ABSTRACT
This study aimed to evaluate some changes in the metabolism of papaya fruits submitted to damage by impact, abrasion and compression. Injuries were caused in two areas of 15 cm2 each; in diametrically opposite positions in the equatorial region of ‘Improved Sunrise Solo Line 72/12’ papaya fruits in ripening stage 3 (fruits with 25-40% of yellow skin). After damage, fruits were stored at 15 ± 1 °C and 85 ± 5% RH, and samples were taken at intervals of two days to evaluate skin color index, incidence of diseases, loss of fresh weight, leak of solutes, pulp firmness, soluble solids content and pectinmethylesterase and polygalacturonase activities. The respiratory rate was measured at intervals of 4, 8, 12, 24, 48 and 72 hours after damage. The results show the suppressive effects of mechanical damage on the final quality and also on fruit shelf life. Mechanically damaged fruits anticipated ripening, with skin color indexes higher than control fruits. Abrasion was the damage that caused more dramatic effects, showing, at the end of the study period, loss of fresh weight and leak of solutes of 27% and 18.7%, respectively, higher than control fruits. In addition, fruits submitted to this type of damage have higher respiratory rate and also higher rot incidence. The pectinmethylesterase and polygalacturonase activity was not consistently changed in relation to damage. Similarly, there was no difference in soluble solids content.
Index terms
Carica papaya L.; impact; abrasion; compression; post-harvest
RESUMO
Objetivou-se avaliar algumas alterações no metabolismo dos frutos do mamoeiro submetidos aos danos de impacto, abrasão e compressão. Os danos foram provocados em duas áreas de 15 cm2 cada, em posições diametralmente opostas, na região equatorial de mamões ‘Improved Sunrise Solo Line 72/12’ no estádio 3 de amadurecimento (frutos com 25 a 40% da área superficial da casca amarela). Após os danos, os frutos foram armazenados a 15 ± 1 ºC e 85 ± 5% de UR, sendo as amostragens feitas em intervalos de dois dias para avaliação de índice de cor da casca, incidência de podridões, perda de massa fresca, vazamento de solutos, firmeza da polpa, teor de sólidos solúveis e atividade das enzimas pectinametilesterase e poligalacturonase. A taxa respiratória foi avaliada nos intervalos de 4, 8, 12, 24, 48 e 72 horas após os danos. Os resultados evidenciaram os efeitos depreciativos dos danos mecânicos na qualidade final e também no tempo de vida de prateleira dos mamões. Os frutos mecanicamente danificados anteciparam o amadurecimento, apresentando índices de cor da casca maiores do que os dos frutos controle. O dano por abrasão foi o que provocou efeitos mais drásticos, por apresentar, no final do período avaliado, perda de massa média e vazamento de solutos 27% e 18,7%, respectivamente, maiores do que nos frutos controle. Além disso, os frutos submetidos a esse dano apresentaram uma maior taxa respiratória e também maior incidência de podridões. A atividade das enzimas pectinametilesterase e poligalacturonase não foi alterada de forma consistente em função dos danos. Da mesma forma, não se observou diferenças no teor de sólidos solúveis.
Termos para indexação
Carica papaya L.; impacto; abrasão; compressão; pós-colheita
INTRODUCTION
Papaya is one of the tropical fruits most consumed in Brazil, with high economic importance in the national fruit production chain (LUZ et al.,2015 LUZ, L.N. PEREIRA, M.G.; BARROS, F.R.; BARROS, G.B.; FERREGUETTI, G.A. Novos híbridos de mamoeiro avaliados nas condições de cultivo tradicional e no semiárido Brasileiro. Revista Brasileira de Fruticultura, Jaboticabal, v. 37, n. 1, p. 159-171, 2015. ). In 2012, papaya world production was about 12 million tons, approximately 435,000 hectares harvested. Brazil stands out as the second world producer, producing that year, about 1.5 million tons in 31,000 hectares harvested, besides being the world’s third largest exporter (FAO, 2015 FAO - Food and Agriculture Organization of the United Nations. FaoStat. 2015. Disponível em: http://faostat.fao.org/site/339;defaut.aspx. Acesso em: 16 abr. 2015.
http://faostat.fao.org/site/339;defaut.a...
). Available data from 2014 indicate that the states of Bahia, Espirito Santo, Ceara and Minas Gerais account for 86% of national production (IBGE, 2015 IBGE - Instituto Brasileiro de Geografia e Estatística. SIDRA - Sistema IBGE de Recuperação Automática. Disponível em: http://www.sidra.ibge.gov.br. Acesso em: 16 abr. 2015.
http://www.sidra.ibge.gov.br...
).
At any ripeness degree, papaya is susceptible to mechanical damage, especially abrasion, impact, compression and cuts (MORETTI, 2002 MORETTI, C.L. Danos mecânicos. In: FOLGATTI, M.I.; SILVEIRA; MATSUURA, F.C. A.U. Mamão pós-colheita. Brasília, DF: Embrapa Informação Tecnológica, 2002. 59 p. (Frutas do Brasil, 21). ). The fruit requires special care during harvest and post-harvest procedures for being quite perishable, having thin skin, tender pulp and climacteric respiratory pattern (PAULL, 1993 PAULL, R.E. Pineapple and papaya, In: SEYMOUR, G.B.; TAYLOR, J.E.; TUCKER, G.A. Biochemistry of fruit ripening. London: Chapman & Hall, 1993, p.291-323. ), with a relatively short post-harvest life, completing its maturation in a few days.
