Abstract
Refractory hypertension (RfH) is an extreme phenotype of resistant hypertension (RH), being considered an uncontrolled blood pressure besides the use of 5 or more antihypertensive medications, including a long-acting thiazide diuretic and a mineralocorticoid antagonist. RH is common, with 10-20% of the general hypertensives, and its associated with renin angiotensin aldosterone system hyperactivity and excess fluid retention. RfH comprises 5-8% of the RH and seems to be influenced by increased sympathetic activity. RH patients are older and more obese than general hypertensives. It is strongly associated with diabetes, obstructive sleep apnea, and hyperaldosteronism status. RfH is more frequent in women, younger patients and Afro-americans compared to RFs. Both are associated with increased albuminuria, left ventricular hypertrophy, chronic kidney diseases, stroke, and cardiovascular diseases. The magnitude of the white-coat effect seems to be higher among RH patients. Intensification of diuretic therapy is indicated in RH, while in RfH, therapy failure imposes new treatment alternatives such as the use of sympatholytic therapies. In conclusion, both RH and RfH constitute challenges in clinical practice and should be addressed as distinct clinical entities by trained professionals who are capable to identify comorbidities and provide specific, diversified, and individualized treatment.
Keywords:
Resistant Hypertension, Refractory Hypertension; Sympathetic Nervous System; Hyperaldosteronism
Resumo
A Hipertensão Arterial Refratária (HARf) representa um fenótipo extremo da hipertensão arterial resistente (HAR), sendo considerada a falência ao tratamento apesar do uso de 5 ou mais classes de anti-hipertensivos, incluindo um diurético tiazídico de longa ação e um antagonista mineralocorticoide. A HAR é comum (10-20%) entre os hipertensos em geral, sendo decorrente de hiperatividade do Sistema Renina Angiotensina Aldosterona e retenção hidrossalina. Aqueles com HARf correspondem a 5-8% dos resistentes e parecem sofrer maior influência catecolaminérgica. Os resistentes tendem a ter maior idade, ao sobrepeso e à obesidade. Comorbidades incluem diabetes, apneia obstrutiva do sono e status de hiperaldosteronismo. Refratários são afro-americanos em maior proporção, mais jovens e, predominantemente, mulheres. Ambos são fortemente associados à elevada albuminúria, HVE, doenças cardio e cerebrovasculares, além da doença renal crônica. O fenômeno do jaleco branco parece ser mais evidente nos resistentes. Quanto ao tratamento, a intensificação da terapia diurética está indicada nos resistentes, enquanto na HARf, a falência à terapia impôs novas alternativas de tratamento ("simpaticolíticas"). Em conclusão, tanto a HAR quanto a HARf constituem-se desafios na prática clínica e devem ser abordadas como entidades clínicas distintas por profissionais especialistas que identifiquem comorbidades e venham a prover um tratamento específico, diversificado e individualizado.
Palavras-chave:
Hipertensão Resistente, Hipertensão Refratária; Sistema Nervoso Simpático; Hiperaldosteronismo
Introduction
Resistant hypertension (RH) has been studied in several different populations since the end of the 20th century. Nevertheless, it was only in 2008 that the American Heart Association published the first guidelines on RH, standardizing its definition and establishing the main risk factors, secondary causes, and the diagnostic and therapeutic approach to these patients11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90. Thenceforth, many studies have demonstrated the high cardiovascular morbidity and mortality and begun to advocate for new therapeutic regimens (i.e. adding definitively spironolactone as the fourth-line drug choice), as well as new interventional therapies seeking for a better blood pressure (BP) control.
In an effort to define a subgroup of high-risk patients who should benefit the most from these new therapies, the refractory hypertension (RfH) definition was established in 201222 Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12. for individuals with worst BP control and, possibly, the worst cardiovascular outcomes.
Despite the final common pathway of an increased sympathetic tonus and hydrosaline retention, the current literature suggests the existence of different clinical phenotypes with different prognoses. These phenotypes would range from arterial hypertension that is responsive to initial treatment to RH and, more recently, to RfH.22 Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12.
Although RfH seems to be an extreme phenotype of RH, recent studies have suggested different pathophysiological mechanisms. Whereas an increased sympathetic activity plays a more important role in the former, inappropriate hydrosaline retention due to a renin-angiotensin-aldosterone system (RAAS) hyperactivity is a major factor in RfH.33 Leibowitz A, Schiffrin EL. Immune mechanisms in hypertension. Curr Hypertens Rep 2011;13:465-72.
4 Modolo R, de Faria AP, Almeida A, Moreno H. Resistant or refractory hypertension: are they different? Curr Hypertens Rep 2014;16:485.
5 Dudenbostel T, Acelajado MC, Pisoni R, Li P, Oparil S, Calhoun D. Refractory Hypertension: Evidence of Heightened Sympathetic Activity as a Cause of Antihypertensive Treatment Failure. Hypertension 2015;66:126-33.-66 Yugar-Toledo JC, Modolo R, de Faria AP, Moreno H. Managing resistant hypertension: focus on mineralocorticoid-receptor antagonists. Vasc Health Risk Manag 2017;13:403-11. Therefore, although hypertension is frequently understood as part of a continuum, a better comprehension about the prevalence of RfH in different populations, as well as its clinical and prognostic differences from RH is essential, especially in the post-mineralocorticoid-antagonist-receptor era77 de Souza F, Muxfeldt ES, Fiszman R, Salles GF. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 2010;55:147-52.
8 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.-99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83..
Discussion
Definition
RH is defined as an office BP that remains above the goal despite the use of 3 or more anti-hypertensive agents of different mechanisms of action at optimal doses, preferentially including a diuretic agent. Patients with controlled office BP on 4 or more drugs are also considered RH.11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90,1010 Braam B, Taler SJ, Rahman M, Fillaus JA, Greco BA, Forman JP, et al. Recognition and Management of Resistant Hypertension. Clin J Am Soc Nephrol 2017;12:524-35.
In parallel to this, the definition of RfH has been evolving since 2012,22 Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12. being currently regarded as the failure of office BP control despite the use of 5 or more anti-hypertensive agents including a long-acting thiazide-like diuretic (ideally chlorthalidone) or a loop diuretics, according to estimated glomerular filtration rate (eGFR), besides a mineralocorticoid receptor antagonist (e.g. spironolactone) as the fourth drug.77 de Souza F, Muxfeldt ES, Fiszman R, Salles GF. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 2010;55:147-52.
Epidemiology
The prevalence of RH, as estimated by multiple multicenter cohorts, lies between 10-20% of all treated individuals.1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.
