Abstract
PURPOSE:
To evaluate the effects of copaiba oil on jaw defects repair in Wistar rats treated with bioglass or adipose tissue.
METHODS:
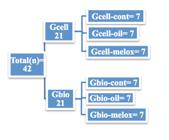
A jaw defect was randomly created in forty-two rats and filled with bioglass or adipose tissue. The two groups (Gbio and Gcell) were subdivided in three subgroups with seven animals each according to gavage administration: control (distillated water), oil (copaiba oil) and melox (meloxicam). Euthanasia was performed after forty post-operative days. The bone formation was analyzed regarding the histological aspects.
RESULTS:
The osteoclasts activity was observed only in four subgroups (p=0.78). Regarding the osteoblasts presence, it was very similar between the subgroups, the difference was due to Gcell-melox (p=0.009) that presented less osteoblastic activity. The inflammatory cells were more evident in Gcell-melox subgroup, however, there was no difference in comparison with the other subgroups (p=0.52). Bone formation was observed in all subgroups, just two animals showed no bone formation even after 40 days. More than 50% of bone matrix mineralization was observed in 56% (23 animals) of the analyzed areas. The bone matrix mineralization was not different between subgroups (p=0.60).
CONCLUSIONS:
The subgroups that received copaiba oil showed bone repair, although not statistically significant in comparison to subgroups treated whit meloxicam or controls. Copaiba oil administered by gavage had no effect on bone repair in this experimental model.
Plants, Medicinal; Bone Regeneration; Anti-Inflammatory Agents; Rats
Introduction
Bone substitutes are frequently used in dentistry to augment bone tissue or to fill bone
defects. Among bone grafts, various types of hydroxyapatite, deproteinized bovine bone
matrix and synthetic glasses have been largely tested on bone tissue engineering11. Carmagnola D, Abati S, Celestino S, Chiapasco M, Bosshardt D, Lang
NP. Oral implants placed in bone defects treated with Bio-Oss, Ostim-Paste or
PerioGlas: an experimental study in the rabbit tibiae. Clin Oral Implants Res. 2008
Dec;19(12):1246-53. doi: 10.1111/j.1600-0501.2008.01584.x.
https://doi.org/10.1111/j.1600-0501.2008...
. Due to its limited availability and morbidity,
the autogenous bone graft as well as an inadequate potential osteoinductive and
osteoconductive of the allograft, research has been conducted to develop new bone
substitutes to improve bone healing22. Oliveira LC, Giovanini AF, Abuabara A, Klug LG, Gonzaga CC, Zielak
JC, Urban CA, Deliberador TM. Fragmented adipose tissue graft for bone healing:
histological and histometric study in rabbits' calvaria. Med Oral Patol Oral Cir
Bucal. 2013 May;18(3):e510-5. doi: 10.4317/medoral.18407.
https://doi.org/10.4317/medoral.18407...
.
Bioglass is a bioactive particulate material made of granules consisting of silicon,
sodium, calcium and phosphorus, able to bond to bone and connective tissue. It may allow
for more rapid repair and regeneration of bone than other synthetic grafting materials
by forming a layer of hydroxyl-carbonate apatite on its surface equivalent to the
mineral phase of human bone. It has been used in a few experimental and clinical studies
to treat different types of bone defect and it was claimed that bioglass promoted bone
formation11. Carmagnola D, Abati S, Celestino S, Chiapasco M, Bosshardt D, Lang
NP. Oral implants placed in bone defects treated with Bio-Oss, Ostim-Paste or
PerioGlas: an experimental study in the rabbit tibiae. Clin Oral Implants Res. 2008
Dec;19(12):1246-53. doi: 10.1111/j.1600-0501.2008.01584.x.
https://doi.org/10.1111/j.1600-0501.2008...
,
33. Stavropoulos A, Kostopoulos L, Nyengaard JR, Karring T. Deproteinized
bovine bone (Bio-Oss) and bioactive glass (Biogran) arrest bone formation when used
as an adjunct to guided tissue regeneration (GTR): an experimental study in the rat.
