Abstracts
The authors conducted a mycologic, immunochemical and molecular biology study on two strains of Paracoccidioides brasiliensis, one of them, called IBIÁ, isolated from soil in the municipality of IBIÁ (Minas Gerais) by Silva-Vergara et al. (l996,1998)20,21, and the other, BAT, cultivated from a human case of paracoccidioidomycosis in Ribeirão Preto (São Paulo/Brazil) by Freitas da Silva (l996)6. Both strains showed cotton-like (M) and yeast-like (Y) forms and were pathogenic for testicularly inoculated guinea pigs, producing granulomatous and/or suppurative orchitis. Immunochemically was demonstrated the presence of gp43 by double immunodiffusion, immunoelectrophoresis and immunoblotting.
Paracoccidioides brasiliensis; Paracoccidioidomycosis; Immunochemical study
Os Autores estudaram do ponto de vista micológico, imunoquímico e de sua biologia molecular, duas amostras de Paracoccidioides brasiliensis, uma isolada do solo, no município de IBIÁ (MG) por Silva-Vergara et al. (l996,1998)20,21 denominada IBIÁ e outra, BAT, cultivada de um caso humano de paracoccidioidomicose em Ribeirão Preto (SP) por Freitas da Silva (l996)6. Tais amostras apresentam colônias cotonosa (M) e leveduriforme (L ou Y), sendo patogênicas para cobaios inoculados por via testicular, produzindo orquite granulomatosa e/ou supurativa. Do ponto de vista imunoquímico, através de provas de Imunodifusão dupla, Imunoeletroforese e Western Blotting, foi demonstrada a presença da gp43. A sequência de nucleotídeos do DNA de tais amostras, através do seqüenciamento de 761 bases, revelou homologia de 100% com amostra padrão de P. brasiliensis, o mesmo ocorrendo com três amostras humanas, uma isolada de fezes de pinguim e outra de ração alimentar para cães contaminada com terra. São discutidos vários aspectos dos resultados obtidos, comparando-os com alguns dados da literatura.
Paracoccidioides brasiliensis.
A MYCOLOGIC AND IMMUNOCHEMICAL STUDY OF TWO STRAINS
Carlos da Silva LACAZ(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ), Mônica Scarpelli Martinelli VIDAL(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ), Elisabeth Maria HEINS-VACCARI(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ), Natalina Takahashi de MELO(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ), Gilda Maria Barbaro DEL NEGRO(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ), Giovana Letícia Hernandez ARRIAGADA(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. ) & Roseli dos Santos FREITAS(1 (1 ) Laboratório de Micologia Médica do Instituto de Medicina Tropical de São Paulo/LIM 53 HCFMUSP, São Paulo, SP, Brasil. )
SUMMARY
The authors conducted a mycologic, immunochemical and molecular biology study on two strains of Paracoccidioides brasiliensis, one of them, called IBIÁ, isolated from soil in the municipality of IBIÁ (Minas Gerais) by Silva-Vergara et al. (l996,1998)20,21, and the other, BAT, cultivated from a human case of paracoccidioidomycosis in Ribeirão Preto (São Paulo/Brazil) by Freitas da Silva (l996)6. Both strains showed cotton-like (M) and yeast-like (Y) forms and were pathogenic for testicularly inoculated guinea pigs, producing granulomatous and/or suppurative orchitis. Immunochemically was demonstrated the presence of gp43 by double immunodiffusion, immunoelectrophoresis and immunoblotting.
KEYWORDS:Paracoccidioides brasiliensis; Paracoccidioidomycosis; Immunochemical study.
INTRODUCTION
Several strains of P. brasiliensis isolated from human sources, as well as from soil, from armadillo organs (Dasypus novemcinctus) and from the feces of a penguin (Pygoscelis adeliae) from the Uruguayan Antarctic have been studied from the mycologic and immunochemical aspects, and also in terms of molecular biology. The methodology consist on obtaining the M and Y forms of the fungus inoculated into the testis of guinea pigs for the reproduction of specific orchitis, identification of gp43 by double immunodiffusion, immunoelectrophoresis and immunoblotting, and sequencing of the ribosomal gene 28 S of P. brasiliensis.
Biochemical tests are not routinely utilized because they do not permit the identification of P. brasiliensis.
In Brazil, tests with strains isolated from dog food contaminated with soil in Uberlândia (Minas Gerais state) by FERREIRA et al. (1990)5, from the feces of an Antarctic penguin (GARCIA et al., 1993)7, and from armadillo organs (VIDAL et al., 1995)23, as well as SILVA-VERGARA (l996)20 and SILVA-VERGARA (1998)21 on strains isolated from the soil of a coffee plantation in IBIÁ (Minas Gerais state), showed homogeneity in nucleotide sequencing (SANDHU et al., 1997)17.
