Abstracts
The objectives of this work were to evaluate two greenhouse screening methods for sudden death syndrome (SDS) and to determine which one is best correlated with field resistance of soybean genotypes. The evaluations were done with three sets of genotypes that were classified as partially resistant, intermediate, and susceptible to SDS based on previous field evaluations. These three sets were independently evaluated for greenhouse SDS reactions using cone and tray inoculation methods. Plants were infected using grains of white sorghum [Sorghum bicolor (L.) Moench] infested with Fusarium solani f. sp. glycines. Foliar symptom severity was rated 21 days after emergence. The cone and field SDS ratings were significantly correlated and ranged from 0.69 for set 1 to 0.51 for set 3. Correlations of SDS ratings of genotypes between field and greenhouse tray ratings were significant for set 1 and not significant for set 2. The cone method showed the highest correlation with field results and is recommended to screen soybean genotypes for SDS resistance.
Glycine max; Fusarium solani; Fusarium virguliforme; cone inoculation method; SDS; symptom severity; tray inoculation method
Os objetivos deste trabalho foram avaliar dois métodos de inoculação, em casa de vegetação, e determinar o que apresenta maior correlação com resultados de reação à síndrome da morte súbita (SDS - "sudden death syndrome") obtidos em soja, no campo. Três grupos de genótipos, classificados previamente, em campo, como parcialmente resistentes, intermediários e suscetíveis, foram avaliados em casa de vegetação, por meio dos métodos de inoculação em cones e em bandeja. As plantas foram contaminadas com grãos de sorgo [Sorghum bicolor (L.) Moench] infestados com Fusarium solani f. sp. glycines. As avaliações dos sintomas foliares foram realizadas 21 dias após a emergência. As correlações entre os resultados pelo método de cones e os obtidos em campo foram significativas e variaram de 0,69 para o grupo 1 a 0,51 para o grupo 3. As correlações entre os resultados pelo método da bandeja e os obtidos em campo foram de 0,54 para o grupo 1, e não significativas para o grupo 2. O método de cones apresentou a maior correlação com os resultados em campo e é recomendado para avaliação de genótipos de soja quanto à resistência à SDS.
Glycine max; Fusarium solani; Fusarium virguliforme; método de inoculação em cone; SDS; severidade de sintomas; método de inoculação em bandeja
FITOSSANIDADE
Austeclinio Lopes de Farias NetoI; Michael SchmidtII; Glen Lee HartmanIII; Shuxian LiIV; Brian Willians DiersIII
IEmbrapa Cerrados, Rodovia BR-020, Km 17, CEP 73310-970 Planaltina, DF, Brazil. E-mail: auster@cpac.embrapa.br
IISouthern Illinois University, Department of Plant, Soil and General Agriculture, Zipcode 62901 Carbondale, IL, USA. E-mail: mesch@siu.edu
IIIUniversity of Illinois, Department of Crop Sciences, Zipcode 61801 Urbana, IL, USA. E-mail: hartman@uiuc.edu, bdiers@uiuc.edu
IVUnited States Department of Agriculture, Agricultural Research Service, Crop Genetics and Production Research Unit, Zipcode 38776 Stoneville, MS, USA. E-mail: shuxian.li@ars.usda.gov
ABSTRACT
The objectives of this work were to evaluate two greenhouse screening methods for sudden death syndrome (SDS) and to determine which one is best correlated with field resistance of soybean genotypes. The evaluations were done with three sets of genotypes that were classified as partially resistant, intermediate, and susceptible to SDS based on previous field evaluations. These three sets were independently evaluated for greenhouse SDS reactions using cone and tray inoculation methods. Plants were infected using grains of white sorghum [Sorghum bicolor (L.) Moench] infested with Fusarium solani f. sp. glycines. Foliar symptom severity was rated 21 days after emergence. The cone and field SDS ratings were significantly correlated and ranged from 0.69 for set 1 to 0.51 for set 3. Correlations of SDS ratings of genotypes between field and greenhouse tray ratings were significant for set 1 and not significant for set 2. The cone method showed the highest correlation with field results and is recommended to screen soybean genotypes for SDS resistance.
Index terms:Glycine max, Fusarium solani, Fusarium virguliforme, cone inoculation method, SDS, symptom severity, tray inoculation method.