Post-harvest loss of papaya is high in Brazil, especially due to the distances between producing areas and distribution centers, inadequate packaging, additional cost for the implementation of adequate post-harvest infrastructure and low consumer demand in relation to the quality of products (Figueiredo Neto et al., 2013 FIGUEIREDO NETO, A.; OLIVER, N.C.; ROJAS, A.B.; SILVA, J.C.; PADILHA, C. Avaliação pós-colheita de mamão variedade ‘Formosa’ submetido a danos mecânicos e ensaios de compressão durante o armazenamento. Revista Ciências Técnicas Agropecuárias, San José de las Lajas, v.22, n.2, p.5-10, 2013. ). The evaluation of mechanical damages has as main function to facilitate decision-making by producer, wholesaler and retailer on the need for investment in damage prevention measures, reducing post-harvest losses and increasing the profitability of the papaya production chain (TEZOTTO et al., 2011 TEZOTTO, J.V.; JACOMINO, A.P.; GODOY, A.E.; CERQUEIRA-PEREIRA, E.C.; GALLON, C.Z. Efeito do corte como dano mecânico na qualidade e na fisiologia de mamões ‘Golden’. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.1, p.241-247, 2011. Número Especial. ).
Lima et al. (2009) LIMA, L.M.; MORAIS, P.L.S.; MEDEIROS, E.V.; MENDONÇA, V.; XAVIER, I.F.; LEITE, G.A. Qualidade pós-colheita do mamão formosa ‘Tainung 01’ comercializado em diferentes estabelecimentos no município de Mossoró-RN. Revista Brasileira de Fruticultura, Jaboticabal, v.31, n.3, p.902-906, 2009. investigated the postharvest quality of ‘Tainung 01’ papaya marketed in different stores in the city of Mossoró-RN and observed high incidence of damage such as stains, deformations and rots that have compromised the external appearance of fruits. Tezotto et al. (2011) TEZOTTO, J.V.; JACOMINO, A.P.; GODOY, A.E.; CERQUEIRA-PEREIRA, E.C.; GALLON, C.Z. Efeito do corte como dano mecânico na qualidade e na fisiologia de mamões ‘Golden’. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.1, p.241-247, 2011. Número Especial. evaluated the effect of cut as mechanical damage on the quality and physiology of ‘Golden’ papaya showed that firmness, soluble solids and ascorbic acid contents, respiratory rate and ethylene production were influenced by the number of cuts, so that the loss of fruit quality was directly proportional to the number of cuts.
Likewise, Godoy et al. (2010) GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010. observed in ‘Golden’ papaya that damages are responsible for significant losses in fruit quality, and damage caused by impact and abrasion were those that most contribute to loss in quality, quickly accelerating ripening, loss of firmness and fresh fruit weight.
Abrasion, in turn, affects the damaged region, especially regarding color, as the damaged region becomes dark, and the impact promotes faster yellowing of fruits. Figueiredo Neto et al. (2013) FIGUEIREDO NETO, A.; OLIVER, N.C.; ROJAS, A.B.; SILVA, J.C.; PADILHA, C. Avaliação pós-colheita de mamão variedade ‘Formosa’ submetido a danos mecânicos e ensaios de compressão durante o armazenamento. Revista Ciências Técnicas Agropecuárias, San José de las Lajas, v.22, n.2, p.5-10, 2013. observed in ‘Formosa’ papaya that damage caused by impact accelerated maturation and caused an increase in titratable acidity and weight loss during storage, resulting in significant losses in fruit quality.
Thus, the aim of this study was to evaluate some changes in metabolism of ‘Improved Sunrise Solo Line 72/12’ papaya submitted to mechanical damage caused by impact, abrasion and compression.
MATERIAL AND METHODS
Papayas (Carica papaya L.) ‘Improved Sunrise Solo Line 72/12 (ISS 72/12)’ cultivar were harvested at maturity stage 1 (BALBINO, 1997 BALBINO, J.M.S. Efeitos de hidrotermia, refrigeração e ethephon na qualidade pós-colheita do mamão (Carica papaya L.). 1997. 104 f. Dissertação (Doutorado em Fisiologia Vegetal) - Universidade Federal de Viçosa, Viçosa, 1997. ), in the morning in a commercial orchard in the city of Sooretama, ES. Selection for size, maturity stage, physical defects and physiological disorders was performed. Then, fruits were packed in cardboard boxes and transported to the Post-harvest Laboratory of the Federal University of Viçosa, Viçosa, MG.
In the laboratory, fruits with approximately 390 g were selected. Two areas of 15 cm2 were marked on the opposite sides of the equatorial region of fruits and then they were immersed in sodium hypochlorite solution (100 mL L-1) for 5 minutes and rinsed. After, fruits were packed in plastic boxes covered with bubble wrap and stored in cold chamber (15 ±1 °C and 85 ± 5%RH) until they reached maturity stage 3, being then submitted to damage (day zero of assessments).
