Abstract
PURPOSE:
To investigate the development of a laparoscopy technique for local injection into the X-linked muscular dystrophy (mdx) diaphragm.
METHODS:
It was used 10 mice Balb/C57 and 5 mdx mice and three differents decubitus type were tested: the right lateral, supine, and supine decubitus with 20 degrees elevation of the forelimb. Abdominal caudal face and the 10 intercostal space were tested as spot to introduce the needle into the diaphragm.
RESULTS:
Supine position with elevation of 20 degrees forelimb and the 10th intercostal space are the beneficial position to apply a local injection.
CONCLUSION:
It was proved to be possible to perform the laparoscopy technique in the X-linked muscular dystrophy diaphragm and this requires a specific position and technique during the surgery.
Diaphragm; Injections; Muscular Dystrophy; Duchenne; Laparoscopy; Mice
Introduction
Laparoscopic techniques are a minimally procedures, aiming to reduce the risks associated with conventional surgery, that proved superior to postoperative pain and recovery11. Liem MSL. Comparison of conventional anterior surgery and laparoscopic surgery for inguinal-hernia repair. N Engl J Med. 1997 May;336(22):1541-7. doi: 10.1056/NEJM199705293362201.
https://doi.org/10.1056/NEJM199705293362...
.
Duchenne muscular dystrophy (DMD) is the most devastating X-linked muscle disorders that affect 1 in 3500 male's births, caused by mutation in gene encoding the 427 kDA protein, called dystrophin22. Spencer MJ, Tidball JG. Do immune cells promote the pathology of dystrophin-deficient myopathies? Neuromuscul Disord. 2001 Sep;11(6-7):556-64. PMID: 11525885. , 33. Nakamura A, Takeda S. Mammalian models of Duchenne Muscular Dystrophy: pathological characteristics and therapeutic applications. J Biomed Biotechnol. 2011;2011:184393. doi: 10.1155/2011/184393.
https://doi.org/10.1155/2011/184393...
. The major respiratory symptoms observed in these patients, occurs, when they used the wheelchair. During this period, these patients are affected by scoliosis structural deformities, mainly responsible for the lung function decrease. Due the respiratory muscles weakness and structural deformities, it can observed the hipercarpnia, pulmonary hypoventilation, hypoxemia (secondary hipercarpnia) and therefore, secretion clearance, decreased lung compliance and chest mobility, increasing the mechanical loading sustained every respiratory movement by the weak musculature44. Araujo APQC, Deco MC DE, Klôh BS, Costa MR, Góis FV, Guimarães AFCM. Diagnosis delay of duchenne muscular dystrophy. Rev Bras Saude Mater Infant. 2004 Jun;4(5):179-83. doi.org/10.1590/S1519-38292004000200008.
https://doi.org/10.1590/S1519-3829200400...
.
Considering these respiratory complications, several experimental therapies are being performed to improve the respiratory conditions DMD patients55. Gayraud J, Matecki S, Hnia K, Mornet D, Prefaut C, Mercier J, Michel A, Ramonatxo M. Ventilation during air breathing and in response to hypercapnia in 5 and 16 month-old mdx and C57 mice. J Muscle Res Cell Motil. 2007 Apr;28(1):29-37. PMID: 17431804.
6. Rafael-Fortney JA, Chimanji NS, Schill KE, Martin CD, Murray JD, Gangul YR, Stangland JE, Tran T, Xu Y, Canan BD, Mays TA, Delfín DA, Janssen PM, raman SV. Early treatment with lisinopril and spironolactone preserves cardiac and skeletal muscle in Duchenne muscular dystrophy mice. Circulation. 2011 Apr;124(5):582-8. doi: 10.1161/CIRCULATIONAHA.111.031716.
https://doi.org/10.1161/CIRCULATIONAHA.1...
- 77. Huang P, Cheng G, Lu H, Aronica M, Ransohoff RM, Zhou L. Impaired respiratory function in mdx and mdx/utrn+/− mice. Muscle Nerve. 2011 Feb;43(2):263-7. doi: 10.1002/mus.21848.
https://doi.org/10.1002/mus.21848...
, evidencing the importance of developing new techniques and advances to resources for experimental therapies, e.g pharmacological and stem cells. Since it extend the resources of DMD respiratory muscles intervention, the chances of these patients maintain their activities of daily living without the use of noninvasive ventilatory resources and patient survival increases.
