Abstract
To determine in vitro anti-aging activity and identify the active chemical constituents in Ginkgo leaf extracts. The antioxidant properties of Ginkgo biloba leaves extracts were evaluated using 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) scavenging and 2,2’-azino-bis (3-ethylbenzothiazoline)-6 sulfonic acid radical (ABTS) scavenging assays. The inhibitory effects of extracts and chemical constituents on reactive oxygen species, matrix metalloproteinase-1 (MMP-1) and collagen tpye I level were tested using human dermal fibroblasts (HDFs). Reverse-phase liquid chromatography (RPLC) and ultra-performance liquid chromatography tandem mass spectroscopy (UPLC/MS/MS) were used to quantitatively analyze flavonoids and terpene trilactones in the extraction. The established quantitative analysis method was validated according to the regulatory guidelines. The extraction and compounds kaempferol 3-O-β-D-glucopyranoside, isorhamnetin-3-O-glucoside, myricetin, ginkgolide A and bilobalide could have potential anti-aging activities of G. biloba. The results suggest that G. biloba could be used as anti-aging products and as a cosmetic and medicinal raw material.
Keywords:
Ginkgo leaf; antioxidant; matrix metalloproteinase-1; collagen I; chemical analysis
1 Introduction
Skin aging is a complex biological phenomenon due to intrinsic and extrinsic factors. Ultraviolet (UV) exposure causes physical changes in the skin through complex pathways and ultimately results in the generation of reactive oxygen species (ROS) and secretion of matrix metalloproteinases (MMPs) and elastase (Demeule et al., 2000Demeule, M., Brossard, M., Page, M., Gingras, D., & Beliveau, R. (2000). Matrix metalloproteinase inhibition by green tea catechins. Biochimica et Biophysica Acta, 1478(1), 51-60. http://dx.doi.org/10.1016/S0167-4838(00)00009-1. PMid:10719174.
http://dx.doi.org/10.1016/S0167-4838(00)...
). ROS directly cause skin aging via oxidative damage of lipids, proteins, and DNA of the skin. They can also indirectly induce the production of MMPs via the MAP kinase pathway (Fisher et al., 2002Fisher, G. J., Kang, S., Varani, J., Bata-Csorgo, Z., Wan, Y., Datta, S., & Voorhees, J. J. (2002). Mechanisms of Photoaging and Chronological Skin Aging. Archives of Dermatology, 138(11), 1462-1470. http://dx.doi.org/10.1001/archderm.138.11.1462. PMid:12437452.
http://dx.doi.org/10.1001/archderm.138.1...
). MMPs are a family of zinc-dependent endopeptidases. MMP-1 is primarily responsible for dermal collagen degradation during the aging process (Kang et al., 2018Kang, C. H., Rhie, S. J., & Kim, Y. C. (2018). Antioxidant and Skin Anti-Aging Effects of Marigold Methanol Extract. Toxicological Research, 34(1), 31-39. http://dx.doi.org/10.5487/TR.2018.34.1.031. PMid:29371999.
http://dx.doi.org/10.5487/TR.2018.34.1.0...
), and MMP-2 and MMP-9 degrade the extracellular matrix (ECM) proteins that influence skin thickness and wrinkle formation (Lu et al., 2013Lu, C., Kim, B. M., Lee, D., Lee, M. H., Kim, J. H., Pyo, H., & Chai, K. Y. (2013). Synthesis of lipoic acid–peptide conjugates and their effect on collagen and melanogenesis. European Journal of Medicinal Chemistry, 69, 449-454. http://dx.doi.org/10.1016/j.ejmech.2013.09.011. PMid:24090916.
http://dx.doi.org/10.1016/j.ejmech.2013....
).
Ginkgo biloba L. (Ginkgoaceae), popularly known as a living fossil (Gong et al., 2008Gong, W., Chen, C., Dobeš, C., Fu, C., & Koch, M. A. (2008). Phylogeography of a living fossil: Pleistocene glaciations forced Ginkgo biloba L. (Ginkgoaceae) into two refuge areas in China with limited subsequent postglacial expansion. Molecular Phylogenetics and Evolution, 48(3), 1094-1105. http://dx.doi.org/10.1016/j.ympev.2008.05.003. PMid:18554931.
http://dx.doi.org/10.1016/j.ympev.2008.0...
), have been used for 5000 years in traditional Chinese medicine. The leaf extract of G. biloba is composed of flavonoids (24%), terpene trilactones (6%), proanthocyanidins, organic acids, and other constituents (Ihl, 2013Ihl, R. (2013). Effects of Ginkgo biloba extract EGb 761® in dementia with neuropsychiatric features: review of recently completed randomised, controlled trials. International Journal of Psychiatry in Clinical Practice, 17(Suppl. 1), 8-14.). The extract has been reported to protect against chronic cerebral hypoperfusion (Kim et al., 2016Kim, M., Bang, J. H., Lee, J., Han, J., Baik, T. G., & Jeon, W. K. (2016). Ginkgo biloba L. extract protects against chronic cerebral hypoperfusion by modulating neuroinflammation and the cholinergic system. Phytomedicine, 23(12), 1356-1364. http://dx.doi.org/10.1016/j.phymed.2016.07.013. PMid:27765355.
http://dx.doi.org/10.1016/j.phymed.2016....
), improve cognitive function and regulate inflammatory responses (Wan et al., 2016Wan, W., Zhang, C., Danielsen, M., Li, Q., Chen, W., Chan, Y., & Li, Y. (2016). EGb761 improves cognitive function and regulates inflammatory responses in the APP/PS1 mouse. Experimental Gerontology, 81, 92-100. http://dx.doi.org/10.1016/j.exger.2016.05.007. PMid:27220811.
http://dx.doi.org/10.1016/j.exger.2016.0...
), and prevent UVB-induced photoaging (Chen et al., 2014Chen, C., Chiang, A., Liu, H., & Chang, Y. (2014). EGb-761 prevents ultraviolet B-induced photoaging via inactivation of mitogen-activated protein kinases and proinflammatory cytokine expression. Journal of Dermatological Science, 75(1), 55-62. http://dx.doi.org/10.1016/j.jdermsci.2014.04.001. PMid:24802711.
http://dx.doi.org/10.1016/j.jdermsci.201...