J Clin Periodontol. 2003 Jul;30(7):636-43. doi:
10.1034/j.1600-051X.2003.00093.x.
https://doi.org/10.1034/j.1600-051X.2003...
.
Mesenchymal stem cells reside in many tissues, including bone marrow, adipose tissue,
synovial fluid, dermis and muscle. They can differentiate along a variety of cell
lineages that can be used to regenerate bone and other tissues. Adipose-derived
mesenchymal stem cells have been identified as a source of multipotent cells that have
osteogenic differentiation potential in vitro and in vivo and is the most abundant and
accessible source of adult stem cells, which is significantly higher in adipose tissue
in comparison to bone marrow (2% vs. 0.002%)2. Moreover, they have been
reported to have low immunogenicity. The studies have demonstrated that adipose tissue
stem cells are capable of exhibiting good attachment properties to most material
surfaces and have the capacity to differentiate into osteoblastic-like cells in vitro
and in vivo44. Shi Y, Niedzinski JR, Samaniego A, Bogdansky S, Atkinson BL.
Adipose-derived stem cells combined with a demineralized cancellous bone substrate
for bone regeneration. Tissue Eng Part A. 2012 Jul;18(13-14):1313-21. doi:
10.1089/ten.tea.2011.0357.
https://doi.org/10.1089/ten.tea.2011.035...
,
55. Liu G, Zhang Y, Liu B, Sun J, Li W, Cui L. Bone regeneration in a
canine cranial model using allogeneic adipose derived stem cells and coral scaffold.
Biomaterials. 2013 Apr;34(11):2655-64. doi:
10.1016/j.biomaterials.2013.01.004.
https://doi.org/10.1016/j.biomaterials.2...
.
Copaiba oil has been widely targeted research in order to prove its therapeutic characteristics. In several studies it had effective pharmacological properties in surgical procedures, wound healing, inflammatory processes, such as antioxidant, antitumor and even as a bacteriostatic substance6-126. Carmona GB, Teixeira RKC, Brito MVH, Pontes FSC, Andrade EHA, Fonseca FP, Costa RMB, Carvalho FM. Effect of andiroba oil on periodontitis in Wistar rats. Acta Cir Bras. 2013 Jun;28(6):430-4. PMID: 23743680. . Yasojima et al. 1111. Yasojima EY, Teixeira, RKC, Houat AP, Costa FLS, Silveira EL, Brito MVH, Lopes Filho GJ. Effect of copaiba oil on correction of abdominal wall defect treated with the use of polypropylene/polyglecaprone mesh. Acta Cir Bras. 2013 Feb;28(2):131-5. PMID: 23370927. analyzed the effect of copaiba oil in correction of abdominal wall defects treated with polypropylene/polyglecaprone meshes in rats. The authors concluded that copaiba oil administered by gavage reduced the amount of abdominal adhesions and accelerated the formation of collagen fibers without damaging the early stages of healing. Nogueira Neto et al. 1212. Nogueira Neto J, Lindoso MJS, Coelho LF, Carvalho RAF, Rodrigues TGPM, Araújo AGP, Girão MJBC, Schor E. Changes in the volume and histology of endometriosis foci in rats treated with copaiba oil (Copaifera langsdorffii). Acta Cir Bras. 2011;26 Suppl 2:20-4. PMID: 22030810. evaluated changes that occurred in rats with experimental endometriosis treated with copaiba oil. They observed a significant increase between the end and the initial volume the control group, whereas the group treated with copaiba oil had a marked reduction in endometrial growth. These results led the authors to conclude that copaiba oil can be an alternative potential treatment for endometriosis.
Many anti-inflammatory substances are currently used in clinical practice after bone
grafting and fractures to minimize clinical postoperative symptoms, nevertheless their
effect in bone tissue can minimize or delay the incorporation process and affect bone
tissue repair13-1813. Li J, Wang X, Zhou C, Liu L, Wu Y, Wang D, Jiang H. Perioperative
glucocorticosteroid treatment delays early healing of a jaw wound by inhibiting
osteogenic differentiation. Injury. 2012 Aug;43(8):1284-9. doi:
10.1016/j.injury.2012.04.014.
https://doi.org/10.1016/j.injury.2012.04...
