ABSTRACT
Zootherapy (the use of the therapeutic potential of animals) is at least 6,000 years old, and has been kept active throughout generations until modern days. Animal fat is commonly used in the zootherapeutic folk medicine from South America, specially the green anaconda’s fat, which is widely promoted as a natural medicine to treat wounds, even though there is no scientific evidence showing its efficacy. In this study we compared the total healing time and the proportional daily reduction of dorsal epithelial incisions in adult male Wistar rats treated with nitrofural (a commercial cicatrizing ointment) and with anaconda fat. We applied the treatments every two days and measured the incision diameter daily, during ten consecutive days. Differences among the groups in the total healing time and in the proportional daily reduction of the incision consistently showed that the fat-based treatment resulted in a faster healing process compared to the commercial ointment tested. The literature suggests that the efficacy of animal fat on healing may be primarily related to the presence of fatty acids, which have been widely demonstrated to be important for biochemical reactions involved in healing processes. Our findings are widely socially relevant, considering that traditional Amazonian communities that use anaconda fat in folk medicine do not have easy access to pharmacies and hospitals.
Keywords:
ethnozoology; folk medicine; snakes; Wistar
RESUMO
Práticas ancestrais de zooterapia (uso das potencialidades terapêuticas da fauna) existem há pelo menos 6000 anos, e têm sido mantidas ao longo de gerações até os tempos modernos. Gordura de animais é comumente utilizada na zooterapia tradicional da América do Sul, e a gordura de serpentes sucuris é amplamente comercializada como cicatrizante natural, embora não existam evidências científicas que mostrem sua eficácia terapêutica. Neste estudo comparamos o tempo de cicatrização epitelial total e a redução proporcional diária de uma incisão dorsal em ratos Wistar machos adultos, entre um grupo controle, um grupo tratado com nitrofural (uma pomada cicatrizante comercial) e um grupo tratado com gordura de sucuri. Nós aplicamos os tratamentos a cada dois dias e medimos o diâmetro da incisão diariamente por dez dias consecutivos. Diferenças entre os grupos no tempo de cicatrização total e na redução proporcional diária da incisão consistentemente mostraram que gordura de sucuri teve ação cicatrizante mais eficiente que a pomada comercial testada. A literatura sugere que a eficácia de gordura animal em processos de cicatrização pode estar principalmente relacionada à presença de ácidos graxos, os quais têm sido amplamente demonstrados como importantes para reações bioquímicas envolvidas em processos de cicatrização. Os resultados têm ampla relevância social, considerando que as comunidades tradicionais amazônicas que utilizam a gordura de sucuris como zooterápico não têm acesso fácil a farmácias e hospitais.
Palavras-chave:
etnozoologia; medicina popular; serpentes; Wistar
INTRODUCTION
Human health and biodiversity have been intrinsically connected over thousands of years through popular phytotherapy and zootherapy practices (Gray 1997Gray, J.S. 1997. Marine biodiversity: patterns, threats and conservation needs. Biodiversity and Conservation, 6:153-175.). In many cultures around the world, native people have learned to use regional biodiversity for purposes beyond food, such as sources of medicine and protection (Alves et al. 2013aAlves, R.R.N.; Oliveira, T.P.R.; Rosa, I.L. 2013a. Wild Animals Used as Food Medicine in Brazil. Evidence-Based Complementary and Alternative Medicine, 2013: 1-12.), although ancestral practices of folk medicine are often associated with mythological beliefs (MacKinney 1946; Chemas 2010Chemas, R.C. 2010. A zooterapia no âmbito da medicina civilizada. I. Organoterapia humana e animal stricto sensu.Zooterapia: Os Animais na Medicina Popular Brasileira, 2: 75-102.). Zootherapy may have been originated after the incorporation of animal protein into the human diet (Holzman 2003Holzman, D. 2003. Meat eating is an old human habit. New Scientist, 179: 19-40.), but accurate estimates regarding time of origin are hampered by the lack of archaeological evidence (Wing 2000Wing, E.S. 2000. Animals used for food in the past: as seen by their remains excavated from archeological sites. In: Kiple, K.F.; Ornelas, K.C. (Ed.) The cambridge world history of food, Cambridge University Press, Cambridge, p.51-58.). Zootherapy may also have played an important role in the developing of non-nomadic human cultures, because, in addition to providing treatments for humans, it has also been used to treat diseases in domestic animals (Souto et al. 2011aSouto, W.M.S.; Mourão, J.S.; Barboza, R.R.D.; Alves, R.R.N. 2011a. Parallels between zootherapeutic practices in ethnoveterinary and human complementary medicine in NE Brazil. Journal of Ethnopharmacology, 134: 753-767., b; Ferreira et al. 2012Ferreira, F.S.; Albuquerque, U.P.; Coutinho, H.D.M.; Almeida, W.O.; Alves, R.R.N. 2012. The Trade in Medicine Animals in Northeastern Brazil. Evidence-based Complementary and Alternative Medicine, 2012: 1-20. , Ferreira et al. 2013; Alves et al. 2013a, b; Souto et al. 2013).
Evidence of ancient folk medicine is available in modern times, since popular medicine practices are maintained over generations in remote regions such as the rainforests in South America (e.g.Costa-Neto 2000Costa-Neto, E.M.A. 2000. Etnzoologia no Brasil: um panorama bibliográfico. Bioikos, 14: 31-45.; Alves and Souto 2011Alves, R.R.N.; Souto, W.M.S. 2011. Ethnozoology in Brazil: current status and perspectives. Journal of Ethnobiology and Ethnomedicine, 7: 22. doi: 10.1186/1746-4269-7-22.
https://doi.org/10.1186/1746-4269-7-22...