RESUMO
Os objetivos deste trabalho foram avaliar dois métodos de inoculação, em casa de vegetação, e determinar o que apresenta maior correlação com resultados de reação à síndrome da morte súbita (SDS - "sudden death syndrome") obtidos em soja, no campo. Três grupos de genótipos, classificados previamente, em campo, como parcialmente resistentes, intermediários e suscetíveis, foram avaliados em casa de vegetação, por meio dos métodos de inoculação em cones e em bandeja. As plantas foram contaminadas com grãos de sorgo [Sorghum bicolor (L.) Moench] infestados com Fusarium solani f. sp. glycines. As avaliações dos sintomas foliares foram realizadas 21 dias após a emergência. As correlações entre os resultados pelo método de cones e os obtidos em campo foram significativas e variaram de 0,69 para o grupo 1 a 0,51 para o grupo 3. As correlações entre os resultados pelo método da bandeja e os obtidos em campo foram de 0,54 para o grupo 1, e não significativas para o grupo 2. O método de cones apresentou a maior correlação com os resultados em campo e é recomendado para avaliação de genótipos de soja quanto à resistência à SDS.
Termos para indexação:Glycine max, Fusarium solani, Fusarium virguliforme, método de inoculação em cone, SDS, severidade de sintomas, método de inoculação em bandeja.
Introduction
Sudden death syndrome (SDS) is a soybean disease caused by the soilborne fungus Fusarium solani f. sp. glycines (FSG) (syn. Fusarium virguliforme Akoi, O'Donnell, Homma and Lattanzi) (Roy, 1997). Severe losses due to SDS have been reported in soybeans grown in the USA (Hartman et al., 1995), Argentina, and Brazil (Rupe & Hartman, 1999). The fungus infects plants through the roots, and severely infected plants exhibit blackened and rotted taproots with few lateral roots (Stephens et al., 1993). Symptoms also include interveinal chlorosis and necrosis of leaves, vascular discoloration of the lower part of the stem, premature defoliation and pod abortion (Rupe, 1989). Recently, Aoki et al. (2005) reported that sudden death syndrome of soybean in North and South Americas is caused by phylogenetically and morphologically distinct species. In North America, SDS is caused by Fusarium virguliforme sp. nov., formally known as F. solani f. sp. glycines, and in South America, SDS is caused by four species of Fusarium: Fusarium brasiliense sp. nov., F. cuneirostrum sp. nov., F. tucumaniae, and F. virguliforme.
Screening for SDS reactions of soybean genotypes has been done under field conditions (Hartwig et al., 1996; Schmidt et al., 1999; Farias Neto et al., 2006, 2007), and under greenhouse conditions (Hartman et al., 1997; Mueller, 2001; Njiti et al., 2001). Selection for SDS resistance in the field is difficult, because the disease occurrence is unpredictable due to the sensitivity of symptom development to environmental factors (Njiti et al., 1996). In addition, the evaluation of a great number of lines in the field is time-consuming and expensive.
Researchers have evaluated SDS resistance under greenhouse conditions by inoculating plants with FSG infested oat (Stephens et al., 1993) or sorghum seeds (Hartman et al., 1997; Mueller, 2001), cornmeal (Njiti et al., 2001) or toothpicks (Klingelfuss et al., 2002). Stephens et al. (1993) evaluated the SDS reactions of 12 soybean cultivars, infected with FSG infested oat seeds, in pots, in a greenhouse and in the field, under natural and under inoculation conditions. The correlation between field and greenhouse SDS ratings of cultivars ranged from 0.60 to 0.90. Njiti et al. (2001) tested soybean genotypes for SDS reactions in a greenhouse, using different rates of inoculum mixed to the growth medium. They found that an inoculum rate of 4,000 FSG spores cm-3 of plant growth medium resulted in the best correlation with the field results.
Hashmi et al. (2005) compared the SDS field reactions of soybean genotypes with greenhouse reactions. The genotypes were infected in a greenhouse by layering inoculum in trays, mixing inoculum to the soil, in trays, and layering inoculum in tubes that were kept under precise soil temperature control with a water bath. They obtained the greatest correlation between field and greenhouse results with the water bath system.
Although successes in predicting SDS field reactions with greenhouse methods has been reported; there is a need to evaluate other greenhouse methods that could predict field reactions more efficiently. Inoculation process, generally, is time-consuming and difficult to be implemented if many genotypes are being evaluated.
The objective of this study was to compare two greenhouse inoculation methods to determine if either can efficiently predict field SDS reactions of soybean genotypes.
