ABSTRACT
The neonatal period is a highly vulnerable time for an infant. The high neonatal morbidity and mortality rates attest to the fragility of life during this period. The incidence of birth trauma is 0.8%, varying from 0.2-2 per 1,000 births. The aim of this study is to describe brain traumas, and their mechanism, anatomy considerations, and physiopathology of the newborn traumatic brain injury.
Methods
A literature review using the PubMed data base, MEDLINE, EMBASE, Science Direct, The Cochrane Database, Google Scholar, and clinical trials. Selected papers from 1922 to 2016 were studied. We selected 109 papers, through key-words, with inclusion and exclusion criteria.
Discussion
This paper discusses the risk factors for birth trauma, the anatomy of the occipito-anterior and vertex presentation, and traumatic brain lesions.
Conclusion
Birth-related traumatic brain injury may cause serious complications in newborn infants. Its successful management includes special training, teamwork, and an individual approach.
Newborn; brain injury; birth Injuries; pediatrics
RESUMO
O período neonatal é um período vulnerável para o recém nascido. As altas taxas de morbidade e mortalidade neonatal atestam a fragilidade da vida durante esta fase. Trauma durante o nascimento é de 0,8%, variando de 0,2 a 2 por 1000 nascimentos. O objetivo deste estudo é descrever o tocotraumatismo, seu mecanismo, considerações anatômicas e fisiopatologia da lesão em recém nascido.
Métodos
Revisão da literatura utilizando base de dados PubMed, MEDLINE, EMBASE, Science Direct, The Cochrane Detabase, Google Scolar, ensaios clínicos. Os trabalhos selecionados foram de 1922 a 2016. Foram selecionados 109 trabalhos, através de palavras-chave, inclusão e critérios de exclusão.
Discussão
Este artigo discute os fatores de risco para o trauma do nascimento, a anatomia da apresentação do vértex occipto-anterior e as lesões traumáticas cerebrais.
Conclusão
Lesão cerebral traumática no nascimento pode causar complicações graves nos recém-nascidos. O tratamento desta condição deve ser especializado, envolvendo trabalho, equipe e abordagem individualizado
Recém-nascido; lesão cerebral; nascimento Lesões; pediatria
During birth the infant’s head is exposed to contractions of the uterine muscles and to intra-abdominal pressure11. Abroms JF, Bresnan MJ, Zuckerman JE, Fischer EG, Strand R. Cervical cord injuries secondary to hyperextension of the head in breech presentations. Obstet Gynecol. 1973;41(3):369-78.. These mechanical influences are the cause of transient physiological or enduring pathological changes to the skull. The skull deformities differ according to the size of the pelvic opening22. Stoll BJ, Kliegman RM. Delivery room emergencies. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson Text book of pediatrics. 17th ed. Philadelphia, PA: Saunders; 2004. p. 569-72..
The neonatal period is a highly vulnerable time for an infant. The high neonatal morbidity and mortality rates attest to the fragility of life during this period. Some of the special problems in newborn infants are related to the adverse effects of delivery. Mechanisms that can cause birth trauma are mechanical (compressive and traction forces) or hypoxic-ischemic. The most important risk factors for birth trauma include: instrument delivery (e.g. midforceps or vacuum extraction), primiparity, cephalopelvic disproportion, birth weight more than 4 kg or less than 2.5 kg, oligohydramnios, prolonged delivery, prolonged or unusually rapid labor, maternal pelvic anomalies, fetus malformations, abnormal presentations such as breech presentation, and prematurity. The incidence of birth trauma has been assessed in most countries and is estimated at two to seven per 1,000 live births in the world. The incidence of birth trauma is 0.82%; prevalence 9.5 per 1,000 livebirths33. Robert MK, Bonita S, Joseph SG, Nina FS. Fetus and the neonatal birth injury. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson text book of pediatrics. 15th ed. Philadelphia, PA: Saunders; 1996. p. 561-6..
The aim of this study is to describe the mechanisms of neonatal brain trauma, and the different consequences to the brain.
HISTORICAL REVIEW
It was not until the beginning of the 19th century that attention was drawn to the fact that there might be a connection between head injuries occurring during delivery and pathological conditions of infancy. In ancient Greek medicine, Hippocrates (460-377 B.C.) distinguished between epilepsy and infantile convulsions. Cerebral palsy was recognized long before and it attracted the interest of the medical profession33. Robert MK, Bonita S, Joseph SG, Nina FS. Fetus and the neonatal birth injury. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson text book of pediatrics. 15th ed. Philadelphia, PA: Saunders; 1996. p. 561-6.. Shakespeare (1564-1616) indicated the pathogenesis, as well as the clinical aspects, of cerebral palsy in his portrayal of King Richard (it is a fact that this king was born prematurely in a foot presentation and that difficulties were involved in resuscitating the infant) 33. Robert MK, Bonita S, Joseph SG, Nina FS. Fetus and the neonatal birth injury. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson text book of pediatrics. 15th ed. Philadelphia, PA: Saunders; 1996. p. 561-6.,44. Hankins GD, Clark SM, Munn MB. Cesarean section on request at 39 weeks: impact on shoulder dystocia, fetal trauma, neonatal encephalopathy, and intrauterine fetal demise. Semin Perinatol. 2006;30(5):276-87. https://doi.org/10.1053/j.semperi.2006.07.009
https://doi.org/10.1053/j.semperi.2006.0...
,55. Henderson SG, Sherman LS. The Roentgen anatomy of the skull in the newborn infant. Radiology. 1946;46(7):107-18. https://doi.org/10.1148/46.2.107
https://doi.org/10.1148/46.2.107...
.
Little (1819-1894), in 1843 proposed that cerebral diplegia in children must be considered in relation to birth55. Henderson SG, Sherman LS. The Roentgen anatomy of the skull in the newborn infant. Radiology. 1946;46(7):107-18. https://doi.org/10.1148/46.2.107
https://doi.org/10.1148/46.2.107...
. He considered prematurity and asphyxia as predisposing factors. Gowers (1888)66. Lindgren L. The lower parts of the uterus during the first stage of labour in occipito-anterior vertex presentation: studies by means of intrauterine tokography. Acta Obstet Gynecol Scand. 1955;34(Suppl 2):1-79. https://doi.org/10.3109/00016345509154530
https://doi.org/10.3109/0001634550915453...
fully supported Little’s concept of cerebral palsy. He reported a number of cases with the following etiology: first-born delivery, asphyxia, complicated (breech) delivery. Salomonsen (1928)77. Moloy HC. Studies on head molding during labor. Amer J Obstet Gynec. 1942;44(5):762-82. https://doi.org/10.1016/S0002-9378(15)30603-7
https://doi.org/10.1016/S0002-9378(15)30...
emphasized that even a normal delivery represents a trauma to the infant: “The traumatic effects of labor upon the brain give rise to demonstrable lesions in such a large proportion of cases that it attains the character of a physical law”. Ehrenfest in 1931, in his extensive monograph, found nystagmus in 35% of “normally” born infants and retinal hemorrhages in 12%. He considered these findings indicative of “physiological birth injuries”44. Hankins GD, Clark SM, Munn MB. Cesarean section on request at 39 weeks: impact on shoulder dystocia, fetal trauma, neonatal encephalopathy, and intrauterine fetal demise. Semin Perinatol. 2006;30(5):276-87. https://doi.org/10.1053/j.semperi.2006.07.009
https://doi.org/10.1053/j.semperi.2006.0...
.