Damage by impact was performed according to methodology of Dadzie and Orchard (1997) DADZIE, B.K.; ORCHARD, J.E. Routine postharvest screening of banana/plantain hybrids: criteria and methods. Inibap Technical Guidelines 2. Montpellier: International Network for the Improvement of Banana and Plantains, 1997. 63 p. . Fruits were individually damaged with a steel ball of 66 grams loose from a height of 1.2 meters on previously demarcated areas. Fruits were placed on foam to minimize additional damage on the opposite side of the test. Each fruit suffered three impacts, totaling six impacts per fruit. Damage by abrasion was caused with the help of a sandpaper of 80 mesh and the entire damaged area was previously marked. Damage by compression was caused with the aid of a modified horizontal fruit leveler (CALBO; NERY, 1995 CALBO, A.G.; NERY, A.A. Medida de firmeza em hortaliças pela técnica de aplanação. Horticultura Brasileira, Brasília, DF, v. 13, n. 1, p. 14-18, 1995. ).
Fruits were placed in a wooden block weighting 3 kg that remained on fruits for 5 min. The fruit leveler contained two wooden wedges, above and below the fruit simulating corners present in wood boxes.
After damage, fruits were packed in plastic boxes and stored in cold room with temperature of 15 ± 1 °C and 85 ± 5% RH for ten days with samplings every two days.
The experiment was conducted in a split plot scheme, plots were treatments (three types of damage and undamaged fruits) and subplots were evaluation periods (0, 2, 4, 6, 8 and 10 days) in a completely randomized design with four replicates and two fruits per sampling unit. Samplings were initiated after fruits being damaged. Respiratory rate was assessed using three replicates and one fruit per sampling unit measured at intervals of 4, 8, 12, 24, 48 and 72 hours after damage.
Fruits were evaluated for skin color index, weight loss, respiratory rate, leak of solutes, pulp firmness, soluble solids content, activity of pectinmethylesterase and polygalacturonase enzymes and rot incidence.
Skin color index was determined by visual scale described by Balbino (1997) BALBINO, J.M.S. Efeitos de hidrotermia, refrigeração e ethephon na qualidade pós-colheita do mamão (Carica papaya L.). 1997. 104 f. Dissertação (Doutorado em Fisiologia Vegetal) - Universidade Federal de Viçosa, Viçosa, 1997. , assigning score 1 for fruits with up to 10% of yellow skin, score 2 for fruits with 10 to 25%, score 3 for fruits with 25 to 40%, score 4 for fruits with 40 to 55%, score 5 for fruits with 55 to 70%, score 6 for fruits with 70 to 85% and score 7 for fruits with 85 to 100% of yellow skin. The reduction in fresh weight was gravimetrically determined with results being expressed as percentage losses.
The leak of solutes was evaluated according to methodology described by Serek et al. (1995) SEREK, M.; TAMARI, G.; SISLER, E.C.; BOROCHOV, A. Inhibition of ethylene-induced cellular senescence symptoms by 1-methylcyclopropene, a new inhibitor of ethylene action. Physiologia Plantarum, Malden, v.94, n.2, p.229-232, 1995. .
At the site of damage of each fruit, a pulp cylinder was collected (1.14 cm diameter x 1.5 cm length) after skin removal. Cylinders were washed with deionized water and dried with absorbent paper and incubated for 2 hours in 30 ml of deionized water at ambient conditions. After this period, the electrical conductivity of the solution was measured with Tecnopon - CA 150 conductivity meter. Next, samples were autoclaved (121 ° C at 1.5 atm) for 30 min and the conductivity was again measured.
Results were expressed as the ratio between the first and second measurement, multiplied by 100.
Pulp firmness was determined by the resistance to penetration of the tip of EFFEGI type penetrometer with diameters of 5/16 “or 7/16” after removing a surface portion of the skin of each fruit in the damaged area, and results were expressed in kPa.
Soluble solids content was determined in a portable Atago model N1 digital refractometer, with reading in the range from 0 to 32 ° Brix.
The extraction and analysis of enzymes were in accordance with Ruiz (2003) RUIZ, G.A.C. Relação entre componentes da parede celular e atividade enzimática no pedicelo e a suscetibilidade de bananas ao despencamento natural. 2003. 48f. Dissertação (Mestrado em Fitotecnia) - Universidade Federal de Viçosa, Viçosa, 2003. . Five grams of pulp were removed from the damaged spot, added of 20 mL of 2 M sodium chloride and triturated in a plant tissue homogenizer (Polytron) for one minute. Then, the volume was completed to 30 ml with NaCl, adjusting the pH to 6.0, remaining under stirring por1 hour. All of these processes occur in ice bath, at temperatures between 0 and 4 ° C. After stirring, the macerated was centrifuged for 30 minutes at 9500 g at 4°C and the supernatant was used to determine the activity of pectinmethylesterase (PME) and polygalacturonase (PG) enzymes.