Materials and Methods
Three sets of genotypes were used in this study. Set 1 included 30 recombinant inbred lines (RILs), selected by Njiti et al. (2001) from a population of 100 lines derived from a cross of the SDS susceptible cultivar Essex with the SDS partially resistant cultivar Forrest (Hartwig & Epps, 1977). The RILs were selected based on previous evaluations of foliar symptoms in five field environments naturally infested with FSG as reported by Njiti et al. (2001) and Hnetkovsky et al. (1996). In that study, the field plots were rated for disease incidence (DI) and disease severity (DS). Disease index (DX) values were calculated from these two scores according to Njiti et al. (1998). Disease incidence was taken as an estimate of the plants percentage in each plot with foliar symptoms. Foliar DS was recorded as: 1, 0-10% chlorosis or 1-5% necrosis; 2, 10-20% chlorosis, or for 6-10% necrosis; 3, 20-40% chlorosis, or for 10-20% necrosis; 4, 60% chlorosis, or for 20-40% necrosis; 5, rates greater than 60% chlorosis or greater than 40% necrosis; 6, defoliation 33%; 7, defoliation up to 66%; 8, defoliation greater than 66%; 9, premature death of the plant (Njiti et al., 2001). A disease index (DX; 0-100) was calculated as (DIxDS)/9. The ten most resistant lines from the population were placed in a partially resistant (PR) class, eight of these lines were significantly more resistant than Forrest; the intermediate (IN) class was composed of the ten lines with resistance ratings closest to the population mean; and susceptible (S) class included the ten least resistant lines, all of them significantly more susceptible than Essex (Njiti et al., 2001).
Set 2 included 24 soybean cultivars and lines with characterized SDS field resistance. These genotypes were previously evaluated for resistance in at least three field environments, and were rated for DI as described previously. Because not all cultivars and lines were evaluated in the same field tests, due to maturity differences among lines, the DI scores were adjusted relatively to resistant and susceptible controls. The relative DI scores were employed to place the lines into partially resistant, intermediate and susceptible classes.
Set 3 included 30 RILs selected from a set of 92, which were derived from the cross of the SDS partially resistant cultivar Ina (Nickell et al., 1999) with the SDS susceptible experimental line LN91-1695. The RILs were evaluated for SDS symptoms in a field in Urbana, IL, during 2003 and 2004 (Farias Neto et al., 2006).
The FSG-1 isolate (Hartman et al., 1995), originated from Monticello, IL, was used to produce all inoculum used in this study, according to Huang & Hartman (1996), with modifications described by Farias Neto et al. (2006). Since 2000, the isolate was inoculated onto soybean and reisolated annually. The isolate was grown on white sorghum seeds, which were soaked in water overnight and autoclaved twice in 1-L flasks. Each flask with 300 g of sorghum seeds was infested with 4 mm diameter plugs of fungal mycelium and, then, incubated for two weeks. The colony forming units (CFU) of the infested sorghum inoculum was determined, as previously described on hairy roots (Li et al., 2008), with modifications described by Farias Neto et al. (2006). Briefly, 1 g of sorghum inoculum was soaked in a 250-mL Erlenmeyer flask containing 100 mL of sterile distilled water. The flasks were shaken at 150 rpm, on an orbital shaker, for 30 min and, then, serially diluted 10 fold with sterile distilled water, for two times, resulting in a 100 fold dilution. From each dilution, 100 µL of inoculum was spread on an agar plate (100x15 mm) containing FSG semi-selective medium (Huang & Hartman, 1996). Six plates were used for each inoculum dilution. The plates were incubated at room temperature (25±2ºC) for 10 days. Colonies of FSG were identified as described previously (Li et al., 2000). The number of colonies on each plate was counted and used to calculate the number of colony-forming units per gram of sorghum. The experiment was run twice. The infested sorghum used as inoculum in the greenhouse experiments averaged 2.4x105 CFU g-1.
In the cone method, all three sets of soybean genotypes were evaluated for SDS reactions in SC-10 type cones, containing a layer of FSG inoculum, as described by Farias Neto et al. (2007). The cones were filled with 100 mL of steam-treated soil mix (2:1 sand:soil) topped with 5 mL (3 g) of FSG infested white sorghum seeds. Twenty mL of soil mix were added to cover the infested sorghum seeds, and three soybean seeds were added to each cone, which were covered with another 20 mL of soil mix. After emergence, seedlings were thinned, and one seedling was left per cone. The soil was maintained near to water-holding capacity by flooding the cones twice daily.
