ABSTRACT
Resource partitioning is important for species coexistence. Species with similar ecomorphology are potential competitors, especially when phylogenetically close, due to niche conservatism. The aim of this study was to investigate the resource partitioning among populations of two species of lebiasinids (Copella nigrofasciata and Pyrrhulina aff. brevis) that co-occur in a first-order Amazonian stream, analyzing the trophic ecology, feeding strategies and ecomorphological attributes related to the use of food and space by these species. Fish were captured in May and September 2010. The stomach contents of 60 individuals were analyzed and quantified volumetrically to characterize the feeding ecology of both species. Eleven morphological attributes were measured in 20 specimens and combined in nine ecomorphological indices. Both species had an omnivorous-invertivorous diet and consumed predominantly allochthonous items. Both showed a tendency to a generalist diet, but intrapopulational variation in resource use was also detected. Overall feeding niche overlap was high, but differed between seasons: low during the rainy season and high in the dry season. In the latter, the food niche overlap was asymmetric because C. nigrofasciata consumed several prey of P. aff. brevis, which reduced its food spectrum. The ecomorphological analysis suggests that C. nigrofasciata has greater swimming capacity (greater relative length of caudal peduncle) than P. aff. brevis, which has greater maneuverability and tendency to inhabit lentic environments (greater relative depth of the body). Our results demonstrate that these species have similar trophic ecology and suggest a spatial segregation, given by morphological differences related to locomotion and occupation of habitat, favoring their coexistence.
Keywords:
Feeding; coexistence; spatial segregation; Copella nigrofasciata; Pyrrhulina aff
RESUMO
A partilha de recursos é importante para a coexistência das espécies. Aquelas com ecomorfologia similar tem alto potencial competitivo, especialmente quando próximas filogeneticamente, devido ao conservantismo do nicho. O objetivo deste estudo foi investigar a partilha de recursos entre populações de duas espécies de lebiasinídeos (Copella nigrofasciata e Pyrrhulina aff. brevis) que coocorrem em um igarapé amazônico de primeira ordem, analisando a ecologia trófica, estratégias alimentares e atributos ecomorfológicos relacionados ao uso de recursos alimentares e espaciais. Os peixes foram capturados em maio e setembro/2010. O conteúdo estomacal de 60 indivíduos foi analisado e os itens quantificados volumetricamente para caracterizar a ecologia trófica das espécies. Foram medidos 11 atributos morfológicos, combinados em nove índices ecomorfológicos. As espécies apresentaram dieta onívora-invertívora com predomínio de itens alóctones, tendência ao generalismo e variação intrapopulacional no uso de recursos alimentares. A sobreposição de nicho trófico foi alta, mas diferiu entre os períodos, sendo baixa na chuva e alta na estiagem. Na última foi assimétrica, pois a dieta de C. nigrofasciata se sobrepôs à de P. aff. brevis, que reduziu seu espectro alimentar. A análise ecomorfológica diferenciou as espécies, indicando que C. nigrofasciata apresenta maior capacidade natatória (maior Comprimento Relativo do Pedúnculo Caudal) do que P. aff. brevis, que apresenta maior manobrabilidade e tendência a habitar ambientes lênticos (maior Altura Relativa do Corpo). Estes resultados demonstram que as espécies têm ecologia trófica semelhante e sugerem uma segregação espacial, dada por diferenças morfológicas relacionadas à locomoção e ocupação de hábitat, favorecendo sua coexistência.
Palavras-chave:
Alimentação; coexistência; segregação espacial; Copella nigrofasciata; Pyrrhulina aff. brevis.
INTRODUCTION
Resource partitioning is one of the main processes underlying species coexistence and community structure on local scale. Several factors may influence the ecological segregation among species, and dietary and spatial resources are considered the predominant factors in structuring populations and communities of vertebrates (Schoener 1974Schoener, T.W. 1974. Resource partitioning in ecological communities. Science, 185: 27-39.). For fish communities, trophic segregation is considered more important than habitat segregation (Ross 1986Ross, S.T. 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia: 352-388.).
Resource partitioning in neotropical fish communities has been well documented (Corrêa et al. 2009Corrêa, C.E.; Hahn, N.S.; Delariva, R.L. 2009. Extreme trophic segregation between sympatric fish species: the case of small sized bodyAphyocharax in the Brazilian Pantanal. Hydrobiologia, 635: 57-65.; Silva et al. 2012Silva, J.C.; Delariva, R.L.; Bonato, K.O. 2012. Food-resource partitioning among fish species from a first-order stream in northwestern Paraná, Brazil. Neotropical Ichthyology, 10: 389-399.) and often related to ecomorphological and/or behavioral differences that influence the use of space (micro-habitat) and food resources (Piorski et al. 2005Piorski, N.M.; Alves, J.R.L.; Machado, M.R.B.; Correia, M.M.F. 2005. Alimentação e ecomorfologia de duas espécies de piranhas (Characiformes: Characidae) do lago de Viana, Estado do Maranhão, Brasil. Acta Amazonica, 35: 63-70.; Lima et al.2008Lima, S.M.Q.; Cunha, A.A.; Sánchez-Botero, J.I.; Caramaschi, E.P. 2008. Vertical segregation of two species of Hyphessobrycon(Characiformes: Characidae) in the Cabiúnas coastal lagoon, southeastern Brazil. Neotropical Ichthyology, 6: 683-688.; Brazil-Sousa et al. 2009Brazil-Sousa, C.; Marques, R.M.; Albrecht, M.P. 2009. Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé (RJ). Biota Neotropica, 9: 1-7.). Morphological features might be indicators of species life habits, types of resource use or adaptations to occupy the environment (Gatz 1979). Thus, ecomorphology aims to access the relationship between shape and function that exists between morphological and ecological characters and to relate it with ecological concepts (Cunico and Agostinho 2006Cunico, A.M.; Agostinho, A.A. 2006. Morphological patterns of fish and their relationships with reservoirs hydrodynamics. Brazilian Archives of Biology and Technology, 49:125-134.).