The mdx (X- linked muscular dystrophy mouse) is an animal model commonly used to DMD studies. These animal can be easily and reliably reproduced and due to a spontaneous mutation in the dystrophin gene and lack of dystrophin expression in muscle tissues88. Lynch GS, Hinkle RT, Faulkner JA. Force and power output of diaphragm muscle strips from mdx and control mice after clenbuterol treatment. Neuromuscul Disord. 2001 Mar;11(2):192-6. PMID: 11257477. , 99. Gosselin LE, Barkley JE, Spencer MJ, McCormick KM, Farkas GA. Ventilatory dysfunction in mdx mice: impact of tumor necrosis factor-alpha deletion. Muscle Nerve. 2003 Sep;28(3):336-43. doi: 10.1002/mus.10431.
https://doi.org/10.1002/mus.10431...
. In the mdx, the limb skeletal muscle exhibits an initial periods of severe necrosis at 3-4weeks of age lifespan, however the limb muscles of mdx mice undergo sufficient muscle fiber regeneration to compensate for the continued injury to fibers and muscle fiber degeneration. Although the diaphragm muscle exhibits progressive deterioration, similar to human DMD55. Gayraud J, Matecki S, Hnia K, Mornet D, Prefaut C, Mercier J, Michel A, Ramonatxo M. Ventilation during air breathing and in response to hypercapnia in 5 and 16 month-old mdx and C57 mice. J Muscle Res Cell Motil. 2007 Apr;28(1):29-37. PMID: 17431804. , 1010. Hartel JV, Granchelli JA, Hudecki MS, Pollina CM, Gosselin LE. Impact of prednisone on TGF-β1 and collagen in diaphragm muscle from mdx mice. Muscle Nerve. 2001 Mar;24(3):428-32. PIMD: 11353432. , 1111. Yeung EW, Whitehead NP, Suchyna TM, Gottlieb PA, Sachs F, Allen DG. Effects of stretch-activated channel blockers on [Ca2+]i and muscle damage in the mdx mouse. J Physiol. 2005;562(Pt 2):367-80..
Considering the mdx features cited by the authors above, the benefits of laparoscopy and the possibility to expand access techniques, for respiratory muscles in patients with DMD, this study, aimed to develop a viable a mdxlaparoscopy technique, that allowed the protocol development for an injection spot in the diaphragm muscle of this clinical model.
Methods
This research was certified by the Ethical Principles in Animal Research adopted by "Ethic Committee in the use of animals" of the School of Veterinary Medicine and Animal Science of University of Sao Paulo, protocol number 2045/2010.
For this research, we used 10 mice Balb/C57 of the Vivarium of Pathology, School of Veterinary Medicine Animal Science, University of Sao Paulo and 5 mdx mice the Vivarium of the ABC School of Medicine.
Laparoscopy
To perform the laparoscopic procedure and the establishment of a favorable decubitus for the diaphragm muscle viewing, were tested three types of position: the right lateral decubitus, supine and supine with 20 degrees of elevation of the forelimb. In all positioning tested were used the same anesthetic protocol and laparoscopic.
Anesthetic protocol
The animals were anesthetic with the combination of 50mg/Kg ketamine (Ketamin-S(r), Cristália) and 2mg/kg xylazine (Calmiun(r), Agener União), remained the animals in the holding spontaneous breath.
Laparoscopic protocol
After anesthesia, was realized an incision of 0.5 cm in the lower abdomen at median sagittal plane for laparoscopic input. The 2,7mm optic was introduced and stabilized with circular sutures with wire of 4-0 poligalactina. The pneumoperitoneum was preceded to the pressure of 2 mmHg through a 25X7G needle placed parallel the optical imput.
Protocol for diaphragm muscle injection
Two protocols were tested to define an effective perform to the local injection in the diaphragm muscle
Protocol 1
The injection in the diaphragm costal face was performed in nine animals Balb/C57 being used three animals in each decubitus cited above. In this protocol, the needle was introduced into the right antimere of the abdomen caudal face, near the midline. We used a needle with 24G caliber plugged into a disposable insulin syringe containing 0.2 ml of Tryplan Blue. When it was introduced, the needle bevel was directed to the diaphragm caudal face, in order to chart a cranial path and upward (Figure 1, Protocol 1).