The studies about P. brasiliensis strains isolated from patients, from animals (JOHNSON & LANG, 1977 9, GROSE & TAMSITT 1965 8, PERAÇOLI et al., 1992 16, BAGAGLI et al., 1998) 1 and from soil (MENDELOVICI et al., 1974 14, FERREIRA, 1990 5, SILVA-VERGARA, 199619,20, 199821) will permit the comparison about virulence towards isogenic animals, as well as the determination of their antigenic variability, already studied by NICODEMOS (1996)15 and MATTAR et al. (1997)13, and their karyotype variability, previously evaluated by CANO et al. (1998)4.
MATERIAL AND METHODS
The methodology used in identification of this two strains was:
Inoculation: Cultures of the IBIÁ and BAT strains maintained on agar-Sabouraud at room temperature were utilized to prepare a suspension in saline solution. Two guinea pigs per strain were inoculated with 0.25 ml of the suspension by the intratesticular route (approximate weight of the animals: 200 g).
Histopathology: The histological sections were stained with hematoxylin-eosin (HE) and by the Gomori-Grocott method.
Antigen preparation: The IBIÁ and BAT strains were cultivated on agar NGTA (1.6% neopeptone, 1% glucose, 0.01% thiamine and 0.02% asparagine) at 37º C for 10 days under constant shaking. After growth, the cultures were killed by adding timerosal at a final concentration of 1:5000 and filtered through no. l Whatman filter paper. The filtered cultures were concentrated 10 times with polyethyleneglycol (PEG) and dialyzed for 48 hours with destilated water. Proteins were determined by the method of Lowry et al.11. The antigens were kept at 4°C until the time for use.
Ouchterlony Double Immunodifusion: The antigens were evaluated with: a) sera from patients with paracoccidioidomycosis, b) polyclonal anti-gp43 rabbit serum c) anti-P. brasiliensis rabbit serum.
To evaluate cross-reactions were used histoplasmosis' and aspergillosis' patients sera.
Immunoelectrophoresis: The Siqueira technique (1982)22 was applied to polyclonal anti-P. brasiliensis rabbit serum for observation of the E arc of YARZÁBAL24.
SDS-PAGE: Electrophoresis on 12.5% gel was performed by the Laemmli's method10. We applied 40 µl of antigen through each well and electrophoresis was run at 100 volts for five hours. The gel was stained with silver nitrate by the Maniatis' method12.
Immunoblotting: The protein fractions obtained by SDS-PAGE were transferred to nitrocellulose paper, according to the MANIATIS12. We used three paracoccidioidomycosis' patient sera at 1:20 dilution. The conjugate used was a human anti-lgG labeled with peroxidase. Diaminobenzidine was used as a developer.
RESULTS
1. Macro- and microscopic aspects of the colonies: The IBIÁ and BAT strains are dimorphic and the M and Y forms were visible on Sabouraud-agar at 25ºC, as well as on Fava Netto-agar at 37°C ( Fig. 1 and 2). Microscopic examination of the M and Y forms revealed septate mycelial filaments with intercalated and/or terminal chlamydoconidia, and yeast-like cells with single or multiple buds, characteristic of P. brasiliensis.
Paracoccidioides brasiliensis (IBIÁ strain). Aspect of the colony growing at 25°C on Sabouraud-agar (M form) and at 37°C on Fava Netto agar medium (L form).
- Paracoccidioides brasiliensis (BAT strain). Aspect of the colony growing at 25°C on Sabouraud-agar (M-form) and at 37°C on Fava Netto-agar medium (L form).
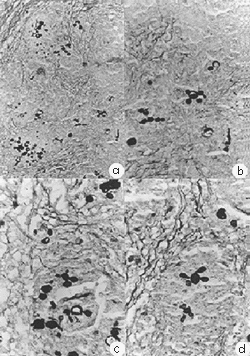
Fig. 3 - Paracoccidioides brasiliensis (IBIÁ strain). Histopathological examination of a guinea pig testis stained with Gomori a) 200X b) 400X c) 630X d) 630X.
Fig. 4 - Paracoccidioides brasiliensis (BAT strain). Histopathological examination of a guinea pig testis stained with Gomori a) 200X b) 400X c) 630X d) 630X.