To analyze the PME activity, 2 mL of enzymatic extract were added to 30 mL of 1% citrus pectin solution in 0.2 M NaCl, pH 7.5. The reaction medium acidified by the action of PME was titrated with 0.005 M NaOH for five minutes to pH 7.0, at room temperature. The enzymatic activity was expressed in micromoles of hydrolysed esters per gram of fresh pulp mass per minute (mmol ester [g FM]-1 min-1), being regarded as one unit (U).
To determine the PG activity, 380 mL of 50 mM sodium acetate buffer (pH 5.0) plus 100 mL of 0.2% polygalacturonic acid (prewashed with 80% ethanol) were added to 20 mL of enzyme extract, totaling 500 mL. The reaction mixture was incubated at 37 ° C for 12 hours. After this period, 1 ml of dinitrosalicylic acid (DNS) was added to the reaction medium, followed by immersion in boiling water bath for 10 minutes. After this time, 5 mL of distilled water were added per tube, determining the absorbance at 540 nm. The enzymatic activity was expressed in micromoles of galacturonic acid produced per gram of fresh pulp mass per minute (mmol galacturonic acid [g FM]-1 min-1), being regarded as one unit (U).
CO2 production was determined by gas chromatography in Gow Mac Series 550 chromatograph with thermal conductivity detector equipped with aluminum column filled with Porapak Q and thermal conductivity detector. The working conditions were: carrier gas flow (helium) of 40 ml min-1; electric current of 100 mA; temperature of column, detector and injector of 50, 70 and 80 ° C respectively. Results were expressed as mg CO2 kg-1 h-1.
Rot incidence was determined only on the damaged area from the onset of symptoms, by counting the number of infected fruits. The number of days between treatments and onset of symptoms was also verified.
Data were submitted to analysis of variance and regression. Qualitative factors were compared by Tukey test at 5% probability. For quantitative factors, models were chosen based on the significance of regression coefficients using the ‘t’ test at 5% probability in the determination coefficient and potential to explain the phenomenon under study.
For skin color index and rot incidence, descriptive statistics was performed.
RESULTS AND DISCUSSION
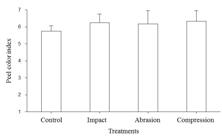
Fruits submitted to damage obtained higher skin color indexes compared to control fruits at the end of the evaluation period. The color indexes of fruits submitted to three types of damage were similar, averaging 6.26 against 5.75 of undamaged fruits (Figure 1). Higher color index indicates acceleration in the maturation process and hence shorter shelf life.
Other types of stress can cause anticipation in papaya ripening. Pimentel et al. (2007) PIMENTEL, R.M.A.; MARCONDES, Y.E.M.; WALDER J.M.M. Qualidade do mamão cv. Solo submetido ao choque térmico e tratamento quarentenário por radiação gama. Revista Brasileira de Fruticultura, Jaboticabal, v.29, n.3, p.483-487, 2007. observed that in fruits irradiated with gamma radiation, skin color change was faster and, in the second day of storage, irradiated fruits were fully yellow. For the authors, anticipation was due to the high production of ethylene as a result of the stress caused by irradiation.
In fruits damaged by impact, three depressions corresponding to the injuries caused by steel ball were observed in each demarcated area. On the other hand, compression caused no symptoms. In areas damaged by abrasion, in addition to the characteristic wrinkling caused by water loss, intensely dark green was observed, indicating an inhibition of the chlorophyll degradation in this region. Godoy et al. (2010) GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010. observed that the area damaged by abrasion did not develop yellow-orange color in ‘Golden’ papaya; Furthermore, the authors reported the occurrence of skin darkening in the abrasion region already on the 2nd day of storage. According to Moretti (2002) MORETTI, C.L. Danos mecânicos. In: FOLGATTI, M.I.; SILVEIRA; MATSUURA, F.C. A.U. Mamão pós-colheita. Brasília, DF: Embrapa Informação Tecnológica, 2002. 59 p. (Frutas do Brasil, 21). , this darkening is due to enzymatic oxidation of the constituents of damaged cells.
Weight loss for all treatments increased linearly during the ten days of evaluation and injury by abrasion was the only to stand out from the others (Figure 2.1).
Fresh weight loss did not differ significantly in the first two days of evaluation for all treatments (Table 1). From the fourth day of evaluation, fruits submitted to damage by abrasion showed greater weight loss. At the end of the study period, the average weight loss caused by this damage was 27% higher than in undamaged fruits. Fruits submitted to other types of damage did not differ from undamaged fruits. The greater weight loss caused by abrasion damage is due to the fact that this damage exposes the internal tissues of fruits by removing the dermal system that has the function of regulating fruit water loss. These results corroborate those obtained by Godoy et al. (2010) GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010. , who observed that abrasion led to greater weight loss, 16% higher than in control fruits. This fact results in acceleration in the product senescence rate, reducing the shelf life (KAYS, 1991 KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p. ).
Papaya is sold by weight unit, and fresh weight losses result in lower yields. According to Cenci et al. (2002) CENCI, S.A.; FONSECA, M.J.; OLIVEIRA; FREITAS-SILVA, O. Procedimentos pós-colheita. In: FOLEGATTI, M.I.; SILVEIRA; MATSUURA, F.C.A.U. Mamão: pós-colheita. Brasília, DF: Embrapa Informação Tecnológica, 2002. p. 24-38 (Frutas do Brasil, 21). , fresh weight losses greater than 5% is enough for the depreciation of papaya and often, these losses are neglected in the marketing chain.