Species with similar ecomorphological characters have high competitive potential (Gatz 1979), especially when phylogenetically close, due to niche conservatism (Wiens et al. 2010Wiens, J.J.; Ackerly, D.D.; Allen, A.P.; Anacker, B.L.; Buckley, L.B; Cornell, H.V.; et al 2010. Niche conservatism as an emerging principle in ecology and conservation biology. EcologyLetters, 13: 1310-1324.). Thus, it can be expected that these species, especially when syntopic, exhibit differences in resource use that might minimize competition (Herder and Freyhof 2006Herder, F.; Freyhof, J. 2006. Resource partitioning in a tropical stream fish assemblage. Journal of Fish Biology, 69: 571-589.). For example, in the size of consumed prey, use of microhabitats and feeding strategy (Esteves and Aranha 1999ESTEVES, K.E. and ARANHA, J.M.R. 1999. Ecologia trófica de peixes de riachos, p. 157182. In: E.P. CARAMASCHI; R. MAZZONI & P.R. PERES NETO (Eds). Ecologia de Peixes de Riachos. Rio de Janeiro, Série Oecologia Brasiliensis, PPGEUFRJ.; Piorski et al. 2005Piorski, N.M.; Alves, J.R.L.; Machado, M.R.B.; Correia, M.M.F. 2005. Alimentação e ecomorfologia de duas espécies de piranhas (Characiformes: Characidae) do lago de Viana, Estado do Maranhão, Brasil. Acta Amazonica, 35: 63-70.; Brazil-Sousa et al. 2009Brazil-Sousa, C.; Marques, R.M.; Albrecht, M.P. 2009. Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé (RJ). Biota Neotropica, 9: 1-7.).
Copella nigrofasciata (Meinken, 1952) and Pyrrhulina sp. aff. P. brevis Steindachner, 1876 (called P. aff. brevisthereafter) belong to the Lebiasinidae family, highly representative in Amazonian streams (Montag et al. 2008Montag, L.F.A.; Freitas, T.M.S.; Wosiacki, W.B.; Barthem, R.B. 2008. Os peixes da Floresta Nacional de Caxiuanã (Município de Melgaço e Portel, Pará - Brasil). Boletim do Museu Paraense Emílio Goeldi (Série Ciências Naturais), 3: 11-34.). Lebiasinids usually have an omnivorous diet based on allochthonous arthropods (Silva 1993Silva, C. 1993. Alimentação e distribuição espacial de algumas espécies de peixe do igarapé do Candiru, Amazonas, Brasil. Acta Amazônica, 23: 271-185.; Arias and Rossi 2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.; Oliveira 2013Oliveira, E.S. 2013. Variáveis ambientais influenciam na dieta de um lebiasinídeo na Amazônia Oriental? Trabalho de Conclusão de Curso, Universidade Federal do Pará, Belém, Pará. 21p.) and inhabit predominantly the surface of the water column in marginal areas with low flow (Sabino and Zuanon 1998Sabino, J.; Zuanon, J. 1998. A stream fish assemblage in Central Amazonia: distribution, activity patterns and feeding behavior. Ichthyological Exploration of Freshwaters, 8: 201-210.). Similarities in the use of habitat and food resources demand mechanisms that minimize interspecific competition to promote the coexistence of these species. The aim of this study was to investigate the resource partitioning among populations of two species of lebiasinids (Copella nigrofasciata and Pyrrhulina aff. brevis) that co-occur in a first-order Amazonian stream, analyzing the trophic ecology, feeding strategies and ecomorphological attributes related to the use of food and space by these species.
MATERIALS AND METHODS
Specimens of Copella nigrofasciata and Pyrrhulina aff.brevis were collected in a first order stream (UTM 554624/9802314) where these two species co-occur and are abundant. This stream belongs to the Araticum micro-basin, located within the Saracá-Taquera National Forest (FLONA) - PA, Northern Brazil (1º 20'and 1º 55' S, 56º 00' and 57º 15'). Samplings were carried out in the rainy season (May/2010) and in the dry season (September/10) during the daytime, using manual trawl (3mm mesh) and screens (55 cm diameter, 0.36 mm mesh) over a stretch of 50 m, which was blocked both upstream and downstream with nets (5 mm mesh). The collected specimens were anesthetized with menthol and fixed in 10% formaldehyde, and taken to the Laboratory of Fish Ecology (UFRJ), where they were transferred to 70° ethanol and identified. Voucher specimens of both species were deposited in the collection of the National Museum of Rio de Janeiro (P. aff. brevis MNRJ 42035; C. nigrofasciata MNRJ 42002).
Trophic ecology
Stomach contents of 30 individuals of each species were analyzed using a stereoscopic microscope. Food items were identified to the lowest taxonomic level with the aid of specialized literature, and were quantified by volumetric method as described in Albrecht and Caramaschi (2003Albrecht, M.P.; Caramaschi, E.P. 2003. Feeding ecology of Leporinus friderici(Teleostei; Anostomidae) in the upper Tocantins river, Central Brazil, before and after installation of a hydroelectric plant. Studies on Neotropical Fauna and Environment, 38: 33-40.).
For the qualitative characterization of the diet, the identified items were grouped into seven categories: 1) Algae - filamentous algae; 2) Sediment and debris - debris mixed with sediments; 3) Aquatic Invertebrates - adult and immature forms of aquatic insects and other arthropods; 4) Terrestrial Invertebrates - adult and immature insects and other terrestrial arthropods; 5) Organic Matter Unidentified - materials of organic origin not identified as animal or vegetal source; 6) Plant Material - parts of leaves, fruits and stems; and 7) Insects Remains - many digested parts difficult to attribute to a taxonomic group or origin (aquatic or terrestrial). The importance of these categories for species' diets was estimated through an Index that combines percentual volume and frequency of occurrence of consumed items (IAi - Kawakami and Vazzoler 1980Kawakami, E.; Vazzoler, G. 1980. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Boletim do Instituto Oceanográfico de São Paulo 29: 205-207.).