2. Testicular inoculation of guinea pigs: Histopathological examination of both testis inoculated with the IBIÁ and BAT strains gave essentially similar results. Both testis were enlarged, measuring on average 0.1 x 0.7 cm in diameter. Tissues exhibited areas of necrosis and suppuration which appeared as yellowish nodules or cystic cavities against a uniform whitish background. Microscopic examination showed extensive destruction of the testicular parenchyma which was infiltrated by many epithelioid granulomas, usually with suppurative centers. An acute and chronic nonspecific inflammation was also observed, the latter composed of lymphocytes, plasma cells and macrophages, with areas of fibrosis. More commonly present at the suppurative centers of the granulomas, but also observed inside the cytoplasm of macrophages, were typical forms of P. brasiliensis, most of them viable. Typical reproductive forms, occurring as large, budding, yeast mother-cells with distinctive ship's wheel morphology were also present.
3. Ouchterlony Double Immunodiffusion: The IBIÁ and BAT metabolic antigens showed precipitation lines with anti-gp43 rabbit serum, as well as with two paracoccidioidomycosis' patients sera. This was not observed for the two histoplasmosis' and two aspergillosis' patients sera (Fig 5).
- Double immunodiffusion. 1, 4 and 5) Paracoccidioidomycosis patients sera; 2) Polyclonal anti-gp43 rabbit serum; 3) Polyclonal anti P-brasiliensis total produced in rabbit; 6) Metabolic antigen from IBIÁ strain; 7) Metabolic antigen from BAT strain.
4. lmmunoelectrophoresis: The same antigens showed the presence of a precipitation arc with cathodal migration (E arc) in the presence of polyclonal anti-P. brasiliensis rabbit serum (Fig 6).
- Double immunodiffusion. 1) Paracoccidioidomycosis patient serum; 2 and 3) Histoplasmosis' patients sera; 4 and 5) Aspergillosis' patients sera; 6) Metabolic antigen from IBIÁ strain; 7) Metabolic antigen from BAT strain.
5. SDS-PAGE: When submitted to 12.5% polyacrylamide gel electrophoresis and stained with silver nitrate, this antigens of both strains revealed the presence of gp43 (Fig 7).
- Immunoelectrophoresis. 1) Metabolic antigen from IBIÁ strain; 2) Metabolic antigen from BAT strain; 3) Polyclonal anti-P. brasiliensis rabbit serum.
6. Immunoblotting: The tested antigens confirmed the presence of gp43, recognized by three paracoccidioidomycosis' patients sera (Fig 8).
- SDS PAGE, 12% gel, silver staining, exhibiting the presence of gp43 and other band (approximately 52kDa). 1) Molecular weight standard; 2) Metabolic antigen from IBIÁ strain; 3) Metabolic antigen from BAT strain.
Fig. 9 - Immunoblotting showing the reactivity of gp43 present on metabolic antigen from IBIÁ and BAT strains against paracoccidioidomycosis' patients sera. 1, 2 and 3 Paracoccidioidomycosis' patients sera; P molecular weight standard.
7. Protein determination: The IBIÁ and BAT strains showed protein levels of 4.31 mg/ml and 3.96 mg/ml , respectively.
DISCUSSION
The IBIÁ strain, isolated from the soil of a coffee plantation in IBIÁ, in the state of Minas Gerais, Brazil, according to the method of SILVA-VERGARA (1996)20 and SILVA-VERGARA et al. (1998)21, has been well described by these researchers, who noted that the antigen obtained revealed a low production of gp43. In contrast, in the present study we clearly demonstrated the presence of this glycoprotein by double immunodiffusion, SDS-PAGE and immunoblotting tests. Perhaps the technique used in the preparation of our antigen (culture filtrate), different from that studied by SILVA-VERGARA20,21 (sonicated suspension), explains the difference in results.
As to the pathogenicity for guinea pigs, this strain appears to be quite virulent considered the high number of parasites in the lesions, many of them showing multiple budding.
According to SANDHU et al. (1997)17 the sequencing of 761 bases of DNA nucleotides from the two strains revealed 100% homogeneity with a standard strain of P. brasiliensis, the same occurring with three human strains, an isolate from penguin feces and another from dog food contaminated with soil.
In the BAT strain, already well-studied by FREITAS DA SILVA et al. (l996)6, besides the glycoprotein component of 43 kDa (gp43), other fractions were also detected including one with a molecular weight of approximately 160 kDa. In our study, we did not detect such a fraction, but rather another one of approximately 52 kDa, not recognized by paracoccidioidomycosis' patients sera through the immunoblotting technique, the same occurred with IBIÁ strain.