The reduction of pulp firmness occurred abruptly from first to second evaluation stabilizing thereafter (Figure 2.2), corroborating Pinto et al.(2011) PINTO, L.K.A.; MARTINS, M.L.L.; RESENDE, E.D.; THIÈBAUT, J.T.L. Atividade da pectina metilesterase e da ß-Galactosidase durante o amadurecimento do mamão cv. Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p. 713-722, 2011. and Souza et al. (2014 SOUZA, A.F.; SILVA, W.B.; GONÇALVES, Y.S.; SILVA, M.G.; OLIVEIRA, J.G. Fisiologia do amadurecimento de mamões de variedades comercializadas no Brasil. Revista Brasileira de Fruticultura, Jaboticabal, v.36, n.2, p.318-328, 2014. ).This marked reduction in firmness observed in all treatments is due to fruit ripening. Differences were significant only in the first day of evaluation (Table 1), and damage by abrasion resulted in lower firmness, which was also observed by Godoy et al. (2010) GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010. . This lower firmness may be related to greater weight loss caused by this damage, because according to Kays (1991) KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p. , fresh weight loss results in less hydration of cells and hence lower pulp firmness.
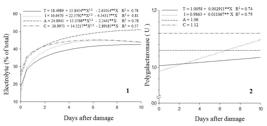
The leak of solutes showed sharp increase from the first to the second assessment in all treatments (Figure 3.1). This may also be related to the higher degree of fruit ripening. Throughout the study period, all forms of damage promoted greater leakage of solutes in relation to control fruits, except for the first day for damage by impact. Damage by abrasion and compression stood out throughout the study period, with average percentage of leak of solutes 19.9% and 12.2% higher, respectively, compared to undamaged fruits (Table 2). Kays (1991) KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p. reported that the first response of fruits to damage is a rapid change in membrane permeability and bioelectric potential.
The response of fruits to damage was immediate, since the leak of solutes on the first day of evaluation caused by compression was 46.4%, and by abrasion, 35.1% higher than in control fruits due to the disruption of cells caused by mechanical damage. On the other hand, on the last day of evaluation, differences compared to control fruits decreased, being 18.7% greater for abrasion and only 3.5% greater for compression. This smaller difference between damaged and undamaged fruits at the end of the study period is due to a series of responses aimed at restoring membranes of damaged tissues and also to the increase in the leak of solutes in undamaged fruits due to ripening and senescence.
This sequence of reactions depends on the damaged tissue and includes desiccation of some cells of the outer parenchyma layer adjacent to the damage, suberization and in some cases lignification of cells below dessicated cells, and acceleration in the process of cell division (KAYS, 1991 KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p. ).
The PG activity was little affected by damage (Table 3). In general, damage by compression caused increased activity, but this did not differ from control fruits and fruits damaged by abrasion.
The PG activity throughout the study period was linear and increasing for control fruits and fruit damaged by impact; however, fruits damaged by abrasion and compression showed no change in PG activity (Figure 3.2).
By comparing enzymatic behavior with firmness (Table 1), it was observed that the sharp decrease in firmness was not accompanied by an increase in the enzymatic activity. According to Paull (1993) PAULL, R.E. Pineapple and papaya, In: SEYMOUR, G.B.; TAYLOR, J.E.; TUCKER, G.A. Biochemistry of fruit ripening. London: Chapman & Hall, 1993, p.291-323. , enzyme activity increases during papaya ripening, showing peak activity when fruits have from 40 to 60% of yellow skin and then, it apparently declines. In this study, high enzyme activity throughout the period of analysis is due to the fact that fruits showed color index 3 (25 to 40% of yellow skin) when they were submitted to damage and therefore supposedly had PG activity close to maximum level.
Silva et al. (2006) SILVA, H.R.F.; RESENDE, E.D.; PINTO, L.K.A.; ALMEIDA, R.F.; MARTINS, M.L.L. Atividade da enzima poligalacturonase em frutos de mamoeiro cv. Golden armazenados sob refrigeração. Revista Brasileira de Armazenamento, Viçosa, MG, v.31, n.2, p.187-191, 2006. found in ‘Golden’ papaya that the PG enzyme has an intense activity in the early ripening of fruits and contributes to pulp softening. Moreover, Lazan et al. (1995) LAZAN, H.; SELAMAT, K; ALI, Z.M. ß-Galactosidase, polygalacturonase and pectinesterase in differential softening and cell wall modification during papaya fruit ripening. Physiologia Plantarum, Malden, v.95, n.1, p.106-112, 1995. found steady increase in PG activity throughout papaya ripening, with the greatest activity in the internal tissue of fruit mesocarp. In the same tissue, the authors observed a three-fold increase in activity when fruits passed from stage of 5% of yellow skin to 100% of yellow skin. On the other hand, pulp firmness only showed a sharp drop when fruits passed from stage 50 to 75% of yellow skin, but no peak was observed in PG activity in the same stage.