). Flavonoids are the main contributors to the anti-inflammatory and antioxidant activity of the extract (Baek et al., 2016Baek, S. H., Lee, J. H., Ko, J., Lee, H., Nam, D., Lee, S. G., Yang, W. M., Um, J., Lee, J., Kim, S., Shim, B. S., & Ahn, K. S. (2016). Ginkgetin Blocks Constitutive STAT3 Activation and Induces Apoptosis through Induction of SHP-1 and PTEN Tyrosine Phosphatases. Phytotherapy Research, 30(4), 567-576. http://dx.doi.org/10.1002/ptr.5557. PMid:27059688.
http://dx.doi.org/10.1002/ptr.5557...
), whereas terpene trilactones are the main contributors to blood microcirculation and neuroprotective effects (Jiang et al., 2014Jiang, M., Li, J., Peng, Q., Liu, Y., Liu, W., Luo, C., Peng, J., Li, J., Yung, K. K. L., & Mo, Z. (2014). Neuroprotective effects of bilobalide on cerebral ischemia and reperfusion injury are associated with inhibition of pro-inflammatory mediator production and down-regulation of JNK1/2 and p38 MAPK activation. Journal of Neuroinflammation, 11(1), 167. http://dx.doi.org/10.1186/s12974-014-0167-6. PMid:25256700.
http://dx.doi.org/10.1186/s12974-014-016...
). In addition, the extract has been demonstrated to be effective in preventing apoptosis and to possess antioxidant activity (Mohanta et al., 2014Mohanta, T. K., Tamboli, Y., & Zubaidha, P. K. (2014). Phytochemical and medicinal importance of Ginkgo biloba L. Natural Product Research, 28(10), 746-752. http://dx.doi.org/10.1080/14786419.2013.879303. PMid:24499319.
http://dx.doi.org/10.1080/14786419.2013....
). However, the relationship between skin anti-aging activity and the chemical constituents in the leaf extract of G. biloba has not been investigated in detail.
The present study was therefore performed to investigate the anti-aging activities of the extraction and main chemical constituents in G. biloba through DPPH, ABTS radical scavenging, ROS scavenging, MMP-1 inhibitory and collagen promoting activities. The chemical constituents in the extraction with anti-aging activity were further elucidated by the established quantitative methods. The results wish to support the use of G. biloba leaf as a raw material in anti-aging cosmetic products in future.
2 Materials and methods
2.1 Chemical and reagents
Ginkgo leaf was collected from Jiangsu province, China (Pizhou medicinal store). 732 cation adsorption resin and HPD5000 macroporous adsorption resin were purchased from Tianjin Nankai University resin Co., Ltd. (Tianjin, China). 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) and 2,2’-azino-bis (3-ethylbenzo thiazoline)-6 sulfonic acid (ABTS) were obtained from Biodee Biotechnology Co., Ltd. (Beijing, China). MMP-1 Kit (No. I21032323) and collagen I Kit were purchased from CUSABIO. ROS Kit was purchased from Beyotime Biotechnology (No. 01191770503). HPLC-grade acetonitrile was purchased from Merck (Germany). Ultra-pure water (18.2 MΩ) was prepared with a Milli-Q water purification system (Millipore, Bedford, MA, USA). Rutin (1), quercetin-3-O-β-glucoside (2), kaempferol 3-O-β-D-glucopyranoside (3), isorhamnetin-3-O-glucoside (4), myricetin (5), apigenin (6), isoginkgetin (7), ginkgetin (8), ginkgolide A (9), ginkgolide B (10), ginkgolide C (11), ginkgolide J (12), and bilobalide (13) were purchased as reference standards from Beijing Simianti Tech. Co., Ltd. (Beijing, China). All of the reference compounds were determined to be greater than 98% pure by high-performance liquid chromatography (HPLC) analysis. All other reagents were of analytical grade.
2.2 Cell culture
HDFs were purchased from the Cell Bank of the Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences. The cells were cultured in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS; BioWhittaker, Walkersville, MD, USA) and 1% penicillin-streptomycin (Gibco BRL, NY, USA), at 37 °C in a humidified atmosphere containing 5% CO2.
2.3 Extraction method
Air-dried samples (0.1 kg) were sieved and extracted with 1.5 L of 95% ethanol (v/v) by ultrasonic extraction for 60 min. In order to remove ginkgolic acids and enrich the active ingredients, the concentrated extraction was eluted through AB-8 macroporous adsorption resin, 732 cation adsorption resin, and HPD5000 macroporous adsorption resin, successively. We got the 60% ethanol (v/v) elution fraction as the tested extraction. The concentrated dry extract was frozen for use in the following chemical and bioactivity assays.
2.4 Determination of DPPH radical scavenging activity
The radical scavenging rates of the samples were measured by the DPPH method (Atta-ur-Rahman et al., 2001Rahman, A. U., Choudhary, M. I., & Thomson, W. J. (2001). Bioassay techniques for drug development. Netherlands: Harwood Academic Publishers.). For each sample, an aliquot of 0.5 mL at different concentrations was added to 1.0 mL DPPH solution (100 μM) for 30 min. Methanol was used as a blank solution. The decrease in absorbance at 517 nm was measured. DPPH radical scavenging activity was expressed as the percentage (%) of the original absorbance as follows (Equation 1):
where AS is the absorbance of the solution after the sample extract has been added and ADPPH is the absorbance of the DPPH solution.
2.5 Determination of ABTS radical scavenging activity
ABTS was dissolved in water to a 7 mM concentration (Re et al., 1999Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology & Medicine, 26(9), 1231-1237. http://dx.doi.org/10.1016/S0891-5849(98)00315-3. PMid:10381194.
http://dx.doi.org/10.1016/S0891-5849(98)...
). ABTS radical cation (ABTS•+) was produced by reacting ABTS stock solution with 2.45 mM potassium persulfate (final concentration) and allowing the mixture to stand in the dark at 25 °C for 12-16 h before use. The ABTS radical cation solution was diluted in ethanol to an absorbance value of 0.70 ± 0.02 at 734 nm and equilibrated at 30 °C. The samples (0.1 mL; final concentrations of 0.1, 0.5, 1.0, 2.0, 4.0, and 6.0 mg/mL) were mixed with 3.9 mL of diluted ABTS radical cation solution, and the absorbance at 734 nm was measured after reaction for 6 min.