Each cone was an experimental unit, and was arranged in a randomized complete blocks design. The sets were evaluated in separate experiments: sets 1 and 3 were each tested in two experiments. Set 2 was tested in a single five-replication experiment.
Plants were rated 21 days after germination for greenhouse disease severity (GDS) using a scale (1 to 6) adapted from Hartman et al. (1997): 1, no symptoms; 2, slight symptom development, with 1 to 20% chlorotic foliage; 3, moderate symptom development, with 21 to 40% chlorotic or necrotic foliage; 4, heavy symptom development, with 41 to 60% chlorotic or necrotic foliage; 5, severe symptom development, with 61 to 80% chlorotic or necrotic foliage; 6, severe symptom development, with more than 80% chlorotic or necrotic foliage.
For the tray method, the SDS reactions of plants from sets 1 and 2 were tested in 37x52 cm galvanized trays, according to Hartman et al. (1997), with modifications. The trays were filled with a steam-treated soil mix (2:1 sand:soil) to a depth of 4 cm. A template was used to make 7 furrows - 36 cm long, 2 cm deep, and 7 cm apart -, and 10 mL of infested sorghum seed was evenly distributed in each furrow. Soil mix was added to cover the infested seeds to a depth of 2 cm. The template was reapplied to make a 2-cm deep furrow directly over the inoculum. Three 12-cm long experimental units were placed in each furrow resulting in each tray holding 21 experimental units, each one sown with five soybean seeds covered with soil mix to a depth of 2 cm.
The soil was maintained near to water-holding capacity by flooding the trays twice daily. The experimental units were arranged in a randomized complete blocks design, with four replicates for set 1 and three replicates for set 2. The plants were rated for SDS symptoms 21 days after germination, using the GDS scale previously described with score based on the mean of the plants per experimental unit.
For both cone and tray greenhouse experiments, noninfected controls were included. The greenhouse experiments were conducted in Urbana, IL, during the winter of 2002/2003, with a 12-hour photoperiod and air temperatures at 25±2ºC.
The analysis of variance was computed for the greenhouse data using PROC MIXED (SAS Institute, 2000). All factors were considered fixed except for blocks. Means were separated using least significance differences (LSD) at 5% probability. Normality and homogeneity of data variances were verified. Preplanned contrasts were computed between the treatments. The CORR PROCEDURE of SAS was used to calculate Pearson correlations between field DX and DI, and greenhouse disease severity (GDS) data and between rankings of genotypes in the field and greenhouse.
Results and Discussion
Typical SDS foliar symptoms for both cone and tray methods were observed on plants of all three sets grown in the greenhouse in soil with inoculation of infested sorghum. The noninoculated control plots showed no SDS foliar symptoms.
For the cone method, analysis of variance for GDS scores across experiments showed that there were significant differences among the resistance classes defined based on field results. Significant differences were detected among genotypes within classes for set 2, but not for sets 1 and 3. Nonsignificant differences between experiments were detected for sets 1 and 3, each one grown in two separate experiments.
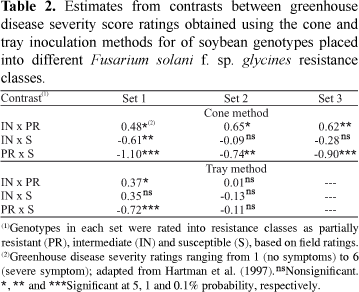
Contrasts between score means of the resistance classes (Table 1), defined by field results, showed that the cone method was able to significantly separate the means of PR and S classes and of PR and IN classes, for the three sets (Tables 1 and 2). However, for sets 2 and 3, the IN class did not differ significantly from the S class. Set 2 was composed by cultivars and lines from different maturity groups, which were not evaluated in the same field experiments; this may have introduced inconsistencies in the field classifications of these genotypes. Besides, genotypes in this set are also from different backgrounds and may respond differently to inoculations under greenhouse conditions, compared to field reactions to the disease. These factors could have made separation of IN and S classes difficult for this set.