Considerations on the mechanisms of births head trauma
At term, the most common way the fetus passes through the maternal pelvis is in a vertex presentation. In the vertex presentation, the head is flexed with the chin resting on the fetal chest, allowing the suboccipitobregamatic diameter to be the smallest diameter presenting in the labor canal33. Robert MK, Bonita S, Joseph SG, Nina FS. Fetus and the neonatal birth injury. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson text book of pediatrics. 15th ed. Philadelphia, PA: Saunders; 1996. p. 561-6.,88. Bhagwanani SG, Price HV, Lawrence KM, Ginz B. Risks and prevention of cervical cord injury in the management of breech presentations with hyperextension of the fetal head. Am J Obst Gynec. 1973;115(8):1159-61. https://doi.org/10.1016/0002-9378(73)90573-5
https://doi.org/10.1016/0002-9378(73)905...
. During the descent of the fetal head into the pelvic cavity, its shape changes to facilitate the relationship between the size of the fetus and the diameters of the maternal pelvis. The parietal bones are pressed out from between the frontal and occipital bones. This causes the formation of a step between the coronal and lambdoid sutures. The parietal bones are symmetrically displaced so that no difference in level occurs within the region of the sagittal suture, no sheering force is exerted upon the sagittal sinus or cortical bridging veins. The trapezoidal form (broader superomedially than inferolaterally) of the parietal bones permits the frontal and occipital bones to approach one another as the parietal bones are displaced, so that the former are pressed underneath the parietal bones. The occipitofrontal diameter may thus be reduced by 1 cm, facilitating passage through the birth canal (Figure 1)55. Henderson SG, Sherman LS. The Roentgen anatomy of the skull in the newborn infant. Radiology. 1946;46(7):107-18. https://doi.org/10.1148/46.2.107
https://doi.org/10.1148/46.2.107...
.
Illustration of skull deformation during birth with vertex presentation. Compression of the frontal and occipital bones (full arrows), the parietal bones are pressed out (dotted arrows).
Other deformations of the skull are not as clinically distinct as these. They are also difficult to observe radiologically. As the parietal bone is displaced toward the vertex, the squamous suture is enlarged. This may cause a difference in the level between the bones that border this suture, with the parietal bones sliding beneath the temporal bones. The degree of deformation changes with the position of the head within the birth canal. It can be shown that the parietal bones are already displaced while the head is in the pelvic inlet. The greatest displacement of both parietal bones occurs in the upper part of the birth canal, however. As the head passes through the bony pelvis into soft tissue, the deformation decreases or disappears. Deformation recurs when the skull passes through the vulva, and the muscular portion of the birth canal compresses it66. Lindgren L. The lower parts of the uterus during the first stage of labour in occipito-anterior vertex presentation: studies by means of intrauterine tokography. Acta Obstet Gynecol Scand. 1955;34(Suppl 2):1-79. https://doi.org/10.3109/00016345509154530
https://doi.org/10.3109/0001634550915453...
,77. Moloy HC. Studies on head molding during labor. Amer J Obstet Gynec. 1942;44(5):762-82. https://doi.org/10.1016/S0002-9378(15)30603-7
https://doi.org/10.1016/S0002-9378(15)30...
.
Malpresentation of the vertex presentation occurs when there is deflection of the fetal head, leading to breech, brow, or face presentations. The deflection of the head is determined by the combined contractile forces of the labor (multiple gestations, grand multiparity), fetal head shape and diameter, and maternal pelvic architecture. The widest diameter of the fetal head forcing the maternal pelvis is the submentobregmatic diameter of face presentation. The fetal head is characteristically deformed with deflected presentations. The parietal bones are pressed inward relative to the frontal and occipital bones, which is opposite to that which occurs in a vertex presentation88. Bhagwanani SG, Price HV, Lawrence KM, Ginz B. Risks and prevention of cervical cord injury in the management of breech presentations with hyperextension of the fetal head. Am J Obst Gynec. 1973;115(8):1159-61. https://doi.org/10.1016/0002-9378(73)90573-5
https://doi.org/10.1016/0002-9378(73)905...
.
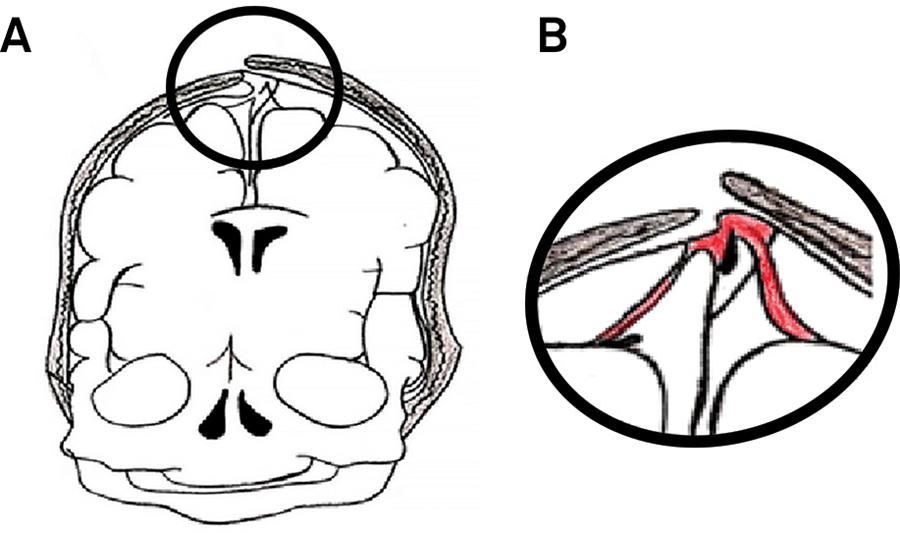
Should deformation occur abruptly and be severe, intracranial bleeding may result from tearing of the falx cerebri and the tentorium cerebelli, or tearing of the bridging veins draining the cortical surface into the venous sinus (Figure 2). An excessive molding effect may cause scalloping of the parietal bones, contributing to the traumatic mechanism to the fetal head (Figure 3). Deformations are more pronounced the longer the birth process and the more the neonatal skull differs from the usual spherical form. Thus, infants with dolichocephaly suffer an especially severe configuration. In such cases, the changes in sutures and fontanels are more distinct. It has been suggested that during breech delivery, occipital osteodiastasis, a separation of the squamous and lateral portions of the developing occipital bone, is caused by pressure from the pubic symphysis against the suboccipital region; however, this may also occur during forcible engagement of the head99. Bucher HU, Boltshauser E, Friderich J, Isler W. Birth injury to the spinal cord. Helv Paediatr Acta. 1979;34(6):517-27.,1010. Byers RK. Spinal-cord injuries during birth. Dev Med Child Neurol. 1975;17(1):103-10. https://doi.org/10.1111/j.1469-8749.1975.tb04967.x
https://doi.org/10.1111/j.1469-8749.1975...
,1111. Cole VA, Durbin GM, Olaffson A, Reynolds EOR, Rivers RPA, Smith JF. Pathogenesis of intraventricular haemorrhage in newborn infants. Arch Dis Child. 1974;49(9):722-8. https://doi.org/10.1136/adc.49.9.722
https://doi.org/10.1136/adc.49.9.722...
.
Illustration of the mechanisms of intracranial hemorrhage during birth. Small arrows indicate tearing of the tent and bleeding points due to rupture of bridging veins.
Illustration of the mechanisms of intracranial hemorrhage during birth. The detail in B highlights the scalloping of the parietal bones at the sagittal suture, causing tearing of the bridging veins.
Various non-life-threatening pathologic changes may occur at the basicranium during passage through the birth canal. Injury to the basicranium may distort synchondroses; thus, weakened during birth, the basicranium might progressively deform during neonatal life until fusion of the elements constituting the base of the skull occurs. In later life, this may be detectable as basilar impression, atlanto-occipital assimilation, or indentation of the occiput behind the foramen magnum, sometimes called a nuchal impression 66. Lindgren L. The lower parts of the uterus during the first stage of labour in occipito-anterior vertex presentation: studies by means of intrauterine tokography. Acta Obstet Gynecol Scand. 1955;34(Suppl 2):1-79. https://doi.org/10.3109/00016345509154530
https://doi.org/10.3109/0001634550915453...