12 Persell SD. Prevalence of resistant hypertension in the United States, 2003-2008. Hypertension 2011;57:1076-80.
13 Daugherty SL, Powers JD, Magid DJ, Tavel HM, Masoudi FA, Margolis KL, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation 2012;125:1635-42.-1414 Sim JJ, Bhandari SK, Shi J, Liu IL, Calhoun DA, McGlynn EA, et al. Characteristics of resistant hypertension in a large, ethnically diverse hypertension population of an integrated health system. Mayo Clin Proc 2013;88:1099-107. The increased prevalence, despite the improvement of anti-hypertensive regimens in the last 30 years, is explained by the progressive ageing of the population and by the obesity pandemic.1515 Egan BM, Zhao Y, Axon RN, Brzezinski WA, Ferdinand KC. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988 to 2008. Circulation 2011;124:1046-58. Analyses excluding pseudo-resistant hypertension are needed to estimate the true prevalence of RH.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,1616 Sarafidis PA, Georgianos P, Bakris GL. Resistant hypertension--its identification and epidemiology. Nat Rev Nephrol 2012;9:51-8. In Brazil, the Brazilian Longitudinal Study of Adult's Health (ELSA) found an 11% RH prevalence among a cohort of more than 15,000 individuals between 35 and 74 years old.1717 Lotufo PA, Pereira AC, Vasconcellos PS, Santos IS, Mill JG, Bensenor IM. Resistant hypertension: risk factors, subclinical atherosclerosis, and comorbidities among adults-the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). J Clin Hypertens (Greenwich) 2015;17:74-80.
The prevalence of RfH has been estimated by a limited number of studies. Of particular importance, is the prospective analysis conducted by Dudenbostel,1818 Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430. which reported a 5% prevalence of RfH among the RH referred to a specialized hypertension clinic. Additionally, in the REGARDS study,1919 Calhoun DA, Booth JN 3rd, Oparil S, Irvin MR, Shimbo D, Lackland DT, et al. Refractory hypertension: determination of prevalence, risk factors, and comorbidities in a large, population-based cohort. Hypertension 2014;63:451-8. similar rates (3.6%) have been described among patients with controlled or uncontrolled RH, highlighting the low prevalence of RfH (0.5%) among the entire population of hypertensive patients. Recently, the analysis of a Spanish ABPM Registry evidenced a prevalence of 8% of RfH among the RH patients (16.9%).2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365.
Mechanisms
RH is mainly attributed to RAAS hyperactivity and consequently to excessive hydrosaline retention, as evidenced by BP reduction with diuretic therapy that is proportional to effective intravascular volume depletion.2121 Siddiqui M, Calhoun DA. Refractory versus resistant hypertension. Curr Opin Nephrol Hypertens 2017;26:14-9. This mechanism appears to be multifactorial, being associated with increasing age, obesity, chronic kidney disease (CKD) and diabetes, Afro-American ethnicity, excessive sodium intake, and, remarkably, to the magnitude of the hyperaldosteronism status.2222 Hwang AY, Dietrich E, Pepine CJ, Smith SM. Resistant Hypertension: Mechanisms and Treatment. Curr Hypertens Rep 2017;19:56.
In contrast, RfH would be less volume-dependent, since, by definition, its treatment with the association of diuretic drugs fails to achieve the BP goals.44 Modolo R, de Faria AP, Almeida A, Moreno H. Resistant or refractory hypertension: are they different? Curr Hypertens Rep 2014;16:485.,55 Dudenbostel T, Acelajado MC, Pisoni R, Li P, Oparil S, Calhoun D. Refractory Hypertension: Evidence of Heightened Sympathetic Activity as a Cause of Antihypertensive Treatment Failure. Hypertension 2015;66:126-33. Thus, refractory hypertensives seem to be under a greater sympathetic influence, having lower levels of plasmatic aldosterone and a reduced 24-hour sodium excretion. Recent studies comparing patients with refractory to resistant hypertension have shown increased markers of sympathetic activity in the former group: higher heart rate, increased 24-hour norepinephrine excretion, and a higher peripheral resistance.44 Modolo R, de Faria AP, Almeida A, Moreno H. Resistant or refractory hypertension: are they different? Curr Hypertens Rep 2014;16:485.,1818 Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430.,2323 Calhoun DA. Refractory and Resistant Hypertension: Antihypertensive Treatment Failure versus Treatment Resistance. Korean Circ J 2016;46:593-600.
Clinical characteristics and comorbidities
Resistant hypertensives tend to be older, overweight, or obese. Commonly associated comorbidities include CKD, diabetes, obstructive sleep apnea, left ventricle hypertrophy (LVH), cardiovascular and cerebrovascular diseases and, lastly, hyperaldosteronism status.11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90,1010 Braam B, Taler SJ, Rahman M, Fillaus JA, Greco BA, Forman JP, et al. Recognition and Management of Resistant Hypertension. Clin J Am Soc Nephrol 2017;12:524-35.,1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.,2424 Muxfeldt ES, Salles GF. Pulse pressure or dipping pattern: which one is a better cardiovascular risk marker in resistant hypertension? J Hypertens 2008;26:878-84.
Refractory patients, compared to their controlled resistant counterparts, are more likely to be younger, Afro-american, and, predominately, females.2121 Siddiqui M, Calhoun DA. Refractory versus resistant hypertension. Curr Opin Nephrol Hypertens 2017;26:14-9. Regarding associated comorbidities, the most common are heart failure1818 Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430., stroke,22 Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12. CKD with moderately increased albuminuria, diabetes, metabolic syndrome, cardiovascular diseases,1919 Calhoun DA, Booth JN 3rd, Oparil S, Irvin MR, Shimbo D, Lackland DT, et al. Refractory hypertension: determination of prevalence, risk factors, and comorbidities in a large, population-based cohort. Hypertension 2014;63:451-8. and left ventricular hypertrophy.22 Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12.
Clinical approach
When assessing a patient with possible RH, we must consider many important factors to define the diagnostic approach (Table 1).