. Despite its proven
anti-inflammatory action, the effect of copaiba oil on bone tissue repair is not well
established2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
. The aim of this experimental
study was to evaluate the copaiba oil influence in experimental bone defects filled with
two bone substitutes in rat´s jaw by evaluating histologically the composition of formed
bone tissue.
Methods
The project was approved by Ethics Committee in the Use of Animals, State University of Para (UEPA) (Protocol 06/2013).
Forty two Wistar male rats (Rattus norvegicus) aged about 120 days and weight ranging from 250 to 300g from the Animal Colony of the Experimental Surgery Laboratory of UEPA were used. They were kept in a controlled environment (light cycle 12/12 h and temperature of 23° C), with food and water ad libitum. The animals were randomly distributed into two groups (Gbio and Gcell) and subdivided in three subgroups each with seven animals (Figure 1).
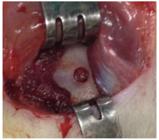
The procedure was performed by a single surgeon. The animals were anesthetized with ketamine hydrochloride (70 mg/Kg) and xylazine hydrochloride (10 mg/Kg), administered intraperitoneally. Once animals' anesthesia was confirmed, the skin was shaved and the surgical site was disinfected. Subsequently, was performed an "U" (7X14mm) incision and a full-thickness flap was raised to expose the external side of jaw. Under continuous saline irrigation, a slowly rotating round burr was used to create circular defects 2.5mm in diameter in the region anterior to the jaw angles on right side of the jaw of each rat. (Figure 2)
Study design
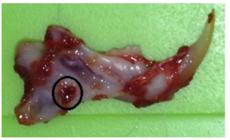
The bone defects were filled with either bioglass (CeelBio) or adipose tissue according to the groups (Figure 3). In Cell subgroups (Gcell-control, Gcell-oil, Gcell-melox), shave, antisepsis and a linear 5mm incision was realized in the inguinal region of the animal and then 9mm3 of the subcutaneous adipose tissue was dissected and removed (Figure 4). The adipose tissue was fragmented and washed with saline solution according to technique described by Gomes et al.1919. Gomes SP, Deliberador TM, Gonzaga CC, Klug LG, Oliveira LC, Urban CA, Zielak JC, Giovanini AF. Bone healing in critical-size defects treated with immediate transplant of fragmented autogenous white adipose tissue. J Craniofac Surg. 2012 Sep;23(5):1239-44. PMID: 22948634. . The bioglass subgroups (Gbio-control, Gbio-oil, Gbio-melox) were grafted with bioglass (granulation of 0.25 mm) provided by the company CeellBio. After the procedures, the wounds were closed with 3-0 nylon sutures.
Gavage procedures
On the fifth postoperative day, the gavage was initiated and performed for seven days, once a day, with three different substances according to subgroups: Gbio-control and Gcell-control received 0.6ml/kg/day distillated water; Gbio-oil and Gcell-oil received 0.6 ml/kg/day copaiba oil in natura (EMBRAPA); Gbio-melox and Gcell-melox received 0.25mg/kg/day meloxicam (Medley(r)) diluted in distillated water 0.6ml/kg (Table 1).
Forty days after surgical procedure the animals were submitted to euthanasia procedure by anesthetic´s overdoses.
The right side of the jaw was removed for histological analysis (Figure 5). The fragments were fixed in 10% formalin for 48 hours and decalcified in 10% nitric acid. So, after included in paraffin, they were processed to obtain 5μm thick serial sections, which were made in buccolingual direction to compose histological blades.
The specimens were stained with hematoxylin/eosin and Masson's trichrome. The quality of new bone and inflammatory response in the defect site were evaluated in histological analysis by light microscopy with x40 magnification (Nikon Eclipse E200, Tokyo, Japan).