2.6 Cell viability assay
Cell viability and proliferation were evaluated using the 3-(4,5-dimethyl thiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay (Lee et al., 2006Lee, M., Lin, Y., Hsu, F., Zhan, G., & Yen, K. (2006). Bioactive constituents of Spatholobus suberectus in regulating tyrosinase-related proteins and mRNA in HEMn cells. Phytochemistry, 67(12), 1262-1270. http://dx.doi.org/10.1016/j.phytochem.2006.05.008. PMid:16782143.
http://dx.doi.org/10.1016/j.phytochem.20...
). HDFs were pretreated with the samples at various concentrations. After incubation for 24 h, MTT solution (final concentration: 5 mg/mL) was added. Next, the cells were incubated at 37 °C for 3 h. Finally, the absorbance of each sample was measured on a microplate reader at 570 nm to obtain the percentage of viable cells.
2.7 ROS measurement
The production of peroxides was measured by staining cells with the fluorescent probe 2,7-dichlorofluorescein diacetate (DCFH-DA) (Zhou et al., 2009Zhou, Y., Zhang, S., Liu, C., & Cai, Y. (2009). The protection of selenium on ROS mediated-apoptosis by mitochondria dysfunction in cadmium-induced LLC-PK1 cells. Toxicology In Vitro, 23(2), 288-294. http://dx.doi.org/10.1016/j.tiv.2008.12.009. PMid:19135140.
http://dx.doi.org/10.1016/j.tiv.2008.12....
). This dye is a stable nonpolar compound that readily diffuses into cells and yields DCFH. Intracellular hydrogen peroxide (H2O2) or OH in the presence of peroxidase changes DCFH to the highly fluorescent compound 2,7-dichlorofluorescein (DCF). Thus, the fluorescence intensity is proportional to the amount of peroxides produced by the cells. After treatment, the cells were harvested, washed with phosphate-buffered saline (PBS: pH 7.2), and incubated with 10 μmol/L DCFH-DA for 0.5 h at 37◦C. Then, ROS generation was measured by the fluorescence intensity (FL-1, 530 nm) of 10,000 cells.
2.8 Enzyme-linked immunosorbent assay (ELISA) for MMP-1 and collagen types I levels
Commercially available ELISA kits were performed to measure MMP-1 and collagen I levels (Du et al., 2017Du, G., Chen, W., Li, X., He, R., & Feng, P. (2017). Induction of MMP-1 and -3 by cyclical mechanical stretch is mediated by IL-6 in cultured fibroblasts of keratoconus. Molecular Medicine Reports, 15(6), 3885-3892. http://dx.doi.org/10.3892/mmr.2017.6433. PMid:28393195.
http://dx.doi.org/10.3892/mmr.2017.6433...
). HDFs were cultured in 6-well plates (5×105/mL cells) with DMEM containing 10% FBS for 24 h, after which the medium was replaced with serum-free medium containing the test samples. After incubation for 24 h, the supernatant was collected from each well and MMP-1 and collagen types I levels were measured using ELISA kits. Each sample was analyzed in triplicate.
2.9 Quantitation of eight flavonoids by HPLC/DAD
Chromatographic separation was performed using the Agilent 1260LC series system (Agilent Technologies, Palo Alto, CA, USA) equipped with an online vacuum degasser, quaternary pump, autosampler, thermostatted column compartment, and DAD. Agilent Technologies Chemstation software for LC (B.02.01) was used. HPLC separation was performed using the Agilent ZORBAX SB-C18 column (4.6 × 250 mm, 5 μm). The detection wavelength was set to 254 nm. The mobile phase consisted of water/phosphoric acid (99.8:0.2, v/v) (A) and acetonitrile (B). The gradient program comprised 15-25% (B) for 0-25 min, 25-35% (B) for 25-35 min, 35-50% (B) for 35-45 min, 50-60% (B) for 45-55 min, 60-70% (B) for 55-60 min, and 70% (B) for 60-70 min. The flow rate was 0.5 mL/min. The column temperature was set to 30 °C and the injection volume was 5.0 μL. The content of the eight flavonoids (compounds 1 to 8) in the active fraction was calculated using an appropriate standard curve. Representative chromatograms of the samples are shown in Figure 1.
The representative chromatogram of standards rutin (1), quercetin-3-O-beta-glucoside (2), kaempferol-3-O-β-D-glucopyranoside (3), isorhamnetin-3-O-glucoside (4), myricetin (5), apigenin (6), isoginkgetin (7), ginkgetin (8).
2.10 Quantitation of five terpene trilactones by UHPLC/MS/MS
Chromatographic analysis was performed on ACQUITYTM UHPLC system (Waters Corp., Milford, MA, USA) with a cooling autosampler and column oven enabling temperature control. The ACQUITY UHPLC® BEH C18 (2.1 × 100 mm, 1.7 μm) was employed, and the column temperature was maintained at 40 °C and a gradient elution with acetonitrile (A)-water (0.1% formic acid, B) was used. The gradient program was as follows: 0-0.5 min, 10% A, 0.5-4.0 min, 90% A. The flow rate was set at 0.3 mL/min. The autosampler was conditioned at 10 °C, and the injection volume for analysis was 2.0 μL. XEVO TQD triple quadruple mass spectrometer (Waters Corp.) equipped with an electrospray ionization source (ESI) was used for mass spectrometric detection. The detection was operated in the multiple reaction monitoring (MRM) mode under unit mass resolution in the mass analyzers. The dwell time was set to 200 ms for each MRM transition. The MRM transitions were m/z 409→345, 425→361, 441→324, 425→308, and 325→163 for ginkgolide A (9), ginkgolide B (10), ginkgolide C (11), ginkgolide J (12), and bilobalide (13), respectively. After optimization, the source parameters were set as follows: curtain gas, 35 psi; nebulizer gas, 50 psi; turbo gas, 60 psi; ion spray voltage, 3.5 kV; and temperature, 500 °C (Table 1). The Masslynx 4.1 software (Waters Corp.) was used for data acquisition and instrument control. Representative chromatograms are shown in Figure 2. Each calibration curve was performed in duplicate, with at least six concentrations of the analyte. The limit of detection (LOD) and limit of quantitation (LOQ) were determined based on S/N ratios of 3:1 and 10:1, respectively.