The first step is to exclude common reasons for pseudo-resistance: inaccurate measurement of BP (special attention should be payed to the adequate size of the cuff for obese patients), poor adherence to both pharmacological and nonpharmacological therapy (i.e. low-sodium diet, physical activity, and weight loss), and an inadequate therapeutic regimen, especially in relation to the use and dosage of the diuretic agents prescribed.11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90,2727 Muxfeldt ES, de Souza F, Salles GF. Resistant hypertension: a practical clinical approach. J Hum Hypertens 2013;27:657-62.,2828 Muxfeldt ES, Salles GF. How to use ambulatory blood pressure monitoring in resistant hypertension. Hypertens Res 2013;36:385-9. Once the pseudo-resistance is excluded, the following steps are recommended:
a) Ambulatory blood pressure monitoring (ABPM)
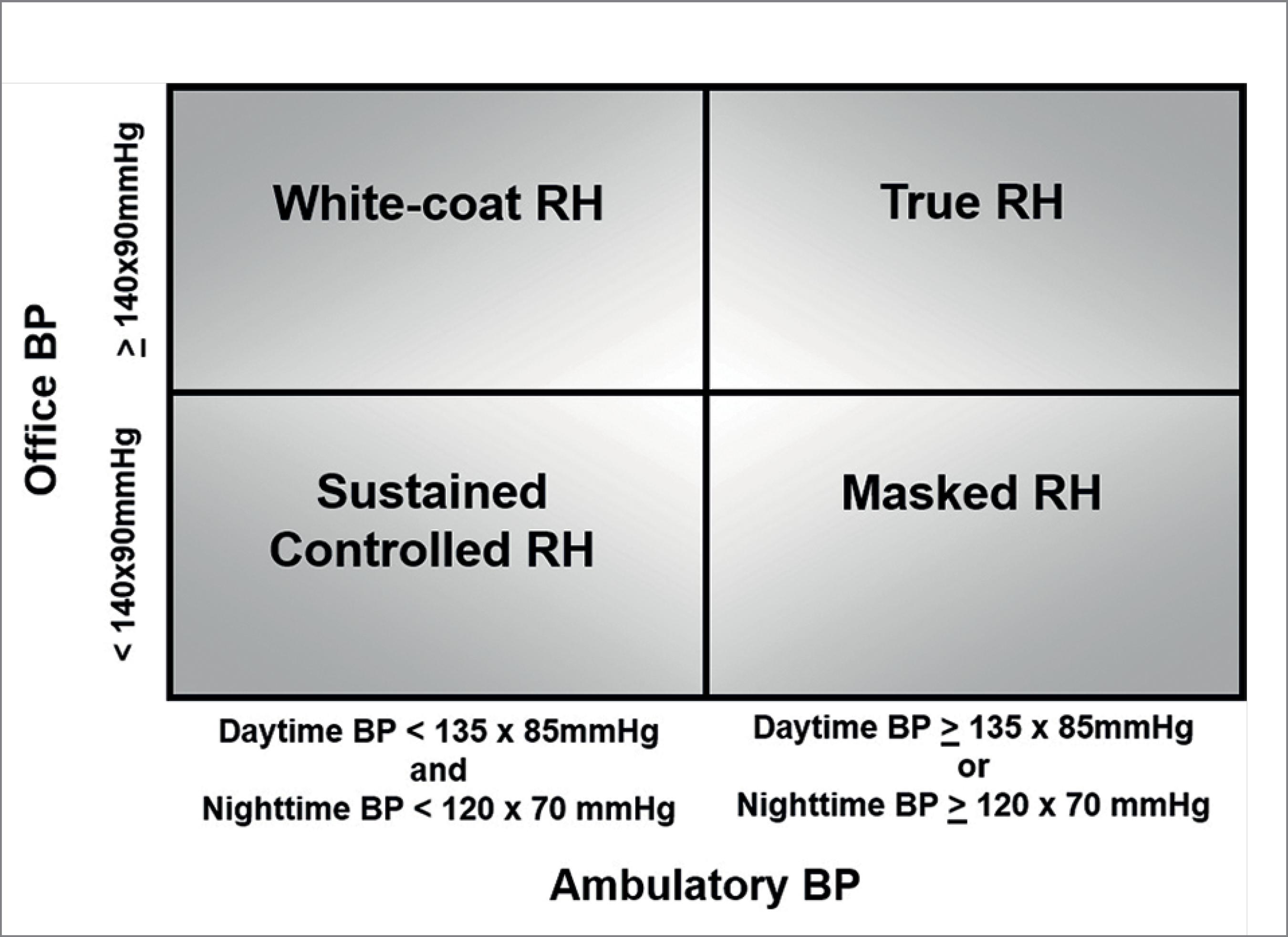
Even though the definitions of both RH and RfH rely on the office BP measurement higher than 140/90 mmHg, the ABPM is a crucial tool in the diagnosis and follow-up of these patients due to the high prevalence (37% in different series) and magnitude of the white-coat effect observed in these patients.1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.,2828 Muxfeldt ES, Salles GF. How to use ambulatory blood pressure monitoring in resistant hypertension. Hypertens Res 2013;36:385-9. (Table 2) Moreover, the ABPM allows patients to be classified into 4 distinct groups (Figure 1) that will determine the subsequent diagnostic evaluation and management: true RH (office BP ≥ 140/90 mmHg and either daytime BP ≥ 135/85 mmHg or night time BP ≥ 120/70 mmHg), white-coat RH (office BP ≥ 140/90 mmHg and either daytime BP < 135/85 mmHg and night time BP < 120/70 mmHg), masked RH (office BP < 140/90 mmHg and either daytime BP ≥ 135/85 mmHg or night time BP ≥ 120/70 mmHg), and controlled RH (office BP < 140/ 90 mmHg and either daytime BP < 135/85 mmHg and night time BP < 120/70 mmHg).1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.,2828 Muxfeldt ES, Salles GF. How to use ambulatory blood pressure monitoring in resistant hypertension. Hypertens Res 2013;36:385-9.,2929 Wei FF, Zhang ZY, Huang QF, Staessen JA. Diagnosis and management of resistant hypertension: state of the art. Nat Rev Nephrol 2018;14:428-41.
Classification of resistant hypertension into four subgroups according to office and ambulatory blood pressure measurements: controlled, masked, white-coat, and true resistant hypertension (authorized reproduction from Muxfeldt et al2828 Muxfeldt ES, Salles GF. How to use ambulatory blood pressure monitoring in resistant hypertension. Hypertens Res 2013;36:385-9.)
On the other hand, among RfH patients, the white-coat phenomenon has not yet been adequately studied. In an analysis of the Spanish ABPM Registry2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365., the prevalence of the white-coat effect was lower among refractory when compared to resistant hypertensives (26.7% versus 37.1%, p < 0.001). In a recent small prospective study with patients with RfH, a prevalence of only 6.5% was found,3030 Siddiqui M, Judd EK, Oparil S, Calhoun DA. White-Coat Effect Is Uncommon in Patients With Refractory Hypertension. Hypertension 2017;70:645-51. suggesting that this phenomenon is much less common among these patients.