Bone formation parameters (scores of osteoclasts, osteoblasts, inflammatory cells and bone matrix) were established to histological evaluation. The osteoprogenitor bone cells presence (osteoblasts) and activity (osteoclasts - 1: inactivity, 2: little, 3: much activity), the inflammatory infiltrate (1: absent, 2: mild, 3: moderate, 4: intense) and the mineralization of bone matrix (1: absence, 2: 0 to 50% bone formation, 3: >50% bone formation) were analyzed and categorized for each subgroup. A previous trained, single blinded examiner was able to establish the scores for each variable set.
Statistical analysis
The non-parametric Kruskal-Wallis test was used to evaluate the statistical differences between the subgroups, followed by Dunn´s test considering a p-value less than 0.05 as significant. The software Bioestat 5.3(r) was used for statistical analysis.
Results
During the research, just one animal died after surgical procedure. There were no other complications like dehiscence, fistulae or infection.
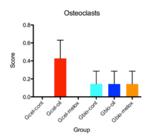
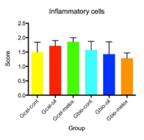
The osteoclasts activity was observed only in four groups, it was more evident in Gcell-oil (p=0.78), however this was not statistically significant (Figure 6). Regarding the osteoblasts presence, the difference was due to Gcell-melox (p=0.009) that presented less osteoblastic activity (Figure 7). The inflammatory cells were present in all groups, however there was no statistical significance (p=0.52) (Figure 8).
Bone formation was observed in all groups, just two animals showed no bone formation even after 40 days. More than 50% of bone matrix mineralization was observed in 56% (23 animals) of the analyzed areas. The bone matrix mineralization was not different between groups (p=0.60) (Figures 9 and 10).
Masson´s trichrome staining. Histological specimen analysis of cell (A) and bioglass (B) group. x40 magnification demonstrating presence of remnant material (yellow arrow), osteoblastic activity (black arrow) and newly formed bone matrix (green arrow).
Discussion
The results from the present study showed that rat jaw grafted with two different biomaterials (bioglass or adipose tissue), obtained, in the 40-day postoperative period, a larger extent of bone matrix deposition, indicating that the groups had similar amounts of mineral deposition in this period of the repair.
The two grafting materials seemed to perform in a similar way concerning their
contribution to bone formation which is consistent whit other studies11. Carmagnola D, Abati S, Celestino S, Chiapasco M, Bosshardt D, Lang
NP. Oral implants placed in bone defects treated with Bio-Oss, Ostim-Paste or
PerioGlas: an experimental study in the rabbit tibiae. Clin Oral Implants Res. 2008
Dec;19(12):1246-53. doi: 10.1111/j.1600-0501.2008.01584.x.
https://doi.org/10.1111/j.1600-0501.2008...
,
22. Oliveira LC, Giovanini AF, Abuabara A, Klug LG, Gonzaga CC, Zielak
JC, Urban CA, Deliberador TM. Fragmented adipose tissue graft for bone healing:
histological and histometric study in rabbits' calvaria. Med Oral Patol Oral Cir
Bucal. 2013 May;18(3):e510-5. doi: 10.4317/medoral.18407.
https://doi.org/10.4317/medoral.18407...
,
44. Shi Y, Niedzinski JR, Samaniego A, Bogdansky S, Atkinson BL.
Adipose-derived stem cells combined with a demineralized cancellous bone substrate
for bone regeneration. Tissue Eng Part A. 2012 Jul;18(13-14):1313-21. doi:
10.1089/ten.tea.2011.0357.
https://doi.org/10.1089/ten.tea.2011.035...
,
55. Liu G, Zhang Y, Liu B, Sun J, Li W, Cui L. Bone regeneration in a
canine cranial model using allogeneic adipose derived stem cells and coral scaffold.
Biomaterials. 2013 Apr;34(11):2655-64. doi:
10.1016/j.biomaterials.2013.01.004.
https://doi.org/10.1016/j.biomaterials.2...