,99. Bucher HU, Boltshauser E, Friderich J, Isler W. Birth injury to the spinal cord. Helv Paediatr Acta. 1979;34(6):517-27..
Secondary suture fusion may be the result of birth trauma across the neonatal skull sutures. As stated earlier, the above-mentioned studies on head molding during labor were based mainly on x-ray investigations. The increasing sophistication of ultrasonic techniques and computed axial tomography should provide more precise data on the physiologic and pathologic changes of the infant’s head during the birth process1212. Ford FR. Breech delivery in its possible relation to injury of the spinal cord with special reference to infantile paraplegia. Arch Neurol Psychiat. 1925;14(6):742-50. https://doi.org/10.1001/archneurpsyc.1925.02200180013002
https://doi.org/10.1001/archneurpsyc.192...
.
The birth process is a blend of compression, contractions, torques, and traction. When fetal size, presentation, or neurologic immaturity complicates this event, these intrapartum forces may lead to tissue damage, edema, hemorrhage, or fracture in the neonate. The use of obstetric instrumentation may further amplify the effects of such forces or may induce injury themselves. Under certain conditions, Cesarean delivery can be an acceptable alternative but does not guarantee an injury-free birth. The sutures are malleable and birth canal movement can, depending on the aggressiveness, break bridging veins, causing hematomas, and the effect of inertia can cause intracranial lesions.
Factors predisposing injury include: primigravida, cephalopelvic disproportion, small maternal stature, maternal pelvic anomalies, prolonged or rapid labor, transverse arrest of descent of the presenting part of the fetus, oligohydramnios, abnormal presentation (breech), use of midcavity forceps or vacuum extraction, versions and extractions, very-low-birth-weight infant or extreme prematurity, fetal macrosomia, large fetal head, and other fetal anomalies (Figure 4) 77. Moloy HC. Studies on head molding during labor. Amer J Obstet Gynec. 1942;44(5):762-82. https://doi.org/10.1016/S0002-9378(15)30603-7
https://doi.org/10.1016/S0002-9378(15)30...
,88. Bhagwanani SG, Price HV, Lawrence KM, Ginz B. Risks and prevention of cervical cord injury in the management of breech presentations with hyperextension of the fetal head. Am J Obst Gynec. 1973;115(8):1159-61. https://doi.org/10.1016/0002-9378(73)90573-5
https://doi.org/10.1016/0002-9378(73)905...
,4040. Gowers WR. Clinical lecture on birth palsies. Lancet. 1888;131(3372):709-11. https://doi.org/10.1016/S0140-6736(02)15842-9
https://doi.org/10.1016/S0140-6736(02)15...
.
TYPES OF PERINATAL TRAUMA
The scalp has five layers: (1) skin, (2) subcutaneous connective tissue, (3) galea aponeurotica, (4) loose areolar connective tissue, and (5) pericranium (periosteum). The skull comprises 22 separated bones, mostly connected by fibrous sutures. The cranium has several parts; the calvarial vault, cranial base, and facial skeleton (Figure 5)1111. Cole VA, Durbin GM, Olaffson A, Reynolds EOR, Rivers RPA, Smith JF. Pathogenesis of intraventricular haemorrhage in newborn infants. Arch Dis Child. 1974;49(9):722-8. https://doi.org/10.1136/adc.49.9.722
https://doi.org/10.1136/adc.49.9.722...
,1212. Ford FR. Breech delivery in its possible relation to injury of the spinal cord with special reference to infantile paraplegia. Arch Neurol Psychiat. 1925;14(6):742-50. https://doi.org/10.1001/archneurpsyc.1925.02200180013002
https://doi.org/10.1001/archneurpsyc.192...
.
Illustration of the five layers of the scalp: skin, subcutaneous connective tissue, galea aponeurotica, loose areolar tissue connective tissue, pericranium. Extracranial traumatic lesions.
There are three types of perinatal trauma, each with various subtypes. These are: (1) extracranial hemorrhage; (2) skull fracture; and (3) intracranial hemorrhage.
Extracranial traumatic lesions
There are three different types of extracranial hemorrhages: (1) caput succedaneum; (2) subgaleal hemorrhage; (3) cephalohematoma (Figure 6).
Caput succedaneum
This type of edematous swelling of the skin, dense and loose connective tissue, and galea is very common following vaginal delivery. It is a subcutaneous collection of extra-periosteal fluid over the presenting part in a vertex presentation, caused by compression exerted mainly by the cervix. The edema is soft and superficial, and does not involve the periosteum1313. Freud S. Die Infantile CerebralHihmung: Specielle Pathologie und Therapie. Wien;1897.. No treatment is necessary, as the edema resolves in a few days. It extends beyond the suture lines, is usually associated with molding of the skull bones, appears at birth and resolves in few days, requiring no treatment1414. Nicholson L. Caput succedaneum and cephalohematoma: the Cs that leaves bumps on the head. Neonatal Netw. 2007;26(5):277-81. https://doi.org/10.1891/0730-0832.26.5.277
https://doi.org/10.1891/0730-0832.26.5.2...
.
Subgaleal hemorrhage
This lesion is much less common than the caput succedaneum. It is subaponeurotic (under skull aponeurosis); spreads rapidly over the head downward to the eye; is very uncommon; may be associated with serious blood loss; may be associated with intracranial hemorrhage; and is 90% associated with vacuum extraction. It requires no treatment unless there is shock or intracranial hemorrhage that requires blood transfusion. The hemorrhage is beneath the aponeurosis (galea), peripheral to the periosteum. Coagulapathies may be present, so emergency coagulation and bleeding studies should be performed. The lesion frequently enlarges after birth and presents as a fluctuant, firm mass. The blood is liquid, and epicranial aspiration is frequently necessary. The procedure is easily performed under local anesthesia after the area has been shaved. Aspiration must be done with all the necessary surgical asepsis. Following aspiration, a compressive dressing is applied. Blood transfusion may be necessary since the subgaleal space is large. Repeated aspirations may occasionally be necessary1313. Freud S. Die Infantile CerebralHihmung: Specielle Pathologie und Therapie. Wien;1897.,1414. Nicholson L. Caput succedaneum and cephalohematoma: the Cs that leaves bumps on the head. Neonatal Netw. 2007;26(5):277-81. https://doi.org/10.1891/0730-0832.26.5.277
https://doi.org/10.1891/0730-0832.26.5.2...
,1515. Harcke HT Jr, Naeye RL, Storch A, Blanc WA. Perinatal cerebral intraventricular hemorrhage. J Pediatr. 1972;80(1):37-42. https://doi.org/10.1016/S0022-3476(72)80450-5
https://doi.org/10.1016/S0022-3476(72)80...
.
Cephalohematoma
Cephalohematoma is confined to the space between the outer table of the skull and the periosteum, and is located between the cranial sutures. It is present in 1% to 2% of “normal” deliveries. Years ago, when midforcep deliveries were common, the incidence of cephalohematoma was higher. Midforceps are rarely used in modern obstetrical practice; thus, the incidence of cephalohematoma is decreasing. The hemorrhage in cephalohematoma is subperiosteal, which explains why the hematoma is confined by the sutures. Most hematomas are unilateral and located over the parietal bone. From 10% to 25% of the cases are caused by an underlying skull fracture. Both skull fracture and stripping of the periosteum from the underlying skull result from the crushing and sheering forces that uterine compression and squeezing of the skull within the pelvic outlet cause (Figure 6)1616. Harwood-Nash DC, Hendrick EB, Hudson AR. The significance of skull fractures in children: a study of 1,187 patients. RadioIogy. 1971;101(1):151-5. https://doi.org/10.1148/101.1.151
https://doi.org/10.1148/101.1.151...
,1717. Korobkin R. The relationship between head circumference and the development of communicating hydrocephalus in infants following intraventricular hemorrhage. Pediatrics. 1975;56(1):74-7..