BAT strain was studied, together with eight other strains of P. brasiliensis from Brazil, by CALGAGNO et al. (l998)3 by randomly amplified polymorphic DNA analysis (RAPD). Analysis of 33 strains of this fungus from South America showed that they produced amplification products sufficiently polymorphic (they showed significant differences in their amplification products) for the identification of 5 different primers, resulting in an elevated discriminatory index. These strains were classified into five distinct groups: Group 1, Venezuelan strains, group 2, Brazilian strains, group 3, Peruvian strains, group 4, Colombian strains, and group 5, Argentine strains. These authors think that the proposed method will be useful for epidemiologic studies of Paracoccidioides brasiliensis.
RESUMO
Paracoccidioides brasiliensis. Estudo de duas amostras sob o ponto de vista micológico e imunoquímico
Os Autores estudaram do ponto de vista micológico, imunoquímico e de sua biologia molecular, duas amostras de Paracoccidioides brasiliensis, uma isolada do solo, no município de IBIÁ (MG) por Silva-Vergara et al. (l996,1998)20,21 denominada IBIÁ e outra, BAT, cultivada de um caso humano de paracoccidioidomicose em Ribeirão Preto (SP) por Freitas da Silva (l996)6. Tais amostras apresentam colônias cotonosa (M) e leveduriforme (L ou Y), sendo patogênicas para cobaios inoculados por via testicular, produzindo orquite granulomatosa e/ou supurativa. Do ponto de vista imunoquímico, através de provas de Imunodifusão dupla, Imunoeletroforese e Western Blotting, foi demonstrada a presença da gp43.
A sequência de nucleotídeos do DNA de tais amostras, através do seqüenciamento de 761 bases, revelou homologia de 100% com amostra padrão de P. brasiliensis, o mesmo ocorrendo com três amostras humanas, uma isolada de fezes de pinguim e outra de ração alimentar para cães contaminada com terra.
São discutidos vários aspectos dos resultados obtidos, comparando-os com alguns dados da literatura.
Correspondence to: Prof.Dr. Carlos da Silva Lacaz, Laboratório de Micologia Médica IMTSP, Av. Dr. Enéas de Carvalho Aguiar 500, 05403-000 São Paulo, SP, Brasil. Phone 55 11 3066 7443 / FAX: 55 11 852 3622. Email: valacaz@usp.com.br
Received: 17 December 1998.
Accepted: 08 March 1999.
- 1. BAGAGLI, E.; SANO, A. COELHO, K.I. et al. - Isolation of Paracoccidioides brasiliensis from armadillos (Dasypus novencinctus) captured in an endemic area of paracoccidioidomycosis. Amer. J. trop. Med. Hyg., 58: 505-512, 1998.
- 2. BRITO, T. de; SANDHU, G.S.; KLINE, B.C. et al. - ln situ hybridization in paracoccidioidomycosis. Med. Mycol., 39, 1999 (In press).
- 3. CALGAGNO, A.M.; NINO-VEJA, G.; SAN-BLAS, F. & SAN-BLAS, G. - Geographic discrimination of P. brasiliensis strains by randomly amplified polymorphic DNA analysis. J. clin. Microbiol, 36: 1733-1736, 1998.
- 4. CANO, M.I.N.; CISALPINO, P.S.; GALINDO, I. et al. - Electrophoretic karyotypes and genome sizing of the pathogenic fungus Paracoccidioides brasiliensis. J. clin. Microbiol., 36: 742-747, 1998.
- 5. FERREIRA, M.S.; FREITAS, L.H.; LACAZ, C. da S. et al. - lsolation and characterization of a Paracoccidioides brasiliensis strain from a dogfood probably contaminated with soil in Uberlândia, Brazil. J. med. vet. Mycol., 28: 253-256, 1990.
- 6. FREITAS DA SILVA, G.; ROQUE-BARREIRA, M.C.; BRAGHETO, I.C.; MALTA, M.H.B. & MARTINEZ, R. - BAT: uma amostra de Paracoccidioides brasiliensis com composiçăo adequada para o sorodiagnóstico e acompanhamento de pacientes com paracoccidioidomicose. In: ENCUENTRO INTERNACIONAL SOBRE PARACOCCIDIOIDOMICOSIS, 6., Montevideo, 1996. p. 124.
- 7. GARCIA, N.M.; DEL NEGRO, G.M.B.; HEINS-VACARRI, E.M. et al. - Paracoccidioides brasiliensis, nova amostra isolada de fezes de pinguim (Pygocelis adeliae). Rev. Inst. Med. trop. S. Paulo, 35: 227-235, 1993.
- 8. GROSE, E. & TAMSITT, J.R. - Paracoccidioides brasiliensis recovered from the intestinal tract of three bats (Artibeus lituratus) in Colombia, South America. Sabouraudia, 4: 124-125, 1965.