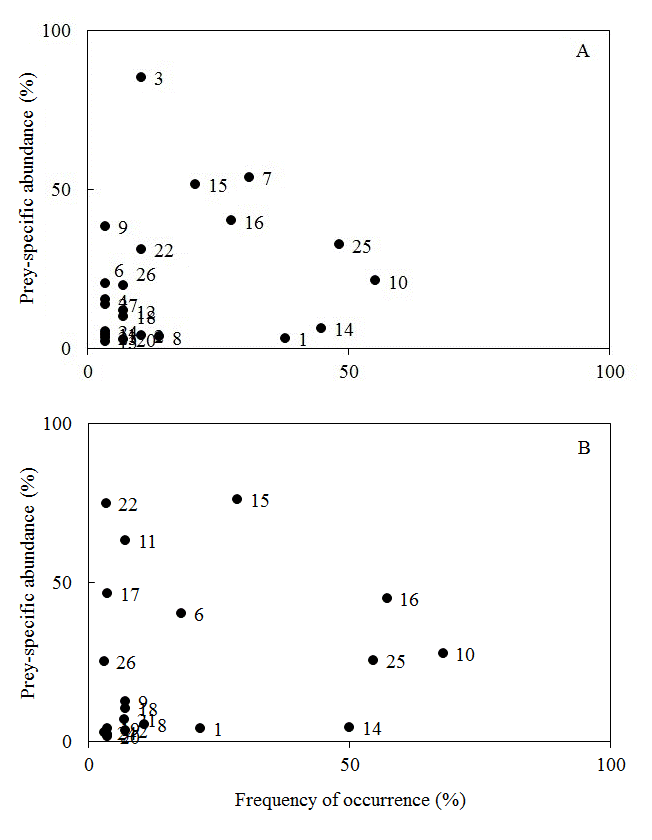
Feeding strategies were characterized using the graphical method proposed by Amundsen et al. (1996Amundsen, P.A.; Gabler, H.M.; Staldvik, F.J. 1996. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. Journal of Fish Biology, 48: 607-614.), which relates prey-specific abundance and frequency of occurrence. The distribution of points (food items) along the graph axes indicates the importance of prey for the population (rare or dominant), the feeding strategy of the predator (generalist or specialist), and the contribution of the between-phenotype (variation in resource use among individuals of the population) and within-phenotype components (when individuals tend to use similar resources) to the niche breadth.
The degree of diet overlap between species was calculated using the Pianka pair-wise niche overlap index (Pianka 1973Pianka, E.R. 1973. The structure of lizard communities. Annual Review of Ecologyand Systematics, 4: 53-74.), which ranges from 0 (no overlap) to 1 (complete overlap). We considered two situations: (1) "overall overlap" between species without distinction between seasons, which can be interpreted as a 'potential' overlap, independent of temporal and/or spatial coexistence; and (2) the "temporal overlap" calculated for each season sample separately. The results of interspecific overlap were interpreted according to Grossman (1986Grossman, G.D. 1986. Food partitioning in a rocky intertidal fish assemblage. Journal of Zoology , 1: 317-355.): high (> 0.6), intermediate (0.4 to 0.6) or low (< 0.4).
We used an Analysis of Similarities (ANOSIM) to evaluate possible differences in diet composition among species. Data were log-transformed and matrices of dissimilarity between individuals were constructed based on the coefficient of Bray-Curtis distance. A Similarity Percentage Analysis (SIMPER) was used to identify which food items contributed to the dissimilarity. These analyses were also performed in two situations: (1) considering the overall spectrum of food consumed by the population, and (2) considering the food resources consumed by populations in each season separately. Both analyses were performed on PRIMER (r)6 (Clarke and Gorley 2006Clarke, K.R.; Gorley, R.N. 2006. PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth, 192pp.).
Ecomorphology
To investigate the morphological differences associated with habitat use and feeding habits, 11 morphological measurements were taken on 20 specimens of each species using a digital caliper with 0.01 mm accuracy. The following morphological measurements were quantified following Piorski et al. (2005Piorski, N.M.; Alves, J.R.L.; Machado, M.R.B.; Correia, M.M.F. 2005. Alimentação e ecomorfologia de duas espécies de piranhas (Characiformes: Characidae) do lago de Viana, Estado do Maranhão, Brasil. Acta Amazonica, 35: 63-70.) and Mazzoni et al. (2010Mazzoni, R.; Moraes, M.; Rezende, C.F.; Miranda, J.C. 2010. Alimentação e padrões ecomorfológicos das espécies de peixes de riacho do alto rio Tocantins, Goiás, Brasil. Iheringia, Série Zoologia, 100: 162-168.): 1) Standard length (SL); 2) Digestive tract length (DTL); 3) Mouth width (MW); 4) Maximum body width (MBW); 5) Eye height (EH); 6) Head depth (HD); 7) Head length (HL); 8) Caudal peduncle length (CPL); 9) Caudal peduncle depth (CPD); 10) Maximum body depth (MBD); 11) Mouth depth (MD).
The ecomorphological indices used were: 1) relative length of the digestive tract (RLDT = DTL/SL) - High values occurs in detritivorous or herbivorous species, while low values are related to carnivorous habits; 2) relative width of the mouth (RWM = MW/MBW) - Higher values indicate ability to ingest larger prey; 3) relative depth of the mouth (RDM = MD/HD) - Higher values indicate ability to ingest larger prey; 4) eye position (EP = EH/HD) - Higher values occur in benthic species, while lower values occur in nektonic species; 5) relative length of the head (RLH = HL/SL) - Higher values indicate ability to ingest larger prey; 6) compression index (CI = MBD/MBW) - Higher values indicate a laterally compressed body adapted to inhabit lentic environments; 7) relative depth of the body (RDB = MBD/SL) - Higher values are related to high maneuverability and with low hydrodynamic environments; 8) relative length of the caudal peduncle (RLCP = CPL/SL) - Higher values occur in predators with good swimming capacity; and 9) relative depth of caudal peduncle (RDCP = CPD/MBD) - Higher values indicate good swimming potential.
The ecomorphological indices were applied to a Principal Component Analysis (PCA) using broken-stick as a stopping rule, and only the extreme variables with the highest correlations with the selected axes were interpreted. A one-way ANOVA was used to test for ecomorphological differences among species, using the principal axes values as ecomorphological predictors. PCA was performed on PC-Ord (McCune and Mefford 2011McCune, B.; Mefford, M.J. 2011. PC-ORD. Multivariate analysis of Ecological Data, Version 6.0 for Windows.) whereas ANOVAs were performed on R (R Development Core Team 2014R Core Team. 2014. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/.
http://www.R-project.org/...
).
RESULTS
Trophic ecology
Of the 60 individuals whose stomachs were examined, only three had empty stomachs (one C. nigrofasciata and two P.aff. brevis). The results of IAi showed that both species are omnivores with a tendency towards an invertivorous habit, consuming predominantly allochthonous items. The diet of C. nigrofasciata was primarily composed of Terrestrial Invertebrates (IAi = 27.10%), Insects Remains (24.53%), Debris and Sediment (14.59%) and Plant Material (13.17%), while the population of P. aff. brevisconsumed mainly Terrestrial Invertebrates (IAi = 34.37%), Plant Material (36.55%) and Insects Remains (21.82%) (Figure 1). A complete list of food items consumed by the two species is shown in Appendix 1.