The lesion presents as a tense, firm mass after birth, frequently enlarging during the first few days. It may be painful due to the stretching of the periosteum during the first days. Typically, the hematoma is contained by the periosteum adhesions to the cranial sutures (Figures (6, (7). Palpated by a finger along the margin of the lesion, the elevated periosteum can be felt as a ridge with a recessed center, a configuration that is often mistaken for a depressed skull fracture. From a neurosurgical point of view, cephalohematoma is rarely of clinical importance unless an intracranial complicating lesion is present. Infected cephalohematomas and meningitis are extremely rare. Usually, cephalohematomas resolve within a few weeks but it takes a longer time than a subgaleal hemorrhage1313. Freud S. Die Infantile CerebralHihmung: Specielle Pathologie und Therapie. Wien;1897..
Calcifications may occur, but they gradually disappear over several months. In most cases no treatment is necessary. In rare cases aspiration is indicated. This procedure is performed in the same way as for subgaleal hematomas1818. Krishnamoorthy KS, Shannon DC, DeLong GR, Todres ID, Davis KR. Neurologic sequelae in the survivors of neonatal intraventricular hemorrhage. Pediatrics. 1979;64(2):233-7..
SKULL FRACTURES
There are three different types of skull fractures: (1) linear, (2) depressed, and (3) occipital osteodiastasis.
Linear fractures
Linear fractures are most commonly located in the parietal bones (Figure 8). The identification of this type of fracture depends on the frequency of x-ray examination. Even with a picture of good quality, a linear fracture may not be detected in about 20% of the cases examined. Consequently, it is very difficult to determine the precise incidence. Associated lesions include cephalohematoma, epidural hematoma, subdural hematoma, dural tears, and cerebral contusion. Except for cephalohematomas, the other lesions are rare. Linear fractures are all of traumatic origin1919. Little WJ. Course of lectures on the deformities of the human frame. Lancet. 1844;41(1072):809-15. https://doi.org/10.1016/S0140-6736(02)64521-0
https://doi.org/10.1016/S0140-6736(02)64...
,2020. McDonald MM, Koops BL, Johnson ML. Timing and etiology of intracranial hemorrhage in the newborn. In: Second Special Laboratories Conference on Perinatal Intracranial Hemorrhage. Columbus, Ohio: Ross Laboratories; 1982. p. 221-32.,2121. Ment LR, Duncan CC, Scott DT, Ehrenkranz RA. Posthemorrhagic hydrocephalus: low incidence in very low birth weight neonates with intraventricular hemorrhage. J Neurosurg. 1984;60(2):343-7. https://doi.org/10.3171/jns.1984.60.2.0343
https://doi.org/10.3171/jns.1984.60.2.03...
.
Linear fractures are of no clinical importance and no therapy is indicated unless any of the above-mentioned associated lesions are present. Follow up is necessary when the skull fracture present with diastasis between its edges. We must be cognisant of growing skull fractures.
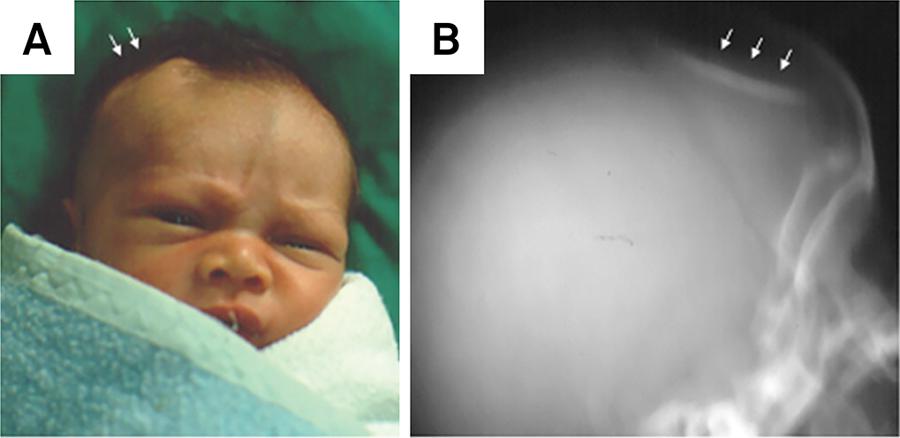
Depressed fractures
These fractures are commonly referred to as “ping-pong ball” lesions. They may be compared to a “green-stick” fracture of the long bones, as there is no loss of continuity. In fact, the depressed fracture is not a real fracture but an inward buckling of the unusually soft bone. Little has been written about the incidence of depressed fractures among a random group of deliveries, although 1/1,000 seems to be a reasonable figure. The parietal bone is the most common site for this lesion, followed by the frontal bone. Associated lesions are extremely rare. This “ping-pong ball” fracture is caused by a compressing force on the skull, such as forceps, the thumb, or the pressure of the head against the pelvic structures during delivery2222. Neville IS, Amorim RL, Paiva WS, Sanders FH, Teixeira MJ, Andrade AF. Early surgery does not seem to be a pivotal criterion to improve prognosis in patients with frontal depressed skull fractures. Biomed Res Int. 2014;2014:879286. https://doi.org/10.1155/2014/879286
https://doi.org/10.1155/2014/879286...
,2323. Natelson SE, Sayers MP. The fate of children sustaining severe head trauma during birth. Pediatrics. 1973;51(2):169-74..
Spontaneous elevation has usually corrected the depressed bone by the three-month follow-up. Usually, there is no need for surgery or any other form of therapy. There have been reports of elevation by vacuum extractor, breast pump, and digital pressure. The use of a vacuum extractor is not without danger and is not recommended. When neurosurgical expertise is available and digital compression or breast pumping has been unsuccessful, operative elevation should be undertaken. This is easily done under local anesthesia after a small area over the nearest suture line has been shaved. An incision of 1 cm in the skin down to the bone exposes the suture. A sharp periostal elevator is then gently introduced under the bone. The tip of the elevator is moved toward the center of the fracture; the impression is then easily elevated, but the surgeon must take care not to use the edge of the skull as a fulcrum lest he extend the depression2323. Natelson SE, Sayers MP. The fate of children sustaining severe head trauma during birth. Pediatrics. 1973;51(2):169-74.,2424. Roberts MH. The spinal fluid in new-born with special reference to intracranial hemorrhage. JAMA. 1925;85(7):500-3. https://doi.org/10.1001/jama.1925.02670070020006
https://doi.org/10.1001/jama.1925.026700...
(Figure 9).
Depressed skull fracture. A. Newborn demonstrating a “ping-pong fracture” in the right frontal bone; B. Corresponding x-ray of the skull (white arrows indicate the depressed area).
Occipital osteodiastasis
This lesion consists of a separation of the squamous and basal portions of the occipital bone. It may result in tentorial laceration, posterior fossa epidural hematoma, cerebellar contusion, or medullary compression. Prior to the use of CT scans, this lesion was detected only postmortem2525. Zalatimo O, Ranasinghe M, Dias M, Iantosca M. Treatment of depressed skull fractures in neonates using percutaneous microscrew elevation. J Neurosurg Pediatr. 2012;9(6):676-9. https://doi.org/10.3171/2012.2.PEDS11304
https://doi.org/10.3171/2012.2.PEDS11304...
(Figure 10).
Occipital osteodiatasis. Axial CT of the skull indicating bilateral osseous diastasis associated with focal subjacent hemorrhage.
INTRACRANIAL HEMORRHAGES
There are eight different types of intracranial hemorrhages: (1) epidural hemorrhage, (2) subdural hemorrhage, (3) infratentorial hemorrhages, (4) subarachnoid hemorrhage, (5) intraventricular hemorrhage, (6) intracerebellar hemorrhage, (7) cerebral contusion, (8) cerebellar contusion2525. Zalatimo O, Ranasinghe M, Dias M, Iantosca M. Treatment of depressed skull fractures in neonates using percutaneous microscrew elevation. J Neurosurg Pediatr. 2012;9(6):676-9. https://doi.org/10.3171/2012.2.PEDS11304
https://doi.org/10.3171/2012.2.PEDS11304...