- 9. JOHNSON, W.D. & LANG, C.M. - Paracoccidioidomycosis (South American blastomycosis) in squirrel monkey (Saimiri sciureus). Vet. Path., 14: 368-371, 1977.
- 10. LAEMMLI, V.K. - Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (Lond.), 227: 680-685, 1970.
- 11. LOWRY, O.H.; ROSEBROUGH, N.J.; FARR, A.L. & RANDALL, R.J. - Protein measurement with the folin phenol reagent. J. biol. Chem., 193: 265-275,1951.
- 12. MANIATIS, T. - Molecular cloning: a laboratory manual. Boston, Cold Spring Harbor Lab. Press, 1989.
- 13. MATTAR, R.; AZEVEDO, M.O; PEREIRA, M. et al. - Expression of glycoprotein gp43 in stage-specifíc forms and during dimorphic differentiation of Paracoccidioides brasiliensis. J. med. vet. Mycol., 35: 341-345, 1997.
- 14. MENDELOVICI, M. de; SALFELDER, K.; MENDELOVICI, E. & ROMAN, A R. de - Intento de aislamento del Paracoccidioides brasiliensis del suelo. Mycopathologia (Den Haag), 52: 45-53, 1974.
- 15. NICODEMOS, C.R. - Variabilidade antigęnica em Paracoccidioides brasiliensis e sua possível aplicaçăo no imunodiagnóstico e patogenia. Săo Paulo, 1996. (Dissertaçăo de Mestrado - Escola Paulista de Medicina).
- 16. PERAÇOLI, M.T.S.; SUGIZAKI, M.F.; MENDES, R.P. et al. - Estudo da virulęncia da amostra de Paracoccidioides brasiliensis isolada de tatu (Dasipus novencinctus). Rev. argent. Micol., 15: 41,1992.
- 17. SANDHU, G.S.; ALEFF, R.A.; KLINE, B.C. & LACAZ, C. da S. - Molecular detection and identification of Paracoccidioides brasiliensis. J. clin. Microbiol., 35: 1894-1896, 1997.
- 18. SANDHU, G.S.; KLINE, B.C.; STOCKMAN, L. & ROBERTS, G.D. - Molecular probes for diagnosis of fungal infections. J. clin. Microbiol., 33: 2913-2919, 1995.
- 19. SILVA-VERGARA, L.M. - Contribuiçăo ao estudo epidemiológico da paracoccidioidomicose: estudo em área de cultura de café. Ribeirăo Preto, 1996. (Tese de Doutoramento - Faculdade de Medicina de Ribeirăo Preto da Universidade de Săo Paulo.)
- 20. SILVA-VERGARA, M.L.; MARTINEZ, R.; CHADU, M.A. et al. - Isolamento de nova amostra do Paracoccidioides brasiliensis em plantaçăo de café. In: ENCUENTRO INTERNACIONAL SOBRE PARACOCCIDIOIDOMICOSIS, 6., Montevideo, 1996. p. 80.
- 21. SILVA-VERGARA, M.L.; MARTINEZ, R.; CHADU, A. et al. - Isolation of a Paracoccidioides brasiliensis strain from the soil of a coffee plantation in lbiá, state of Minas Gerais, Brazil. J. med. vet. Mycol., 36: 37-42, 1998.
- 22. SIQUEIRA, A.M. de - Avaliaçăo da sensibilidade e especificidade de algumas provas sorológicas no diagnóstico, prognóstico e controle de cura da paracoccidioidomicose. Caracterizaçăo imunoquímica do antígeno E2 do Paracoccidioides brasiliensis. Săo Paulo, 1982. (Tese de Doutoramento - Instituto de Cięncias Biomédicas da Universidade de Săo Paulo).
- 23. VIDAL, M.S.M.; MELO, N.T. de; DEL NEGRO, G.M.B. et al. - Paracoccidioides brasiliensis. A mycologic and immunochemical study of a sample isolated from an armadillo (Dasipus novencinctus). Rev. Inst. Med. trop. S. Paulo, 37: 43-49, 1995.
- 24. YARZÁBAL, L.A.; BOUT, D.; NAQUIRA, F.; FRUIT, J. & ANDRIEWS, S. - Identification and purification of the specific antigen of Paracoccidioides brasiliensis responsable for immunoelectrophoretic band E. Sabouraudia, 15: 79-85, 1977.
Publication Dates
-
Publication in this collection
02 July 1999 -
Date of issue
Mar 1999
History
-
Accepted
08 Mar 1999 -
Received
17 Dec 1998