Correlations between field DX or DI scores and greenhouse cone GDS scores were significant for the three sets (Table 3). The greatest correlation between greenhouse and field scores was observed for set 1, while the lowest ones was observed for set 2. The correlation for set 3 genotypes was 0.68, when only the selected 30 lines were used in the analysis, and dropped to 0.51 when all 94 lines in the population were included. This lower correlation was expected, because the 30 selected lines were weighted towards those having the greatest and the least resistance. The rank correlation values were greater than the correlations of the scores. The rank correlations between the field and GDS scores were 0.74 for set 1, 0.61 for set 2 and 0.65 for set 3. A rank correlation of 0.50 was detected for set 3, when all the 94 lines from the cross Ina by LN91-1695 were included in the analysis.
The good association between field and greenhouse cone scores is illustrated by a comparison of scores for genotypes in set 1 (Table 4), set 2 (Table 5) and set 3 (Table 6). For example, of the 11 genotypes placed in the partially resistant class in set 2 based on field DI, eight were rated as partially resistant based on the cone ratings. Of the seven genotypes placed in the susceptible class based on field DI, five were rated as susceptible, based on the cone ratings (Table 5).
For the tray method, analysis of variance of GDS scores revealed significant differences an disease severity among field based resistance classes for set 1, and nonsignificant effects for set 2. This method significantly separated PR class from S class and IN from PR classes, for set 1 (Table 1). For set 2, the DS averages of the three classes were similar (Table 2), and there were no significant differences among these classes. No significant differences among genotypes within classes were observed for set 1, while highly significant differences were found for set 2.
Correlations between field and tray scores were lower than the ones between field and cone scores (Table 3). For set 1, both correlations between field DI and tray GDS scores and rank correlation were 0.54. For set 2, no significant correlations were found between field DI and tray GDS scores.
When cone and tray GDS scores were compared, the correlation between these methods was significant for set 1 (0.48) but not for set 2. These correlations between greenhouse methods were lower than the ones between field DI and GDS scores, for either method.
The greenhouse inoculation methods, especially the cone method, were successful in predicting field SDS ratings. Because of the difficulty in achieving consistent SDS symptoms in the field, these greenhouse methods could prove useful for evaluating the level of SDS resistance of soybean genotypes. For example, breeding populations could be first screened for resistance in the greenhouse, followed by the verification of resistance ratings of selected lines in the field.
The correlation between GDS scores obtained using the tray and cone methods was lower than the correlation of the GDS scores for either method with field scores. It is surprising that GDS scores from the two greenhouse methods were not more highly correlated since for both methods, the same inoculum, greenhouse, planting depth, and watering regime were used.
A greater correlation was observed between GDS scores and field scores for the cones method than for the trays method. A reason for the lower correlation for the tray method may be that roots too frequently escaped infection on this method compared to the cone method. This is because a continuous layer of infested grain was placed in the cones, whereas for the tray method, infested grain was only placed under the seed. This allowed the roots to potentially grow around the inoculum in the tray method, thus escaping the disease.
Some lines partially resistant in the field were susceptible with both greenhouse methods, showing that the inoculum overcame the resistance of these lines, as observed by Njiti et al. (2001). In addition, genotypes in set 2 were from different backgrounds and may respond differently to inoculations under greenhouse conditions compared to field reactions to the disease.
The cone method requires more resources than the tray one for conducting resistance evaluations. This is because each cone has to be prepared separately and contains only one plant. In contrast, 21 experimental units, planted with five seeds each, were grown in each tray. Because of the fewer plants in each experimental unit with the cone method, we used more replications with this method than for the tray one.
The associations obtained between the greenhouse cone method and field scores were not as great as observed by Njiti et al. (2001) or Hashmi et al. (2005). Set 1 lines and field data used in the present work were the same used by Njiti et al. (2001). They evaluated these lines in a greenhouse test in which plants were grown in pots with inoculum mixed into the growth medium with low (3.3 x 103 spores cm-3 of grow medium), moderate (5 x 103), and high (104) inoculum levels. The moderate inoculum level resulted in the greatest correlation with field results and the R2 value from the regression was 0.60 which is greater than the R2 from the cone test, which was 0.48.
Hashmi et al. (2005) achieved a correlation of 0.81 between field and greenhouse inoculations using the same set 2 genotypes and field data used in this work. In cone method used in this work and in the method used by Hashmi et al. (2005), plants were grown in tubes and inoculated with a layer of inoculum that the roots needed to grow through. The main difference between these two methods is that in Hashmi et al. (2005) greater soil temperature control were obtained by the use of a water bath system whereas our soil temperature was regulated only by the air temperature in the greenhouse. This greater soil temperature regulation may be a major factor leading to the high correlation between field and greenhouse results observed by Hashmi et al. (2005) Further research is needed to investigate the role of soil temperature on SDS development in the greenhouse.