,2626. Weiner EJ, McIntosh MS, Joseph MM, Maraqa N, Davis PG. Neonatal scalp abscess: is it a benign disease? J Emerg Med. 2011;40(5):e97-101. https://doi.org/10.1016/j.jemermed.2009.08.019
https://doi.org/10.1016/j.jemermed.2009....
.
Traumatic epidural hemorrhage
This hemorrhage occurs between the dura and the periosteum on the inner side of the bone. The lesion is rare in neonates. According to autopsy reports, only about 2% of neonatal intracranial hemorrhages are an epidural hematoma. The source of bleeding is one of the branches from the middle meningeal artery, a major venous sinus, or the diploe secondary to skull fracture. A linear skull fracture is usually present. If an infant, after experiencing a difficult labor or delivery, is eventually delivered by forceps or vacuum extractor and presents signs of increased intracranial pressure (bulging fontanels or separation of the sutures), an epidural hematoma should be suspected. An emergency CT scan should be performed, followed by careful judgment if evacuation of the hematoma is necessary. Small hematomas should be observed, depending on the time from the birth and the progression of the hematoma2626. Weiner EJ, McIntosh MS, Joseph MM, Maraqa N, Davis PG. Neonatal scalp abscess: is it a benign disease? J Emerg Med. 2011;40(5):e97-101. https://doi.org/10.1016/j.jemermed.2009.08.019
https://doi.org/10.1016/j.jemermed.2009....
,2727. Collins KA, Byard RW. Birth trauma. In: Collins KA, Byard RW. Forensic pathology of infancy and childhood. New York: Springer; 2014. p. 139-68.,2828. Habibi Z, Meybodi AT, Haji Mirsadeghi SM, Miri SM. Burr-hole drainage for the treatment of acute epidural hematoma in coagulopathic patients: a report of eight cases. J Neurotrauma. 2012;29(11):2103-7. https://doi.org/10.1089/neu.2010.1742
https://doi.org/10.1089/neu.2010.1742...
.
Traumatic subdural hemorrhage
Subdural hemorrhage may be caused by rupture of the bridging veins, tearing of the dural tent or foix, and deep cerebral veins. Damage to these vessels involves excessive deformations of the skull during the birth. Usually, the degree of hemorrhage is not severe, due to the low pressure system of the intracranial venous structures. Distribution of the subdural hemorrhage is very peculiar in birth trauma. It is usually found at the parieto-occipital convexity, the suboccipital area close to the tent, or the inter-hemispheric space. Cerebral contusion is a frequent finding with subdural hemorrhage, usually at the occipital lobe. Infratentorial hemorrhage is predominantly caused by tentorial laceration or occipital osteodiastasis. The clot may very rapidly result in lethal compression of the brain stem. Subdural hemorrhage in the newborn is always a traumatic lesion. The percentages of full-term babies and premature infants with subdural hemorrhage are now similar2929. Jung SW, Kim DW. Our experience with surgically treated epidural hematomas in children. J Korean Neurosurg Soc. 2012;51(4):215-8. https://doi.org/10.3340/jkns.2012.51.4.215
https://doi.org/10.3340/jkns.2012.51.4.2...
,3030. Rabelo NN, Silveira Filho LJ, Silva BNB, Cunha CD, Furtado IS, Valli DAB et al. Differential diagnosis between neoplastic and non-neoplastic brain lesions in radiology. Arq Bras Neurocir. 2016;35(1):45-61. https://doi.org/10.1055/s-0035-1570362
https://doi.org/10.1055/s-0035-1570362...
.
The symptoms and signs depend upon the extent of the hemorrhage. Irritability may be the only symptom of a minor hemorrhage. Bulging of the fontanel or sudden episodes of staring may be the initial presenting symptoms. Nuchal rigidity and opisthotonus, followed by bradycardia, pupil dilatation, respiratory failure and finally, cardiac arrest, may follow a massive hemorrhage3131. Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367..
Progression of a subdural supratentorial hemorrhage may extend to the infratentorial compartment leading to an infratentorial blood collection. There is usually no brain tissue involved in the origin of the bleeding. More commonly, bleeding originates from the tearing of the tent or from the deep draining veins and venous sinus of the brain. As this is usually a passive accumulation of blood, the clot has no compressive effect on the posterior fossa nervous structures. A CT scan and real-time sonography are definitive means of diagnosing the extent and severity of a subdural hemorrhage. Commonly, the prognosis is good3232. Swanson AE, Veldman A, Wallace EM, Malhotra A. Subgaleal hemorrhage: risk factors and outcomes. Acta Obstet Gynecol Scand. 2011;91(2):260-3. https://doi.org/10.1111/j.1600-0412.2011.01300.x
https://doi.org/10.1111/j.1600-0412.2011...
.
Traumatic subarachnoid hemorrhage
Primary subarachnoid hemorrhage refers to blood in the subarachnoid space, which is not an extension from a subdural, intracerebral, or intraventricular hemorrhage, nor from vascular anomalies. This kind of hemorrhage is very common in premature infants. There is a close correlation between bloody cerebrospinal fluid (CSF) in the newborn and retinal hemorrhage. Thus, extensive retinal hemorrhage generally indicates severe subarachoid hemorrhage3333. Gupta SN, Kechli AM, Kanamalla US. Intracranial hemorrhage in term newborns: management and outcomes. Pediatr Neurol. 2009;40(1):1-12. https://doi.org/10.1016/j.pediatrneurol.2008.09.019
https://doi.org/10.1016/j.pediatrneurol....
.
According to Volpe, the frequency of periventricular and intraventricular hemorrhage is much higher than that of primary subarachnoid hemorrhage. In infants weighing less than 2000 g, Volpe’s findings were 63% versus 29%, respectively2727. Collins KA, Byard RW. Birth trauma. In: Collins KA, Byard RW. Forensic pathology of infancy and childhood. New York: Springer; 2014. p. 139-68.,3131. Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367..
The source of bleeding in primary subarachnoid hemorrhage is presumably venous – that is, bridging veins within the subarachnoid space or small vessels in the meningeal plexus2424. Roberts MH. The spinal fluid in new-born with special reference to intracranial hemorrhage. JAMA. 1925;85(7):500-3. https://doi.org/10.1001/jama.1925.02670070020006
https://doi.org/10.1001/jama.1925.026700...
.
Complications from primary subarachnoid hemorrhage in the newborn are difficult to identify, except for hydrocephalus. Adhesions around the cisterna magna and the fourth ventricle may result in an obstruction to CSF circulation. Adhesions over the cerebral convexities may also impair CSF absorption or flow. Late sequelae from a primary subarachnoid hemorrhage are extremely difficult to prove. There is, however, some indication that children who sustain such hemorrhages have a higher incidence of pathological EEG, lower school performances, and higher incidence of minimal brain dysfunction. From an intellectual point of view, it is difficult to accept that even a small subarachnoid hemorrhage is of no importance for the growing brain, particularly for immature or premature infants. No extensive reports have been published in this field so far, however2525. Zalatimo O, Ranasinghe M, Dias M, Iantosca M. Treatment of depressed skull fractures in neonates using percutaneous microscrew elevation. J Neurosurg Pediatr. 2012;9(6):676-9. https://doi.org/10.3171/2012.2.PEDS11304
https://doi.org/10.3171/2012.2.PEDS11304...
.