). For instance, data from questionnaires and experimental tests have shown that more than 350 animal species are used as sources of zootherapeutic products in Brazil (Alves et al. 2013a). These studies have shown that the ancestral zootherapy in Brazil consists mainly in the use of fat extracted from animals, such as jaguar (Panthera onca) and black caiman (Melanosuchus niger) for treating respiratory diseases, electric eel (Electrophorus electrus) for treating rheumatism, giant South American turtle (Podocnemis expansa) for protection against sunburnt, tucuxi Amazon dolphin (Sotalia fluviatilis) for treating asthma, and anaconda snake to heal epithelial wounds (Silva 2008Silva, A.L. 2008. Animais medicinais: conhecimentos e uso entre as populações ribeirinhas do rio Negro, Amazonas, Brasil. Boletim do Museu Paraense Emílio Goeldi Ciências Humanas, 3: 343-357.). The fat from some South American native reptiles has been experimentally demonstrated to have effective anti-inflammatory action (Ferreira et al. 2010Ferreira, S.F.; Brito, S.V.; Saraiva, R.A.; Araruna, M.K.A.; Menezes, I.R.A.; Costa, J.G.M.; Coutinho, H.D.M.; Almeida, W.O.; Alves, R.R.N. 2010. Topical anti-inflamatory activity of body fat from the lizard Tupinambis merianae. Journal of Etthnopharmacology, 130: 514-520.; Ferreira et al. 2013), but the potential pharmacological properties of anaconda fat has never been tested experimentally.
The green anacondaEunectes murinus (Boidae) is a giant aquatic snake, widely distributed through South America (Murphy and Henderson 1997Murphy, J.C.; Henderson, R.W. 1997. Tales of Giant Snakes: a Historical Natural History of Anacondas and Pythons. Krieger Company Malabar, Florida, 221p.). Due to its impressively large body size, many legends are maintained through generations of Amerindians and mestizos, as stories told by the oldest people in the villages (e.g., Silva 1999Silva, W.M.D.A. 1999. Lendas e mitos dos índios brasileiros ( Lendas e mitos dos índios brasileiros (www.mardehistoriassemfim.blogspot.com.br/2011/04/lendasindigenas.html
). Accessed on 18/02/2016.
www.mardehistoriassemfim.blogspot.com.br...
; Cascudo 2001Cascudo, L.D.C. 2001. Lendas Brasileiras. Global, São Paulo, 176p.; Taylor 2008Taylor, S. 2008. Cobra Grande: Histórias da Amazônia. Edições SM, São Paulo, 60p.). Anacondas eat relatively large prey (Fraga et al. 2013Fraga, R.; Lima, A.P.; Prudente, A.L.; Magnusson, W.E. 2013. Guia de cobras da região de Manaus - Amazônia Central. Instituto Nacional de Pesquisa da Amazônia, Manaus, 113p.), which generates energy accumulation in large fat deposits that are commonly used throughout Amazonia as a homemade natural therapy (Alves and Pereira-Filho 2007Alves, R.R.N.; Rosa, I.M.L. 2007. Biodiversity, traditional medicine and public health: where do they meet? Journal of Ethnobiology and Ethnomedicine, 3: 14. doi: 10.1186/1746-4269-3-14.
https://doi.org/10.1186/1746-4269-3-14....
). In fact, anaconda fat is the most commercialized product for zootherapeutic use in public markets in the larger Amazonian cities (Pinto and Maduro 2002Pinto, A.A.D.C.; Maduro, C.B. 2002 Produtos e subprodutos da medicina popular comercializados na cidade de Boa Vista, Roraima. Acta Amazonica, 33: 281-290.; Silva 2008). Considering folk medicine practices as a major cause for an unsustainable global biodiversity overexploitation (Alves and Rosa 2007), long-term trade may generate concerns regarding the impact of overexploitation on wild populations (Alves and Pereira-Filho 2007; Alves et al. 2008).
According to the traditional knowledge in the Amazon, it is believed that the anaconda fat has anti-inflammatory properties, which optimize healing processes (Alves et al. 2007Alves, R.R.N.; Rosa, I.M.L. 2007. Biodiversity, traditional medicine and public health: where do they meet? Journal of Ethnobiology and Ethnomedicine, 3: 14. doi: 10.1186/1746-4269-3-14.
https://doi.org/10.1186/1746-4269-3-14....
; Ferreira et al. 2013Ferreira, F.S.; Fernandes-Ferreira, H.; Neto, N.A.L.; Brito, S.V.; Alves, R.R.N. 2013. The Trade of medicinal animals in Brazil: current status and perspectives, Biodiversity and Conservation, 22: 839-870.). In this study we test some of the potential pharmacological properties of the anaconda fat through an experiment with rats. Specifically, we propose a comparison between anaconda fat and an industrialized and widely marketed ointment based on nitrofural regarding total epithelial healing time and the progressive reduction of dorsal epithelial incisions in Wistar rats.
MATERIALS AND METHODS
Extracting anaconda fat
All the anaconda fat used in the experiment was collected by veterinarians of the Brazilian Army, from an adult individual who died in captivity at the CIGS (Centro de Instruções de Guerra na Selva) Zoo, kept by the Brazilian Army in Manaus, Amazonas, Brazil. The anaconda fat was experimentally used by permission of the Instituto de Proteção Ambiental do Amazonas - IPAAM (process nr. 2972/T/16), and all procedures were approved by the Ethics Committee on Animal Use of the Instituto Nacional de Pesquisas da Amazônia - CEUA/INPA (process nr. 033/2016).
