Abstracts
PURPOSE: To investigate whether N-acetylcysteine, a free radicals scavenger has a protective effect against lung injury as a remote organ after skeletal muscle ischemia-reperfusion. METHODS: Twenty Wistar male rats were divided randomly into two experimental groups: group ischemia-reperfusion (group I) and group ischemia-reperfusion + N-acetylcysteine (group II). All animals were undergone two hours of ischemia by occlusion femoral artery and 24h of reperfusion. Before clamped the femoral artery, 250 IU heparin was administered via the jugular vein to prevent clotting. Rats that were treated with N-acetylcysteine given IV at a dose of 150 mgkg-¹, immediately before reperfusion. After 24h of reperfusion, animals were euthanized and left lung harvested for histopathological analysis under light microscopy. RESULTS: In the group I, tissues showed histological changes with intra-alveolar edema, intra-alveolar hemorrhage and neutrophilic infiltration. Histopathologically, there was a significant difference (P = 0.005) between two groups. CONCLUSION: Administration of N-acetylcysteine treatment significantly decreased lung injury induced by skeletal muscle ischemia reperfusion according to histological findings.
Acetylcysteine; Muscle, Skeletal; Lung; Ischemia; Reperfusion; Reperfusion Injury; Histology; Rats
OBJETIVO: Investigar se N-acetilcisteína, neutralizador de radicais livres, tem efeito protetor contra dano pulmonar como um órgão remoto após isquemia-reperfusão de músculo esquelético. MÉTODOS: Vinte ratos machos Wistar, foram aleatóriamente distribuídos em dois grupos: grupo isquemia-reperfusão (grupo I) e grupo isquemia-reperfusão +N-acetilcisteína (grupo II). Todos os animais foram submetidos a duas horas de ischemia pela oclusão artéria femoral e 24 horas de reperfusão. Antes de ocluir a artéria femoral, foi administrado 250 IU de heparina pela veia jugular para prevenir coagulação. A N-acetilcisteína foi administrada por via intravenosa, na uma dose de 150 mgkg-1, imediatamente antes de reperfusão. Após 24 horas de reperfusão, os animais foram eutanasiados e o pulmão esquerdo foi removido para análise histológica em microscopia óptica. RESULTADOS: No grupo I, os tecidos mostraram alterações histológicas com edema e hemorragia intra-alveolar e infiltração neutrofílica. Houve diferença histopatológica significante (P = 0.005) entre os dois grupos. CONCLUSÃO: O tratamento com a N-acetilcisteína diminuiu significantemente o dano pulmonar induzido por isquemia-reperfusão de músculo esquelético.
Acetilcisteína; Músculo Esquelético; Pulmão; Isquemia; Reperfusão; Traumatismo por Reperfusão; Histologia; Ratos
12- ORIGINAL ARTICLE
ISCHEMIA-REPERFUSION
Effect of N-acetylcysteine on lung injury induced by skeletal muscle ischemia-reperfusion. Histopathological study in rat model1 Correspondence: Mohammad Ashrafzadeh Department of Veterinary Surgery Faculty of Specialized Veterinary Sciences Sciences and Research Branch Islamic Azad University Hesarak, Tehran, Iran Phone: 1477893855 Dr_ashrafzadeh@yahoo.com
Efeito de N-acetilcisteína em dano pulmonar induzido por isquemia-reperfusão de músculo esquelético. Estudo histopatólogico em modelo de rato
Amir SotoudehI, Mohammad Ashrafzadeh TakhtfooladiII, Amirali JahanshahiIII, Adel Haghighi Khiabanian AslIV, Hamed Ashrafzadeh TakhtfooladiV, Mohammadreza KhansariVI
IAssistant Professor, Faculty of Veterinary Science, Kahnooj Branch, Islamic Azad University, Kerman, Iran. Design, supervised all phases of the study, analysis and interpretation of data, manuscript writing
IIFellow PhD degree, Department of Surgery, Faculty of Specialized Veterinary Sciences, Science and Research Branch, Islamic Azad University, Tehran, Iran. Conception, design and scientific content of study, analysis and interpretation of data, manuscript writing.