Alimentary index (IAi) for the categories of diet items consumed by Copella nigrofasciata and Pyrrhulina aff. brevis sampled in a first order stream located inside the Saracá-Taquera National Forest - PA. Codes: Al - algae; SD - Sediment and Debris; AI - Aquatic Invertebrates; TI - Terrestrial Invertebrates; OM - Organic Matter Unidentified; PM - Plant Material; IR - Insects Remains.
Both species had an overall generalist diet, as no item was found to be dominant in their diets (consumed in high abundance by most individuals). The prevalence of rare items in the diet of both populations and the presence of groups of individuals who behaved as specialists (consuming few items in large quantities), indicate a high influence of the between-phenotype component in the niche width of both species (Figure 2). Hymenoptera (especially Formicidae) was the only item consumed by a little more than 50% of the population of C. nigrofasciata, but in low abundance (Figure 2a). Pyrrhulina. aff. brevis consumed other items in high frequencies and relative abundances besides hymenopterans (Plant Material, Insects Remains and Diptera Larvae), indicating that the feeding strategy of this species is also under influence of the within-phenotype component (Figure 2b).
Graphical method of Amundsen et al. (1996), with diet items consumed by Copella nigrofasciata (A) and Pyrrhulina aff. brevis (B) in a first order stream located inside the Saracá-Taquera National Forest - PA. Codes: 1- Aquatic Acarina (Hydracarina); 2- Terrestrial Acarina; 3 - Filamentous Algae; 4- Araneae; 5- Blattodea; 6- Coleoptera (Terrestrial Adult); 7- Debris; 8- Diptera Adult; 9 - Hemiptera; 10 - Hymenoptera; 11- Isoptera; 12 - Aquatic Coleoptera Larvae; 13 - Terrestrial Coleoptera Larvae; 14 - Diptera Larvae; 15 - Organic Matter Non-Identified; 16 - Plant Matter; 17 - Debris and Sediment; 18 -Ephemeroptera Nymphs; 19 -Odonata Nymphs; 20 -Plecoptera Nymphs; 21 - Opiliones; 22 - Invertebrate Eggs; 23 - Psocoptera; 24 - Diptera Pupae; 25 - Insects Remains; 26 - Seeds; 27 - Tysanoptera.
The calculated degree of niche overlap between species was high (0.83), and feeding dissimilarity was low, albeit significant (ANOSIM; R = 0.07; p = 0.001). Copella nigrofasciata consumed Debris and Algae, contrary to P. aff. brevis. The latter, in turn, consumed, in larger quantities, items present in the diets of both species (Table 1), such as Plant Matter, Insect Remains and Hymenoptera.
Similarity Percentage Analysis (SIMPER) showing the contribution of the principal items that are responsible for the dissimilarity between the diets of Copella nigrofasciata (C_nig) and Pyrrhulina aff. brevis (P_brev) in a first order stream located inside the Saracá-Taquera National Forest - PA. Codes: Av. Diss - Average Dissimilarity; Diss/SD - Standard Deviation of Dissimilarity; Cont. (%) - Percentage contribution for each diet item; Cum. (%) - Cumulative contribution.
Niche overlap between the two species was high in the dry season (0.75), and no significant differences were found in the proportion of items consumed (ANOSIM; R = 0.008, p = 0.527). During the dry season, the number of items consumed by P. aff. brevis was lower (13) than that consumed by C. nigrofasciata (17), and, with the exception of Opiliones and allochthonous Coleoptera, all items consumed by P.aff. brevis were also present in the diet of C. nigrofasciata (Table 2).
Alimentary indices (IAi) for the diet items consumed by Copella nigrofasciata(C_nig) and Pyrrhulinaaff. brevis (P_bre) in a first order stream located inside the Saracá-Taquera National Forest - PA, for both rainy and dry season. Codes: A - Aquatic; T - Terrestrial;
In the rainy season niche overlap had an intermediate value (0.52), and the proportion of consumed items was significantly different between species (ANOSIM; R = 0.13; p = 0.014). Copella nigrofasciata and P.aff. brevis consumed similar number of items (18 and 19, respectively), but types and relative proportions were different (Table 2). The major items that contributed to the dissimilarity between the diets of the species during rainy season were Plant Material, Hymenoptera and Insect Remains which were consumed by both species, but in higher quantities by P. aff. brevis, and Algae and Debris, which were consumed exclusively by C. nigrofasciata (SIMPER - Table 3).
Similarity Percentage Analysis (SIMPER) showing the contribution of the principal items responsible for the dissimilarity between the diets of Copella nigrofasciata(C_nigand Pyrrhulina aff.brevis (P_bre) in a first order stream located inside the Saracá-Taquera National Forest - PA in the rainy season. Codes: Av. Diss - Average Dissimilarity; Diss/SD - Standard Deviation of Dissimilarity; Cont. (%) - Percentage contribution for each diet item; Cum. (%) - Cumulative contribution.;(T) - Terrestrial;
Ecomorphology
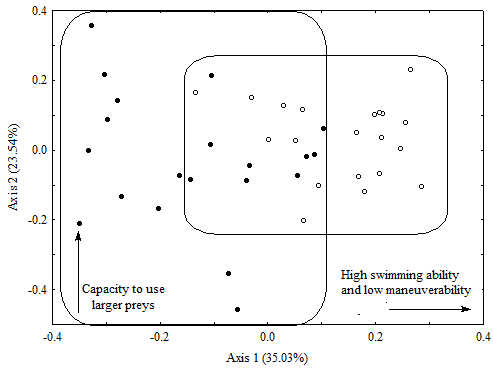
The standard length (SL) of C. nigrofasciataranged from 19.26 to 33.7 mm (26.27 ± 4.30 standard deviation), and SL of P. aff. brevis from 17.55 to 55.06 mm (30.64 ± 11.69 standard deviation). The first three PCA axes were selected by the broken-stick criteria and accounted for 76.01% of the total variation in the ecomorphological space. However, we interpret only PC1 and PC2 (58.6% of variation, Table 4). PC3 was not interpreted because it showed intrapopulational variations rather than diferences between species. Furthermore, indices with high scores on both PC2 and PC3 (RDM and RWM, respectively) indicate similar ecological features (ability to ingest larger prey). The first principal component explained most of the variation (35.03%) and showed interspecific differences related to the relative depth of the body (RDB) and to the relative length of the caudal peduncle (RLCP). The second axis showed mainly intraspecific variations related to relative length of the head (RLH) and to the relative depth of the mouth (RDM) (Table 4; Figure 3). The species ordination on the first axis formed two groups (T = 6.51; d. f. = 38; p < 0.001), but some overlap could be observed in the ecomorphological space (Figure 3). Copella nigrofasciata had usually higher scores on the first axis, showing higher values of RLCP, whereas P. aff. brevis had lower values in the axis, showing higher values of RDB.