We used a generic method for the preparation of animal fat-based remedies, which consists of a topical ointment made by liquefied and filtered fat (Alves and Rosa 2007Alves, R.R.N.; Rosa, I.M.L. 2007. Biodiversity, traditional medicine and public health: where do they meet? Journal of Ethnobiology and Ethnomedicine, 3: 14. doi: 10.1186/1746-4269-3-14.
https://doi.org/10.1186/1746-4269-3-14....
; Alves and Alves 2011). We removed the solid fat in natura and we liquefied it in a pan over fire (~70 oC). The liquid fat was filtered using a 2 mm sieve, stored and decanted in a sealed plastic container, at non ar-conditioned room temperature. This method is widely used in folk medicine, and it seems to converge among different cultures around the world, including Amerindians (see Dias 2013Dias, D.Q. 2013. Estudo zooterápico do óleo fixo de Phrynops geoffroanus (Scweigger, 1812) (Testudines: Chelidae) do nordeste do Brasil, com análise química e farmacológica (in vitro e in vivo). Master’s dissertation, Universidade Regional do Cariri, Crato, Ceará. 86p.; Ferreira et al. 2014Ferreira S.F.; Brito S.V.; Sales D.L.; Menezes I.R.A.; Coutinho, H.D.M.; Souza, E.P. 2014. Anti-inflammatory potential of zootherapeutics derived from animals used in Brazilian traditional medicine. Pharmaceutical Biology, 52: 1403-1410.; Verma et al. 2014Verma, A.K.; Prasad, S.B.; Rongpi, T.; Arjun, J. 2014. Traditional healing with animals (zootherapy) by the major ethnic group of Karbi Anglong district of Assam, India. International Journal of Pharmacy and Pharmaceutical Sciences, 6: 593-600.).
Experiment with rats
We used three groups of Wistar rats, each containing eight adult males. Each rat was anesthetized with intraperitoneal ketamine-hydrochloride injection at 82.5 mg kg-1 and xylazine-hydrochloride at 5.75 mg kg-1. The anesthetized rats were immobilized in ventral decubitus and we used disposable punch and sterile forceps to make an epithelial dorsal incision, with 3.75-5.48 mm in diameter and 2-2.5 mm in depth. This method is commonly used in pharmacological testing, because it prevents fast healing of the superficial skin layers, which may bias the estimated difference in healing time between treatments, and does not cause serious inflammatory processes (Janning et al. 2011Janning, D.; Albuquerque, C.A.C.; Barauna, S.C. 2011. Avaliação preliminar do extrato hidroalcoólico de Tabernaemontana catharinensis no progresso de cicatrização de feridas em pele de ratos (Rattus novergicus). Revista Eletrônica de Farmácia, 8: 53-64.). Rats were individually housed in bed cages with constant feed and water supply, and postoperative analgesia was done using Ketofen-Citoprofen 50 mg kg-1 for each rat. The control-group received no external stimulus for healing, but asepsis was done every two days with 70% ethanol to reduce the probability of infection. One of the treatment-groups had a topical 2-mg nitrofural wound-healing medication applied every two days on the wound, in an amount sufficient to cover the incision. Nitrofural has antibiotic effects, being primarily indicated for burn treatments, and also widely used for general epithelial healing by stimulating granulation and tissue re-epithelization. Nitrofural-based drugs have been reported to be more efficient for epithelial healing compared to iodopovidone, silver sulfadiazine (Weber et al. 1977Weber, C.E.; Glanges, E.; Crenshaw, C.A. 1977. Treatment of second degree burns: nitrofurazone, povidone-iodine, and sulfadiazine. Journal of the American College of Emergency Physicians, 6: 486-490.), vaseline (Jeffords and Hagerty 1957Jeffords, J.V.; Hagerty, R.F. 1957. The healing of donor sites: a quantitative comparison of Furacin® soluble dressing and Vaseline® impregnated fine mesh gauze. Annals of Surgery, 145: 169-174.), and hexachlorophene soap (Sandifer 1970Sandifer, S.H. 1970. Clinical trial of topical nitrofurazone, with or without hydrocortisone, in 252 children with skin infections. Journal of the South Carolina Medical Association, 66: 363-365.). The other treatment-group had anaconda fat applied every two days on the wound in an amount sufficient to cover the incision.
We monitored the healing process daily and measured the incision diameter using a digital caliper (0.01 mm accuracy). The experiment was completed in 10 days, when all rats were completely healed. The experiment was carried out in a sterile environment of the vivarium, and the whole procedure was monitored by a veterinarian and a technologist from the INPA Central Vivarium (Manaus, Brazil).
Data analysis
The difference in time to complete healing among the three groups was analyzed using a Kruskal-Wallis test. We opted for a non-parametric test because the diameter values of the incision did not follow a normal distribution (Shapiro-Wilk W = 0.83; p = 0.001). In addition, we used a Tukey test to compare the healing time between pairs of groups. One of the rats in the nitrofural group had the initial diameter of the incision considerably larger than the average (5.48 mm, average = 3.99 mm). So, to avoid biased results, we repeated the same analyses excluding this observation.
Because there was variation in the initial incision diameter, we converted the daily values of incision diameter to proportions in relation to the initial diameter. To compare the daily grading processes of healing between the groups, we used averages between proportional values of incision diameter per rat and averages between rats per group. Differences in daily gradual healing process between groups were tested by paired t-test.
RESULTS
At the beginning of the postoperative treatment some rats presented inflammatory processes and behavioral expressions of discomfort. Such response probably resulted from stress, but all the rats recovered quickly and remained alive until the end of the experiment. We observed adverse reactions in the nitrofural and control groups throughout the first four days of the experiment, such as the presence of inflammatory secretions and blood clots on the incision. The group treated with anaconda fat had no visible signs of inflammatory processes.