In addition, ABPM is essential in the follow-up of these patients at high cardiovascular risk, since it is the only available tool to assess nocturnal blood pressure. In clinical practice, this information allows adjustments to therapeutic regimens based on chronotherapy.3131 Hermida RC, Ayala DE, Mojón A, Fernández JR. Effects of time of antihypertensive treatment on ambulatory blood pressure and clinical characteristics of subjects with resistant hypertension. Am J Hypertens 2010;23:432-9.,3232 Hermida RC, Ayala DE, Fernández JR, Mojón A, Crespo JJ, Ríos MT, et al. Bedtime Blood Pressure Chronotherapy Significantly Improves Hypertension Management. Heart Fail Clin 2017;13:759-73. It is recommended that patients with RH use at least one of their anti-hypertensive drugs at bedtime.3131 Hermida RC, Ayala DE, Mojón A, Fernández JR. Effects of time of antihypertensive treatment on ambulatory blood pressure and clinical characteristics of subjects with resistant hypertension. Am J Hypertens 2010;23:432-9.,3232 Hermida RC, Ayala DE, Fernández JR, Mojón A, Crespo JJ, Ríos MT, et al. Bedtime Blood Pressure Chronotherapy Significantly Improves Hypertension Management. Heart Fail Clin 2017;13:759-73. It has been demonstrated that chronotherapy was capable of reversing the non-dipper pattern in these patients.3333 Hermida RC, Ayala DE, Fernández JR, Calvo C. Chronotherapy improves blood pressure control and reverts the nondipper pattern in patients with resistant hypertension. Hypertension 2008;51:69-76.
It is known that the non-dipper status is the most common pattern among patients with resistant hypertension, affecting up to 65% of these patients.1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902. Furthermore, it is considered an important prognostic marker, especially for coronary artery disease.3434 Muxfeldt ES, Cardoso CR, Salles GF. Prognostic value of nocturnal blood pressure reduction in resistant hypertension. Arch Intern Med 2009;169:874-80. In addition, ambulatory blood pressure during the three periods, but especially at nighttime, are strong predictors of stroke.3535 Salles GF, Cardoso CR, Muxfeldt ES. Prognostic influence of office and ambulatory blood pressures in resistant hypertension. Arch Intern Med 2008;168:2340-6.
The Spanish ABPM registry compared resistant with refractory hypertensives and identified higher ambulatory BP levels in the latter group, with a smaller nocturnal BP reduction. The prevalence of the non-dipper and of the riser patterns was 42.7% and 19.3% among RH patients and of 45.2% and 26.0% among refractory hypertensives, respectively.2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365.
b) Laboratory exams
At first evaluation, it is necessary to assess the metabolic profile and the renal function (serum creatinine, calculation of the eGFR, and albuminuria dosage) (Table 2).
Patients with RfH have a higher prevalence of diabetes mellitus (48.1% versus 33.5%, p < 0.001) and dyslipidemia (61.9% versus 51.7%, p < 0.001) than patients with RH.2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365. The association between CKD and RH is well established, as both a cause and a consequence of therapeutic failure. Besides, moderately increased albuminuria and a reduction in the GFR3636 Salles GF, Cardoso CR, Pereira VS, Fiszman R, Muxfeldt ES. Prognostic significance of a reduced glomerular filtration rate and interaction with microalbuminuria in resistant hypertension: a cohort study. J Hypertens 2011;29:2014-23. identify patients with a high cardiovascular risk3737 Oliveras A, Armario P, Sierra C, Arroyo JA, Hernández-del-Rey R, Vazquez S, et al. Urinary albumin excretion at follow-up predicts cardiovascular outcomes in subjects with resistant hypertension. Am J Hypertens 2013;26:1148-54.,3838 da Costa PM, Cortez AF, de Souza F, Mares GS, dos Santos BDM, Muxfeldt ES. Prognostic impact of baseline urinary albumin excretion rate in patients with resistant hypertension: a prospective cohort study. J Hum Hypertens 2018;32:139-49. and albuminuria reduction may be used as a therapeutic goal in these patients.3939 Salles GF, Cardoso CR, Fiszman R, Muxfeldt ES. Prognostic importance of baseline and serial changes in microalbuminuria in patients with resistant hypertension. Atherosclerosis 2011;216:199-204.
A higher prevalence of a eGFR < 60 mL/min/1.73 m2 (32.1% versus 23.6%, p < 0.001) and of moderately increased albuminuria (38.3% versus 24.5%, p < 0.001) was identified in RfH patients when compared to resistant hypertensives in an analysis of the Spanish ABPM registry.2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365.
c) Electrocardiogram (ECG)
The 12-lead ECG is a useful tool of low-cost and widely available even in primary health units (Table 2). Left ventricular hypertrophy identified on ECG is an important prognostic marker indicating that a subclinical lesion is under development, even in patients who seem to have a well-controlled office BP. These patients may be experiencing masked RH or isolated nocturnal RH.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83. The diagnosis of LVH will guide the choice of the therapeutic regimen. Preferentially, an inhibitor of the RAAS should be chosen, aiming for the regression of the LVH.4040 Salles GF, Cardoso CR, Fiszman R, Muxfeldt ES. Prognostic impact of baseline and serial changes in electrocardiographic left ventricular hypertrophy in resistant hypertension. Am Heart J 2010;159:833-40. The electrocardiographic diagnosis of LVH implies an increase in the cardiovascular risk and its prevention or regression aims to improve the prognosis.4040 Salles GF, Cardoso CR, Fiszman R, Muxfeldt ES. Prognostic impact of baseline and serial changes in electrocardiographic left ventricular hypertrophy in resistant hypertension. Am Heart J 2010;159:833-40.
On the Spanish ABPM registry, electrocardiographically-diagnosed LVH was more prevalent among patients with RfH than in those with RH (27.6% versus 14.9%, p < 0.001).2020 Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365.
d) Causes of secondary hypertension
By definition, in all individuals with suspected RH or RfH, secondary hypertension should be excluded. The most prevalent causes are obstructive sleep apnea, primary aldosteronism, renovascular hypertension, renal parenchymal disease, and pheochromocytoma (Table 3).4141 Pedrosa RP, Drager LF, Gonzaga CC, Sousa MG, de Paula LK, Amaro AC, et al. Obstructive sleep apnea: the most common secondary cause of hypertension associated with resistant hypertension. Hypertension 2011;58:811-7. Other causes as coarctation of the aorta, Cushing's syndrome, hypo or hyperthyroidism, and acromegaly should be investigated only in situations where there are stigmas of the disease.