IIIFellow PhD degree, Department of Surgery, Faculty of Specialized Veterinary Sciences, Science and Research Branch, Islamic Azad University, Tehran, Iran. Helped with technical procedures, collection and processing of study informations, analysis and interpretation of data
IVAssistant Professor, Department of Pathology, Faculty of Specialized Veterinary Sciences, Science and Research Branch, Islamic Azad University, Tehran, Iran. Histological analysis
VGraduate student, Faculty of Veterinary Sciences, Karaj Branch, Islamic Azad University, Alborz, Iran. Helped with technical procedures and collection of study informations
VIMaster, Department of Physiology, Faculty of Specialized Veterinary Sciences, Science and Research Branch, Islamic Azad University, Tehran, Iran. Statistical analysis
Corresponde Correspondence: Mohammad Ashrafzadeh Department of Veterinary Surgery Faculty of Specialized Veterinary Sciences Sciences and Research Branch Islamic Azad University Hesarak, Tehran, Iran Phone: 1477893855 Dr_ashrafzadeh@yahoo.com
ABSTRACT
PURPOSE: To investigate whether N-acetylcysteine, a free radicals scavenger has a protective effect against lung injury as a remote organ after skeletal muscle ischemia-reperfusion.
METHODS: Twenty Wistar male rats were divided randomly into two experimental groups: group ischemia-reperfusion (group I) and group ischemia-reperfusion + N-acetylcysteine (group II). All animals were undergone two hours of ischemia by occlusion femoral artery and 24h of reperfusion. Before clamped the femoral artery, 250 IU heparin was administered via the jugular vein to prevent clotting. Rats that were treated with N-acetylcysteine given IV at a dose of 150 mgkg-1, immediately before reperfusion. After 24h of reperfusion, animals were euthanized and left lung harvested for histopathological analysis under light microscopy.
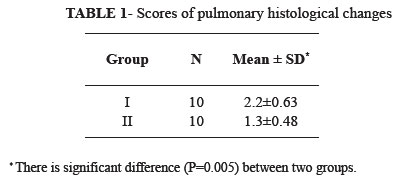
RESULTS: In the group I, tissues showed histological changes with intra-alveolar edema, intra-alveolar hemorrhage and neutrophilic infiltration. Histopathologically, there was a significant difference (P = 0.005) between two groups.
CONCLUSION: Administration of N-acetylcysteine treatment significantly decreased lung injury induced by skeletal muscle ischemia reperfusion according to histological findings.
Key words: Acetylcysteine. Muscle, Skeletal. Lung. Ischemia. Reperfusion. Reperfusion Injury. Histology. Rats.
RESUMO
OBJETIVO: Investigar se N-acetilcisteína, neutralizador de radicais livres, tem efeito protetor contra dano pulmonar como um órgão remoto após isquemia-reperfusão de músculo esquelético.
MÉTODOS: Vinte ratos machos Wistar, foram aleatóriamente distribuídos em dois grupos: grupo isquemia-reperfusão (grupo I) e grupo isquemia-reperfusão +N-acetilcisteína (grupo II). Todos os animais foram submetidos a duas horas de ischemia pela oclusão artéria femoral e 24 horas de reperfusão. Antes de ocluir a artéria femoral, foi administrado 250 IU de heparina pela veia jugular para prevenir coagulação. A N-acetilcisteína foi administrada por via intravenosa, na uma dose de 150 mgkg-1, imediatamente antes de reperfusão. Após 24 horas de reperfusão, os animais foram eutanasiados e o pulmão esquerdo foi removido para análise histológica em microscopia óptica.
RESULTADOS: No grupo I, os tecidos mostraram alterações histológicas com edema e hemorragia intra-alveolar e infiltração neutrofílica. Houve diferença histopatológica significante (P = 0.005) entre os dois grupos.
CONCLUSÃO: O tratamento com a N-acetilcisteína diminuiu significantemente o dano pulmonar induzido por isquemia-reperfusão de músculo esquelético.
Palavras-chave: Acetilcisteína. Músculo Esquelético. Pulmão. Isquemia. Reperfusão. Traumatismo por Reperfusão. Histologia. Ratos.
Introduction
Many surgical procedures, such as limb revascularization and free-flap reconstruction, involve prolonged ischemia of skeletal muscle. Ischemic damage can lead to severe postoperative complications1. There are two components to the reperfusion syndrome, which follows extremity ischemia. The local response follows reperfusion and the systemic response, which results in multiple organ failure and death2.
Ischemia reperfusion of the lower extremities causes lung injury because of polymorphonuclear sequestration in pulmonary microvasculature, increased endothelial permeability and interstitial edema3. Ischemic damage results from a decrease in the blood flow of the organ and reperfusion injury results from the enhanced generation of oxygen radicals4.
Several agents, such as caffeic acid phenethyl ester or Erdosteine are proposed to be useful against lung injury induced by oxidative stress damage, including ischemia-reperfusion or endotoxin instillation5,6.