Protocol 1. Laparoscopy image; caudal view of the diaphragm muscle. Observe the needle placed into the diaphragm costal face, (1) liver, (2) lung and (3) diaphragm costal face.
Protocol 2
The protocol 2 was utilized after the following criteria: the determination of the most favorable position for surgery (data obtained using protocol 1) and after checking the effectiveness of the protocol 1.
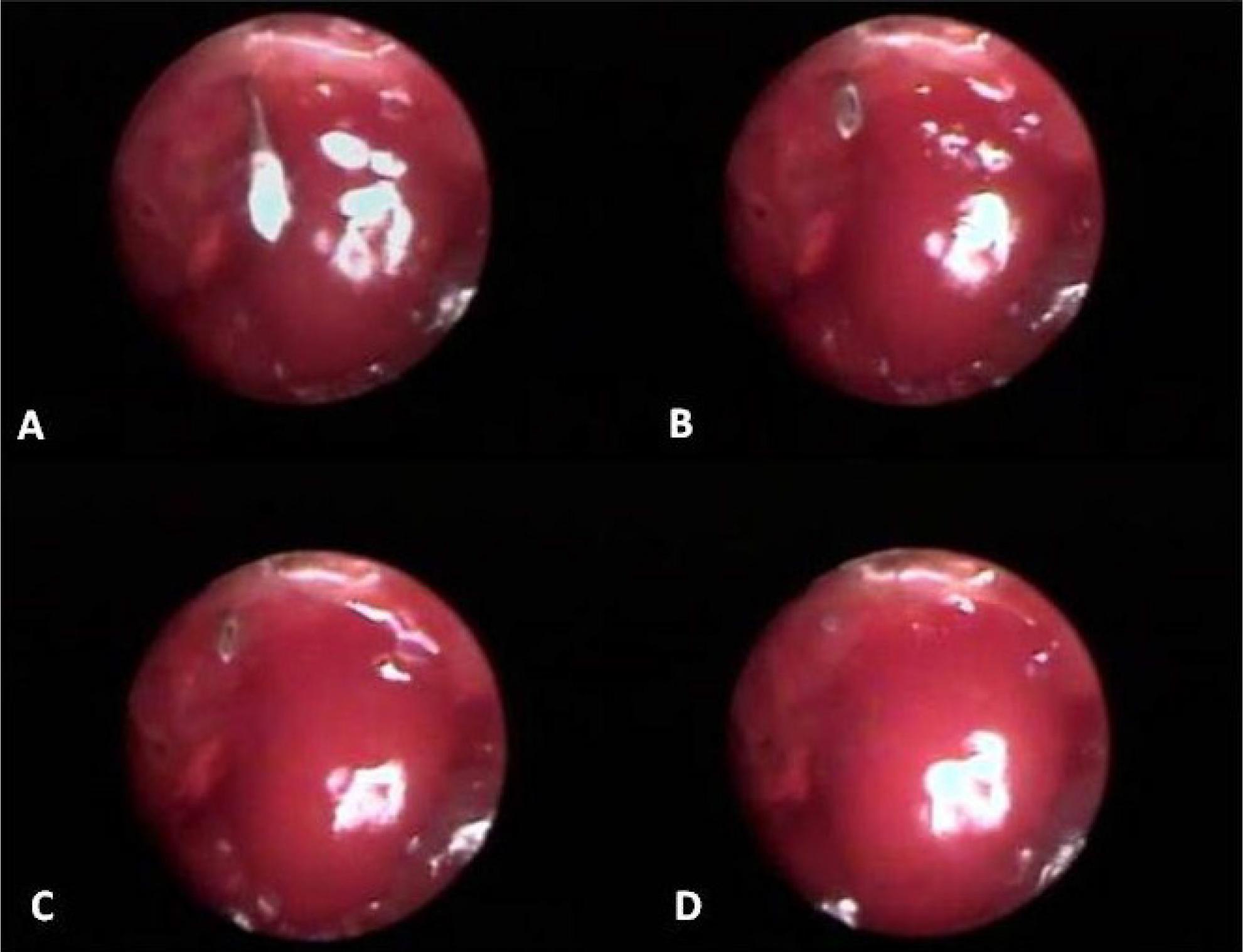
The protocol 2 was realized in supine position with elevation of 20 degrees of the thoracic limbs in 1 Balb/C57 and in 5 mdx. It was used a needle with 24G caliber plugged into the disposable insulin syringe with 0.2 ml de Tryplan Blue. This one was introduced in the 10 intercostal space, piercing the intercostal muscle and the place where the diaphragm is inserted. When the bevel of the needle was visualized on the monitor, this was retreated until the entire needle orifice stay completely within the muscle, occupying a portion of the diaphragm and another in intercostal muscle (Figure 2, Protocol 2).