Therapeutic approach
Nonpharmacological strategies
Obesity, as well as physical inactivity, high sodium intake, smoking, and alcoholism are strongly associated with anti-hypertensive treatment failure, all of them considered important risk factors for RH.11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90,1111 de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.,3434 Muxfeldt ES, Cardoso CR, Salles GF. Prognostic value of nocturnal blood pressure reduction in resistant hypertension. Arch Intern Med 2009;169:874-80.,4242 Brambilla G, Bombelli M, Seravalle G, Cifkova R, Laurent S, Narkiewicz K, et al. Prevalence and clinical characteristics of patients with true resistant hypertension in central and Eastern Europe: data from the BP-CARE study. J Hypertens 2013;31:2018-24. In this way, it is imperative to reinforce the importance of lifestyle changes:88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.
-
Reduction of dietary sodium intake: (below 2 g/day of sodium, corresponding to 5 g/day of salt);
-
DASH diet: use of the Dietary Approaches to Stop Hypertension;4343 Sacks FM, Svetkey LP, Vollmer WM, Appel LJ, Bray GA, Harsha D, et al.; DASH-Sodium Collaborative Research Group. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med 2001;344:3-10.
-
Weight loss: preferentially a BMI < 25 kg/m2;
-
Physical activity: practicing aerobic exercises, dynamic resistance training, and isometric resistance training weekly (at least 30 minutes on 5-7 days per week), after cardiology evaluation;
-
Smokers: quitting smoking, preferentially with assistance;
-
Alcohol: reduce the consumption;
-
Avoidance of drugs that increase blood pressure.
Pharmacological strategies
The initial cornerstone of resistant hypertension treatment is the association of at least three classes of different drugs: i) an appropriate diuretic, preferentially a long-action thiazide diuretic (ex. chlorthalidone) in patients with normal renal function, or loop diuretics should replace thiazides if eGFR is < 30 mL/min/1.73m2 or in other edematous state; ii) a RAAS inhibitor (angiotensin-converting enzyme inhibitors and angiotensin receptor blockers; iii) long-acting dihydropyridines calcium-channel blockers.11 Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90,88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83.,2727 Muxfeldt ES, de Souza F, Salles GF. Resistant hypertension: a practical clinical approach. J Hum Hypertens 2013;27:657-62. Even though hydrochlorothiazide is the most widely prescribed diuretic, chlorthalidone is the diuretic of choice because of its long-acting effect with higher efficacy.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83. For patients with CKD stage 4 or 5 (eGFR lower than 30 mL/min/1.73m2), loop diuretics must be prescribed and administered at least twice a day.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83.
Coronary artery diseases, heart failure, and arrhythmias are special situations when beta-blockers can substitute calcium antagonists at the initial therapeutic scheme with 3 drugs.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83.,2727 Muxfeldt ES, de Souza F, Salles GF. Resistant hypertension: a practical clinical approach. J Hum Hypertens 2013;27:657-62.
RH treatment should be based on diuretic therapy intensification, with special emphasis in the use of spironolactone as a fourth drug, because its association with thiazides provides additive effect in reducing BP.66 Yugar-Toledo JC, Modolo R, de Faria AP, Moreno H. Managing resistant hypertension: focus on mineralocorticoid-receptor antagonists. Vasc Health Risk Manag 2017;13:403-11.,77 de Souza F, Muxfeldt ES, Fiszman R, Salles GF. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 2010;55:147-52.,1818 Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430.,4444 Dudenbostel T, Calhoun DA. Use of Aldosterone Antagonists for Treatment of Uncontrolled Resistant Hypertension. Am J Hypertens 2017;30:103-9. The ASPIRANT Trial4545 Václavík J, Sedlák R, Plachy M, Navrátil K, Plásek J, Jarkovsky J, et al. Addition of spironolactone in patients with resistant arterial hypertension (ASPIRANT): a randomized, double-blind, placebo-controlled trial. Hypertension 2011;57:1069-75.
46 Vaclavik J, Sedlak R, Jarkovsky J, Kocianova E, Taborsky M. Effect of spironolactone in patients with resistant arterial hypertension in relation to age and sex: insights from the aspirant trial. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2014;158:384-90.-4747 Vaclavik J, Sedlak R, Jarkovsky J, Kocianova E, Taborsky M. The effect of spironolactone in patients with resistant arterial hypertension in relation to baseline blood pressure and secondary causes of hypertension. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2013;157:50-5. showed that the addition of spironolactone (25 mg/day) versus placebo lowers systolic BP significantly, especially in older patients. Even in resistant hypertensives with CKD, the spironolactone may be used, except in cases of hyperkalemia.4848 Abolghasmi R, Taziki O. Efficacy of low dose spironolactone in chronic kidney disease with resistant hypertension. Saudi J Kidney Dis Transpl 2011;22:75-8.,4949 Pisoni R, Acelajado MC, Cartmill FR, Dudenbostel T, Dell'Italia LJ, Cofield SS, et al. Long-term effects of aldosterone blockade in resistant hypertension associated with chronic kidney disease. J Hum Hypertens 2012;26:502-6.
Recently, the ReHOT study - a Brazilian multicenter study comparing spironolactone versus clonidine as a fourth-drug therapy in RH - found that both drugs achieved office and ambulatory BP control in similar rates, but spironolactone promoted greater decreased in systolic and diastolic 24-hour BP and diastolic daytime BP, without nighttime BP difference. Nevertheless, spironolactone was considered preferable as the fourth-drug therapy because of its easier posology, less adverse effects, and consequently better long-term adherence.5050 Krieger EM, Drager LF, Giorgi DMA, Pereira AC, Barreto-Filho JAS, Nogueira AR, et al.; ReHOT Investigators. Spironolactone Versus Clonidine as a Fourth-Drug Therapy for Resistant Hypertension: The ReHOT Randomized Study (Resistant Hypertension Optimal Treatment). Hypertension 2018;71:681-90.
If after the four-drug scheme ambulatory BP remains uncontrolled, a fifth-line drug should be added. Possible fifth or sixth drugs are beta-blockers (preferentially the ones with vasodilation effect, as carvedilol, bisoprolol5151 Williams B, MacDonald TM, Morant S, Webb DJ, Sever P, McInnes G et al. British Hypertension Society s PATHWAY Study Group. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet 2015;386:2059-2068 or nebivolol), central alfa1-agonists (clonidine or doxazosin5151 Williams B, MacDonald TM, Morant S, Webb DJ, Sever P, McInnes G et al. British Hypertension Society s PATHWAY Study Group. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet 2015;386:2059-2068), and direct vasodilators (hydralazine or minoxidil). The latter two are capable to lowering BP although they do not reduce cardiovascular morbidity and mortality.88 Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.,99 Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83.