The total healing time per rat ranged from seven to 10 days, with lowest average time for the group treated with anaconda fat (Table 1). The average time to complete healing was significantly different between groups (Kruskal-Wallis chi -square = 8.3; p = 0.01). This finding was mainly related to the lowest healing time for the group treated with anaconda fat (Figure 1). The exclusion of the observation from the nitrofural group that had excessively larger value of initial incision diameter did not affect the results (Kruskal-Wallis chi-square = 7.92; p = 0.01).
Healing process indicators of epithelial incisions in two treatment groups and a control-group of Wistar rats. Each group contained eight adult male rats. The total duration of the experiment was ten days, and the treatments were applied every two days. Values are averages followed by the standard deviation.
Total time (days) for healing of epithelial incisions in three groups of Wistar rats (two treatments and one control). Each group contained eight adult male rats. The bold line indicates the median, the boxes show the upper and lower quartiles, and the whiskers indicate the data ranges.
Comparisons between pairs of groups revealed a significant difference in the total healing time between anaconda fat and nitrofural (p = 0.009) and between anaconda fat and control (p = 0.01). However, we found no significant difference between the nitrofural and control groups (p = 0.96). Results were very similar when we removed the extreme observation from the nitrofural group with initial incision diameter larger than the average, with significant differences between the anaconda fat and nitrofural groups (p = 0.01) and the anaconda fat and control groups (p = 0.03), and non-significant difference between the nitrofural and control groups (p = 0.89) (Table 2).
Summary of a Tukey test applied to compare the time for total healing of epithelial incisions in three groups of Wistar rats (control, treated with nitrofural, treated with anaconda fat). Each group contained eight adult male rats.
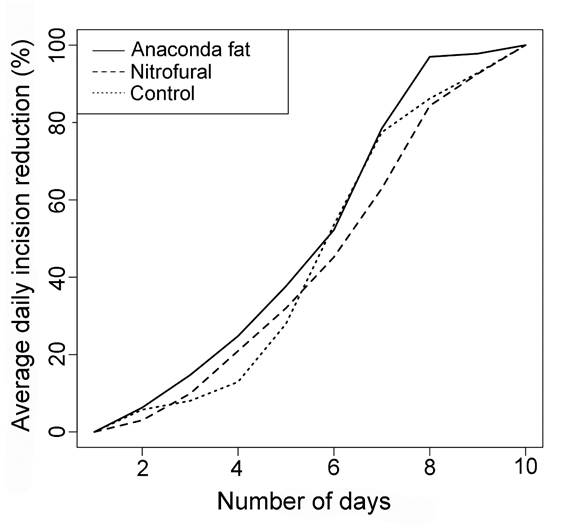
Anaconda fat was also more efficient as estimated by the daily mean proportional reduction of the incision, although the differences between the groups were not very large. The average proportional reduction in the incision diameter was significantly higher for the anaconda fat when compared to the nitrofural group (t = 3.69, p = 0.004) and control (t = 2.77, p = 0.02). However, there was no significant difference between the nitrofural and control groups (p = 0.5). We observed that the slope of the progression curve of the daily reduction in the proportional incision diameter was consistently steeper for the anaconda fat group throughout the experiment (Figure 2). In the nitrofural group the curve tended to be steep in the first days of the experiment, but the progression of the incision reduction was lower than the control group between the sixth and seventh days of the experiment. During this period the control group showed similar results to the group treated with anaconda fat.
Progressive reduction in the average diameter of epithelial incisions in three groups of Wistar rats (one control and two treatments). Each group contained eight adult males.
DISCUSSION
Our results showed that anaconda fat can be efficiently used as a homemade medicine for healing epithelial wounds. In general, the fat-induced healing process was more efficient compared to a widely-used industrialized drug and a control-group. This finding is expected to be associated with anti-inflammatory properties of fatty acids acting on healing, which has been experimentally demonstrated to be present in the fat of some reptiles such as Boa constrictor, rattlesnakes Crotalus durissus (Ferreira et al. 2013Ferreira, F.S.; Fernandes-Ferreira, H.; Neto, N.A.L.; Brito, S.V.; Alves, R.R.N. 2013. The Trade of medicinal animals in Brazil: current status and perspectives, Biodiversity and Conservation, 22: 839-870.) and tegu lizards Salvator merianae (Ferreira et al. 2010). We have no reason to suggest that the therapeutic potential of anaconda fat also includes antibiotic properties, because studies have shown that reptilian fats by themselves lack effective action in immune responses against bacterial infection (Ferreira et al. 2009; Ferreira et al. 2011).