According to the authors, fruit softening is related to pectin depolymerization of and hemicelluloses and PG plays an important role in the solubilization and not in pectin depolymerization; therefore, only the action of the PG may not be sufficient to induce fruit softening.
Damage virtually did not change the PME activity (Table 3). There were no significant differences between treatments during the first eight days of evaluation, with differences only on the tenth day after treatment application, and damage by compression induced higher enzyme activity, differing from control. Pinto et al. (2011) PINTO, L.K.A.; MARTINS, M.L.L.; RESENDE, E.D.; THIÈBAUT, J.T.L. Atividade da pectina metilesterase e da ß-Galactosidase durante o amadurecimento do mamão cv. Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p. 713-722, 2011. found high PME activity at the beginning of storage of ‘Golden’ papaya, which decreased sharply until the third day, coinciding with the early fruit ripening. Similar results were found by Fontes et al. (2008) FONTES, R.V.; SANTOS, M.P.; FALQUETO, A.R.; SILVA, D.M. Atividade da pectinametilesterase e sua relação com a perda de firmeza da polpa de mamão cv. Sunrise Solo e Tainung. Revista Brasileira de Fruticultura, Jaboticabal, v.30, n.1, p.54-58, 2008. , in which the PME activity of ‘Sunrise Solo’ papaya was high on the first day after harvest and reduced gradually until the 4th day, when it reached its lowest value.
It was not possible to make a correlation between PME activity and loss of firmness (Table 1). Pinto et al. (2011) PINTO, L.K.A.; MARTINS, M.L.L.; RESENDE, E.D.; THIÈBAUT, J.T.L. Atividade da pectina metilesterase e da ß-Galactosidase durante o amadurecimento do mamão cv. Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p. 713-722, 2011. concluded that PME enzymes and ß-galactosidase (ß-Gal) are responsible for pectin depolymerization and consequent reduction of firmness of ‘Golden’ papaya during ripening.
PME is more effective at the beginning of ripening and ß-Gal presents abrupt increase at the beginning of ripening, with greater activity in the intermediate and late ripening periods. According to the authors, fluctuations in the intensity of activity of these enzymes occur during ripening, and periods of higher PME intensity precede periods of increased ß-Gal activity. Lazan et al. (1995) LAZAN, H.; SELAMAT, K; ALI, Z.M. ß-Galactosidase, polygalacturonase and pectinesterase in differential softening and cell wall modification during papaya fruit ripening. Physiologia Plantarum, Malden, v.95, n.1, p.106-112, 1995. also reported that the loss of papaya pulp firmness is directly linked to the ß-galactosidase enzyme activity.
Average soluble solids levels ranged from 11.15 to 12.41 °Brix on the first day of evaluation (Table 4). Throughout the study period and comparing treatments, it was not possible to establish any behavior pattern for this variable. The differences between treatments observed on days zero, two and six were apparently random. In studies by Figueredo Neto et al. (2013) FIGUEIREDO NETO, A.; OLIVER, N.C.; ROJAS, A.B.; SILVA, J.C.; PADILHA, C. Avaliação pós-colheita de mamão variedade ‘Formosa’ submetido a danos mecânicos e ensaios de compressão durante o armazenamento. Revista Ciências Técnicas Agropecuárias, San José de las Lajas, v.22, n.2, p.5-10, 2013. , Tezotto et al.(2011) TEZOTTO, J.V.; JACOMINO, A.P.; GODOY, A.E.; CERQUEIRA-PEREIRA, E.C.; GALLON, C.Z. Efeito do corte como dano mecânico na qualidade e na fisiologia de mamões ‘Golden’. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.1, p.241-247, 2011. Número Especial. , Pimentel, et al. (2007) PIMENTEL, R.M.A.; MARCONDES, Y.E.M.; WALDER J.M.M. Qualidade do mamão cv. Solo submetido ao choque térmico e tratamento quarentenário por radiação gama. Revista Brasileira de Fruticultura, Jaboticabal, v.29, n.3, p.483-487, 2007. and Balbino (1997) BALBINO, J.M.S. Efeitos de hidrotermia, refrigeração e ethephon na qualidade pós-colheita do mamão (Carica papaya L.). 1997. 104 f. Dissertação (Doutorado em Fisiologia Vegetal) - Universidade Federal de Viçosa, Viçosa, 1997. , stress caused by compression, cuts, gamma radiation and heat, respectively, were not sufficient to change the soluble solids content in papaya. However, Godoy et al. (2010) GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010. observed reduction of soluble solids content during storage of papaya damaged by abrasion, cut and wrinkled. This demonstrates that the soluble solids content is not a good parameter to evaluate mechanical damage to papaya.
The respiratory rate of fruits was changed due to the stress caused by damage. Damage by abrasion and compression increased the respiratory rate, significantly differing from undamaged fruits four and eight hours after damage (Table 5). At the time of twelve hours, no difference between treatments was observed. Moreover, in time of twenty-four hours, significant differences were again observed, and damage by abrasion stood out, which provided respiratory rate 146% higher than control fruits. In other times, no difference between treatments was observed, despite the higher respiratory rates of damaged fruits.