Regarding RfH with failure in controlling BP despite the use of optimized therapeutic scheme with 5 or even 6 drugs, new interventions have emerged, as sympatholytic therapies.1818 Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430.,2424 Muxfeldt ES, Salles GF. Pulse pressure or dipping pattern: which one is a better cardiovascular risk marker in resistant hypertension? J Hypertens 2008;26:878-84. Among these new strategies, we highlight the following:
Baroreflex activation therapy
The Rheos system is a programmable device that consists of a battery-powered implantable generator that works by electrically activating the carotid baroreflex. The Rheos Pivotal Trial did not identify long-term benefits.5252 Bakris GL, Nadim MK, Haller H, Lovett EG, Schafer JE, Bisognano JD. Baroreflex activation therapy provides durable benefit in patients with resistant hypertension: results of long-term follow-up in the Rheos Pivotal Trial. J Am Soc Hypertens 2012;6:152-8.
Renal sympathetic denervation
The renal denervation procedure uses radiofrequency energy to ablate the nerves within the main renal arteries. This therapy was evaluated by three studies called SYMPLICITY.5353 Bakris GL, Townsend RR, Liu M, Cohen SA, D'Agostino R, Flack JM, et al.; SYMPLICITY HTN-3 Investigators. Impact of renal denervation on 24-hour ambulatory blood pressure: results from SYMPLICITY HTN-3. J Am Coll Cardiol 2014;64:1071-8. Different meta-analyses, including a Cochrane's revision, showed that the procedure was safe, but did not significantly decrease BP.5454 Elmula FF, Jin Y, Larstorp AC, Persu A, Kjeldsen SE, Staessen JA. Meta-analysis of five prospective and randomized trials of renal sympathetic denervation on office and ambulatory systolic blood pressure in treatment resistant hypertension. J Hypertens 2015;33:e107.
55 Fadl Elmula F, Jin Y, Yang WY, Thijs L, Lu YC, Larstorp AC, et al.; European Network Coordinating Research On Renal Denervation (ENCOReD) Consortium. Meta-analysis of randomized controlled trials on renal denervation in treatment-resistant hypertension. Blood Press 2015;24:263-74.-5656 Coppolino G, Pisano A, Rivoli L, Bolignano D. Renal denervation for resistant hypertension. Cochrane Database Syst Rev 2017;2:CD011499. The authors advised to await further trials with next-generation catheters, longer follow-up and bigger sample sizes, and especially with standardized procedures.5454 Elmula FF, Jin Y, Larstorp AC, Persu A, Kjeldsen SE, Staessen JA. Meta-analysis of five prospective and randomized trials of renal sympathetic denervation on office and ambulatory systolic blood pressure in treatment resistant hypertension. J Hypertens 2015;33:e107.
Continuous positive airway pressure (CPAP)
Although the benefits in BP control with CPAP use in resistant hypertensives with moderate-severe sleep apnea are not well established with controversy results in different populations,5757 Iftikhar IH, Valentine CW, Bittencourt LR, Cohen DL, Fedson AC, Gíslason T, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a meta-analysis. J Hypertens 2014;32:2341-50. the CPAP should be indicated as an adjuvant treatment, in so far as the adherence is greater than 4 hours per night, improving the quality of life and probably reestablishing the dipper pattern.5858 Martínez-García MA, Capote F, Campos-Rodríguez F, Lloberes P, Díaz de Atauri MJ, Somoza M, et al.; Spanish Sleep Network. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: the HIPARCO randomized clinical trial. JAMA 2013;310:2407-15.
Central Iliac arteriovenous anastomosis
The ROX Medical arteriovenous coupler is a stent-like device that exhibits shape memory to self-expand, forming an AV anastomosis in central iliac. The ROX control HTN demonstrated significant BP decrease, possibly reducing cardiovascular morbidity in those patients.5959 Lobo MD, Ott C, Sobotka PA, Saxena M, Stanton A, Cockcroft JR, et al. Central Iliac Arteriovenous Anastomosis for Uncontrolled Hypertension: One-Year Results From the ROX CONTROL HTN Trial. Hypertension 2017;70:1099-105. Notwithstanding, this is an isolated study and more clinical evidence is necessary.
Table 4 summarizes the main differences between resistant and refractory hypertension observed in various populations.
Conclusion
Despite the common final pathway of hypertension encompassing hydrosaline retention and increased sympathetic tonus, the existence of many phenotypes with distinct clinical paths and prognosis, a broad spectrum ranging from easily controlled hypertension to RH, and more recently, RfH has been suggested.
Even though these two entities are frequently considered a continuum of the same process, it is interesting to observe that they seem to have different pathophysiological mechanisms, suggesting two distinct conditions.
RH patients compared with general hypertensives, are older and more obese. The principal associated comorbidities are established CKD, diabetes, sleep apnea, stroke, and cardiovascular diseases, all of them involving the hyperaldosteronism status. ABPM is mandatory in the diagnosis and follow-up of those patients because of a high magnitude of the white-coat effect.
Moreover, refractory hypertensives compared with controlled RH are younger, predominantly women, and Afro-american. They also have a high prevalence of heart failure, stroke, and CKD with moderately increased albuminuria and LVH. The white-coat effect seems to be less evident in those patients.
In RH, the therapeutic scheme should be based on the intensification of diuretic therapy, emphasizing the spironolactone as the fourth drug associated with a long-acting thiazide, as chlorthalidone. On the other hand, as RfH usually fails all used classes of anti-hypertensives including association of different diuretics, RfH treatment is not well established and new therapies have been proposed such as sympatholytic intervention.
The unfavorable cardiovascular and renal prognosis of RH patients is well established, but future longitudinal studies are necessary to define the morbidity and mortality of RfH.
Resistant and refractory hypertension are challenges in clinical practice and should be addressed as different entities, ideally by specialized professionals capable of identifying comorbidities and to provide diversified and individualized treatment.
References
-
1Carey RM, Calhoun DA, Bakris GLO, Brook RD, Daugherty SL, Dennison-Himmerlfarb CR et al. Resistant hypertension: detection, evaluation and management. A scientific statement from the American Heart Association. Hypertension 2018;72:e53-e90
-
2Acelajado MC, Pisoni R, Dudenbostel T, Dell'Italia LJ, Cartmill F, Zhang B, et al. Refractory hypertension: definition, prevalence, and patient characteristics. J Clin Hypertens 2012;14:7-12.