Representative MRM chromatograms of standards and the extraction. Standards: Ginkgolide A (7), Ginkgolide B (8), Ginkgolide C (9), Ginkgolide J (10), Bilobalide (11).
2.11 Data analysis
The statistical significance of the differences between the mean measurements of each treated group and that of the control group were determined using Dunnett’s t-test. A p-value < 0.05 was considered statistically significant. The data were also analyzed by one-way analysis of variance (ANOVA) using SPSS 17.0 (IBM Corp., Armonk, NY, USA).
3 Results and discussion
3.1 Antioxidant activities of different extracts
The results showed that the extraction showed significant DPPH and ABTS radical scavenging activities with IC50 of 0.103 mg/mL and 0.052 mg/mL. In literature, the extract of G. biloba has been reported to have their potent antioxidant properties. Flavonoids, such as quercetin and kaempferol, can directly quench free radicals (Sadowska-Krępa et al., 2017Sadowska-Krępa, E., Kłapcińska, B., Pokora, I., Domaszewski, P., Kempa, K., & Podgórski, T. (2017). Effects of six-week ginkgo biloba supplementation on aerobic performance, blood pro/antioxidant balance, and serum brain-derived neurotrophic factor in physically active men. Nutrients, 9(8), 803. http://dx.doi.org/10.3390/nu9080803. PMid:28933745.
http://dx.doi.org/10.3390/nu9080803...
).
3.2 Evaluation of the extraction on ROS and MMP-1 in HDFs
The effects of the extraction against ROS production and MMP-1 activity were evaluated. MTT assay was used to determine the maximum permissible level (MPL) of the extracts in HDFs at concentrations of 0.125, 0.25, 0.5, and 1.0 mg/mL. F0 represents the fluorescence value of the control group, and F represents the fluorescence value of the sample group, and the greater the FO/F ratio, the stronger ROS scavenging ability. At the tested concentration (0.2 mg/mL), the extraction showed significant ROS scavenging activity (FO/F value 1.38±0.0025) compared with the control group (FO/F value 1±0.043), and ROS control group (FO/F value 0.84±0.023). Moreover, the extraction also showed significant MMP-1 inhibitory activity at the concentration of 0.2 mg/mL (MMP-1 level 0.22±0.03 ng/mL) and the concentration of 0.1 mg/mL (MMP-1 level 1.115±0.25 ng/mL) compared with the control group (MMP-1 level 8.98±0.06 ng/mL). ROS are thought to be a major factor in the destruction of collagen, which is a hallmark of photoaging (Dong et al., 2008Dong, K. K., Damaghi, N., Picart, S. D., Markova, N. G., Obayashi, K., Okano, Y., Masaki, H., Grether-Beck, S., Krutmann, J., Smiles, K. A., & Yarosh, D. B. (2008). UV-induced DNA damage initiates release of MMP-1 in human skin. Experimental Dermatology, 17(12), 1037-1044. http://dx.doi.org/10.1111/j.1600-0625.2008.00747.x. PMid:18459971.
http://dx.doi.org/10.1111/j.1600-0625.20...
). The maintenance of structural integrity of the connection between the epidermis and dermis, called the dermal-epidermal junction, is important to prevent skin aging (Amano et al., 2005Amano, S., Ogura, Y., Akutsu, N., Matsunaga, Y., Kadoya, K., Adachi, E., & Nishiyama, T. (2005). Protective effect of matrix metalloproteinase inhibitors against epidermal basement membrane damage: skin equivalents partially mimic photoageing process. British Journal of Dermatology, 153(s2, Suppl. 2), 37-46. http://dx.doi.org/10.1111/j.1365-2133.2005.06968.x. PMid:16280020.
http://dx.doi.org/10.1111/j.1365-2133.20...
). MMP-1 is considered a key cellular regulator that plays a prominent role in the breakdown of the dermal extracellular matrix, in particular collagen types I and III, during the photoaging process, resulting in collagen deficiency and wrinkling. In vitro studies with cultured HDFs exposed to UV radiation showed an increase in ROS production and a decrease in type I collagen synthesis (Buechner et al., 2008Buechner, N., Schroeder, P., Jakob, S., Kunze, K., Maresch, T., Calles, C., Krutmann, J., & Haendeler, J. (2008). Changes of MMP-1 and collagen type Iα1 by UVA, UVB and IRA are differentially regulated by Trx-1. Experimental Gerontology, 43(7), 633-637. http://dx.doi.org/10.1016/j.exger.2008.04.009. PMid:18524517.
http://dx.doi.org/10.1016/j.exger.2008.0...
).
3.3 Validation of the established UHPLC/MS/MS method
The linearity for each compound was established by plotting the peak area (y) versus the concentration (x) of each analyte, as demonstrated by the equations reported in Table 2. All the calibration curves showed good linearity (r2>0.998) within the ranges tested. Stability was evaluated by repeated analysis of the same sample solution every 4 h for 24 h. The results (RSD<3%) showed that the sample solution was stable during the analysis. To assess repeatability, six different solutions of the same sample were analyzed. The RSDs were all less than 3%, which indicates that the method has good repeatability. The LOD (ng), LOQ (ng), precision, and repeatability for all the analytes are summarized in Table 2. The established UHPLC/MS/MS method was identified as a fast, accurate, and suitable method for quantitative analysis of the samples.
3.4 Quantitative analysis of the main constituents in different fractions
To elucidate the main chemical constituents responsible for the antioxidant and anti-MMP-1 activities in the extraction, eight flavonoids (compounds 1 to 8) and 5 terpene trilactones (compounds 9 to 13) in the extraction were quantified by the established HPLC/DAD and UHPLC/MS/MS, respectively. The active extraction contained the flavonoids rutin (1, 4.02±0.022 µg/mg), quercetin-3-O-β-glucoside (2, 1.19±0.016 µg/mg), kaempferol 3-O-β-D-glucopyranoside (3, 0.45±0.003µg/mg), isorhamnetin-3-O-glucoside (4, 0.54±0.003 µg/mg), myricetin (5, 4.71±0.009 µg/mg). These flavonoids are mainly with a glycosidic bond at C3 position. The main flavonoid myricetin has been reported to have various biological activities, including antioxidant, anticancer (Phillips et al., 2011Phillips, P. A., Sangwan, V., Borja-Cacho, D., Dudeja, V., Vickers, S. M., & Saluja, A. K. (2011). Myricetin induces pancreatic cancer cell death via the induction of apoptosis and inhibition of the phosphatidylinositol 3-kinase (PI3K) signaling pathway. Cancer Letters, 308(2), 181-188. http://dx.doi.org/10.1016/j.canlet.2011.05.002. PMid:21676539.
http://dx.doi.org/10.1016/j.canlet.2011....