N-acetylcysteine is not simply an antioxidant drug. It acts as a glutathione precursor, as a chemical reductant of oxidized thiols, as a scavenger of radical oxygen species, as a vasodilator and also improves microcirculation by restoring the decreased activity of endothelium-derived relaxing factor and may have antiaggregan features7,8. There is growing evidence regarding its beneficial effects in ameliorating lung ischemia-reperfusion injury9. However its role in reducing the damage in lung after skeletal muscle ischemia-reperfusion has not been addressed yet.
In this experimental study, we aimed to examine the protective effect of N-acetylcysteine on lung injury induced by the skeletal muscle ischemia-reperfusion by evaluation of histopathology in rat model.
Methods
Research performed in accordance with the Islamic Azad University Law on animal experimentation.
Twenty Wistar male rats weighing 220250 g (5-7 weeks old) were used in this study. Animals were housed at an average ambient temperature of 22ºC under a 12h/12h light/dark cycle before the experiment and were fed on a standard rodent diet and filtered tap water ad libitum. Animals were divided randomly into two experimental groups of ten rats each: group ischemia-reperfusion (group I) and group ischemia-reperfusion + N-acetylcysteine (group II). Anesthesia was induced using intramuscular ketamine (50 mgkg-1) (ketamine Hcl 10% AlfasoonWoerden-Holland) plus xylazine (10 mgkg-1) (2% AlfasoonWoerden-Holland). After induction of anesthesia, the left hind limb was completely clipped with an electric shaver. After clipping, disinfecting and dropping (using a sterile technique), a skin incision was made on medial surface of the left hind limb. After isolated the femoral artery and vein from the surrounding structures, femoral artery was exposed and clamped with a mini bulldog forceps.
Before clamped the femoral artery, 250 IU heparin (Heparodic® 5000 Caspian tamin Rasht-Iran) was administered via the jugular vein to prevent clotting. All animals were undergone 2h of ischemia by occlusion femoral artery with a vascular clamp and 24h of reperfusion10. Rats were maintained in a dorsal recumbency and kept anesthetized throughout the duration of the ischemic period. Additional doses were given as necessary to maintain anesthesia during the experiment. Body temperature was maintained with a heating pad under anesthesia. In group I N-acetylcysteine (150 mgkg-l) was injected intravenous immediately before reperfusion. Following the ischemic period, the vascular forceps was removed and then surgical site was routinely closed. During the reperfusion period, rats were returned to their cages with food and water ad libitum. After 24h of reperfusion, rats were euthanized with an overdose of intraperitoneal pentobarbital injection (300 mgkg-l) and left lung harvested rapidly for histopathological analysis under light microscopy. Lung tissues were placed in 10% formalin solution and processed routinely by embedding in paraffin then tissues were sectioned in 6 µm pieces and stained with Hematoxylin-Eosin stain. An experienced pathologist who was unaware of the treatment conditions made histopathological assessments. Pulmonary injury was graded into four grades as follows: grade 0, no diagnostic change: grade 1, mild neutrophil leukocyte infiltration and mild to moderate interstitial congestion; grade 2, moderate neutrophil leukocyte infiltration, perivascular edema formation, and partial destruction of pulmonary architecture; and grade 3, included dense neutrophil leukocyte infiltration and complete destruction of pulmonary structure11. A total of four slides from each lung sample were randomly screened and the mean was accepted as the representative value of the sample. Statistical analyses were carried out using SPSS statistical software (version 18). Results were expressed as the mean +/- standard deviation. The Mann-Whitney U-test was employed to analyze two groups consecutively. Values of P<0.05 were considered as statistically significant.
Results
All of rats tolerated operation and survived until the final study period. Figure 1 illustrates representative photomicrograph of the lung tissues from group I and Figure 2 illustrates representative photomicrograph of the lung tissues from group II that obtained 24h after reperfusion. In the group I, tissues showed histological changes with intra-alveolar edema, intra-alveolar hemorrhage and neutrophilic infiltration. These pathological changes, particularly neutrophilic infiltration, were much less in the group II than in the group I. Histopathologically, there was a significant difference (P=0.005) between two groups (Table 1).