The effectiveness of the anaconda fat as a natural medicine to healing wounds is probably related to the presence of fatty acids in its biochemical composition, which stimulate different processes involved in wound healing, such as muscle contraction, chemotaxis, adhesion, diapedesis, activation and cell death (Hatanaka and Curi 2007Hatanaka, E.; Curi, R. 2007. Ácidos graxos e cicatrização: uma revisão. Revista Brasileira de Farmácia, 88: 53-58. ). Additionally, fatty acids are important modulators of cell signaling for calcium (Soldati et al. 2002Soldati, L.; Lombardi, C.; Adamo, D.; Terranegra, A.; Bianchin, C.; Bianchin, G.; Vezzoli, G. 2002. Arachidonic acid increases intracellular calcium in erythrocytes. Biochemical and biophysical research communications, 293: 974-978.) and protein kinase C (May and Calder 1993May, C.L.; Calder, P.C. 1993. Unsatured fatty acids inhibit lymphocyte protein kinase c activity. Biochemical Society Transactions, 21: 377.), and they are precursors of lipid mediators such as prostaglandins, thromboxanes and leukotrienes, that stimulate blood vessel constriction and platelet aggregation (O’Shea et al. 2004O’Shea, M.; Bassaganya, J.; Mohede, I. C. 2004. Immunomodulaty properties of conjugated linoleic acid. American Journal of Clinical Nutrition, 79: 1199-1206.). Fatty acids also accelerate tissue granulation by leukocyte stimuli (Silva et al. 2002Silva, R.R.; Oliveira, T.T.; Nagem, T.J.; Leão, M.A. 2002. Efeito de flavonoides no metabolismo do ácido araquidônico. Medicina Ribeirão Preto, 35: 127-133.; Magalhães et al. 2008Magalhães, M.S.F.; Fechine, F.V.; Macedo, F.N.; Monteiro, D.L.S.; Oliveira, C.C.; Brito, G.A.C.; Moraes, M.E.A.; Moraes, M.O. 2008. Effect of a combination of medium chain triglycerides, linoleic acid, soy lecithin and vitamins A and E on wound healing in rats. Acta Cirurgica Brasileira, 23: 262-269.; Manhezi et al. 2008Manhezi, A.C.; Bachion, M.M.; Pereira, A.L. 2008. Utilização de ácidos graxos essenciais no tratamento de feridas. Revista Brasileira de enfermagem, 61: 620-629.), and they are also precursors of arachidonic acid, which induces gene expression in B lymphocytes. These cells are directly involved in repair of damaged tissues (Verlengia et al. 2003Verlengia, R.R.; Kanunfre, C.C.; Bordin, S.; De Lima, T.M.; Newsholme, P.; Curi, R. 2003. Genes Regulated by Arachidonic and Oleic Acid in Raji Cells. Lipids, 38: 1157-1165.). Fatty acids have been found to be effective in topical treatments for healing wounds (Calder 2003), because their rapid tissue uptake and metabolism stimulate neutrophils to secrete cytokines responsible for apoptosis, which reduces the amount of necrosed cell around the incision (Hatanaka et al. 2006). We emphasize that chemical analyses (e.g. gas and high performance liquid cromatography) are necessary to properly test the pharmacological properties of fatty acids from anaconda fat.
Although the sample size was relatively small (eight rats per group), it was sufficient to capture differences in the healing process among the groups, which were consistent across different statistical approaches. This result shows that it is possible to obtain reliable results on pharmacological tests, without submitting large amounts of rats to experimentation, although larger samples may lead to lower standard deviations (e.g.Barros et al. 2014Barros, E.M.L.; Lira, R.S.R.; Lemos, S.I.A.; Barros, T.L.; Rizo, M.S. 2014. Estudo do creme de buriti (Mauritia flexuosa L.) no processo de cicatrização. ConScientiae e Saúde, 13: 603-610.). The variation in the initial incision diameter also did not represent a sampling bias, since results were consistent even when we removed the observation with an outlier value for the initial incision diameter. Finally, similarities in the healing processes between the nitrofural and the control groups may be explained by the posology adopted in this study, which differs from the posology suggested in the nitrofural ointment package. The nitrofural manufacturer suggests applying an ointment-soaked gauze on the lesion. However, nitrofural has been identified as most effective for healing epithelial wounds in cases with severe inflammation, and it may show no difference in efficacy when compared to simple sterilization by povidone-iodine in cases without complication (Weber et al. 1977Weber, C.E.; Glanges, E.; Crenshaw, C.A. 1977. Treatment of second degree burns: nitrofurazone, povidone-iodine, and sulfadiazine. Journal of the American College of Emergency Physicians, 6: 486-490.). Although some rats in the nitrofural group showed visual signs of inflammatory processes, these symptoms lasted for up to four days, and no severe inflammation was observed.
The average total healing time differed slightly between groups, less than two days. This finding shows that anaconda fat is not much more efficient than an industrialized healing ointment, considering the overall process of epithelial healing. However, rats treated with anaconda fat showed no signs of postoperative inflammatory process and their wounds healed significantly faster, which was probably related to fatty acids present in the anaconda fat, as discussed above. These results are important from a social point of view because the anaconda fat is mainly used in folk medicine by traditional Amazonian people, who generally live in remote regions without easy access to hospitals and pharmacies. Natural-resource based folk medicine practices have been shown to be preferred among traditional people, because they are cheaper and more affordable than the orthodox medicine (Sofowora 1993Sofowora, A. 1993. Medicinal plants and traditional medicine in Africa. Spectron books limited, Nova Deli, 289p.; Luoga et al. 2000Luoga, E.J.; Witkowski, E.T.F.; Balkwill, K. 2000. Differential utilization and ethnobotany of trees in Kitulanghalo forest reverse and surrounding communal lands, eastern Tanzania. Society for Economic Botany, 54: 343.), or simply because the strong social and cultural relationships that traditional people maintain with natural resources (Tabuti et al. 2003Tabuti, J.R.S.; Lye, K.A.; Dhillions, S.S. 2003. Traditional medicine in Bulomogi county, Uganda: its practitiones uses and viability. Journal Ethnopharmacol, 85: 119-129.) generate resistance against modern urban medicine (Boltanski 1989Boltanski, L. 1989. As classes sociais e o corpo. 2da ed. Graal, Rio de Janeiro, 191p.). In fact, folk medicine may be expected to be a relevant practice even among urbanized populations, because about 80% of the world’s population primarily depends on animal and mainly plant-based medicines (WHO, IUCN and WWF 1993).