), anti-inflammatory (Tong et al., 2009Tong, Y., Zhou, X., Wang, S., Yang, Y., & Cao, Y. (2009). Analgesic activity of myricetin isolated from Myrica rubra Sieb. et Zucc. leaves. Archives of Pharmacal Research, 32(4), 527-533. http://dx.doi.org/10.1007/s12272-009-1408-6. PMid:19407970.
http://dx.doi.org/10.1007/s12272-009-140...
), antibacterial (Xu et al., 2001Xu, H., Ziegelin, G., Schroder, W., Frank, J., Ayora, S., Alonso, J. C., Lanka, E., & Saenger, W. (2001). Flavones inhibit the hexameric replicative helicase RepA. Nucleic Acids Research, 29(24), 5058-5066. http://dx.doi.org/10.1093/nar/29.24.5058. PMid:11812837.
http://dx.doi.org/10.1093/nar/29.24.5058...
), antiviral (Pasetto et al., 2014Pasetto, S., Pardi, V., & Murata, R. M. (2014). Anti-HIV-1 Activity of Flavonoid Myricetin on HIV-1 Infection in a Dual-Chamber In Vitro Model. PLoS One, 9(12), e115323. http://dx.doi.org/10.1371/journal.pone.0115323. PMid:25546350.
http://dx.doi.org/10.1371/journal.pone.0...
), and antidiabetic effects (Li & Ding, 2012Li, Y., & Ding, Y. (2012). Minireview: Therapeutic potential of myricetin in diabetes mellitus. Food Science and Human Wellness, 1(1), 19-25. http://dx.doi.org/10.1016/j.fshw.2012.08.002.
http://dx.doi.org/10.1016/j.fshw.2012.08...
). The terpene trilactones ginkgolide A (9, 10.71±0.07 µg/mg), ginkgolide B (10, 6.54±0.09 µg/mg), ginkgolide C (11, 9.09±1.14 µg/mg), ginkgolide J (12, 1.49±0.03 µg/mg), and bilobalide (13, 51.56±2.07 µg/mg) were also quantified in the active extraction of G. biloba. The extraction contained 79.39 µg/mg total lactones. Bilobalide was present at relatively higher levels than other lactones. These lactones were reported to have blood microcirculation and neuroprotective effects.
3.5 Evaluation of the active constituents on MMP-1 and CollageIlevels by ELISA
To elucidate the active chemical structure with anti-aging activity, we further evaluated the 10 chemical constituents tested in the active extraction on MMP-1 level by ELISA. In HDFs, except for quercetin-3-O-β-glucoside, the other 9 components showed significant inhibition on MMP-1 level at concentrations of 0.2 mg/mL (p <0.01). Kaempferol 3-O-β-D-glucopyranoside, isorhamnetin-3-O-glucoside, myricetin, and bilobalide showed significant inhibition on MMP-1 level at a concentration of 0.1 mg/mL (p <0.01) (Figure 3). In literature, these compounds have been reported to possess significant antioxidant and anti-inflammatory activities (Wu et al., 2016Wu, Z., Zheng, X., Gong, M., & Li, Y. (2016). Myricetin, a potent natural agent for treatment of diabetic skin damage by modulating TIMP/MMPs balance and oxidative stress. Oncotarget, 7(44), 71754-71760. http://dx.doi.org/10.18632/oncotarget.12330. PMid:27765936.
http://dx.doi.org/10.18632/oncotarget.12...
; Ko, 2012Ko, S. (2012). Myricetin suppresses LPS-induced MMP expression in human gingival fibroblasts and inhibits osteoclastogenesis by downregulating NFATc1 in RANKL-induced RAW 264.7 cells. Archives of Oral Biology, 57(12), 1623-1632. http://dx.doi.org/10.1016/j.archoralbio.2012.06.012. PMid:22795564.
http://dx.doi.org/10.1016/j.archoralbio....
). Treatment of isorhamnetin 3-O-β-D-glucoside in human fibrosarcoma (HT1080) cells significantly inhibited MMP-9 and MMP-2 activities (Kong et al., 2008Kong, C., Kim, Y. A., Kim, M., Park, J., Kim, J., Kim, S., Lee, B., Nam, T. J., & Seo, Y. (2008). Flavonoid glycosides isolated from Salicornia herbacea inhibit matrix metalloproteinase in HT1080 cells. Toxicology In Vitro, 22(7), 1742-1748. http://dx.doi.org/10.1016/j.tiv.2008.07.013. PMid:18715546.
http://dx.doi.org/10.1016/j.tiv.2008.07....
). Myricetin stimulated procollagen synthesis, and it was proposed that myricetin may utilize signaling mediation through the JNK pathway, a potent inducer of procollagen production (Fisher et al., 2000Fisher, G. J., Datta, S., Wang, Z., Li, X. Y., Quan, T., Chung, J. H., Kang, S., & Voorhees, J. J. (2000). c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. The Journal of Clinical Investigation, 106(5), 663-670. http://dx.doi.org/10.1172/JCI9362. PMid:10974019.
http://dx.doi.org/10.1172/JCI9362...
). In addition, myricetin can downregulate the expression of catalase and superoxide dismutase, the two main enzymes involved in the induction of cellular oxidative stress. Therefore, myricetin is an important material for anti-aging process. Ginkgolide A can also inhibit the expression of MMP-1 at the concentration of 0.1 mg/mL (p<0.05). Ginkgolide A reduces inflammatory response in high-glucose-stimulated human umbilical vein endothelial cells through STAT3-mediated pathway (Zhao et al., 2015Zhao, Q., Gao, C., & Cui, Z. (2015). Ginkgolide A reduces inflammatory response in high-glucose-stimulated human umbilical vein endothelial cells through STAT3-mediated pathway. International Immunopharmacology, 25(2), 242-248. http://dx.doi.org/10.1016/j.intimp.2015.02.001. PMid:25681539.
http://dx.doi.org/10.1016/j.intimp.2015....