-
3Leibowitz A, Schiffrin EL. Immune mechanisms in hypertension. Curr Hypertens Rep 2011;13:465-72.
-
4Modolo R, de Faria AP, Almeida A, Moreno H. Resistant or refractory hypertension: are they different? Curr Hypertens Rep 2014;16:485.
-
5Dudenbostel T, Acelajado MC, Pisoni R, Li P, Oparil S, Calhoun D. Refractory Hypertension: Evidence of Heightened Sympathetic Activity as a Cause of Antihypertensive Treatment Failure. Hypertension 2015;66:126-33.
-
6Yugar-Toledo JC, Modolo R, de Faria AP, Moreno H. Managing resistant hypertension: focus on mineralocorticoid-receptor antagonists. Vasc Health Risk Manag 2017;13:403-11.
-
7de Souza F, Muxfeldt ES, Fiszman R, Salles GF. Efficacy of spironolactone therapy in patients with true resistant hypertension. Hypertension 2010;55:147-52.
-
8Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, Burnier M et al. 2018 European Society for Cardiology (ESC)/European Society of Hypertension (ESH) guidelines for the management of arterial hypertension. J Hypertens 2018;36(10):1953-2041.
-
9Malachias MVB, Souza WKSB, Plavnik FL, Rodrigues CIS, Brandão AA, Neves MFT, et al. 7ª Diretriz Brasileira de Hipertensão Arterial. Arq Bras Cardiol 2016;107:1-83.
-
10Braam B, Taler SJ, Rahman M, Fillaus JA, Greco BA, Forman JP, et al. Recognition and Management of Resistant Hypertension. Clin J Am Soc Nephrol 2017;12:524-35.
-
11de la Sierra A, Segura J, Banegas JR, Gorostidi M, de la Cruz JJ, Armario P, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension 2011;57:898-902.
-
12Persell SD. Prevalence of resistant hypertension in the United States, 2003-2008. Hypertension 2011;57:1076-80.
-
13Daugherty SL, Powers JD, Magid DJ, Tavel HM, Masoudi FA, Margolis KL, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation 2012;125:1635-42.
-
14Sim JJ, Bhandari SK, Shi J, Liu IL, Calhoun DA, McGlynn EA, et al. Characteristics of resistant hypertension in a large, ethnically diverse hypertension population of an integrated health system. Mayo Clin Proc 2013;88:1099-107.
-
15Egan BM, Zhao Y, Axon RN, Brzezinski WA, Ferdinand KC. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988 to 2008. Circulation 2011;124:1046-58.
-
16Sarafidis PA, Georgianos P, Bakris GL. Resistant hypertension--its identification and epidemiology. Nat Rev Nephrol 2012;9:51-8.
-
17Lotufo PA, Pereira AC, Vasconcellos PS, Santos IS, Mill JG, Bensenor IM. Resistant hypertension: risk factors, subclinical atherosclerosis, and comorbidities among adults-the Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). J Clin Hypertens (Greenwich) 2015;17:74-80.
-
18Dudenbostel T, Siddiqui M, Gharpure N, Calhoun DA. Refractory versus resistant hypertension: Novel distinctive phenotypes. J Nat Sci 2017;3:pii: e430.
-
19Calhoun DA, Booth JN 3rd, Oparil S, Irvin MR, Shimbo D, Lackland DT, et al. Refractory hypertension: determination of prevalence, risk factors, and comorbidities in a large, population-based cohort. Hypertension 2014;63:451-8.
-
20Armario P, Calhoun DA, Oliveras A, Blanch P, Vinyoles E, Banegas JR, et al. Prevalence and Clinical Characteristics of Refractory Hypertension. J Am Heart Assoc 2017;6:pii: e007365.
-
21Siddiqui M, Calhoun DA. Refractory versus resistant hypertension. Curr Opin Nephrol Hypertens 2017;26:14-9.
-
22Hwang AY, Dietrich E, Pepine CJ, Smith SM. Resistant Hypertension: Mechanisms and Treatment. Curr Hypertens Rep 2017;19:56.
-
23Calhoun DA. Refractory and Resistant Hypertension: Antihypertensive Treatment Failure versus Treatment Resistance. Korean Circ J 2016;46:593-600.
-
24Muxfeldt ES, Salles GF. Pulse pressure or dipping pattern: which one is a better cardiovascular risk marker in resistant hypertension? J Hypertens 2008;26:878-84.
-
25Departamento de Hipertensão Arterial da Sociedade Brasileira de Cardiologia. I Posicionamento Brasileiro sobre hipertensão arterial resistente. Arq Bras Cardiol 2012;99:576-85.
-
26de Souza F, Muxfeldt ES, Salles GF. Prognostic factors in resistant hypertension: implications for cardiovascular risk stratification and therapeutic management. Expert Rev Cardiovasc Ther 2012;10:735-45.
-
27Muxfeldt ES, de Souza F, Salles GF. Resistant hypertension: a practical clinical approach. J Hum Hypertens 2013;27:657-62.
-
28Muxfeldt ES, Salles GF. How to use ambulatory blood pressure monitoring in resistant hypertension. Hypertens Res 2013;36:385-9.
-
29Wei FF, Zhang ZY, Huang QF, Staessen JA. Diagnosis and management of resistant hypertension: state of the art. Nat Rev Nephrol 2018;14:428-41.
-
30Siddiqui M, Judd EK, Oparil S, Calhoun DA. White-Coat Effect Is Uncommon in Patients With Refractory Hypertension. Hypertension 2017;70:645-51.
-
31Hermida RC, Ayala DE, Mojón A, Fernández JR. Effects of time of antihypertensive treatment on ambulatory blood pressure and clinical characteristics of subjects with resistant hypertension. Am J Hypertens 2010;23:432-9.
-
32Hermida RC, Ayala DE, Fernández JR, Mojón A, Crespo JJ, Ríos MT, et al. Bedtime Blood Pressure Chronotherapy Significantly Improves Hypertension Management. Heart Fail Clin 2017;13:759-73.
-
33Hermida RC, Ayala DE, Fernández JR, Calvo C. Chronotherapy improves blood pressure control and reverts the nondipper pattern in patients with resistant hypertension. Hypertension 2008;51:69-76.
-
34Muxfeldt ES, Cardoso CR, Salles GF. Prognostic value of nocturnal blood pressure reduction in resistant hypertension. Arch Intern Med 2009;169:874-80.
-
35Salles GF, Cardoso CR, Muxfeldt ES. Prognostic influence of office and ambulatory blood pressures in resistant hypertension. Arch Intern Med 2008;168:2340-6.