Intraventricular ischemic-hypoxic hemorrhage
Subependymal and intraventricular hemorrhages are caused mainly by hypoxic/ischemic factors. Before the introduction of CT and ultrasound, information about the extent and incidence of intracranial hemorrhage was obtainable only through postmortem examination. With modern real-time ultrasonography, information concerning cerebral blood flow has been greatly improved. These sonar investigations are noninvasive and without side effects. Disturbances of autoregulation in asphyxia of premature infants is of importance in understanding the pathogenesis of intraventricular hemorrhage3434. Brittain C, Muthukumar P, Job S, Sanka S. “Ping pong” fracture in a term infant. BMJ Case Rep. 2012;2012:pii: bcr0120125631. https://doi.org/10.1136/bcr.01.2012.5631
https://doi.org/10.1136/bcr.01.2012.5631...
.
The anatomical factors in the pathogenesis of subependymal intraventricular hemorrhage now seem more clear. The localization of the hemorrhage is dependent upon the brain’s degree of development. During the first 24 to 32 weeks of gestation, the function of the basal ganglia, midbrain, and brain stem has developed. There are many newly-formed vessels in this part of the brain. The capillaries and veins here are very fragile, so that the hemorrhage occurs within this subependymal germinal matrix network. In infants of less than 28 weeks’ gestation, the hemorrhage usually starts below the head of the caudate nucleus and the thalamus2020. McDonald MM, Koops BL, Johnson ML. Timing and etiology of intracranial hemorrhage in the newborn. In: Second Special Laboratories Conference on Perinatal Intracranial Hemorrhage. Columbus, Ohio: Ross Laboratories; 1982. p. 221-32.. After 32 weeks, subependymal intraventricular hemorrhages are relatively rare. Hemorrhage in cortical and white matter increases with the length of gestation. The disturbance of hemodynamics in the cerebral vessels is of utmost importance in the pathogenesis of ventricular hemorrhage in premature infants. Normally, the cerebral blood flow is essentially independent of arterial pressure. This vascular autoregulation is disturbed in premature infants, however. A sudden increase in arterial blood pressure may lead to a rupture of the fragile capillaries in the germinal matrix. Especially important is perinatal asphyxia, which can lead to hypoxia, hypercapnia, and acidosis. Depressed autoregulation will finally produce cerebral vascular dilation. Increased cerebral perfusion leads to rupture of the vessels. In contrast, decreased venous return may induce an increase of the central venous pressure. This in turn increases cerebral perfusion pressure, thereby causing venous stasis, followed by rupture of vessels3030. Rabelo NN, Silveira Filho LJ, Silva BNB, Cunha CD, Furtado IS, Valli DAB et al. Differential diagnosis between neoplastic and non-neoplastic brain lesions in radiology. Arq Bras Neurocir. 2016;35(1):45-61. https://doi.org/10.1055/s-0035-1570362
https://doi.org/10.1055/s-0035-1570362...
,3131. Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367.,3232. Swanson AE, Veldman A, Wallace EM, Malhotra A. Subgaleal hemorrhage: risk factors and outcomes. Acta Obstet Gynecol Scand. 2011;91(2):260-3. https://doi.org/10.1111/j.1600-0412.2011.01300.x
https://doi.org/10.1111/j.1600-0412.2011...
. All of the factors that may disturb cerebral perfusion can induce intracerebral hemorrhage in the premature infant: soft cranial bones, traumatic delivery, asphyxia, hypertension, hypotonia, apnea, patent ductus arteriosus, hypoxia, hypercapnia, fast expansion of volume, respiratory disturbance, rupture of pulmonary alveoli, or careless transport and handling of the baby. About 50% of infants with a gestational age of 30 weeks or less have intracerebral hemorrhage. The incidence decreases after 30 weeks of gestation. The reason for this is possibly the maturing of the vascular system and better autoregulation. Hemorrhaging takes place in about 80% of infants within 72 hours following delivery. Later hemorrhages are generally rare3030. Rabelo NN, Silveira Filho LJ, Silva BNB, Cunha CD, Furtado IS, Valli DAB et al. Differential diagnosis between neoplastic and non-neoplastic brain lesions in radiology. Arq Bras Neurocir. 2016;35(1):45-61. https://doi.org/10.1055/s-0035-1570362
https://doi.org/10.1055/s-0035-1570362...
.
A frequent complication following hemorrhage is post-hemorrhagic hydrocephalus. Sonographical investigations have shown the following: (1) transitory hydrocephalus; (2) persistent but not progressive hydrocephalus; (3) persistent, fast, and progressive hydrocephalus; (4) cerebral atrophy3131. Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367.. Sonography has further shown that the hydrocephalic process starts about two weeks after the hemorrhage. It is important to realize that the process starts about two weeks before a pathological increase of the head circumference can be measured. Sonographically, post-hemorrhagic hydrocephalus can be visualized while the head circumference still follows the normal curve3636. Ekéus C, Högberg U, Norman M. Vacuum assisted birth and risk for cerebral complications in term newborn infants: a population-based cohort study. BMC Pregnancy Childbirth. 2014;14(1):36. https://doi.org/10.1186/1471-2393-14-36
https://doi.org/10.1186/1471-2393-14-36...
,3737. Reichard R. Birth injury of the cranium and central nervous system. Brain Pathol. 2008;18(4):565-70. https://doi.org/10.1111/j.1750-3639.2008.00205.x
https://doi.org/10.1111/j.1750-3639.2008...
. A typical lesion found in the immature brain is caused by hypoxia/ischemia in the periventricular areas. Hypoperfusion starts this process. Secondly, hemorrhagic infarctions are formed, which later may be transformed into cysts and porencephalic cavities. Such lesions close to the ventricles affect the pyramidal tracts in the internal capsule, resulting in a spastic diplegia3131. Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367..
Intracerebellar hemorrhage
This lesion is a relatively common postmortem finding in premature babies. Recent reports indicate an increasing frequency of such findings in modern neonatal intensive care units. The reason for this seems to be the active attitude toward treatment of low birthweight premature infants. The neuropathology and pathogenesis is very similar to those for cerebral hemorrhage and there seems to be a strong correlation between intraventricular hemorrhage and cerebellar hemorrhage3535. Kerr HA. Closed head injury. Clin Sports Med. 2013;32(2):273-87. https://doi.org/10.1016/j.csm.2012.12.008
https://doi.org/10.1016/j.csm.2012.12.00...
.
Clinically, there is a strong association between respiratory distress and perinatal asphyxia. The most consistent signs are rapid deterioration with bradycardia, apnea, hemorrhagic CSF, and falling hematocrit3434. Brittain C, Muthukumar P, Job S, Sanka S. “Ping pong” fracture in a term infant. BMJ Case Rep. 2012;2012:pii: bcr0120125631. https://doi.org/10.1136/bcr.01.2012.5631
https://doi.org/10.1136/bcr.01.2012.5631...
. The suspicion of cerebellar hemorrhage is essential for proper diagnosis. A premature infant with brain stem dysfunction and bloody CSF should promptly be referred for a CT scan. The possibility of cerebellar herniation always exists when spinal taps are performed. If a CT scan is immediately available, the spinal tap is preferably deferred. The prognosis is usually very dubious, however. Some infants who survive intracerebellar hemorrhage develop hydrocephalus. Management depends on the CT scan finding. If a well-circumscribed hematoma is visualized on the scan, an emergency suboccipital craniotomy with evacuation of the clot should be performed. Conditions that argue against surgery are associated cerebral lesions, serious pulmonary disorders, or other systemic disorders2626. Weiner EJ, McIntosh MS, Joseph MM, Maraqa N, Davis PG. Neonatal scalp abscess: is it a benign disease? J Emerg Med. 2011;40(5):e97-101. https://doi.org/10.1016/j.jemermed.2009.08.019
https://doi.org/10.1016/j.jemermed.2009....
.
Traumatic brain contusion
The incidence rate for cerebral contusion is unknown; presumably, it is a relatively rare lesion. There is no specific therapy for this perinatal trauma.