).
MMP-1 level of the 10 components rutin (1), quercetin-3-O-β-glucoside (2), kaempferol 3-O-β-D-glucopyranoside (3), isorhamnetin-3-O-glucoside (4), myricetin (5), ginkgolide A (6), ginkgolide B (7), ginkgolide C (8), ginkgolide J (9), and bilobalide (10). *indicates significant difference (p < 0.05); **indicates very significant difference (p < 0.01).
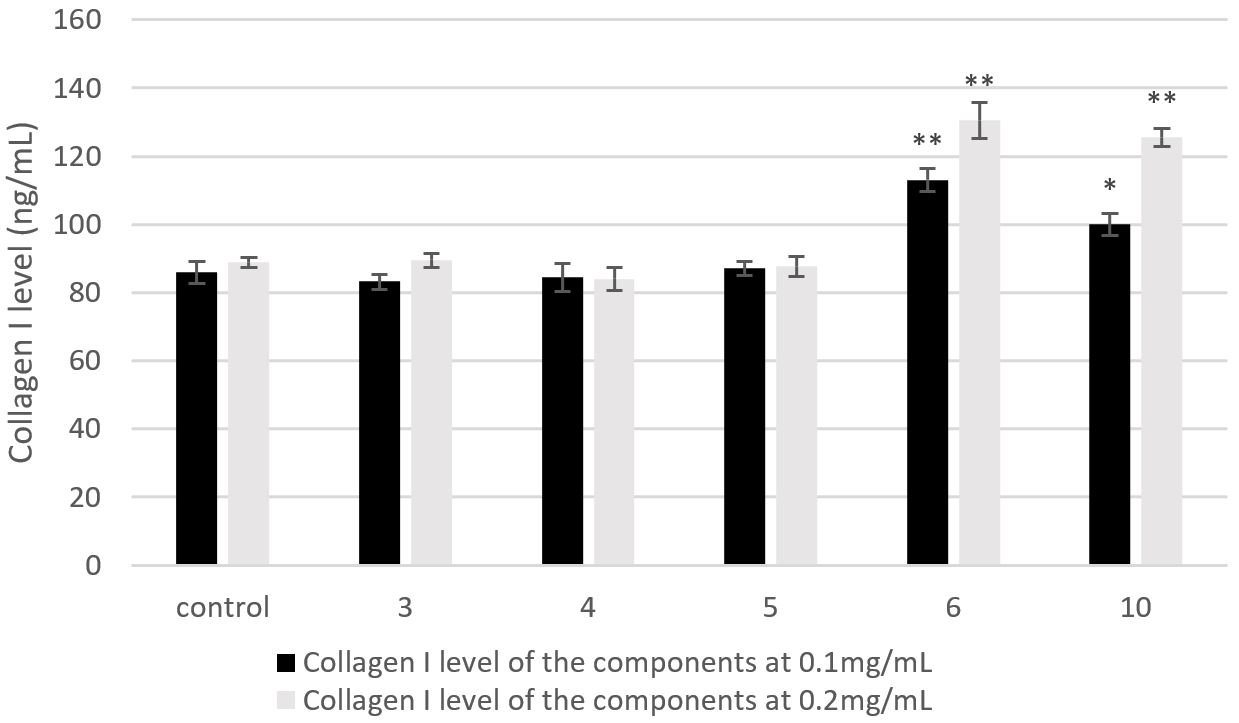
Type I collagen is the major structural component of the ECM and the most abundant protein in skin connective tissue. The individual polypeptide chains of type I collagen are synthesized by dermal fibroblasts from procollagen, which is secreted into the dermal extracellular space. Senescent fibroblasts produce less ECM, such as collagen I and III, and more MMPs, contributing to the observed ageing morphology. In the present study, the effect on Collage I level of the compounds kaempferol 3-O-β-D-glucopyranoside, isorhamnetin-3-O-glucoside, myricetin, ginkgolide A, and bilobalide were further tested (Figure 4). Ginkgolide A and bilobalide showed better collagen promoting activity than other compounds. We should pay more attentions on the lactones like ginkgolide A and bilobalide about their anti-aging activity in future.
Collagen I level of the 5 components kaempferol 3-O-β-D-glucopyranoside (3), isorhamnetin-3-O-glucoside (4), myricetin (5), ginkgolide A (6), and bilobalide (10). *indicates significant difference (p < 0.05); **indicates very significant difference (p < 0.01).
4 Conclusion
The results of this study indicate that the extraction of G. biloba, owing to high levels of flavonoids and lactones, inhibited ROS and MMP-1 degradation in HDFs. The chemical constituents kaempferol 3-O-β-D-glucopyranoside, isorhamnetin-3-O-glucoside, myricetin, ginkgolide A and bilobalide in the active extraction, showed marked MMP-1 inhibitory activity. Ginkgolide A and bilobalide, which have better collagen promoting activity should be studied further for their anti-aging activity. The results provide evidence supporting the use of the leaf extract and active compounds of G. biloba as the anti-aging medicinal and cosmetic raw materials.
Acknowledgements
This study was supported by the Key Laboratory of Cosmetic, China National Light Industry, Beijing Technology and Business University Innovation Fund ID: KLC-2017-YB2. This work was also supported by China Cosmetic Collaborative Innovation Center, the Open Research Fund Program of Beijing Key Lab of Plant Resource Research and Development, BTBU(PRRD-2017-ZD1).
-
Practical Application: Research about anti-aging activities and chemicals of Ginkgo biloba.
References
- Amano, S., Ogura, Y., Akutsu, N., Matsunaga, Y., Kadoya, K., Adachi, E., & Nishiyama, T. (2005). Protective effect of matrix metalloproteinase inhibitors against epidermal basement membrane damage: skin equivalents partially mimic photoageing process. British Journal of Dermatology, 153(s2, Suppl. 2), 37-46. http://dx.doi.org/10.1111/j.1365-2133.2005.06968.x PMid:16280020.
» http://dx.doi.org/10.1111/j.1365-2133.2005.06968.x - Baek, S. H., Lee, J. H., Ko, J., Lee, H., Nam, D., Lee, S. G., Yang, W. M., Um, J., Lee, J., Kim, S., Shim, B. S., & Ahn, K. S. (2016). Ginkgetin Blocks Constitutive STAT3 Activation and Induces Apoptosis through Induction of SHP-1 and PTEN Tyrosine Phosphatases. Phytotherapy Research, 30(4), 567-576. http://dx.doi.org/10.1002/ptr.5557 PMid:27059688.