The present study was based on a widespread traditional knowledge that has been scientifically poorly explored, although it probably existed for thousands of years and persists presently among native people from the Amazon rainforests (Gordon and Neuberger 1949Gordon, B.L.; Neuburger, M. 1949. Medicine throughout antiquity. F.A. Davis Company, Philadelphia, p.313-354.; Lev 2003Lev, E. 2003. Traditional healing with animals (zootherapy): medieval to present-day Levantine practice. Journal of Ethnopharmacol, 85: 107-118.; Lidgard 2005Lidgard, H. 2005. Quackery or complementary medicine - a historical approach to the present situation. Master’s dissertation, University of Lund, Sweden. 72p.). Our results need to be confirmed through further studies involving larger-scale laboratory and clinical tests, including comparative analyses with other commercially available wound-healing and anti-inflammatory products, as well as the evaluation of collateral effects, the chemical analysis of anaconda fat, and the observation of the fat action at histological levels.
CONCLUSIONS
The present study produced evidence that anaconda fat is a more efficient healing method compared to an industrialized nitrofural drug, which was probably related to the presence of fatty acids in the biochemical structure of the fat. Our results showed consistently shorter total healing time and faster daily proportional reduction of epithelial incisions inflicted on Wistar rats as compared to rats treated with nitrofural ointment. Rats treated with anaconda fat also showed no signs of postoperative inflammatory process, which was observed in both the control and nitrofural-treated groups. Although our sample size was small (eight rats per experimental group), low within-group variances suggest that the responses observed were robust. Our results are relevant in the context of rural and traditional communities in the Amazon region, because we provide scientific evidence for the effectiveness of a traditional folk medicine that has been used for centuries to treat wounds among people that have little or no access to hospitals and pharmacies.
ACKNOWLEDGEMENTS
We thank C. Keller, M. Ferrão, D. Fagundes, H. Astolpho and two anonymous reviewers for critical reading of early versions of the manuscript. The Brazilian Army (CIGS Zoo, Manaus) provided us all the anaconda fat used in the experiment. T. Sumita and A. Leitão helped us with the experiments. RF thanks the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for the scholarship provided. FPW thanks financial support from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Amazonas (FAPEAM) and Partnerships for Enhanced Engagement in Research from the U.S. National Academy of Sciences and U.S. Agency of International Development.
- Alves, R.R.N.; Alves, H.N. 2011. The faunal drugstore: Animal-based remedies used in traditional medicines in Latin America. Journal of Ethnobiology and Ethnomedicine, 7: 1-43.
- Alves, R.R.N.; Lima, H.N.; Tavares, M.C.; Souto, W.M.S.; Barboza, R.P.D.; Vasconcellos, A. 2008. Animal-based remedies as complementary medicines in Santa Cruz do Capibaribe, Brasil. BMC Complementary and Alternative Medicine, 8: 44. doi: 10.1186/1472-6882-8-44.
» https://doi.org/10.1186/1472-6882-8-44 - Alves, R.R.N.; Melo, M.F.; Ferreira, F.S.; Trovão, D.M.B.M.; Dias, T.L.P.; Oliveira, J.V.; Lucena, R.F.P. 2015. Healing with animals in a semiarid northeastern area of Brazil. Evironment, Development and Sustainability, 18: 1733-1747.
- Alves, R.R.N.; Oliveira, T.P.R.; Rosa, I.L. 2013a. Wild Animals Used as Food Medicine in Brazil. Evidence-Based Complementary and Alternative Medicine, 2013: 1-12.
- Alves, R.R.N.; Pereira-Filho, G. 2007. Commercialization and use of snakes in North and Northeastern Brazil: implications for conservation and management. Biodiversity and Conservation, 16: 969-985.
- Alves, R.R.N.; Rosa, I.L. 2006. From cnidarians to mammals: the use of animals as remedies in fishing communities in NE Brazil. Journal of Ethnofarmacology, 107: 259-276.
- Alves, R.R.N.; Rosa, I.M.L. 2007. Biodiversity, traditional medicine and public health: where do they meet? Journal of Ethnobiology and Ethnomedicine, 3: 14. doi: 10.1186/1746-4269-3-14.
» https://doi.org/10.1186/1746-4269-3-14. - Alves, R.R.N.; Santana, G.G.; Rosa, I.L. 2013b. The role of animal-derived remedies as complementary medicine in Brazil. In: Alves, R.R.N.; Rosa, I.L. (Ed.) Animals in Traditional Folk Medicine: Implications for Conservation Springer, New York, Dordrecht, London, Berlin, p.289-290.
- Alves, R.R.N.; Souto, W.M.S. 2011. Ethnozoology in Brazil: current status and perspectives. Journal of Ethnobiology and Ethnomedicine, 7: 22. doi: 10.1186/1746-4269-7-22.
» https://doi.org/10.1186/1746-4269-7-22 - Alves, R.R.N.; Vieira, W.L.S.; Santana, G.G.; Vieira, K.S.; Montenegro, P.F.G.P. 2013c. Herpetofauna used in traditional folk medicine: Conservation implications. In: Alves, R.R.N, Rosa, I.L. (Ed.) Animals in Traditional Folk Medicine: Implications for Conservation Springer, Berlin, p.109-133.
- Alves, R.R.N.; Vieira, W.L.S.; Santana, G.G. 2008. Reptiles used in traditional folk medicine: conservation implications. Biodiversity and Conservation, 17: 2037-2049.
- Barros, E.M.L.; Lira, R.S.R.; Lemos, S.I.A.; Barros, T.L.; Rizo, M.S. 2014. Estudo do creme de buriti (Mauritia flexuosa L.) no processo de cicatrização. ConScientiae e Saúde, 13: 603-610.
- Boltanski, L. 1989. As classes sociais e o corpo 2da ed. Graal, Rio de Janeiro, 191p.
- Calder, P.C. 2003. Long-chain n-3 fatty acids and inflammation: potential application in surgical and trauma patients. Brazilian Journal of Medical and Biological Research, 36: 433-446.
- Cascudo, L.D.C. 2001. Lendas Brasileiras Global, São Paulo, 176p.