Discussion
In the lung the end-result of systemic or remote ischemia-reperfusion injury is non-cardiogenic pulmonary oedema, or adult respiratory distress syndrome2. There is evidence to suggest that neutrophils are activated directly by metabolites (e.g. leukotriene B4, thromboxane A2, complement C5), that are produced by the ischaemic tissue and then go on to be trapped in the pulmonary circulation12. There is also evidence to suggest that neutrophils are activated locally in the lung by the pulmonary endothelial production of platelet activating factor, thromboxane A2, leukotriene B4, and oxygen free radicals, which are neutrophil chemo-attractants13. The local release of thromboxane A2 also causes pulmonary vasoconstriction, further increasing the chance of neutrophil-endothelial interaction and the no-reflow phenomenon. Interleukin-8 which is produced by alveolar macrophages and pulmonary endothelial cells and released into the systemic circulation during ischemia-reperfusion is also thought to play a role in activating neutrophils within the pulmonary pool, and the rest of the circulation14.
N-acetylcysteine exerts its effect both as a source of sulfhydryl groups (repletion of intracellular reduced glutatione) and through a direct reaction with hydroxyl radical15. Recently, a number of studies in animals suggest benefits from acetylcysteine in the context of systemic inflammatory response syndrome caused by severe sepsis model. In a pig gram-negative sepsis model, an infusion of acetylcysteine reduced pulmonary capillary leak without reducing mortality16. Acetylcysteine also beneficially modulates inflammatory cell function in animals16. Endothoxin-induced neutrophil activation in sheep lung is reduced by Acetylcysteine16. Additional protective effects of N-acetylcysteine may lie within the ability of this compound to reduce oxyradical-related oxidant processes by either directly interfering with the oxidants, or up-regulating antioxidant systems such as superoxide dismutase15 or enhancing the catalytic activity of glutathione peroxidase17.Nacetylcysteine has antioxidant property18 and as a sulfhydryl donor, may contribute to the regeneration of endothelium-derived relaxing factor and glutathione12. Increasing evidence indicates that the action of N-acetylcysteineis pertinent to microcirculatory blood flow and tissue oxygenation. N-acetylcysteinewas shown to enhance oxygen consumption via increased oxygen extraction in patients 18h after the onset of fulminant liver failure12.
In an experimental study Boerjesson et al.19 showed that treatment with N-acetylcysteine prevents intestinal ischemia- reperfusion induced over activation of pulmonary macrophages and decreases pulmonary content. This observation was supported by Weinbraum et al.9, which demonstrated, pretreatment of the lungs with N-acetylcysteineduring reperfusion period with ischemic reperfused liver effluent prevents acute lung injury. In a randomized clinical trial on patients with acute respiratory distress syndrome, N-acetylcysteine enhanced recovery from acute lung injury20. There are also several studies concerning beneficial role of N-acetylcysteine in ameliorating myocardial and brain injury21,22.
In the current study, we tested the hypothesis that N-acetylcysteine could protect the lungs from remote organ injury after skeletal muscle ischemia-reperfusion. Our data demonstrate that N-acetylcysteine significantly decreases the severity of acute lung injury, decreases the infiltration of macrophages and polymorphonuclear leukocytes in the lungs, decreases pulmonary vascular permeability and inhibits cellular apoptosis in the lungs after skeletal muscle ischemia-reperfusion injury.
Conclusions
The present study demonstrates that temporary occlusion of the femoral artery of the rats resulted in altered pulmonary endothelial barrier function, with increased protein concentration and neutrophil infiltration. However, administration of the N-acetylcysteine treatment significantly decreased lung injury induced by skeletal muscle ischemia-reperfusion according to our histological findings. These results suggest the possibility of clinical application of N-acetylcysteine in ischemia-reperfusion injury of the lung. Different dosages, alternate time protocols and way of N-acetylcysteine administration for lung injury induced by skeletal muscle ischemia-reperfusion should be investigated in future studies.
Received: September 20, 2011
Review: November 22, 2011
Accepted: December 19, 2011
Conflict of interest: none
Financial source: none
1 Research performed at Department of Veterinary Surgery, Faculty of Specialized Veterinary Sciences, Islamic Azad University, Tehran, Iran.
- 1. Beyersdorf F, Unger A, Wildhirt A. Studies of reperfusion injury in skeletal muscle: preserved cellular viability after extended periods of warm ischemia. J Cardiovasc Surg. 1991;32:664-76.
- 2. Blaisdell FW. The pathophysiology of skeletal muscle ischemia and the reperfusion syndrome: a review. Cardiovasc Surg. 2002;10: 62030.
- 3. Welbourn CR, Goldman G, Paterson IS, Valeri CR, Shepro D, Hechtman HB. Pathophysiology of ischaemia reperfusion injury: central role of the neutrophil. Br J Surg. 1991;78:651-5.