The cone method used in this study has a number of advantages compared to other methods. Although the correlations with field results were not as great compared to the methods described by Nijti et al. (2001) or Hashmi et al. (2005), the cone method is less complicated to set up than these other methods and does not require a water bath system. This makes it a good choice when researchers need a relatively simple system to rate the SDS resistance levels of genotypes in genetic mapping studies, breeding programs and cultivar testing.
Conclusions
1. The cone method showed the highest correlations with field results and can be used to screen soybean genotypes for sudden death syndrome resistance.
2. The tray method is a good option for screening soybean germplasm for sudden death syndrome resistance, when many genotypes need to be tested.
Acknowledgements
To Coordenação de Aperfeiçoamento de Pessoal de Nível Superior and to Embrapa, for support; to the Illinois Soybean Association and to the United Soybean Board, for research support and grants; to Dr. David Lightfoot, Southern Illinois University, Carbondale, IL, USA, for providing the recombinant inbreed lines (RILs) of set 1.
Received on May 6, 2008 and accepted on July 31, 2008
- AOKI, T.; O'DONNELL, K.; SCANDIANNI, M.M. Sudden death syndrome of soybean in South America is caused by four species of Fusarium: Fusarium brasiliense sp. nov., F. cuneirostrum sp. nov., F. tucumaniae, and F. virguliforme Mycoscience, v.46, p.162-183, 2005.
- FARIAS NETO, A.L. de; HARTMAN, G.L.; PEDERSEN, W.L.; LI, S.; BOLLERO, G.A.; DIERS,W.B. Irrigation and inoculation treatments that increase the severity of soybean sudden death syndrome in the field. Crop Science, v.46, p.2547-2554, 2006.
- FARIAS NETO, A.L. de; HASHMI, R.; SCHMIDT, M.; CARLSON, S.R.; HARTMAN, G.L.; LI, S.; NELSON, R.L.; DIERS, B.W. Mapping and confirmation of a new sudden death syndrome resistance QTL on linkage group D2 from the soybean genotypes PI 567374 and Ripley. Molecular Breeding, v.20, p.53-62, 2007.
- FARIAS NETO, A.L.; SOUZA, P.I.M.; MOREIRA, C.T.; DIANESE, A.C.; SILVA, N.S.; SILVA, S.A. Reação de genótipos de soja à síndrome da morte súbita, causada pelo fungo Fusarium solani f. sp. glycines In: REUNIÃO DE PESQUISA DE SOJA DA REGIÃO CENTRAL DO BRASIL, 29., 2007, Campo Grande. Anais Londrina: Embrapa Soja; Campo Grande: Uniderp, 2007. p.128-130.
- HARTMAN, G.L.; HUANG, Y.H.; NELSON, R.L.; NOEL, G.R. Germplasm evaluation of Glycine max for resistance to Fusarium solani, the causal organism of sudden death syndrome. Plant Disease, v.81, p.515-518, 1997.
- HARTMAN, G.L.; NOEL, G.R.; GRAY, L.E. Occurrence of soybean sudden death syndrome in East-Central Illinois and associated yield losses. Plant Disease, v.79, p.314-318, 1995.
- HARTWIG, E.E.; EPPS, J.M. Registration of 'Forrest' soybeans. Crop Science, v.13, p.287, 1977.
- HARTWIG, E.E.; YOUNG, L.D.; GIBSON, P. Registration of soybean 'D83-3349' resistant to sudden death syndrome, soybean cyst nematode, and two root-knot nematode. Crop Science, v.36, p.212, 1996.
- HASHMI, R.Y.; BOND, J.P.; SCHMIDT, M.E.; KLEIN, J.H. A temperature-controlled water bath method for evaluating soybean reaction to sudden death syndrome (SDS). Plant Health Progress, p.1-9, 2005. Available at: <http://www.plantmanagementnetwork.org/pub/php/research/2005/sds/>. Accessed on: 12 Jul. 2008. doi:10.1094/PHP-2005-0906-01-RS, 2005.
- HNETKOVSKY, N.; CHANG, S.J.C.; DOUBLER, T.W.; GIBSON, P.T.; LIGHTFOTT, D.A. Genetic mapping of loci underlying field resistance to soybean sudden death syndrome (SDS). Crop Science, v.36, p.393-400, 1996.