- Chemas, R.C. 2010. A zooterapia no âmbito da medicina civilizada. I. Organoterapia humana e animal stricto sensu.Zooterapia: Os Animais na Medicina Popular Brasileira, 2: 75-102.
- Costa-Neto, E.M.A. 2000. Etnzoologia no Brasil: um panorama bibliográfico. Bioikos, 14: 31-45.
- Dias, D.Q. 2013. Estudo zooterápico do óleo fixo de Phrynops geoffroanus (Scweigger, 1812) (Testudines: Chelidae) do nordeste do Brasil, com análise química e farmacológica (in vitro e in vivo) Master’s dissertation, Universidade Regional do Cariri, Crato, Ceará. 86p.
- Ferreira, F.S.; Albuquerque, U.P.; Coutinho, H.D.M.; Almeida, W.O.; Alves, R.R.N. 2012. The Trade in Medicine Animals in Northeastern Brazil. Evidence-based Complementary and Alternative Medicine, 2012: 1-20.
- Ferreira, F.S.; Brito, S.V.; Ribeiro, S.C.; Almeida, W.O.; Alves, R.R.N. 2009. Zootherapeutics utilized by residents of the community Poço Dantas, Crato-CE, Brazil. Journal of Ethnobiology and Ethnomedicine, 5: 1-10.
- Ferreira S.F.; Brito S.V.; Sales D.L.; Menezes I.R.A.; Coutinho, H.D.M.; Souza, E.P. 2014. Anti-inflammatory potential of zootherapeutics derived from animals used in Brazilian traditional medicine. Pharmaceutical Biology, 52: 1403-1410.
- Ferreira, S.F.; Brito, S.V.; Saraiva, R.A.; Araruna, M.K.A.; Menezes, I.R.A.; Costa, J.G.M.; Coutinho, H.D.M.; Almeida, W.O.; Alves, R.R.N. 2010. Topical anti-inflamatory activity of body fat from the lizard Tupinambis merianae Journal of Etthnopharmacology, 130: 514-520.
- Ferreira, F.S.; Fernandes-Ferreira, H.; Neto, N.A.L.; Brito, S.V.; Alves, R.R.N. 2013. The Trade of medicinal animals in Brazil: current status and perspectives, Biodiversity and Conservation, 22: 839-870.
- Ferreira, F.S.; Silva, N.L.G.; Matias, E.F.F.; Brito, S.V.; Oliveira, F.G.; Costa, J.G.M.; Coutinho, H.D.M.; Almeida, W.O.; Alves, R.R.N. 2011. Potentiation of aminoglycoside antibiotic activity using the body fat from the snake Boa Constrictor Brazilian Journal of Pharmacognosy, 21: 503-509.
- Fraga, R.; Lima, A.P.; Prudente, A.L.; Magnusson, W.E. 2013. Guia de cobras da região de Manaus - Amazônia Central Instituto Nacional de Pesquisa da Amazônia, Manaus, 113p.
- Gordon, B.L.; Neuburger, M. 1949. Medicine throughout antiquity F.A. Davis Company, Philadelphia, p.313-354.
- Gray, J.S. 1997. Marine biodiversity: patterns, threats and conservation needs. Biodiversity and Conservation, 6:153-175.
- Hatanaka, E.; Curi, R. 2007. Ácidos graxos e cicatrização: uma revisão. Revista Brasileira de Farmácia, 88: 53-58.
- Hatanaka, E.; Leonardo, M.P.; Martins, E.F. Liberti, E.A.; Farsky, S.H.P.; Curi, R.; Pithon-Curi, T.C. 2006. Evidências do efeito pró-inflamatório dos tecidos oleico e linoleico no processo de cicatrização em ratos. Proceedings of the II Simpósio sobre ácidos graxos e saúde, p.70-72.
- Holzman, D. 2003. Meat eating is an old human habit. New Scientist, 179: 19-40.
- Janning, D.; Albuquerque, C.A.C.; Barauna, S.C. 2011. Avaliação preliminar do extrato hidroalcoólico de Tabernaemontana catharinensis no progresso de cicatrização de feridas em pele de ratos (Rattus novergicus). Revista Eletrônica de Farmácia, 8: 53-64.
- Jeffords, J.V.; Hagerty, R.F. 1957. The healing of donor sites: a quantitative comparison of Furacin® soluble dressing and Vaseline® impregnated fine mesh gauze. Annals of Surgery, 145: 169-174.
- Lev, E. 2003. Traditional healing with animals (zootherapy): medieval to present-day Levantine practice. Journal of Ethnopharmacol, 85: 107-118.
- Lidgard, H. 2005. Quackery or complementary medicine - a historical approach to the present situation Master’s dissertation, University of Lund, Sweden. 72p.
- Luoga, E.J.; Witkowski, E.T.F.; Balkwill, K. 2000. Differential utilization and ethnobotany of trees in Kitulanghalo forest reverse and surrounding communal lands, eastern Tanzania. Society for Economic Botany, 54: 343.
- Mackinney, L.C. 1946. Animal substances in materia medica. Journal of History Medicine and Allied Sciences, 1: 149-170.
- Magalhães, M.S.F.; Fechine, F.V.; Macedo, F.N.; Monteiro, D.L.S.; Oliveira, C.C.; Brito, G.A.C.; Moraes, M.E.A.; Moraes, M.O. 2008. Effect of a combination of medium chain triglycerides, linoleic acid, soy lecithin and vitamins A and E on wound healing in rats. Acta Cirurgica Brasileira, 23: 262-269.
- Manhezi, A.C.; Bachion, M.M.; Pereira, A.L. 2008. Utilização de ácidos graxos essenciais no tratamento de feridas. Revista Brasileira de enfermagem, 61: 620-629.