Protocol 2. Laparoscopy image; caudal view of the diaphragm muscle. (A) Observe the needle into the abdominal cavity, which was introduced in the tenth intercostal space; (B and C) needle being retracted; (D) needle positioned between the diaphragm and intercostal muscle.
Postoperative protocol
The postoperative protocol was performed with 2.5 / kg flunixin meglumine for three days, and 2 mg / kg of meperidine hydrochloride three times daily during five days for analgesia. For prophylactic antibiotics were used 10mg/kg of enrofloxacin by intramuscular injection two times per day. After 10 days, all animals that remained alive were euthanized with an overdose of anesthesia and diaphragmatic muscle was dissected.
Results
Protocol 1 - Laparoscopy in supine position
It was observed in this position, the liver of the animal confined large part of diaphragm costal face, hindering the injection spot. After the needle was introduced, we can observe the occurrence of pneumothorax and after, the death of the animal.
At the end of the procedure, the diaphragm was dissected for analysis. The dissection proceeded in antimeres with the removal of the integument and muscles of the chest and abdominal regions, and thus accessed their organs. For visualization of the diaphragm muscle, the abdominal and thoracic organs were removed. It was observed the diaphragm muscle emerging bundles and peripheral ligaments oriented radially, joining the central tendon presenting planed format. The tendons peripherals were connected ventrally in the sternum and at the xiphoid process, laterally to the ribs (7 th, 8 th, 9 th and 10 th, and apices of the 11 th and 12 th) and the correspondents costal cartilages and dorsally three lumbar vertebrae. It was noted the foramen of the vena cava, esophageal hiatus, aortic hiatus and your three divisions: sternal part, and costal part and the left and right pillars of the diaphragm muscle (Figure 3).
(A) Balb/C57 photograph in supine position, we observe the optical with "X" suture. (B) Caudal view of the diaphragm, (1) right costal face, (2) central tendon, (3) left costal face, (4) xiphoid process, (5) foramen vena cava, (6) esophageal hiatus, (7) aortic hiatus, (8) and left diaphragmatic pillars (9) right diaphragmatic pillars. Note the diaphragmatic muscle stained in blue, due to leakage of the Tryplan Blue.
Protocol 1 - Laparoscopy in right lateral decubitus
It was observed in this position, the animal's liver displacement of the diaphragm caudal face and the liver remained hindering the injection spot, but not precluding the injection. After the needle is inserted, we can observe the occurrence of pneumothorax and after, the death of the animal (Figure 4).
(A) Balb/C57 photograph in right lateral decubitus. (B) Laparoscopy image, (1) left costal face and (2) ligament gastrophrenic of the stomach. (C) Injection spot.
At the end of the procedure, the diaphragm was dissected for analyses and it was observe the same results describe above.
Protocol 1 - Laparoscopy in supine position with 20 degrees of the thoracic limbs
During protocol 1, that the supine position with 20 degrees of thoracic limbs was favorable to visualize the diaphragm muscle, because the liver of the animal had a considerable displacement of the diaphragm costal face, facilitating the injection application. After the needle was introduced we can observe the occurrence of pneumothorax and after, the death of the animal. We observed that this decubitus was the most favored to perform the injection spot in the diaphragm. However this protocol has not kept the animals alive.
At the end of the procedure, the diaphragm was dissected for analyses and it was observe the same results describe above.
Protocol 2 - Laparoscopy in supine position with 20 degrees of thoracic limbs
During perform, of the protocol 2, after the needle be introduced, we can observe that there was no extravasation of Tryplan Blue in the abdominal cavity; pneumothorax has not occurred and the animals remained alive after the procedure. These animals were kept alive for 10 days and after all the animals were euthanized and the diaphragm muscle was dissected. The same morphological structures mentioned above were observed and it was verified that there was no rupture in the diaphragm muscle (Figure 5).
Macroscopic photography, caudal view of the diaphragm muscle. Observe the injection spot marked by Trypan Blue.
