palsy and mental retardation. Dev Med Child Neurol 1995;37:776-782.
- 15. Mellits ED, Holden KR, Freeman JM. Neonatal seizures II: a multivariate analysis of factors associated with outcome. Pediatrics 1982;70:177-185.
- 16. Clancy RR, Legido A. Postnatal epilepsy after EEG-confirmed neonatal seizures. Epilepsia 1991;32:69-76.
- 17. Legido A, Clancy RR, Berman PH. Neurologic outcome after electroencefalographically proven neonatal seizures. Pediatrics 1991;88:583-596.
- 18. Volpe JJ. Neonatal seizures. Clinics in Perinatology 1977;4:43-63.
- 19. Mizrahi EM, Kellaway P. Diagnosis and management of neonatal seizures. Philadelphia: Lippincott-Raven, 1998.
- 20. Holanda MRR, Melo AN. Comparative clinical study of preterm and full-term newborn neonatal seizures. Arq Neuropsiquiatr 2006;64: 45-50.
- 21. Capurro H, Konichewsky S, Fonseca D. A simplified method for diagnosis of gestational age in the newborn infant. J Pediatr 1979;93:120-122.
- 22. Ballard JL, Novak KK, Driver MA. A simplified score for assessment of fetal maturation of newborn infants. J Pediatr 1979;95:769-774.
- 23. Nunes ML, Da Costa JC. Manual de EEG e polissonografia neonatal: atlas de traçados. Porto Alegre: EDIPUCRS, 2003.
- 24. Lombroso CT. Neonatal EEG polygraphy in normal and abnormal newborns. In Niedermeyer E, Lopes da Silva F (Eds). Electroencephalography. 3.Ed. Baltimore: Urban & Schwarzenberg, 1994:803-875.
- 25. Volpe JJ. Neurology of the newborn. 4.Ed. Philadelphia: Saunders, 2000.
- 26. Commission on classification and terminology of the International League Against Epilepsy. Proposal for classification of epilepsies and epileptic syndromes. Epilepsia 1989;30:389-399.
- 27. Frankenburg WK, Dodds JB, Archer P, Sharir H, Breusnich. The Denver II: a major revision and restandartization of the Denver developmental screening test. Pediatrics 1992;89:91-97.
- 28. Nunes ML, Batista BHB. Ficha de avaliação neurológica do recém-nascido. In Nunes ML, Marrone ACM (Eds). Semiologia neurológica. Porto Alegre: EDIPUCRS, 2002:597-600.
- 29. Sander JWA, Shorvon SD. Incidence and prevalence studies in epilepsy and their methodological problems: a review. J Neurol Neurosurg Psychiatry 1987;50:829-839.
- 30. Bergman I, Painter MJ, Hirsh RP, Crumine PK, David R. Outcome of neonates with convulsions treated in an intensive care unit. Ann Neurol 1983;14:642-647.
- 31. Lombroso CT. Early myoclonic encephalopathy, early infantile epileptic encephalopathy, and benign and severe myoclonic epilepsies: a critical review and personal contributions. J Clin Neurophysiol 1990;7:380-408.
- 32. Nunes ML, Da Costa JC. Outcome of newborns with neonatal seizures: risk factors and predictors. Current Pediatrics Review 2006;2:315-321.
- 33. Tekgul H, Gauvreau K, Soul J, et al. The current etiologic profile and neurodevelopmental outcome of seizures in term newborn infants. Pediatrics 2006;117:1270-1280.
- 34. Watanabe K. Etiological and preventive aspects of epileptology in the developing nervous system. In Akimato H, Seino M, Ward AA (Eds). Advances in epileptology. New York: Raven Press, 1982:77-82.
Publication Dates
-
Publication in this collection
02 June 2008 -
Date of issue
June 2008
History
-
Accepted
01 Feb 2008 -
Received
11 Sept 2007 -
Reviewed
28 Jan 2008