- HUANG, Y.H.; HARTMAN, G.L. A semi-selective medium for detecting Fusarium solani, the causal organism of soybean sudden death syndrome. Phytopathology, v.86, p.12, 1996.
- KLINGELFUSS, L.H.; YORINORI, J.T.; ARIAS, C.A.A.; DESTRO, D. Reaction of soybean cultivars to sudden death syndrome and disease scoring methods for screening resistance. Crop Breeding and Applied Biotechnology, v.2, p.257-264, 2002.
- LI, S.; HARTMAN, G.L.; LEE, B.S.; WIDHOLM, J.W. Identification of a stress-induced protein in stem exudates of soybean seedlings root-infected with Fusarium solani f. sp. glycines culture filtrates. Plant Physiology and Biochemistry, v.38, p.803-809, 2000.
- LI, S.; LYGIN, A.; ZERNOVA, O.; LOZOVAYA, V.; HARTMAN, G.L.; WIDHOLM, J.M. Genotype response of soybean (Glycine max) whole plants and hairy roots to Fusarium solani f. sp. glycines infection. Soybean Science, v.27, p.275-282, 2008.
- MUELLER, D.S. Resistance to Fusarium solani f. sp. glycines, the causal organism of sudden death syndrome of soybean 2001. 119p. Dissertation (Ph.D.) - University of Illinois, Urbana-Champaign.
- NICKELL, C.D.; NOEL, G.R.; CARY, T.R.; THOMAS, D.J.; LEITZ, R.A. Registration of 'Ina' soybean. Crop Science, v.9, p.1533, 1999.
- NJITI, V.N.; JOHNSON, J.E.; TORTO, T.A.; GRAY, L.E.; LIGHTFOOT, D.A. Inoculum rates influence selection for field resistance to soybean sudden death syndrome in the greenhouse. Crop Science, v.41, p.1726-1731, 2001.
- NJITI, V.N.; SHENAUT, M.A.; SUTTNER, R.J.; SCHMIDT, M.E.; GIBSON, P.T. Relationship between soybean sudden death syndrome disease measures and yield components in F6-derived lines. Crop Science, v.38, p.673-678, 1998.
- NJITI, V.N.; SHENAUT, M.A.; SUTTNER, R.J.; SCHMIDT, M.E.; GIBSON, P.T. Soybean response to sudden death syndrome: inheritance influenced by cyst nematode resistance in Pyramid x Douglas progenies. Crop Science, v.36, p.1165-1170, 1996.
- ROY, K.W. Fusarium solani on soybean roots: nomenclature of the causal agent of sudden death syndrome and identity and relevance of F. solani form B. Plant Disease, v.81, p.259-266, 1997.
- RUPE, J.C. Frequency and pathogenicity of Fusarium solani recovered from soybeans with sudden death syndrome. Plant Disease, v.73, p.581-584, 1989.
- RUPE, J.C.; HARTMAN, G.L. Sudden death syndrome. In: HARTMAN, G.L.; SINCLAIR, J.B.; RUPE, J.C. (Ed.). Compendium of soybean diseases 4th ed. St. Paul: APS Press, 1999. p.37-39.
- SAS INSTITUTE. SAS user's guide Cary: SAS Institute, 2000.
- SCHERM, H.; YANG, X.B. Development of sudden death syndrome of soybean in relation to soil temperature and soil water matric potential. Phytopathology, v.86, p.642-649, 1996.
- SCHMIDT, M.E.; SUTTNER, R.J.; KLEIN, J.H.; GIBSON, P.T.; LIGHTFOOT, D.A.; MYERS JUNIOR, O. Registration of LS-G96 soybean germplasm resistant to soybean sudden death syndrome and soybean cyst nematode race 3. Crop Science, v.39, p.598, 1999.
- STEPHENS, P.A.; NICKELL, C.D.; LIM, S.M. Sudden death syndrome development in soybean cultivars differing in resistance to Fusarium solani Crop Science, v.33, p.63-66, 1993.
Inoculation methods under greenhouse conditions for evaluating soybean resistance to sudden death syndrome
Publication Dates
-
Publication in this collection
22 Dec 2008 -
Date of issue
Nov 2008
History
-
Accepted
31 July 2008 -
Received
06 May 2008