A typical lesion found in the immature brain is caused by hypoxia/ischemia in the periventricular areas. Hypoperfusion starts this process. Secondly, hemorrhagic infarctions are formed, which later may be transformed into cysts and porencephalic cavities. Such lesions close to the ventricles affect the pyramidal tracts in the internal capsula, resulting in a spastic diplegia3636. Ekéus C, Högberg U, Norman M. Vacuum assisted birth and risk for cerebral complications in term newborn infants: a population-based cohort study. BMC Pregnancy Childbirth. 2014;14(1):36. https://doi.org/10.1186/1471-2393-14-36
https://doi.org/10.1186/1471-2393-14-36...
.
Cerebellar contusion
This lesion is often found with occipital osteodiastasis. The pathogenesis and pathology are very similar to those of cerebral contusions. Treatment with serial lumbar punctures in order to stop post-hemorrhagic ventricular dilation has been recommended. The beneficial effect is the acceleration of CSF absorption by removal of protein and blood, in addition to the reduction of CSF pressure. It is still unclear whether this procedure is really effective, however; controlled studies have to be performed. In addition to serial lumbar taps, drugs that decrease CSF production (e.g., furosemide, acetazolamide, digitoxin, glycerol, and isosorbide) may be used. Compressive head wrapping to arrest progressive hydrocephalus has been recommended in young infants, particularly when associated with myelomeningocele and Chiari II malformation. Because the premature infant, in particular, has a very compliant skull, cranial distortion may lead to severe hemodynamic alterations. The possibility of provoking an extension of the hemorrhage or lesions is significant; consequently, this treatment not recommended3737. Reichard R. Birth injury of the cranium and central nervous system. Brain Pathol. 2008;18(4):565-70. https://doi.org/10.1111/j.1750-3639.2008.00205.x
https://doi.org/10.1111/j.1750-3639.2008...
.
Facial nerve injury
Facial paralysis refers to weakness of the facial muscles and indicates injury to the facial nerve, that is, involvement of the muscles of the upper, as well as lower, face. Injury to the facial nerve is the most common manifestation of birth trauma. The incidence rate was 6.4% when the child was examined on the second day of life. There was no difference in incidence for children delivered manually versus by forceps3838. Hehir MP, Reidy FR, Wilkinson MN, Mahony R. Increasing rates of operative vaginal delivery across two decades: accompanying outcomes and instrument preferences. Eur J Obstet Gynecol Reprod Biol. 2013;171(1):40-3. https://doi.org/10.1016/j.ejogrb.2013.08.015
https://doi.org/10.1016/j.ejogrb.2013.08...
,3939. Manning JJ, Adour KK, Facial paralysis in children. Pediatrics. 1972;49(1):102-9.. In view of the favorable prognosis it is likely that edema or hemorrhage into the nerve sheath is present. The site of the lesion is at the exit of the nerve from the stylomastoid foramen, where the nerve divides into its two major branches: the temporofacial and the cervicofacial nerves. Hepner’s work presents strong evidence that intrauterine pressure of the mother’s sacral promonotory on the fetal facial nerve is the cause of the injury. In each of his 56 cases of facial paralysis, there was a direct correlation between the affected side of the face and the position of the head in utero3838. Hehir MP, Reidy FR, Wilkinson MN, Mahony R. Increasing rates of operative vaginal delivery across two decades: accompanying outcomes and instrument preferences. Eur J Obstet Gynecol Reprod Biol. 2013;171(1):40-3. https://doi.org/10.1016/j.ejogrb.2013.08.015
https://doi.org/10.1016/j.ejogrb.2013.08...
,3939. Manning JJ, Adour KK, Facial paralysis in children. Pediatrics. 1972;49(1):102-9.,4040. Gowers WR. Clinical lecture on birth palsies. Lancet. 1888;131(3372):709-11. https://doi.org/10.1016/S0140-6736(02)15842-9
https://doi.org/10.1016/S0140-6736(02)15...
.
CONCLUSION
The frequency of birth injuries has steadily diminished in the industrialized world during the last decades. As a consequence of this, an increasing number of children are delivered by Caesarean section today. The frequency of brain damage increases almost proportionally with decreasing birth weight, and is mainly related to ischemic-hypoxic injuries. Traumatic injuries usually have a good prognosis. Real-time ultrasound scanners have shown detailed and depressing pictures of severe intracerebral hemorrhages, particularly in the very low birth weight premature infant. This kind of brain damage is invariably followed by extensive neurological deficits. Issues of birth trauma should be a part of obstetricians’ basic education since its successful management includes special training, teamwork, and an individual approach.
Birth-related traumatic brain injuries cause serious complications and death in newborn infants. The condition also impacts the close relatives. There are three major categories, extracranial hemorrhages, skull fractures and intracranial hemorrhages, that relate to traumatic brain injury. Depending on the region and the extent of the damage, symptoms differ and treatment varies.
References
-
1Abroms JF, Bresnan MJ, Zuckerman JE, Fischer EG, Strand R. Cervical cord injuries secondary to hyperextension of the head in breech presentations. Obstet Gynecol. 1973;41(3):369-78.
-
2Stoll BJ, Kliegman RM. Delivery room emergencies. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson Text book of pediatrics. 17th ed. Philadelphia, PA: Saunders; 2004. p. 569-72.
-
3Robert MK, Bonita S, Joseph SG, Nina FS. Fetus and the neonatal birth injury. In: Behrman RE, Kliegman RM, Jenson HB, editors. Nelson text book of pediatrics. 15th ed. Philadelphia, PA: Saunders; 1996. p. 561-6.
-
4Hankins GD, Clark SM, Munn MB. Cesarean section on request at 39 weeks: impact on shoulder dystocia, fetal trauma, neonatal encephalopathy, and intrauterine fetal demise. Semin Perinatol. 2006;30(5):276-87. https://doi.org/10.1053/j.semperi.2006.07.009
» https://doi.org/10.1053/j.semperi.2006.07.009 -
5Henderson SG, Sherman LS. The Roentgen anatomy of the skull in the newborn infant. Radiology. 1946;46(7):107-18. https://doi.org/10.1148/46.2.107
» https://doi.org/10.1148/46.2.107 -
6Lindgren L. The lower parts of the uterus during the first stage of labour in occipito-anterior vertex presentation: studies by means of intrauterine tokography. Acta Obstet Gynecol Scand. 1955;34(Suppl 2):1-79. https://doi.org/10.3109/00016345509154530
» https://doi.org/10.3109/00016345509154530 -
7Moloy HC. Studies on head molding during labor. Amer J Obstet Gynec. 1942;44(5):762-82. https://doi.org/10.1016/S0002-9378(15)30603-7
» https://doi.org/10.1016/S0002-9378(15)30603-7 -
8Bhagwanani SG, Price HV, Lawrence KM, Ginz B. Risks and prevention of cervical cord injury in the management of breech presentations with hyperextension of the fetal head. Am J Obst Gynec. 1973;115(8):1159-61. https://doi.org/10.1016/0002-9378(73)90573-5
» https://doi.org/10.1016/0002-9378(73)90573-5 -
9Bucher HU, Boltshauser E, Friderich J, Isler W. Birth injury to the spinal cord. Helv Paediatr Acta. 1979;34(6):517-27.
-
10Byers RK. Spinal-cord injuries during birth. Dev Med Child Neurol. 1975;17(1):103-10. https://doi.org/10.1111/j.1469-8749.1975.tb04967.x
» https://doi.org/10.1111/j.1469-8749.1975.tb04967.x -
11Cole VA, Durbin GM, Olaffson A, Reynolds EOR, Rivers RPA, Smith JF. Pathogenesis of intraventricular haemorrhage in newborn infants. Arch Dis Child. 1974;49(9):722-8. https://doi.org/10.1136/adc.49.9.722
» https://doi.org/10.1136/adc.49.9.722 -
12Ford FR. Breech delivery in its possible relation to injury of the spinal cord with special reference to infantile paraplegia. Arch Neurol Psychiat. 1925;14(6):742-50. https://doi.org/10.1001/archneurpsyc.1925.02200180013002
» https://doi.org/10.1001/archneurpsyc.1925.02200180013002 -
13Freud S. Die Infantile CerebralHihmung: Specielle Pathologie und Therapie. Wien;1897.