Discussion
In DMD, the Golden Retriever Muscular Dystrophy (GRMD) has been the most extensively animal model examined and characterized for elucidation of the pathogenesis and assessment of efficacy and toxicity, during the development of therapies33. Nakamura A, Takeda S. Mammalian models of Duchenne Muscular Dystrophy: pathological characteristics and therapeutic applications. J Biomed Biotechnol. 2011;2011:184393. doi: 10.1155/2011/184393.
https://doi.org/10.1155/2011/184393...
. Although the GRMD are a potential model of human disease, these models require a high maintenance cost and treatment, because they are large, which hinders their handling when was used in experiments. Unlike the mdx model that presents easy reproduction, handling, low cost, low emotional involvement with his treater and vice versa, and convenience for experiments in laboratories88. Lynch GS, Hinkle RT, Faulkner JA. Force and power output of diaphragm muscle strips from mdx and control mice after clenbuterol treatment. Neuromuscul Disord. 2001 Mar;11(2):192-6. PMID: 11257477. , 99. Gosselin LE, Barkley JE, Spencer MJ, McCormick KM, Farkas GA. Ventilatory dysfunction in mdx mice: impact of tumor necrosis factor-alpha deletion. Muscle Nerve. 2003 Sep;28(3):336-43. doi: 10.1002/mus.10431.
https://doi.org/10.1002/mus.10431...
.
In recent years, the diaphragm muscle has been widely approached in the DMD studies55. Gayraud J, Matecki S, Hnia K, Mornet D, Prefaut C, Mercier J, Michel A, Ramonatxo M. Ventilation during air breathing and in response to hypercapnia in 5 and 16 month-old mdx and C57 mice. J Muscle Res Cell Motil. 2007 Apr;28(1):29-37. PMID: 17431804.
6. Rafael-Fortney JA, Chimanji NS, Schill KE, Martin CD, Murray JD, Gangul YR, Stangland JE, Tran T, Xu Y, Canan BD, Mays TA, Delfín DA, Janssen PM, raman SV. Early treatment with lisinopril and spironolactone preserves cardiac and skeletal muscle in Duchenne muscular dystrophy mice. Circulation. 2011 Apr;124(5):582-8. doi: 10.1161/CIRCULATIONAHA.111.031716.
https://doi.org/10.1161/CIRCULATIONAHA.1...
- 77. Huang P, Cheng G, Lu H, Aronica M, Ransohoff RM, Zhou L. Impaired respiratory function in mdx and mdx/utrn+/− mice. Muscle Nerve. 2011 Feb;43(2):263-7. doi: 10.1002/mus.21848.
https://doi.org/10.1002/mus.21848...
which demonstrate the concern of researchers in obtaining data about this muscle.
The laparoscopic procedure has been successfully used in interventions in the diaphragm1212. Matecki S, Guibinga GH, Petrof BJ. Regenerative capacity of the dystrophic (mdx) diaphragm after induced injury. Am J Physiol Regul Integr Comp Physiol. 2004 Jan;87(4):R961-8. doi: 10.1113/jphysiol.2004.075275.
https://doi.org/10.1113/jphysiol.2004.07...
, 1313. Matz A, Landau O, Alis M, Charuzi I, Kyzer SL. The role of laparoscopy in the diagnosis and treatment of missed diaphragmatic ruptures. Surg Endosc. 2000 Jun;14(6):537-9. PMID: 10890960.. However, until the present moment there are no studies using laparoscopic procedure for local injection in the mdx diaphragm.
Matecki et al.1212. Matecki S, Guibinga GH, Petrof BJ. Regenerative capacity of the dystrophic (mdx) diaphragm after induced injury. Am J Physiol Regul Integr Comp Physiol. 2004 Jan;87(4):R961-8. doi: 10.1113/jphysiol.2004.075275.
https://doi.org/10.1113/jphysiol.2004.07...
, used the laparoscopy to expose the abdominal surface of the right hemidiaphragm in themdx and wild-type (C57BL10) and induce a massive necrosis with a topical application of notexin. After surgery, the authors reported that there were no complications, confirming the mdx tolerance for this type of intervention. Despite, the local diaphragm access was described above, in this research, the size of the incision and the fact that there was not diaphragm perforation, differs these two techniques. In the laparotomy, the incision is larger than in laparoscopy, a fact that favors the laparoscopic technique for model mdx.