The peak respiratory rate occurred twelve hours after the start of the experiment for all treatments, but this was greater in damaged fruits.
After peak, respiratory rate slowly returned to baseline levels, stabilizing between 24 and 48 hours after damage (Figure 4). The increase in the respiratory rate of damaged fruits is due to a response of fruits to damage. According to Kays (1991) KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p. , the increase in respiratory rate in response to injury is related to the healing process, with the formation of lignin, suberin and in some cases callose. Montero et al (2010) MONTERO, C.R.S.; SANTOS, L.C.; ANDREAZZA, C.S.; BENDER, R.J. Influência do dano mecânico de impacto e compressão sobre a respiração de maçãs cultivares Fuji Suprema e Royal Gala. Semina: Ciências Agrárias, Londrina, v.31, n.4, p.967-974, 2010. found in ‘Fuji’ and ‘Gala’ apples damaged by impact and compression, increased respiratory rate in the first six hours after damage. After 24 hours, there was a reduction in respiratory rates.
The presence of rot was observed only in fruits damaged by impact and abrasion. When damaged by impact, 25% of fruits showed symptoms on the tenth day after damage. However, in damage by abrasion, 25% of fruits presented symptoms already on the sixth day after damage. With the advance of the storage period, the incidence percentage increased, reaching a total of 87.5% of fruits presenting rot on the tenth day after damage. Precocity and high rot incidence caused by damage by abrasion are due to the fact that this damage removes the protective layer of the fruit and, therefore, exposes the internal tissues of fruits to the atmosphere, increasing the attack of fungi.
Papaya skin color index (Carica papaya L.) ten days after application or not of damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH). Vertical bars represent the standard deviation of the mean.
Loss of weight (1) and firmness (2) of papaya submitted or not (T) to mechanical damage by impact (I), abrasion (A) and compression (C) and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Leak of solutes (1) and polygalacturonase activity (2) of papaya submitted or not (T) to mechanical damage by impact (I), abrasion (A) and compression (C) and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Respiratory rate of papaya submitted or not to mechanical damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH) for 72 hours.
Average percentage of fresh weight loss and average firmness of papaya pulp submitted or not to mechanical damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Average percentage of leak of solutes of papaya submitted or not to mechanical damage by impact, abrasion and compression, and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Average polygalacturonase and pectinametilesterase activity papaya submitted or not to mechanical damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Average soluble solids content of papaya submitted or not to mechanical damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH) for 10 days.
Average respiratory rate of papaya submitted or not to mechanical damage by impact, abrasion and compression and stored in cold chamber (15 ° C and 85 ± 5% RH) for 72 hours.
CONCLUSIONS
Mechanical damage, especially by abrasion, accelerate processes related to ripening and senescence, such as respiratory rate, leak of solutes, weight loss, rot incidence and evolution of the skin color of fruits. There was little change in the activity of pectinmethylesterase and polygalacturonase enzymes with mechanical damage. Among damages, abrasion was the one that most depreciated fruits in both in visual quality as in fruit physiology, thus showing the importance of reducing it in order to obtain higher post-harvest life of fruits.
- BALBINO, J.M.S. Efeitos de hidrotermia, refrigeração e ethephon na qualidade pós-colheita do mamão (Carica papaya L.). 1997. 104 f. Dissertação (Doutorado em Fisiologia Vegetal) - Universidade Federal de Viçosa, Viçosa, 1997.
- CALBO, A.G.; NERY, A.A. Medida de firmeza em hortaliças pela técnica de aplanação. Horticultura Brasileira, Brasília, DF, v. 13, n. 1, p. 14-18, 1995.
- CENCI, S.A.; FONSECA, M.J.; OLIVEIRA; FREITAS-SILVA, O. Procedimentos pós-colheita. In: FOLEGATTI, M.I.; SILVEIRA; MATSUURA, F.C.A.U. Mamão: pós-colheita. Brasília, DF: Embrapa Informação Tecnológica, 2002. p. 24-38 (Frutas do Brasil, 21).
- DADZIE, B.K.; ORCHARD, J.E. Routine postharvest screening of banana/plantain hybrids: criteria and methods. Inibap Technical Guidelines 2. Montpellier: International Network for the Improvement of Banana and Plantains, 1997. 63 p.
- FAO - Food and Agriculture Organization of the United Nations. FaoStat. 2015. Disponível em: http://faostat.fao.org/site/339;defaut.aspx Acesso em: 16 abr. 2015.
» http://faostat.fao.org/site/339;defaut.aspx - FIGUEIREDO NETO, A.; OLIVER, N.C.; ROJAS, A.B.; SILVA, J.C.; PADILHA, C. Avaliação pós-colheita de mamão variedade ‘Formosa’ submetido a danos mecânicos e ensaios de compressão durante o armazenamento. Revista Ciências Técnicas Agropecuárias, San José de las Lajas, v.22, n.2, p.5-10, 2013.
- FONTES, R.V.; SANTOS, M.P.; FALQUETO, A.R.; SILVA, D.M. Atividade da pectinametilesterase e sua relação com a perda de firmeza da polpa de mamão cv. Sunrise Solo e Tainung. Revista Brasileira de Fruticultura, Jaboticabal, v.30, n.1, p.54-58, 2008.