,
2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
. They appeared to be osteoconductive, leading
to a mineralized bone matrix with osteoblasts and chronic inflammatory infiltrate.
Through histological evaluation regarding the quality of newly formed bone tissue and
comparing the oil, control and melox groups can be inferred that copaiba oil did not
interfere with bone formation. To date, just one research correlating copaiba oil to the
healing of bone tissue was found. Silva et al.
2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
evaluated the influence of topic and systemic
administration of copaiba oil on the alveolar wound healing after tooth extraction in
rats. They observed an increased bone neoformation in both groups treated with copaiba
oil when compared with placebo group, however, the topic application should be carefully
considered because rats treated with topic administration of copaiba oil presented
ulcerations and large number of inflammatory cells2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
.
Several other substances with anti-inflammatory properties have been subject of research
in this area with contradictory findings1313. Li J, Wang X, Zhou C, Liu L, Wu Y, Wang D, Jiang H. Perioperative
glucocorticosteroid treatment delays early healing of a jaw wound by inhibiting
osteogenic differentiation. Injury. 2012 Aug;43(8):1284-9. doi:
10.1016/j.injury.2012.04.014.
https://doi.org/10.1016/j.injury.2012.04...
,
1515. Dimmen S, Nordsletten L, Madsen JE. Parecoxib and indomethacin delay
early fracture healing: a study in rats. Clin Orthop Relat Res. 2009
Aug;467(8):1992-9. doi: 10.1007/s11999-009-0783-0.
https://doi.org/10.1007/s11999-009-0783-...
,
1818. Cappello T, Nuelle JAV, Katsantonis N, Nauer RK, Lauing KL,
Jagodzinski JE, Callaci JJ. Ketorolac administration does not delay early fracture
healing in a juvenile rat model: a pilot study. J Pediatr Orthop. 2013
Jun;33(4):415-21. doi: 10.1097/BPO.0b013e318288b46f.
https://doi.org/10.1097/BPO.0b013e318288...
. The studies indicates that non-steroidal
anti-inflammatory drugs (NSAIDs) with higher selectivity to COX-2 showed delayed bone
healing and compromised bone density1313. Li J, Wang X, Zhou C, Liu L, Wu Y, Wang D, Jiang H. Perioperative
glucocorticosteroid treatment delays early healing of a jaw wound by inhibiting
osteogenic differentiation. Injury. 2012 Aug;43(8):1284-9. doi:
10.1016/j.injury.2012.04.014.
https://doi.org/10.1016/j.injury.2012.04...
,
1515. Dimmen S, Nordsletten L, Madsen JE. Parecoxib and indomethacin delay
early fracture healing: a study in rats. Clin Orthop Relat Res. 2009
Aug;467(8):1992-9. doi: 10.1007/s11999-009-0783-0.
https://doi.org/10.1007/s11999-009-0783-...
,
1818. Cappello T, Nuelle JAV, Katsantonis N, Nauer RK, Lauing KL,
Jagodzinski JE, Callaci JJ. Ketorolac administration does not delay early fracture
healing in a juvenile rat model: a pilot study. J Pediatr Orthop. 2013
Jun;33(4):415-21. doi: 10.1097/BPO.0b013e318288b46f.
https://doi.org/10.1097/BPO.0b013e318288...
. Even the glucocorticoids, by inhibition of
COX-2 showed a reduced osteogenic differentiation and maturation13 while
non-selective or NSAIDs with low selectivity did not show changes in bone repair1717. Teófilo JM, Giovanini GS, Fracon RN, Lamano T. Histometric study of
alveolar bone healing in rats treated with the nonsteroidal anti-inflammatory drug
nimesulide. Implant Dent. 2011 Apr;20(2):e7-13. doi:
10.1097/ID.0b013e31820fbacf.
https://doi.org/10.1097/ID.0b013e31820fb...