Principal Component Analysis (PCA) of the nine ecomorphological indices calculated for Copella nigrofasciata and Pyrrhulina aff.brevis sampled in a first order stream located inside the Saracá-Taquera National Forest - PA. Hypothesis related to the interpretation of the ecomorphological indices highly correlated to the first and second axis are presented. Codes: Black circles represents P. aff. brevis and empty circles represents C. nigrofasciata.
Pearson correlation coefficients (r) obtained for the first two axes of the Principal Component Analysis (PCA) for the nine ecomorphological indices of Copella nigrofasciata and Pyrrhulina aff. brevis in a first order stream located inside the Saracá-Taquera National Forest - PA. The indices with the highest correlation with the axes are shown in bold type. Codes: RLDT - relative length of the digestive tract; RWM - relative width of the mouth; RDM - relative depth of the mouth; EP - eye position; RLH - relative length of the head; CI - compression index; RDB - relative depth of the body; RLCP - relative length of the caudal peduncle; RDCP - relative depth of caudal peduncle.
DISCUSSION
Copella nigrofasciata and Pyrrhulina aff. brevis showed an overall similar trophic ecology. Both could be classified as omnivores with a tendency for an invertivorous habit, and consumed predominantly allochthonous prey. Their feeding strategy is generalist with high influence of the between-phenotype component on niche breadth; however, the influence of the within-phenotype component cannot be neglected, particularly for P. aff. brevis. The diet niche overlap between species is potentially high, but showed significant temporal variations: it was intermediate in the rainy season and high but asymmetric in the dry season, when C. nigrofasciata consumed several prey types also consumed by P. aff. brevis, which in turn, showed a decrease in the diet spectrum. In contrast to the similarity in trophic ecology, species showed ecomorphological differences that suggest a distinct use of space. Individuals of Copella nigrofasciata showed greater swimming endurance, while individuals of P.aff. brevis showed greater maneuverability and tendency to inhabit slow water pools.
Despite the high diversity of prey consumed, most of the diet of C. nigrofasciata and P. aff. brevis were composed by arthropods, mainly terrestrial, fact that is favored by the terminal upward position of the mouth and occupation of the superficial layer of the water column. The predominance of terrestrial arthropods confirms other studies on these species (Anjos 2005Anjos, M.B. 2005. Estrutura de comunidades de peixes de igarapés de terra firme na Amazônia Central: composição, distribuição e características tróficas. Dissertação de Mestrado, Instituto Nacional de Pesquisas da Amazônia/Universidade Federal do Amazonas, Manaus, Amazonas. 68p.; Carvalho 2008Carvalho, L.N. 2008. História natural de peixes amazônicos: utilizando a abordagem do Conceito do Rio ContínuoTese de Doutorado, Instituto Nacional de Pesquisas da Amazônia/Universidade do Amazonas. Manaus. 142p.) and on other species of lebiasinids (Lebiasina panamensis (Gill, 1877) in a stream of Colombian Amazonia - Román-Valencia 2004Román-Valencia, C. 2004. Sobre la bioecología de Lebiasina panamensis (Pisces: Lebiasinidae) en la cuenca del Río León, Caribe Colombiano. Asociación Colombiana de Ictiólogos - DAHLIA, 7: 33-35.; and Copella arnoldi (Regan, 1912) in streams of eastern Amazonia - Oliveira 2013Oliveira, E.S. 2013. Variáveis ambientais influenciam na dieta de um lebiasinídeo na Amazônia Oriental? Trabalho de Conclusão de Curso, Universidade Federal do Pará, Belém, Pará. 21p.), but contrasts to the findings of Arias and Rossi (2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.), which showed a significant contribution of aquatic arthropods in the diet of Pyrrhulina australis Eigenmann and Kennedy 1903 in lagoons of the middle Paraná River.
The massive consumption of ants by the majority of individuals in the populations of C. nigrofasciata and P. aff. brevis indicates not only that these lebiasinids capture allochthonous insects that fall on the water surface, but also reflects the high representation of ants in the soil macrofauna in Central Amazon (Fittkau and Klinge 1973Fittkau, E.J.; Klinge, H. 1973. On biomass and trophic structure of the Central Amazonian Rain Forest Ecosystem. Biotropica, 5: 2-14.). The importance of these and other resources from adjacent terrestrial ecosystems as food for Amazonian stream fishes was reported (Mendonça et al. 2012Mendonça, M.B.; Montag, L.F.A.; Oliveira-Raiol, R.D.; Wosiacki, W.B. 2012. Feeding ecology of two sympatric species of the genus Jupiaba(Characiformes: Characidae) in streams in the Urucu river basin, AM, Brazil. Uakari, 8: 59-67.; Gonçalves et al.2013Gonçalves, A.F.G.; Prudente, B.S.; Carvalho-Filho, F.S.; Montag, L.F.A. 2013. Feeding ecology of dash-dot tetra Hemigrammus belotti (Steindachner, 1882) (Characiformes: Characidae) in the streams of the Urucu River basin, central Amazonia, Brazil. Biota Neotropica, 13: 141-147.). Terrestrial arthropods that fall into the streams are an important high quality food resource (low C: N ratio) that is directly available to fish (Baxter et al. 2005Baxter C.V.; Fausch, K.D.; Saunders, W.C. 2005. Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshwater Biology, 50: 201-220.; Small et al. 2013Small, G.E.; Torres, P.J.; Schweizer, L.M.; Duff, J.H.; Pringle, C.M. 2013. Importance of terrestrial arthropods as subsidies in lowland Neotropical rain forest stream ecosystems. Biotropica, 45: 80-87.). This fact highlights the importance of riparian vegetation and associated animal resources for the maintenance of aquatic communities. The consumption of these organisms reveals the participation of the studied species in the pathways of matter and energy between terrestrial and aquatic ecosystems (Baxter et al.2005Baxter C.V.; Fausch, K.D.; Saunders, W.C. 2005. Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshwater Biology, 50: 201-220.).
The calculated feeding overlap was high, which could indicate competition for resources between these two phylogenetically and morphologically similar syntopic species. We however interpret it as potential diet overlap value, since other analyses carried out in the present study suggest rather a low overlap due to: (i) different feeding strategies within populations (individuals or groups of individuals); (ii) seasonal variation in the diet; and (iii) minor morphological differences that suggest distinct spatial niche (utilization of microhabitats).