Regarding the parameters used in laparoscopy1414. Costa VA, Oliveira FM, JR Oliveira LC, Carreiro MC, Guimarães P. Modelo experimental de esplenectomia laparoscópica em ratos. Act Cir Bras. 2003 Jun;18(6):553-6. doi: 10.1590/S0102-86502003000600013.
https://doi.org/10.1590/S0102-8650200300...
, to develop a model of splenectomy in rats, used to establish pneumoperitoneum pressure of 7 mmHg and introduced a 5mm trocar. In one study to practice bariatric surgery in rats, the authors used the pressure of 8 mmHg and 11mm trocar. Meyer et al. 16 16. Meyer F, Ioshii SO, Shin EWK, Esser DM, Marcondes RT, Patriani RH, Pimpão BF. Nefrectomia parcial laparoscópica em ratos. Acta Cir Bras. 2007 Feb;22(2):152-6. doi: 10.1590/S0102-86502007000200014.
https://doi.org/10.1590/S0102-8650200700...
to establish a model of partial nephrectomy, used 20 Wistar rats and established pneumoritônio with pressure of 5 mm Hg and a 5mm trocar. Ettinger et al. 1515. Ettinger JE, Santos-Filho PV, Oliveira PD, Ázaro E, Mello CAB, Amaral PCG, Fahel E. Laparoscopic gastric banding in the rat model as a means of videolaparoscopic training. Obes Surg. 2006 Jul;16(7):903-7. PMID: 16839491., as well as the Meyer et al. 1616. Meyer F, Ioshii SO, Shin EWK, Esser DM, Marcondes RT, Patriani RH, Pimpão BF. Nefrectomia parcial laparoscópica em ratos. Acta Cir Bras. 2007 Feb;22(2):152-6. doi: 10.1590/S0102-86502007000200014.
https://doi.org/10.1590/S0102-8650200700...
, used a pressure of 5 mmHg for establishing pneumoperitoneum. However, in our experiment, we used a 2.7-mm of trocar size, because the animals used, has a smaller size and weight than the animals used in Meyeret al.16 16. Meyer F, Ioshii SO, Shin EWK, Esser DM, Marcondes RT, Patriani RH, Pimpão BF. Nefrectomia parcial laparoscópica em ratos. Acta Cir Bras. 2007 Feb;22(2):152-6. doi: 10.1590/S0102-86502007000200014.
https://doi.org/10.1590/S0102-8650200700...
and Ettinger et al. 1515. Ettinger JE, Santos-Filho PV, Oliveira PD, Ázaro E, Mello CAB, Amaral PCG, Fahel E. Laparoscopic gastric banding in the rat model as a means of videolaparoscopic training. Obes Surg. 2006 Jul;16(7):903-7. PMID: 16839491..
Conclusion
It was proved to be possible to perform the laparoscopy technique in the X-linked muscular dystrophy diaphragm and this requires a specific position and technique during the surgery.
References
-
1Liem MSL. Comparison of conventional anterior surgery and laparoscopic surgery for inguinal-hernia repair. N Engl J Med. 1997 May;336(22):1541-7. doi: 10.1056/NEJM199705293362201.
» https://doi.org/10.1056/NEJM199705293362201 -
2Spencer MJ, Tidball JG. Do immune cells promote the pathology of dystrophin-deficient myopathies? Neuromuscul Disord. 2001 Sep;11(6-7):556-64. PMID: 11525885.
-
3Nakamura A, Takeda S. Mammalian models of Duchenne Muscular Dystrophy: pathological characteristics and therapeutic applications. J Biomed Biotechnol. 2011;2011:184393. doi: 10.1155/2011/184393.
» https://doi.org/10.1155/2011/184393 -
4Araujo APQC, Deco MC DE, Klôh BS, Costa MR, Góis FV, Guimarães AFCM. Diagnosis delay of duchenne muscular dystrophy. Rev Bras Saude Mater Infant. 2004 Jun;4(5):179-83. doi.org/10.1590/S1519-38292004000200008.
» https://doi.org/10.1590/S1519-38292004000200008 -
5Gayraud J, Matecki S, Hnia K, Mornet D, Prefaut C, Mercier J, Michel A, Ramonatxo M. Ventilation during air breathing and in response to hypercapnia in 5 and 16 month-old mdx and C57 mice. J Muscle Res Cell Motil. 2007 Apr;28(1):29-37. PMID: 17431804.