-
36Salles GF, Cardoso CR, Pereira VS, Fiszman R, Muxfeldt ES. Prognostic significance of a reduced glomerular filtration rate and interaction with microalbuminuria in resistant hypertension: a cohort study. J Hypertens 2011;29:2014-23.
-
37Oliveras A, Armario P, Sierra C, Arroyo JA, Hernández-del-Rey R, Vazquez S, et al. Urinary albumin excretion at follow-up predicts cardiovascular outcomes in subjects with resistant hypertension. Am J Hypertens 2013;26:1148-54.
-
38da Costa PM, Cortez AF, de Souza F, Mares GS, dos Santos BDM, Muxfeldt ES. Prognostic impact of baseline urinary albumin excretion rate in patients with resistant hypertension: a prospective cohort study. J Hum Hypertens 2018;32:139-49.
-
39Salles GF, Cardoso CR, Fiszman R, Muxfeldt ES. Prognostic importance of baseline and serial changes in microalbuminuria in patients with resistant hypertension. Atherosclerosis 2011;216:199-204.
-
40Salles GF, Cardoso CR, Fiszman R, Muxfeldt ES. Prognostic impact of baseline and serial changes in electrocardiographic left ventricular hypertrophy in resistant hypertension. Am Heart J 2010;159:833-40.
-
41Pedrosa RP, Drager LF, Gonzaga CC, Sousa MG, de Paula LK, Amaro AC, et al. Obstructive sleep apnea: the most common secondary cause of hypertension associated with resistant hypertension. Hypertension 2011;58:811-7.
-
42Brambilla G, Bombelli M, Seravalle G, Cifkova R, Laurent S, Narkiewicz K, et al. Prevalence and clinical characteristics of patients with true resistant hypertension in central and Eastern Europe: data from the BP-CARE study. J Hypertens 2013;31:2018-24.
-
43Sacks FM, Svetkey LP, Vollmer WM, Appel LJ, Bray GA, Harsha D, et al.; DASH-Sodium Collaborative Research Group. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med 2001;344:3-10.
-
44Dudenbostel T, Calhoun DA. Use of Aldosterone Antagonists for Treatment of Uncontrolled Resistant Hypertension. Am J Hypertens 2017;30:103-9.
-
45Václavík J, Sedlák R, Plachy M, Navrátil K, Plásek J, Jarkovsky J, et al. Addition of spironolactone in patients with resistant arterial hypertension (ASPIRANT): a randomized, double-blind, placebo-controlled trial. Hypertension 2011;57:1069-75.
-
46Vaclavik J, Sedlak R, Jarkovsky J, Kocianova E, Taborsky M. Effect of spironolactone in patients with resistant arterial hypertension in relation to age and sex: insights from the aspirant trial. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2014;158:384-90.
-
47Vaclavik J, Sedlak R, Jarkovsky J, Kocianova E, Taborsky M. The effect of spironolactone in patients with resistant arterial hypertension in relation to baseline blood pressure and secondary causes of hypertension. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2013;157:50-5.
-
48Abolghasmi R, Taziki O. Efficacy of low dose spironolactone in chronic kidney disease with resistant hypertension. Saudi J Kidney Dis Transpl 2011;22:75-8.
-
49Pisoni R, Acelajado MC, Cartmill FR, Dudenbostel T, Dell'Italia LJ, Cofield SS, et al. Long-term effects of aldosterone blockade in resistant hypertension associated with chronic kidney disease. J Hum Hypertens 2012;26:502-6.
-
50Krieger EM, Drager LF, Giorgi DMA, Pereira AC, Barreto-Filho JAS, Nogueira AR, et al.; ReHOT Investigators. Spironolactone Versus Clonidine as a Fourth-Drug Therapy for Resistant Hypertension: The ReHOT Randomized Study (Resistant Hypertension Optimal Treatment). Hypertension 2018;71:681-90.
-
51Williams B, MacDonald TM, Morant S, Webb DJ, Sever P, McInnes G et al. British Hypertension Society s PATHWAY Study Group. Spironolactone versus placebo, bisoprolol, and doxazosin to determine the optimal treatment for drug-resistant hypertension (PATHWAY-2): a randomised, double-blind, crossover trial. Lancet 2015;386:2059-2068
-
52Bakris GL, Nadim MK, Haller H, Lovett EG, Schafer JE, Bisognano JD. Baroreflex activation therapy provides durable benefit in patients with resistant hypertension: results of long-term follow-up in the Rheos Pivotal Trial. J Am Soc Hypertens 2012;6:152-8.
-
53Bakris GL, Townsend RR, Liu M, Cohen SA, D'Agostino R, Flack JM, et al.; SYMPLICITY HTN-3 Investigators. Impact of renal denervation on 24-hour ambulatory blood pressure: results from SYMPLICITY HTN-3. J Am Coll Cardiol 2014;64:1071-8.
-
54Elmula FF, Jin Y, Larstorp AC, Persu A, Kjeldsen SE, Staessen JA. Meta-analysis of five prospective and randomized trials of renal sympathetic denervation on office and ambulatory systolic blood pressure in treatment resistant hypertension. J Hypertens 2015;33:e107.
-
55Fadl Elmula F, Jin Y, Yang WY, Thijs L, Lu YC, Larstorp AC, et al.; European Network Coordinating Research On Renal Denervation (ENCOReD) Consortium. Meta-analysis of randomized controlled trials on renal denervation in treatment-resistant hypertension. Blood Press 2015;24:263-74.
-
56Coppolino G, Pisano A, Rivoli L, Bolignano D. Renal denervation for resistant hypertension. Cochrane Database Syst Rev 2017;2:CD011499.
-
57Iftikhar IH, Valentine CW, Bittencourt LR, Cohen DL, Fedson AC, Gíslason T, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a meta-analysis. J Hypertens 2014;32:2341-50.
-
58Martínez-García MA, Capote F, Campos-Rodríguez F, Lloberes P, Díaz de Atauri MJ, Somoza M, et al.; Spanish Sleep Network. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: the HIPARCO randomized clinical trial. JAMA 2013;310:2407-15.
-
59Lobo MD, Ott C, Sobotka PA, Saxena M, Stanton A, Cockcroft JR, et al. Central Iliac Arteriovenous Anastomosis for Uncontrolled Hypertension: One-Year Results From the ROX CONTROL HTN Trial. Hypertension 2017;70:1099-105.
Publication Dates
-
Publication in this collection
06 Dec 2018 -
Date of issue
Apr-Jun 2019
History
-
Received
20 May 2018 -
Accepted
05 Sept 2018