- 4. Schoenberg MH, Beger HG. Reperfusion injury after intestinal ischemia. Crit Care Med. 1993;21:137689.
- 5. Calikoglu M, Tamer L, Sucu N. The effects of caffeic acid phenethyl ester on tissue damage in lung after hindlimb ischemia-reperfusion. Pharmacol Res. 2003;48:397-403.
- 6. Mehmet S, Efkan UZ, Rana S. The effects of erdosteine on lung injury induced by the ischemia- reperfusion of the hind-limbs in rats. J Surg Res. 2008;145:3037.
- 7. Tredger JM. N-acetylcysteine: not a simply glutathione precursor. Transplantation. 2000;69:703-4.
- 8. Arstall MA, Yang J, Stafford I, Betts WH, Horowitz JD. N-acetylcysteine in combination with nitroglycerin and streptokinase for the treatment of evolving acute myocardial infarction. Safety and biochemical effects. Circulation. 1995;92:2855-62.
- 9. Weinbroum AA, Rudick V, Ben-Abraham R, Kachevski E. N-acetyl-L-cysteine for preventing lung reperfusion injury after liver ischemia-reperfusion: a possible dual protective mechanism in a dose-response study. Transplantation. 2000;69(5):853-9.
- 10-Eberlin KR, McCormack MC, Nguyen JT, Tatlidede HS, Randolph MA, Austen Jr W. Ischemic preconditioning of skeletal muscle mitigates remote injury and mortality. J Surg Res. 2008;148:24-30.
- 11. Koksel O, Yildirim C, Cinel L, Tamer L, Ozdulger A, Bastürk M, Degirmenci U, Kanik A, Cinel I. Inhibition of poly (ADPribose) polymerase attenuates lung tissue damage after hind limb ischemiareperfusion in rats. Pharmacol Res. 2005;51(5):453-62.
- 12. Harrison PM, Wendon YA, Gimson AES. Improvement by acetylcisteine of hemodynamics and oxygen transport in fulminant hepatic failure. N Engl J Med. 1991;324:18527.
- 13. Klausner JM, Paterson IS, Kobzik L. Oxygen free radical mediate ischemia-induced lung injury. Surgery. 1989;105:192-9.
- 14. Baggiolini M, Walz A, Kunkel SL. Neutrophil activating peptide-1/ interleukin-8, a novel cytokine that activates neutrophils. J Clin Invest. 1989;84:1045-9.
- 15. Galley HF, Howdle PD, Walker BE. The effect of intravenous antioxidants in patients with septic shock. Free Radic Biol Med. 1997;23:76874.
- 16. Lucht WD, English DK, Bernard GR. Prevention of release of granulocyte aggregates into sheep lung lymph following endotoxemia by N-acetylcysteine. Am J Med Sci. 1987;294:1617.
- 17. Schillier HJ, Reilly PM, Bulkley GB. Antioxidant therapy. Crit Care Med. 1993;21:92102.
- 18. Aruoma OI, Halliwell B, Hoey BM. The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid. Free Radic Biol Med. 1989;6:5937.
- 19. Borjesson A, Wang X, Sun Z, Wallen R, Deng X, Johnson E. Effects of N-acetylcysteine on pulmonary macrophage activity after intestinal ischemia and reperfusion in rats / with invited commentaries. Dig Surg. 2000;17(4):379-87.
- 20. Suter PM, Domenighetti G, Schaller M, Laverriere M, Ritz R, Perret C. N-acetylcysteine enhances recovery from acute lung injury in man. A randomized, double-blind, placebo-controlled clinical study. Chest. 1994;105(1):190-4.
- 21. Cuzzocrea S, Mazzon E, Costantino G, Serraino I, Dugo L, Calabrò G, Cucinotta G, De Sarro A, Caputi AP. Beneficial effects of n-acetylcysteine on ischemic brain injury. Br J Pharmacol. 2000;130(6):1219-26.
- 22. Menasché P, Grousset C, Gauduel Y, Mouas C, Piwnica A. Maintenance of the myocardial thiol pool by N-acetylcysteine. An effective means of improving cardioplegic protection. J Thorac Cardiovasc Surg. 1992;103(5):936-44.
Publication Dates
-
Publication in this collection
05 Mar 2012 -
Date of issue
Feb 2012
History
-
Received
20 Sept 2011 -
Accepted
19 Dec 2011 -
Reviewed
22 Nov 2011