» http://dx.doi.org/10.1002/ptr.5557 - Buechner, N., Schroeder, P., Jakob, S., Kunze, K., Maresch, T., Calles, C., Krutmann, J., & Haendeler, J. (2008). Changes of MMP-1 and collagen type Iα1 by UVA, UVB and IRA are differentially regulated by Trx-1. Experimental Gerontology, 43(7), 633-637. http://dx.doi.org/10.1016/j.exger.2008.04.009 PMid:18524517.
» http://dx.doi.org/10.1016/j.exger.2008.04.009 - Chen, C., Chiang, A., Liu, H., & Chang, Y. (2014). EGb-761 prevents ultraviolet B-induced photoaging via inactivation of mitogen-activated protein kinases and proinflammatory cytokine expression. Journal of Dermatological Science, 75(1), 55-62. http://dx.doi.org/10.1016/j.jdermsci.2014.04.001 PMid:24802711.
» http://dx.doi.org/10.1016/j.jdermsci.2014.04.001 - Demeule, M., Brossard, M., Page, M., Gingras, D., & Beliveau, R. (2000). Matrix metalloproteinase inhibition by green tea catechins. Biochimica et Biophysica Acta, 1478(1), 51-60. http://dx.doi.org/10.1016/S0167-4838(00)00009-1 PMid:10719174.
» http://dx.doi.org/10.1016/S0167-4838(00)00009-1 - Dong, K. K., Damaghi, N., Picart, S. D., Markova, N. G., Obayashi, K., Okano, Y., Masaki, H., Grether-Beck, S., Krutmann, J., Smiles, K. A., & Yarosh, D. B. (2008). UV-induced DNA damage initiates release of MMP-1 in human skin. Experimental Dermatology, 17(12), 1037-1044. http://dx.doi.org/10.1111/j.1600-0625.2008.00747.x PMid:18459971.
» http://dx.doi.org/10.1111/j.1600-0625.2008.00747.x - Du, G., Chen, W., Li, X., He, R., & Feng, P. (2017). Induction of MMP-1 and -3 by cyclical mechanical stretch is mediated by IL-6 in cultured fibroblasts of keratoconus. Molecular Medicine Reports, 15(6), 3885-3892. http://dx.doi.org/10.3892/mmr.2017.6433 PMid:28393195.
» http://dx.doi.org/10.3892/mmr.2017.6433 - Fisher, G. J., Datta, S., Wang, Z., Li, X. Y., Quan, T., Chung, J. H., Kang, S., & Voorhees, J. J. (2000). c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. The Journal of Clinical Investigation, 106(5), 663-670. http://dx.doi.org/10.1172/JCI9362 PMid:10974019.
» http://dx.doi.org/10.1172/JCI9362 - Fisher, G. J., Kang, S., Varani, J., Bata-Csorgo, Z., Wan, Y., Datta, S., & Voorhees, J. J. (2002). Mechanisms of Photoaging and Chronological Skin Aging. Archives of Dermatology, 138(11), 1462-1470. http://dx.doi.org/10.1001/archderm.138.11.1462 PMid:12437452.
» http://dx.doi.org/10.1001/archderm.138.11.1462 - Gong, W., Chen, C., Dobeš, C., Fu, C., & Koch, M. A. (2008). Phylogeography of a living fossil: Pleistocene glaciations forced Ginkgo biloba L. (Ginkgoaceae) into two refuge areas in China with limited subsequent postglacial expansion. Molecular Phylogenetics and Evolution, 48(3), 1094-1105. http://dx.doi.org/10.1016/j.ympev.2008.05.003 PMid:18554931.
» http://dx.doi.org/10.1016/j.ympev.2008.05.003 - Ihl, R. (2013). Effects of Ginkgo biloba extract EGb 761® in dementia with neuropsychiatric features: review of recently completed randomised, controlled trials. International Journal of Psychiatry in Clinical Practice, 17(Suppl. 1), 8-14.
- Jiang, M., Li, J., Peng, Q., Liu, Y., Liu, W., Luo, C., Peng, J., Li, J., Yung, K. K. L., & Mo, Z. (2014). Neuroprotective effects of bilobalide on cerebral ischemia and reperfusion injury are associated with inhibition of pro-inflammatory mediator production and down-regulation of JNK1/2 and p38 MAPK activation. Journal of Neuroinflammation, 11(1), 167. http://dx.doi.org/10.1186/s12974-014-0167-6 PMid:25256700.
» http://dx.doi.org/10.1186/s12974-014-0167-6 - Kang, C. H., Rhie, S. J., & Kim, Y. C. (2018). Antioxidant and Skin Anti-Aging Effects of Marigold Methanol Extract. Toxicological Research, 34(1), 31-39. http://dx.doi.org/10.5487/TR.2018.34.1.031 PMid:29371999.
» http://dx.doi.org/10.5487/TR.2018.34.1.031 - Kim, M., Bang, J. H., Lee, J., Han, J., Baik, T. G., & Jeon, W. K. (2016). Ginkgo biloba L. extract protects against chronic cerebral hypoperfusion by modulating neuroinflammation and the cholinergic system. Phytomedicine, 23(12), 1356-1364. http://dx.doi.org/10.1016/j.phymed.2016.07.013 PMid:27765355.
» http://dx.doi.org/10.1016/j.phymed.2016.07.013 - Ko, S. (2012). Myricetin suppresses LPS-induced MMP expression in human gingival fibroblasts and inhibits osteoclastogenesis by downregulating NFATc1 in RANKL-induced RAW 264.7 cells. Archives of Oral Biology, 57(12), 1623-1632. http://dx.doi.org/10.1016/j.archoralbio.2012.06.012 PMid:22795564.
» http://dx.doi.org/10.1016/j.archoralbio.2012.06.012 - Kong, C., Kim, Y. A., Kim, M., Park, J., Kim, J., Kim, S., Lee, B., Nam, T. J., & Seo, Y. (2008). Flavonoid glycosides isolated from Salicornia herbacea inhibit matrix metalloproteinase in HT1080 cells. Toxicology In Vitro, 22(7), 1742-1748. http://dx.doi.org/10.1016/j.tiv.2008.07.013 PMid:18715546.