,
1818. Cappello T, Nuelle JAV, Katsantonis N, Nauer RK, Lauing KL,
Jagodzinski JE, Callaci JJ. Ketorolac administration does not delay early fracture
healing in a juvenile rat model: a pilot study. J Pediatr Orthop. 2013
Jun;33(4):415-21. doi: 10.1097/BPO.0b013e318288b46f.
https://doi.org/10.1097/BPO.0b013e318288...
.
In the present study, meloxicam, a preferential inhibitor to COX-2, decreased
osteoblasts presence when administered in the group that received adipose tissue graft
(p<0.05), which probably affected the synthesis of bone matrix by mesenchymal stem
cells, being consistent with studies showing that non-steroidal anti-inflammatory drugs
affects the quality of bone repair1313. Li J, Wang X, Zhou C, Liu L, Wu Y, Wang D, Jiang H. Perioperative
glucocorticosteroid treatment delays early healing of a jaw wound by inhibiting
osteogenic differentiation. Injury. 2012 Aug;43(8):1284-9. doi:
10.1016/j.injury.2012.04.014.
https://doi.org/10.1016/j.injury.2012.04...
,
1515. Dimmen S, Nordsletten L, Madsen JE. Parecoxib and indomethacin delay
early fracture healing: a study in rats. Clin Orthop Relat Res. 2009
Aug;467(8):1992-9. doi: 10.1007/s11999-009-0783-0.
https://doi.org/10.1007/s11999-009-0783-...
. However, there was a good mineralization of
bone matrix formed after forty days in this specific group, suggesting the osteoblasts
were present but could have a time-limited activity.
The anti-inflammatory activity of copaiba oil has not yet been clearly elucidated. It is
believed that such activity is due to the sesquiterpenes which are known to inhibit the
activity of receptor activator of nuclear factor kappa b (RANK). RANK is the receptor
for RANK-Ligand (RANKL) and part of a signaling pathway that regulates osteoclast
differentiation and activation. It is associated with bone remodeling and repair, immune
cell function, lymph node development, thermal regulation, and mammary gland
development. Its presence on the osteoclast precursors and mature osteoclasts´ surface
plays an important role in promoting the differentiation and activation of osteoclasts,
leading to bone resorption1111. Yasojima EY, Teixeira, RKC, Houat AP, Costa FLS, Silveira EL, Brito
MVH, Lopes Filho GJ. Effect of copaiba oil on correction of abdominal wall defect
treated with the use of polypropylene/polyglecaprone mesh. Acta Cir Bras. 2013
Feb;28(2):131-5. PMID: 23370927.
,
1212. Nogueira Neto J, Lindoso MJS, Coelho LF, Carvalho RAF, Rodrigues
TGPM, Araújo AGP, Girão MJBC, Schor E. Changes in the volume and histology of
endometriosis foci in rats treated with copaiba oil (Copaifera langsdorffii). Acta
Cir Bras. 2011;26 Suppl 2:20-4. PMID: 22030810.
,
2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
. Silva et al.
2020. Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of
topic and systemic administration of copaiba oil on the alveolar wound healing after
tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi:
10.4317/jced.51104.
https://doi.org/10.4317/jced.51104...
believe that positive influence of copaiba oil on alveolar bone formation is
probably explained by the fact that the sesquiterpenes of copaiba oil inhibited the
action of RANK, facilitating the alveolar bone repair.
In the present study the copaiba oil was administered by gavage and the bone formation was histologically evaluated 40 days after the procedure, these two factors could contribute to the results found. Others ways of copaiba oil administration and assessment in shortest and largest period of time, could contribute to different results of this substance influence in jaw bone repair.
Conclusion
Copaiba oil did not affect the amount of bone formation in jaw defects in rats 40 days after the procedure.
Acknowledgements
CEELBIO for providing the bioglass and EMBRAPA by the copaiba oil.
References
-
1Carmagnola D, Abati S, Celestino S, Chiapasco M, Bosshardt D, Lang NP. Oral implants placed in bone defects treated with Bio-Oss, Ostim-Paste or PerioGlas: an experimental study in the rabbit tibiae. Clin Oral Implants Res. 2008 Dec;19(12):1246-53. doi: 10.1111/j.1600-0501.2008.01584.x.