Niche partitioning is considered a common feature of fish communities (Ross 1986Ross, S.T. 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia: 352-388.), but variable according to environmental conditions, such as seasonality (Bouton et al. 1997Bouton, N.; Seehausen, O.; van Alphen, J.J.M. 1997. Resource partitioning among rock-dwelling haplochromines (Pisces: Cichlidae) from Lake Victoria. Ecology of Freshwater Fish, 6: 224-240.), like it was observed for both species in our study. The differences in feeding overlap between seasons indicate variation in food availability (Deus and Petrere-Junior 2003Deus, C.P.; Petrere-Junior, M. 2003. Seasonal diet shifts on seven fish species in an Atlantic Rainforest stream in southeastern Brazil. Brazilian Journal of Biology, 63: 579-588.), despite the fact that seasonal fluctuations are supposed to be milder upstream in Amazonian streams (Sioli 1984Sioli, H. 1984. Hydrochemistry and geology in the Brazilian Amazon region. Amazoniana, 1: 74-83.). During the rainy season, the flooding of marginal areas carries allochthonous resources into the stream and increases foraging areas, what we believe has led to a higher trophic segregation among P. aff. brevis and C. nigrofasciata, whereas in dry season the opposite was observed, i.e., more resource sharing. The relationship between the availability of resources and niche overlap between populations has shown controversial results. In a review paper, Esteves and Aranha (1999ESTEVES, K.E. and ARANHA, J.M.R. 1999. Ecologia trófica de peixes de riachos, p. 157182. In: E.P. CARAMASCHI; R. MAZZONI & P.R. PERES NETO (Eds). Ecologia de Peixes de Riachos. Rio de Janeiro, Série Oecologia Brasiliensis, PPGEUFRJ.) reported studies that demonstrated higher overlap among fishes in dry seasons, whereas other studies show the opposite pattern.
The high niche overlap observed in the dry season does not necessarily mean a scenario of competition. Since the niche is multidimensional, resource partitioning may be not experienced in the type of consumed preys, but in other dimensions like prey size, different use of space and/or phenotypic characteristics that allow subtle differences in the use of available resources (Esteves and Aranha 1999ESTEVES, K.E. and ARANHA, J.M.R. 1999. Ecologia trófica de peixes de riachos, p. 157182. In: E.P. CARAMASCHI; R. MAZZONI & P.R. PERES NETO (Eds). Ecologia de Peixes de Riachos. Rio de Janeiro, Série Oecologia Brasiliensis, PPGEUFRJ.; Piorski et al. 2005Piorski, N.M.; Alves, J.R.L.; Machado, M.R.B.; Correia, M.M.F. 2005. Alimentação e ecomorfologia de duas espécies de piranhas (Characiformes: Characidae) do lago de Viana, Estado do Maranhão, Brasil. Acta Amazonica, 35: 63-70.; Brazil-Sousa et al. 2009Brazil-Sousa, C.; Marques, R.M.; Albrecht, M.P. 2009. Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé (RJ). Biota Neotropica, 9: 1-7.). Furthermore, when considering average values of resource use within a population we ignore the fact that the individuals composing the same population may use different resources, minimizing an apparent competition between species.
The generalist feeding strategy observed for both P. aff. brevis and C. nigrofasciata was also observed by Arias and Rossi (2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.) for Pyrrhulina australis. This type of feeding strategy is favored by the naturally unstable characteristic of lotic systems, especially in tropical environments (Lowe-McConnell 1999Lowe-McConnell, R.H. 1999. Estudos ecológicos de comunidades de peixes tropicais. EDUSP, São Paulo, 584p.). A generalist population may be formed: (1) by groups of specialist individuals (high between-phenotype component); or (2) all or most part of generalist individuals (high within-phenotype component) (Bolnick et al. 2003Bolnick, D.I.; Svanbäck, R.; Fordyce, J.A.; Yang, L.H.; Davis, J.M.; Hulsey, C.D.; Forister, M.L. 2003. The ecology of individuals: incidence and implications of individual specialization. The American Naturalist, 161: 1-28.), both resulting in a high niche breadth. Copella nigrofasciata and P. aff. brevis appear to have an intermediate strategy between these two extremes, similarly to two species of Heptapteridade in Atlantic Forest streams (Brazil-Sousa et al.2009Brazil-Sousa, C.; Marques, R.M.; Albrecht, M.P. 2009. Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé (RJ). Biota Neotropica, 9: 1-7.).
However, some subtle differences can be found in the feeding strategies of these lebiasinids. Most individuals of C. nigrofasciata behaved as generalists, i.e., each individual or groups of individuals fed on several items. Exceptions to this pattern were a few individuals that consumed filamentous algae in large quantities. On the other hand, P. aff. brevis showed both extremes, being influenced both by between-phenotype and within-phenotype components. Groups of individuals were specialists, consuming invertebrate eggs, termites and organic matter in high amounts (volume), whereas the majority of the population consumed insect remains, plant material, and hymenopterans. The influence of the within-phenotype component was also observed for P. australis in the middle Paraná River (Arias and Rossi 2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.). The importance of the between-phenotype component indicates an intraspecific variation in resource use, suggesting the occurrence of individual specialization (Bolnick et al. 2003Bolnick, D.I.; Svanbäck, R.; Fordyce, J.A.; Yang, L.H.; Davis, J.M.; Hulsey, C.D.; Forister, M.L. 2003. The ecology of individuals: incidence and implications of individual specialization. The American Naturalist, 161: 1-28.). This variation is possibly a reflection of intraspecific morphological variation on prey size, revealed by ecomorphological analyzes in our study.
The ecomorphological analysis of C. nigrofasciata and P. aff. brevis indicated that these species differ in swimming endurance and maneuverability, and consequently in their ability to explore different habitats and resources available in streams. Upstream rivers are usually shallow (mainly first-order streams), but can present high diversity of habitats, such as dens, marginal mudflats, sandy and litter banks, and the channel of the stream itself (Pazin et al. 2006Pazin, V.F.V.; Magnusson, W.E.; Zuanon, J.; Mendonça, F.P. 2006. Fish assemblages in temporary ponds adjacent to "terra-firme" streams in Central Amazonia. Freshwater Biology, 51: 1025-1037.). The largest swimming endurance of C. nigrofasciata enables it to explore different environments available along the water column more easily, allowing it to maintain a diversified diet even during the dry season. Pyrrhulina aff. brevis, in turn, shows a tendency to inhabit lentic environments, and greater maneuverability than C. nigrofasciata, which could facilitate vertical movement, but hampers occupation and horizontal mobility among microhabitats. These results are consistent with the habitats where these species were captured: more than 60% of individuals of P. aff. brevis were captured in flooded marginal areas, while individuals of C. nigrofasciata were captured more evenly across all available microhabitats (~ 30 %).