» http://dx.doi.org/10.1016/j.tiv.2008.07.013 - Lee, M., Lin, Y., Hsu, F., Zhan, G., & Yen, K. (2006). Bioactive constituents of Spatholobus suberectus in regulating tyrosinase-related proteins and mRNA in HEMn cells. Phytochemistry, 67(12), 1262-1270. http://dx.doi.org/10.1016/j.phytochem.2006.05.008 PMid:16782143.
» http://dx.doi.org/10.1016/j.phytochem.2006.05.008 - Li, Y., & Ding, Y. (2012). Minireview: Therapeutic potential of myricetin in diabetes mellitus. Food Science and Human Wellness, 1(1), 19-25. http://dx.doi.org/10.1016/j.fshw.2012.08.002
» http://dx.doi.org/10.1016/j.fshw.2012.08.002 - Lu, C., Kim, B. M., Lee, D., Lee, M. H., Kim, J. H., Pyo, H., & Chai, K. Y. (2013). Synthesis of lipoic acid–peptide conjugates and their effect on collagen and melanogenesis. European Journal of Medicinal Chemistry, 69, 449-454. http://dx.doi.org/10.1016/j.ejmech.2013.09.011 PMid:24090916.
» http://dx.doi.org/10.1016/j.ejmech.2013.09.011 - Mohanta, T. K., Tamboli, Y., & Zubaidha, P. K. (2014). Phytochemical and medicinal importance of Ginkgo biloba L. Natural Product Research, 28(10), 746-752. http://dx.doi.org/10.1080/14786419.2013.879303 PMid:24499319.
» http://dx.doi.org/10.1080/14786419.2013.879303 - Pasetto, S., Pardi, V., & Murata, R. M. (2014). Anti-HIV-1 Activity of Flavonoid Myricetin on HIV-1 Infection in a Dual-Chamber In Vitro Model. PLoS One, 9(12), e115323. http://dx.doi.org/10.1371/journal.pone.0115323 PMid:25546350.
» http://dx.doi.org/10.1371/journal.pone.0115323 - Phillips, P. A., Sangwan, V., Borja-Cacho, D., Dudeja, V., Vickers, S. M., & Saluja, A. K. (2011). Myricetin induces pancreatic cancer cell death via the induction of apoptosis and inhibition of the phosphatidylinositol 3-kinase (PI3K) signaling pathway. Cancer Letters, 308(2), 181-188. http://dx.doi.org/10.1016/j.canlet.2011.05.002 PMid:21676539.
» http://dx.doi.org/10.1016/j.canlet.2011.05.002 - Rahman, A. U., Choudhary, M. I., & Thomson, W. J. (2001). Bioassay techniques for drug development Netherlands: Harwood Academic Publishers.
- Re, R., Pellegrini, N., Proteggente, A., Pannala, A., Yang, M., & Rice-Evans, C. (1999). Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biology & Medicine, 26(9), 1231-1237. http://dx.doi.org/10.1016/S0891-5849(98)00315-3 PMid:10381194.
» http://dx.doi.org/10.1016/S0891-5849(98)00315-3 - Sadowska-Krępa, E., Kłapcińska, B., Pokora, I., Domaszewski, P., Kempa, K., & Podgórski, T. (2017). Effects of six-week ginkgo biloba supplementation on aerobic performance, blood pro/antioxidant balance, and serum brain-derived neurotrophic factor in physically active men. Nutrients, 9(8), 803. http://dx.doi.org/10.3390/nu9080803 PMid:28933745.
» http://dx.doi.org/10.3390/nu9080803 - Tong, Y., Zhou, X., Wang, S., Yang, Y., & Cao, Y. (2009). Analgesic activity of myricetin isolated from Myrica rubra Sieb. et Zucc. leaves. Archives of Pharmacal Research, 32(4), 527-533. http://dx.doi.org/10.1007/s12272-009-1408-6 PMid:19407970.
» http://dx.doi.org/10.1007/s12272-009-1408-6 - Wan, W., Zhang, C., Danielsen, M., Li, Q., Chen, W., Chan, Y., & Li, Y. (2016). EGb761 improves cognitive function and regulates inflammatory responses in the APP/PS1 mouse. Experimental Gerontology, 81, 92-100. http://dx.doi.org/10.1016/j.exger.2016.05.007 PMid:27220811.
» http://dx.doi.org/10.1016/j.exger.2016.05.007 - Wu, Z., Zheng, X., Gong, M., & Li, Y. (2016). Myricetin, a potent natural agent for treatment of diabetic skin damage by modulating TIMP/MMPs balance and oxidative stress. Oncotarget, 7(44), 71754-71760. http://dx.doi.org/10.18632/oncotarget.12330 PMid:27765936.
» http://dx.doi.org/10.18632/oncotarget.12330 - Xu, H., Ziegelin, G., Schroder, W., Frank, J., Ayora, S., Alonso, J. C., Lanka, E., & Saenger, W. (2001). Flavones inhibit the hexameric replicative helicase RepA. Nucleic Acids Research, 29(24), 5058-5066. http://dx.doi.org/10.1093/nar/29.24.5058 PMid:11812837.
» http://dx.doi.org/10.1093/nar/29.24.5058 - Zhao, Q., Gao, C., & Cui, Z. (2015). Ginkgolide A reduces inflammatory response in high-glucose-stimulated human umbilical vein endothelial cells through STAT3-mediated pathway. International Immunopharmacology, 25(2), 242-248. http://dx.doi.org/10.1016/j.intimp.2015.02.001 PMid:25681539.
» http://dx.doi.org/10.1016/j.intimp.2015.02.001 - Zhou, Y., Zhang, S., Liu, C., & Cai, Y. (2009). The protection of selenium on ROS mediated-apoptosis by mitochondria dysfunction in cadmium-induced LLC-PK1 cells. Toxicology In Vitro, 23(2), 288-294. http://dx.doi.org/10.1016/j.tiv.2008.12.009 PMid:19135140.
» http://dx.doi.org/10.1016/j.tiv.2008.12.009
Publication Dates
-
Publication in this collection
03 Feb 2020 -
Date of issue
Apr-Jun 2020
History
-
Received
19 Feb 2019 -
Accepted
16 Sept 2019