» https://doi.org/10.1111/j.1600-0501.2008.01584.x -
2Oliveira LC, Giovanini AF, Abuabara A, Klug LG, Gonzaga CC, Zielak JC, Urban CA, Deliberador TM. Fragmented adipose tissue graft for bone healing: histological and histometric study in rabbits' calvaria. Med Oral Patol Oral Cir Bucal. 2013 May;18(3):e510-5. doi: 10.4317/medoral.18407.
» https://doi.org/10.4317/medoral.18407 -
3Stavropoulos A, Kostopoulos L, Nyengaard JR, Karring T. Deproteinized bovine bone (Bio-Oss) and bioactive glass (Biogran) arrest bone formation when used as an adjunct to guided tissue regeneration (GTR): an experimental study in the rat. J Clin Periodontol. 2003 Jul;30(7):636-43. doi: 10.1034/j.1600-051X.2003.00093.x.
» https://doi.org/10.1034/j.1600-051X.2003.00093.x -
4Shi Y, Niedzinski JR, Samaniego A, Bogdansky S, Atkinson BL. Adipose-derived stem cells combined with a demineralized cancellous bone substrate for bone regeneration. Tissue Eng Part A. 2012 Jul;18(13-14):1313-21. doi: 10.1089/ten.tea.2011.0357.
» https://doi.org/10.1089/ten.tea.2011.0357 -
5Liu G, Zhang Y, Liu B, Sun J, Li W, Cui L. Bone regeneration in a canine cranial model using allogeneic adipose derived stem cells and coral scaffold. Biomaterials. 2013 Apr;34(11):2655-64. doi: 10.1016/j.biomaterials.2013.01.004.
» https://doi.org/10.1016/j.biomaterials.2013.01.004 -
6Carmona GB, Teixeira RKC, Brito MVH, Pontes FSC, Andrade EHA, Fonseca FP, Costa RMB, Carvalho FM. Effect of andiroba oil on periodontitis in Wistar rats. Acta Cir Bras. 2013 Jun;28(6):430-4. PMID: 23743680.
-
7Gelmini F, Beretta G, Anselmi C, Centini M, Magni P, Ruscica M, Cavalchini A, Facino RM. GC-MS profiling of the phytochemical constituents of the oleoresin from Copaifera langsdorffii Desf and a preliminary in vivo evaluation of its antipsoriatic effect. Int J Pharm. 2013 Jan;440(2):170-8. doi: 10.1016/j.ijpharm.2012.08.021.
» https://doi.org/10.1016/j.ijpharm.2012.08.021 -
8Pieri FA, Mussi MCM, Fiorini JE, Moreira MAS, Schneedorf JM. Bacteriostatic effect of copaiba oil (Copaifera officinalis) against Streptococcus mutans. Braz Dent J. 2012;23(1):36-38. PMID: 22460312.
-
9Brito NMB, Brito MVH, Carvalho RKV, Matos LTBM, Lobato RC, Correa SC, Brito RB. The effect of copaiba balsam on Walker 256 carcinoma inoculated into the vagina and uterine cervix of female rats. Acta Cir Bras. 2010 Apr;25(2):176-80. PMID: 20305885.
-
10Silva JJL, Guimarães SB, Silveira ER, Vasconcelos PRL, Lima GG, Torres SM, Vasconcelos RC. Effects of copaifera langsdorffii desf on ischemia-reperfusion of randomized skin flaps in rats. Aesthetic Plast Surg. 2009 Jan;33(1):104-9. doi: 10.1007/s00266-008-9263-2.
» https://doi.org/10.1007/s00266-008-9263-2 -
11Yasojima EY, Teixeira, RKC, Houat AP, Costa FLS, Silveira EL, Brito MVH, Lopes Filho GJ. Effect of copaiba oil on correction of abdominal wall defect treated with the use of polypropylene/polyglecaprone mesh. Acta Cir Bras. 2013 Feb;28(2):131-5. PMID: 23370927.