- May, C.L.; Calder, P.C. 1993. Unsatured fatty acids inhibit lymphocyte protein kinase c activity. Biochemical Society Transactions, 21: 377.
- Murphy, J.C.; Henderson, R.W. 1997. Tales of Giant Snakes: a Historical Natural History of Anacondas and Pythons Krieger Company Malabar, Florida, 221p.
- O’Shea, M.; Bassaganya, J.; Mohede, I. C. 2004. Immunomodulaty properties of conjugated linoleic acid. American Journal of Clinical Nutrition, 79: 1199-1206.
- Pinto, A.A.D.C.; Maduro, C.B. 2002 Produtos e subprodutos da medicina popular comercializados na cidade de Boa Vista, Roraima. Acta Amazonica, 33: 281-290.
- Sandifer, S.H. 1970. Clinical trial of topical nitrofurazone, with or without hydrocortisone, in 252 children with skin infections. Journal of the South Carolina Medical Association, 66: 363-365.
- Silva, A.L. 2008. Animais medicinais: conhecimentos e uso entre as populações ribeirinhas do rio Negro, Amazonas, Brasil. Boletim do Museu Paraense Emílio Goeldi Ciências Humanas, 3: 343-357.
- Silva, R.R.; Oliveira, T.T.; Nagem, T.J.; Leão, M.A. 2002. Efeito de flavonoides no metabolismo do ácido araquidônico. Medicina Ribeirão Preto, 35: 127-133.
- Silva, W.M.D.A. 1999. Lendas e mitos dos índios brasileiros ( Lendas e mitos dos índios brasileiros (www.mardehistoriassemfim.blogspot.com.br/2011/04/lendasindigenas.html ). Accessed on 18/02/2016.
» www.mardehistoriassemfim.blogspot.com.br/2011/04/lendasindigenas.html - Sofowora, A. 1993. Medicinal plants and traditional medicine in Africa Spectron books limited, Nova Deli, 289p.
- Soldati, L.; Lombardi, C.; Adamo, D.; Terranegra, A.; Bianchin, C.; Bianchin, G.; Vezzoli, G. 2002. Arachidonic acid increases intracellular calcium in erythrocytes. Biochemical and biophysical research communications, 293: 974-978.
- Souto, W.M.S.; Mourão, J.S.; Barboza, R.R.D.; Alves, R.R.N. 2011a. Parallels between zootherapeutic practices in ethnoveterinary and human complementary medicine in NE Brazil. Journal of Ethnopharmacology, 134: 753-767.
- Souto, W.M.S.; Mourão, J.S.; Barboza, R.R.D.; Mendonça, L.E.T.; Lucena, R.F.P.; Confessor, M.V.A.; Vieira, W.L.S.; Montenegro, P.F.G.P.; Lopez, L.C.S.; Alves, R.R.N. 2011b. Medicinal animals used in ethnoveterinary practices of the Cariri Paraibano, NE Brazil. Journal of Ethnobiology and Ethnomedicine, 7: 30. doi:10.1186/1746-4269-7-30.
» https://doi.org/10.1186/1746-4269-7-30 - Souto, W.M.S.; Pinto, L.C.; Mendonça, L.E.T.; Mourão, J.S.; Vieira, W.L.S.; Montenegro, P. F. G. P.; Alves, R.R.N. 2013. Medicinal animals in ethnoveterinary practices: A world overview. In: Alves, R.R.N.; Rosa, I.L. (Ed.). Animals in traditional folk medicine Springer, New York, p.43-66.
- Tabuti, J.R.S.; Lye, K.A.; Dhillions, S.S. 2003. Traditional medicine in Bulomogi county, Uganda: its practitiones uses and viability. Journal Ethnopharmacol, 85: 119-129.
- Taylor, S. 2008. Cobra Grande: Histórias da Amazônia Edições SM, São Paulo, 60p.
- Verlengia, R.R.; Kanunfre, C.C.; Bordin, S.; De Lima, T.M.; Newsholme, P.; Curi, R. 2003. Genes Regulated by Arachidonic and Oleic Acid in Raji Cells. Lipids, 38: 1157-1165.
- Verma, A.K.; Prasad, S.B.; Rongpi, T.; Arjun, J. 2014. Traditional healing with animals (zootherapy) by the major ethnic group of Karbi Anglong district of Assam, India. International Journal of Pharmacy and Pharmaceutical Sciences, 6: 593-600.
- Weber, C.E.; Glanges, E.; Crenshaw, C.A. 1977. Treatment of second degree burns: nitrofurazone, povidone-iodine, and sulfadiazine. Journal of the American College of Emergency Physicians, 6: 486-490.
- Wing, E.S. 2000. Animals used for food in the past: as seen by their remains excavated from archeological sites. In: Kiple, K.F.; Ornelas, K.C. (Ed.) The cambridge world history of food, Cambridge University Press, Cambridge, p.51-58.
- World Health Organization (WHO), World Conservation Union (IUCN) and World Wide Fund for Nature (WWF). 1993. Guidelines on the conservation of medicinal plants, Gland, Switzerland, 38p.
-
ERRATA
On page 341, Where it reads: “Leonardo BRANDÃO”: Erika SOUZA1*, Fernanda P. WERNECK², Leonardo BRANDÃO3, Rafael de FRAGA4 Please read as: “Leonardo B. MATOS”: Erika SOUZA1*, Fernanda P. WERNECK², Leonardo B. MATOS3, Rafael de FRAGA4 Acta Amazonica (2018) 48(1): 83-83
Publication Dates
-
Publication in this collection
Oct-Dec 2017
History
-
Received
05 July 2017 -
Accepted
24 Aug 2017