A significant amount of debris was found in the diet of C. nigrofasciata. Although water velocity is usually high in headwaters (Vannote et al. 1980Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Cushing, C.E. 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences, 37: 130-137.), in Amazonian igarapés the low declivity permits the deposition of debris on the bottom substrate. Furthermore, individuals of Copella nigrofasciata were captured on flooded marginal areas, which are small temporary pools that form in valleys during rainy season by the overflowing stream water (Pazin et al. 2006Pazin, V.F.V.; Magnusson, W.E.; Zuanon, J.; Mendonça, F.P. 2006. Fish assemblages in temporary ponds adjacent to "terra-firme" streams in Central Amazonia. Freshwater Biology, 51: 1025-1037.). These pools can be the environment where this species can easily consume detritus. Although P. aff. brevis also occurs in high abundance in these microhabitats, the low amounts of detritus in its diet may indicate that it has a different foraging behavior when compared to C. nigrofasciata.
The consumption of debris has been associated with the behavior of substrate exploration (Mendonça et al. 2012Mendonça, M.B.; Montag, L.F.A.; Oliveira-Raiol, R.D.; Wosiacki, W.B. 2012. Feeding ecology of two sympatric species of the genus Jupiaba(Characiformes: Characidae) in streams in the Urucu river basin, AM, Brazil. Uakari, 8: 59-67.), which can be the case for C. nigrofasciata. Individuals of P. aff. brevis, on the other hand, could capture prey mainly in water column or surface (Arias and Rossi 2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.); a sit-and-wait strategy may be related to the increased maneuverability observed in P. aff. brevis. In a behavioral study in captivity, Arias and Rossi (2005Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.) found that the sit-and-wait is the main strategy adopted by P. australis : during the "waiting period", individuals spent most of the time amidst the vegetation or near the substrate and when moving towards prey demonstrated skill in maneuvering and braking abruptly. Data available for the present study do not allow inferring which type of behavior is predominant for the studied species. However, morphological and diet analyses provide clues about behavioral patterns.
CONCLUSIONS
Albeit the phylogenetically close species P. aff. brevis and C. nigrofasciata belong to the same trophic guild and have a high potential niche overlap, their coexistence in this first order stream seems to be facilitated by a variety of non-mutually exclusive factors, such as seasonal variation in diet, intraspecific variation in resource use, and morphological differences that allow a differentiated exploitation of available habitats. Whether these patterns are consistent across Amazonian streams will only be known by the accumulation of studies or through studies with replicates in larger scales. Our findings in this study, however, provide information on the ecology of these particular species with potential use as ornamental fish and that dwell Amazon streams, which are still poorly understood systems, despite being subject of anthropogenic modifications that may result in species losses even before they are known. Studies on the natural history and ecological relations of fish species are fundamental to provide knowledge on the ecological processes of the Amazon region for monitoring and management actions.
ACKNOWLEDGEMENTS
This research was supported by Mineração Rio do Norte. Authorization (No. 513/2014) for fish capture and transportation was given by IBAMA (DILIC) to Mineração Rio do Norte. The authors are grateful to other participants of the project for obtaining fish and field data, to members of the Laboratório de Ecologia de Peixes/ UFRJ for early discussions about the study design and analyses. NCSS was recipient of a Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) MSc fellowship.
REFERENCES
- Albrecht, M.P.; Caramaschi, E.P. 2003. Feeding ecology of Leporinus friderici(Teleostei; Anostomidae) in the upper Tocantins river, Central Brazil, before and after installation of a hydroelectric plant. Studies on Neotropical Fauna and Environment, 38: 33-40.
- Amundsen, P.A.; Gabler, H.M.; Staldvik, F.J. 1996. A new approach to graphical analysis of feeding strategy from stomach contents data - modification of the Costello (1990) method. Journal of Fish Biology, 48: 607-614.
- Anjos, M.B. 2005. Estrutura de comunidades de peixes de igarapés de terra firme na Amazônia Central: composição, distribuição e características tróficas. Dissertação de Mestrado, Instituto Nacional de Pesquisas da Amazônia/Universidade Federal do Amazonas, Manaus, Amazonas. 68p.
- Arias, J.; Rossi, L. 2005. Dieta y estrategia alimentaria de Pyrrhulina australe(Pisces, Lebiasinidae). Revista FABICIB, 9: 197-205.
- Baxter C.V.; Fausch, K.D.; Saunders, W.C. 2005. Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshwater Biology, 50: 201-220.
- Bolnick, D.I.; Svanbäck, R.; Fordyce, J.A.; Yang, L.H.; Davis, J.M.; Hulsey, C.D.; Forister, M.L. 2003. The ecology of individuals: incidence and implications of individual specialization. The American Naturalist, 161: 1-28.
- Bouton, N.; Seehausen, O.; van Alphen, J.J.M. 1997. Resource partitioning among rock-dwelling haplochromines (Pisces: Cichlidae) from Lake Victoria. Ecology of Freshwater Fish, 6: 224-240.
- Brazil-Sousa, C.; Marques, R.M.; Albrecht, M.P. 2009. Segregação alimentar entre duas espécies de Heptapteridae no Rio Macaé (RJ). Biota Neotropica, 9: 1-7.
- Carvalho, L.N. 2008. História natural de peixes amazônicos: utilizando a abordagem do Conceito do Rio ContínuoTese de Doutorado, Instituto Nacional de Pesquisas da Amazônia/Universidade do Amazonas. Manaus. 142p.
- Clarke, K.R.; Gorley, R.N. 2006. PRIMER v6: User Manual/Tutorial. PRIMER-E, Plymouth, 192pp.
- Corrêa, C.E.; Hahn, N.S.; Delariva, R.L. 2009. Extreme trophic segregation between sympatric fish species: the case of small sized bodyAphyocharax in the Brazilian Pantanal. Hydrobiologia, 635: 57-65.
- Cunico, A.M.; Agostinho, A.A. 2006. Morphological patterns of fish and their relationships with reservoirs hydrodynamics. Brazilian Archives of Biology and Technology, 49:125-134.