-
12Nogueira Neto J, Lindoso MJS, Coelho LF, Carvalho RAF, Rodrigues TGPM, Araújo AGP, Girão MJBC, Schor E. Changes in the volume and histology of endometriosis foci in rats treated with copaiba oil (Copaifera langsdorffii). Acta Cir Bras. 2011;26 Suppl 2:20-4. PMID: 22030810.
-
13Li J, Wang X, Zhou C, Liu L, Wu Y, Wang D, Jiang H. Perioperative glucocorticosteroid treatment delays early healing of a jaw wound by inhibiting osteogenic differentiation. Injury. 2012 Aug;43(8):1284-9. doi: 10.1016/j.injury.2012.04.014.
» https://doi.org/10.1016/j.injury.2012.04.014 -
14Iwamoto J, Seki A, Sato Y, Matsumoto H, Tadeda T, Yeh JK. Vitamin k2 promotes bone healing in a rat femoral osteotomy model with or without glucocorticoid treatment. Calcif Tissue Int. 2010 Mar;86(3):234-41. doi: 10.1007/s00223-010-9333-8.
» https://doi.org/10.1007/s00223-010-9333-8 -
15Dimmen S, Nordsletten L, Madsen JE. Parecoxib and indomethacin delay early fracture healing: a study in rats. Clin Orthop Relat Res. 2009 Aug;467(8):1992-9. doi: 10.1007/s11999-009-0783-0.
» https://doi.org/10.1007/s11999-009-0783-0 -
16Akritopoulos P, Papaioannidou P, Hatzokos I, Haritanti A, Iosifidou E, Kotoula M, Mirtsou-Fidani V. Parecoxib has non-significant long-term effects on bone healing in rats when administered for a short period after fracture. Arch Orthop Trauma Surg. 2009 Oct;129(10):1427-32. doi: 10.1007/s00402-008-0707-6.
» https://doi.org/10.1007/s00402-008-0707-6 -
17Teófilo JM, Giovanini GS, Fracon RN, Lamano T. Histometric study of alveolar bone healing in rats treated with the nonsteroidal anti-inflammatory drug nimesulide. Implant Dent. 2011 Apr;20(2):e7-13. doi: 10.1097/ID.0b013e31820fbacf.
» https://doi.org/10.1097/ID.0b013e31820fbacf -
18Cappello T, Nuelle JAV, Katsantonis N, Nauer RK, Lauing KL, Jagodzinski JE, Callaci JJ. Ketorolac administration does not delay early fracture healing in a juvenile rat model: a pilot study. J Pediatr Orthop. 2013 Jun;33(4):415-21. doi: 10.1097/BPO.0b013e318288b46f.
» https://doi.org/10.1097/BPO.0b013e318288b46f -
19Gomes SP, Deliberador TM, Gonzaga CC, Klug LG, Oliveira LC, Urban CA, Zielak JC, Giovanini AF. Bone healing in critical-size defects treated with immediate transplant of fragmented autogenous white adipose tissue. J Craniofac Surg. 2012 Sep;23(5):1239-44. PMID: 22948634.
-
20Silva MAD, Pereira AC, Marin MCC, Salgado MAC. The influence of topic and systemic administration of copaiba oil on the alveolar wound healing after tooth extraction in rats. J Clin Exp Dent. 2013 Oct;5(4):e169-73. doi: 10.4317/jced.51104.
» https://doi.org/10.4317/jced.51104
-
Financial source: none
-
1Research performed at Experimental Surgery Laboratory (LCE), Para State University (UEPA), Brazil. Part of Master degree thesis, Postgraduate Program in Clinical Dentistry, UFPA. Tutor: Marcus Vinicius Henriques Brito.
Publication Dates
-
Publication in this collection
Feb 2015
History
-
Received
15 Oct 2014 -
Reviewed
17 Dec 2014 -
Accepted
19 Jan 2015