-
6Rafael-Fortney JA, Chimanji NS, Schill KE, Martin CD, Murray JD, Gangul YR, Stangland JE, Tran T, Xu Y, Canan BD, Mays TA, Delfín DA, Janssen PM, raman SV. Early treatment with lisinopril and spironolactone preserves cardiac and skeletal muscle in Duchenne muscular dystrophy mice. Circulation. 2011 Apr;124(5):582-8. doi: 10.1161/CIRCULATIONAHA.111.031716.
» https://doi.org/10.1161/CIRCULATIONAHA.111.031716 -
7Huang P, Cheng G, Lu H, Aronica M, Ransohoff RM, Zhou L. Impaired respiratory function in mdx and mdx/utrn+/− mice. Muscle Nerve. 2011 Feb;43(2):263-7. doi: 10.1002/mus.21848.
» https://doi.org/10.1002/mus.21848 -
8Lynch GS, Hinkle RT, Faulkner JA. Force and power output of diaphragm muscle strips from mdx and control mice after clenbuterol treatment. Neuromuscul Disord. 2001 Mar;11(2):192-6. PMID: 11257477.
-
9Gosselin LE, Barkley JE, Spencer MJ, McCormick KM, Farkas GA. Ventilatory dysfunction in mdx mice: impact of tumor necrosis factor-alpha deletion. Muscle Nerve. 2003 Sep;28(3):336-43. doi: 10.1002/mus.10431.
» https://doi.org/10.1002/mus.10431 -
10Hartel JV, Granchelli JA, Hudecki MS, Pollina CM, Gosselin LE. Impact of prednisone on TGF-β1 and collagen in diaphragm muscle from mdx mice. Muscle Nerve. 2001 Mar;24(3):428-32. PIMD: 11353432.
-
11Yeung EW, Whitehead NP, Suchyna TM, Gottlieb PA, Sachs F, Allen DG. Effects of stretch-activated channel blockers on [Ca2+]i and muscle damage in the mdx mouse. J Physiol. 2005;562(Pt 2):367-80.
-
12Matecki S, Guibinga GH, Petrof BJ. Regenerative capacity of the dystrophic (mdx) diaphragm after induced injury. Am J Physiol Regul Integr Comp Physiol. 2004 Jan;87(4):R961-8. doi: 10.1113/jphysiol.2004.075275.
» https://doi.org/10.1113/jphysiol.2004.075275 -
13Matz A, Landau O, Alis M, Charuzi I, Kyzer SL. The role of laparoscopy in the diagnosis and treatment of missed diaphragmatic ruptures. Surg Endosc. 2000 Jun;14(6):537-9. PMID: 10890960.
-
14Costa VA, Oliveira FM, JR Oliveira LC, Carreiro MC, Guimarães P. Modelo experimental de esplenectomia laparoscópica em ratos. Act Cir Bras. 2003 Jun;18(6):553-6. doi: 10.1590/S0102-86502003000600013.
» https://doi.org/10.1590/S0102-86502003000600013 -
15Ettinger JE, Santos-Filho PV, Oliveira PD, Ázaro E, Mello CAB, Amaral PCG, Fahel E. Laparoscopic gastric banding in the rat model as a means of videolaparoscopic training. Obes Surg. 2006 Jul;16(7):903-7. PMID: 16839491.
-
16Meyer F, Ioshii SO, Shin EWK, Esser DM, Marcondes RT, Patriani RH, Pimpão BF. Nefrectomia parcial laparoscópica em ratos. Acta Cir Bras. 2007 Feb;22(2):152-6. doi: 10.1590/S0102-86502007000200014.
» https://doi.org/10.1590/S0102-86502007000200014
-
Financial sources: Sao Paulo Research Foundation (FAPESP), Coordination of Improvement for Higher Academic Staff (CAPES)
-
1
Research performed at Faculty of Veterinary Medicine and Animal Science, Department of Surgery, University of Sao Paulo (USP), Brazil. Part of Master degree thesis, Postgraduate Program in Anatomy of Domestic and Wild Animals. Tutor: Prof. Dr. Carlos Eduardo Ambrósio.
Publication Dates
-
Publication in this collection
Nov 2014
History
-
Received
12 June 2014 -
Reviewed
14 Aug 2014 -
Accepted
15 Sept 2014