-
14Nicholson L. Caput succedaneum and cephalohematoma: the Cs that leaves bumps on the head. Neonatal Netw. 2007;26(5):277-81. https://doi.org/10.1891/0730-0832.26.5.277
» https://doi.org/10.1891/0730-0832.26.5.277 -
15Harcke HT Jr, Naeye RL, Storch A, Blanc WA. Perinatal cerebral intraventricular hemorrhage. J Pediatr. 1972;80(1):37-42. https://doi.org/10.1016/S0022-3476(72)80450-5
» https://doi.org/10.1016/S0022-3476(72)80450-5 -
16Harwood-Nash DC, Hendrick EB, Hudson AR. The significance of skull fractures in children: a study of 1,187 patients. RadioIogy. 1971;101(1):151-5. https://doi.org/10.1148/101.1.151
» https://doi.org/10.1148/101.1.151 -
17Korobkin R. The relationship between head circumference and the development of communicating hydrocephalus in infants following intraventricular hemorrhage. Pediatrics. 1975;56(1):74-7.
-
18Krishnamoorthy KS, Shannon DC, DeLong GR, Todres ID, Davis KR. Neurologic sequelae in the survivors of neonatal intraventricular hemorrhage. Pediatrics. 1979;64(2):233-7.
-
19Little WJ. Course of lectures on the deformities of the human frame. Lancet. 1844;41(1072):809-15. https://doi.org/10.1016/S0140-6736(02)64521-0
» https://doi.org/10.1016/S0140-6736(02)64521-0 -
20McDonald MM, Koops BL, Johnson ML. Timing and etiology of intracranial hemorrhage in the newborn. In: Second Special Laboratories Conference on Perinatal Intracranial Hemorrhage. Columbus, Ohio: Ross Laboratories; 1982. p. 221-32.
-
21Ment LR, Duncan CC, Scott DT, Ehrenkranz RA. Posthemorrhagic hydrocephalus: low incidence in very low birth weight neonates with intraventricular hemorrhage. J Neurosurg. 1984;60(2):343-7. https://doi.org/10.3171/jns.1984.60.2.0343
» https://doi.org/10.3171/jns.1984.60.2.0343 -
22Neville IS, Amorim RL, Paiva WS, Sanders FH, Teixeira MJ, Andrade AF. Early surgery does not seem to be a pivotal criterion to improve prognosis in patients with frontal depressed skull fractures. Biomed Res Int. 2014;2014:879286. https://doi.org/10.1155/2014/879286
» https://doi.org/10.1155/2014/879286 -
23Natelson SE, Sayers MP. The fate of children sustaining severe head trauma during birth. Pediatrics. 1973;51(2):169-74.
-
24Roberts MH. The spinal fluid in new-born with special reference to intracranial hemorrhage. JAMA. 1925;85(7):500-3. https://doi.org/10.1001/jama.1925.02670070020006
» https://doi.org/10.1001/jama.1925.02670070020006 -
25Zalatimo O, Ranasinghe M, Dias M, Iantosca M. Treatment of depressed skull fractures in neonates using percutaneous microscrew elevation. J Neurosurg Pediatr. 2012;9(6):676-9. https://doi.org/10.3171/2012.2.PEDS11304
» https://doi.org/10.3171/2012.2.PEDS11304 -
26Weiner EJ, McIntosh MS, Joseph MM, Maraqa N, Davis PG. Neonatal scalp abscess: is it a benign disease? J Emerg Med. 2011;40(5):e97-101. https://doi.org/10.1016/j.jemermed.2009.08.019
» https://doi.org/10.1016/j.jemermed.2009.08.019 -
27Collins KA, Byard RW. Birth trauma. In: Collins KA, Byard RW. Forensic pathology of infancy and childhood. New York: Springer; 2014. p. 139-68.
-
28Habibi Z, Meybodi AT, Haji Mirsadeghi SM, Miri SM. Burr-hole drainage for the treatment of acute epidural hematoma in coagulopathic patients: a report of eight cases. J Neurotrauma. 2012;29(11):2103-7. https://doi.org/10.1089/neu.2010.1742
» https://doi.org/10.1089/neu.2010.1742 -
29Jung SW, Kim DW. Our experience with surgically treated epidural hematomas in children. J Korean Neurosurg Soc. 2012;51(4):215-8. https://doi.org/10.3340/jkns.2012.51.4.215
» https://doi.org/10.3340/jkns.2012.51.4.215 -
30Rabelo NN, Silveira Filho LJ, Silva BNB, Cunha CD, Furtado IS, Valli DAB et al. Differential diagnosis between neoplastic and non-neoplastic brain lesions in radiology. Arq Bras Neurocir. 2016;35(1):45-61. https://doi.org/10.1055/s-0035-1570362
» https://doi.org/10.1055/s-0035-1570362 -
31Hockenberry M, Wilson D. Wong’s essentials of pediatric nursing. 9th ed. Saint Louis: Mosby, 2013. Health problems of newborns, p. 274-367.
-
32Swanson AE, Veldman A, Wallace EM, Malhotra A. Subgaleal hemorrhage: risk factors and outcomes. Acta Obstet Gynecol Scand. 2011;91(2):260-3. https://doi.org/10.1111/j.1600-0412.2011.01300.x
» https://doi.org/10.1111/j.1600-0412.2011.01300.x -
33Gupta SN, Kechli AM, Kanamalla US. Intracranial hemorrhage in term newborns: management and outcomes. Pediatr Neurol. 2009;40(1):1-12. https://doi.org/10.1016/j.pediatrneurol.2008.09.019
» https://doi.org/10.1016/j.pediatrneurol.2008.09.019 -
34Brittain C, Muthukumar P, Job S, Sanka S. “Ping pong” fracture in a term infant. BMJ Case Rep. 2012;2012:pii: bcr0120125631. https://doi.org/10.1136/bcr.01.2012.5631
» https://doi.org/10.1136/bcr.01.2012.5631 -
35Kerr HA. Closed head injury. Clin Sports Med. 2013;32(2):273-87. https://doi.org/10.1016/j.csm.2012.12.008
» https://doi.org/10.1016/j.csm.2012.12.008 -
36Ekéus C, Högberg U, Norman M. Vacuum assisted birth and risk for cerebral complications in term newborn infants: a population-based cohort study. BMC Pregnancy Childbirth. 2014;14(1):36. https://doi.org/10.1186/1471-2393-14-36
» https://doi.org/10.1186/1471-2393-14-36 -
37Reichard R. Birth injury of the cranium and central nervous system. Brain Pathol. 2008;18(4):565-70. https://doi.org/10.1111/j.1750-3639.2008.00205.x
» https://doi.org/10.1111/j.1750-3639.2008.00205.x -
38Hehir MP, Reidy FR, Wilkinson MN, Mahony R. Increasing rates of operative vaginal delivery across two decades: accompanying outcomes and instrument preferences. Eur J Obstet Gynecol Reprod Biol. 2013;171(1):40-3. https://doi.org/10.1016/j.ejogrb.2013.08.015
» https://doi.org/10.1016/j.ejogrb.2013.08.015 -
39Manning JJ, Adour KK, Facial paralysis in children. Pediatrics. 1972;49(1):102-9.
-
40Gowers WR. Clinical lecture on birth palsies. Lancet. 1888;131(3372):709-11. https://doi.org/10.1016/S0140-6736(02)15842-9
» https://doi.org/10.1016/S0140-6736(02)15842-9
Publication Dates
-
Publication in this collection
Mar 2017
History
-
Received
08 Nov 2016 -
Accepted
08 Dec 2016