- Deus, C.P.; Petrere-Junior, M. 2003. Seasonal diet shifts on seven fish species in an Atlantic Rainforest stream in southeastern Brazil. Brazilian Journal of Biology, 63: 579-588.
- ESTEVES, K.E. and ARANHA, J.M.R. 1999. Ecologia trófica de peixes de riachos, p. 157182. In: E.P. CARAMASCHI; R. MAZZONI & P.R. PERES NETO (Eds). Ecologia de Peixes de Riachos. Rio de Janeiro, Série Oecologia Brasiliensis, PPGEUFRJ.
- Fittkau, E.J.; Klinge, H. 1973. On biomass and trophic structure of the Central Amazonian Rain Forest Ecosystem. Biotropica, 5: 2-14.
- Gatz Jr., A.J. 1979. Community organization in fishes as indicated by morphological features. Ecology, 60: 711-718.
- Gonçalves, A.F.G.; Prudente, B.S.; Carvalho-Filho, F.S.; Montag, L.F.A. 2013. Feeding ecology of dash-dot tetra Hemigrammus belotti (Steindachner, 1882) (Characiformes: Characidae) in the streams of the Urucu River basin, central Amazonia, Brazil. Biota Neotropica, 13: 141-147.
- Grossman, G.D. 1986. Food partitioning in a rocky intertidal fish assemblage. Journal of Zoology , 1: 317-355.
- Herder, F.; Freyhof, J. 2006. Resource partitioning in a tropical stream fish assemblage. Journal of Fish Biology, 69: 571-589.
- Kawakami, E.; Vazzoler, G. 1980. Método gráfico e estimativa de índice alimentar aplicado no estudo de alimentação de peixes. Boletim do Instituto Oceanográfico de São Paulo 29: 205-207.
- Lima, S.M.Q.; Cunha, A.A.; Sánchez-Botero, J.I.; Caramaschi, E.P. 2008. Vertical segregation of two species of Hyphessobrycon(Characiformes: Characidae) in the Cabiúnas coastal lagoon, southeastern Brazil. Neotropical Ichthyology, 6: 683-688.
- Lowe-McConnell, R.H. 1999. Estudos ecológicos de comunidades de peixes tropicais. EDUSP, São Paulo, 584p.
- Mazzoni, R.; Moraes, M.; Rezende, C.F.; Miranda, J.C. 2010. Alimentação e padrões ecomorfológicos das espécies de peixes de riacho do alto rio Tocantins, Goiás, Brasil. Iheringia, Série Zoologia, 100: 162-168.
- McCune, B.; Mefford, M.J. 2011. PC-ORD. Multivariate analysis of Ecological Data, Version 6.0 for Windows.
- Mendonça, M.B.; Montag, L.F.A.; Oliveira-Raiol, R.D.; Wosiacki, W.B. 2012. Feeding ecology of two sympatric species of the genus Jupiaba(Characiformes: Characidae) in streams in the Urucu river basin, AM, Brazil. Uakari, 8: 59-67.
- Montag, L.F.A.; Freitas, T.M.S.; Wosiacki, W.B.; Barthem, R.B. 2008. Os peixes da Floresta Nacional de Caxiuanã (Município de Melgaço e Portel, Pará - Brasil). Boletim do Museu Paraense Emílio Goeldi (Série Ciências Naturais), 3: 11-34.
- Oliveira, E.S. 2013. Variáveis ambientais influenciam na dieta de um lebiasinídeo na Amazônia Oriental? Trabalho de Conclusão de Curso, Universidade Federal do Pará, Belém, Pará. 21p.
- Pazin, V.F.V.; Magnusson, W.E.; Zuanon, J.; Mendonça, F.P. 2006. Fish assemblages in temporary ponds adjacent to "terra-firme" streams in Central Amazonia. Freshwater Biology, 51: 1025-1037.
- Pianka, E.R. 1973. The structure of lizard communities. Annual Review of Ecologyand Systematics, 4: 53-74.
- Piorski, N.M.; Alves, J.R.L.; Machado, M.R.B.; Correia, M.M.F. 2005. Alimentação e ecomorfologia de duas espécies de piranhas (Characiformes: Characidae) do lago de Viana, Estado do Maranhão, Brasil. Acta Amazonica, 35: 63-70.
- R Core Team. 2014. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/
» http://www.R-project.org/ - Román-Valencia, C. 2004. Sobre la bioecología de Lebiasina panamensis (Pisces: Lebiasinidae) en la cuenca del Río León, Caribe Colombiano. Asociación Colombiana de Ictiólogos - DAHLIA, 7: 33-35.
- Ross, S.T. 1986. Resource partitioning in fish assemblages: a review of field studies. Copeia: 352-388.
- Sabino, J.; Zuanon, J. 1998. A stream fish assemblage in Central Amazonia: distribution, activity patterns and feeding behavior. Ichthyological Exploration of Freshwaters, 8: 201-210.
- Schoener, T.W. 1974. Resource partitioning in ecological communities. Science, 185: 27-39.
- Silva, C. 1993. Alimentação e distribuição espacial de algumas espécies de peixe do igarapé do Candiru, Amazonas, Brasil. Acta Amazônica, 23: 271-185.
- Silva, J.C.; Delariva, R.L.; Bonato, K.O. 2012. Food-resource partitioning among fish species from a first-order stream in northwestern Paraná, Brazil. Neotropical Ichthyology, 10: 389-399.
- Sioli, H. 1984. Hydrochemistry and geology in the Brazilian Amazon region. Amazoniana, 1: 74-83.
- Small, G.E.; Torres, P.J.; Schweizer, L.M.; Duff, J.H.; Pringle, C.M. 2013. Importance of terrestrial arthropods as subsidies in lowland Neotropical rain forest stream ecosystems. Biotropica, 45: 80-87.
- Vannote, R.L.; Minshall, G.W.; Cummins, K.W.; Cushing, C.E. 1980. The river continuum concept. Canadian Journal of Fisheries and Aquatic Sciences, 37: 130-137.
- Wiens, J.J.; Ackerly, D.D.; Allen, A.P.; Anacker, B.L.; Buckley, L.B; Cornell, H.V.; et al 2010. Niche conservatism as an emerging principle in ecology and conservation biology. EcologyLetters, 13: 1310-1324.
Publication Dates
-
Publication in this collection
Mar 2016
History
-
Received
20 Mar 2015 -
Accepted
20 July 2015