- GODOY, A.E.; JACOMINO, A.P.; CERQUEIRA-PEREIRA, E.C.; GUTIERREZ, A.S.D.;VIEIRA, C.E.M.; FORATO, L.A. Injúrias mecânicas e seus efeitos na qualidade de mamões Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.32, n.3, p.682-691, 2010.
- IBGE - Instituto Brasileiro de Geografia e Estatística. SIDRA - Sistema IBGE de Recuperação Automática. Disponível em: http://www.sidra.ibge.gov.br Acesso em: 16 abr. 2015.
» http://www.sidra.ibge.gov.br - KAYS, J.S. Postharvest physiology of perishables plant products. New York: Avi, 1991. 532p.
- LAZAN, H.; SELAMAT, K; ALI, Z.M. ß-Galactosidase, polygalacturonase and pectinesterase in differential softening and cell wall modification during papaya fruit ripening. Physiologia Plantarum, Malden, v.95, n.1, p.106-112, 1995.
- LIMA, L.M.; MORAIS, P.L.S.; MEDEIROS, E.V.; MENDONÇA, V.; XAVIER, I.F.; LEITE, G.A. Qualidade pós-colheita do mamão formosa ‘Tainung 01’ comercializado em diferentes estabelecimentos no município de Mossoró-RN. Revista Brasileira de Fruticultura, Jaboticabal, v.31, n.3, p.902-906, 2009.
- LUZ, L.N. PEREIRA, M.G.; BARROS, F.R.; BARROS, G.B.; FERREGUETTI, G.A. Novos híbridos de mamoeiro avaliados nas condições de cultivo tradicional e no semiárido Brasileiro. Revista Brasileira de Fruticultura, Jaboticabal, v. 37, n. 1, p. 159-171, 2015.
- MONTERO, C.R.S.; SANTOS, L.C.; ANDREAZZA, C.S.; BENDER, R.J. Influência do dano mecânico de impacto e compressão sobre a respiração de maçãs cultivares Fuji Suprema e Royal Gala. Semina: Ciências Agrárias, Londrina, v.31, n.4, p.967-974, 2010.
- MORETTI, C.L. Danos mecânicos. In: FOLGATTI, M.I.; SILVEIRA; MATSUURA, F.C. A.U. Mamão pós-colheita. Brasília, DF: Embrapa Informação Tecnológica, 2002. 59 p. (Frutas do Brasil, 21).
- PAULL, R.E. Pineapple and papaya, In: SEYMOUR, G.B.; TAYLOR, J.E.; TUCKER, G.A. Biochemistry of fruit ripening. London: Chapman & Hall, 1993, p.291-323.
- PIMENTEL, R.M.A.; MARCONDES, Y.E.M.; WALDER J.M.M. Qualidade do mamão cv. Solo submetido ao choque térmico e tratamento quarentenário por radiação gama. Revista Brasileira de Fruticultura, Jaboticabal, v.29, n.3, p.483-487, 2007.
- PINTO, L.K.A.; MARTINS, M.L.L.; RESENDE, E.D.; THIÈBAUT, J.T.L. Atividade da pectina metilesterase e da ß-Galactosidase durante o amadurecimento do mamão cv. Golden. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.3, p. 713-722, 2011.
- RUIZ, G.A.C. Relação entre componentes da parede celular e atividade enzimática no pedicelo e a suscetibilidade de bananas ao despencamento natural. 2003. 48f. Dissertação (Mestrado em Fitotecnia) - Universidade Federal de Viçosa, Viçosa, 2003.
- SEREK, M.; TAMARI, G.; SISLER, E.C.; BOROCHOV, A. Inhibition of ethylene-induced cellular senescence symptoms by 1-methylcyclopropene, a new inhibitor of ethylene action. Physiologia Plantarum, Malden, v.94, n.2, p.229-232, 1995.
- SILVA, H.R.F.; RESENDE, E.D.; PINTO, L.K.A.; ALMEIDA, R.F.; MARTINS, M.L.L. Atividade da enzima poligalacturonase em frutos de mamoeiro cv. Golden armazenados sob refrigeração. Revista Brasileira de Armazenamento, Viçosa, MG, v.31, n.2, p.187-191, 2006.
- SOUZA, A.F.; SILVA, W.B.; GONÇALVES, Y.S.; SILVA, M.G.; OLIVEIRA, J.G. Fisiologia do amadurecimento de mamões de variedades comercializadas no Brasil. Revista Brasileira de Fruticultura, Jaboticabal, v.36, n.2, p.318-328, 2014.
- TEZOTTO, J.V.; JACOMINO, A.P.; GODOY, A.E.; CERQUEIRA-PEREIRA, E.C.; GALLON, C.Z. Efeito do corte como dano mecânico na qualidade e na fisiologia de mamões ‘Golden’. Revista Brasileira de Fruticultura, Jaboticabal, v.33, n.1, p.241-247, 2011. Número Especial.
Publication Dates
-
Publication in this collection
2016
History
-
Received
13 May 2015 -
Accepted
12 Apr 2016