Abstract
Purpose:
To investigate the expression of nitric oxide synthase (NOS) and apoptosis associated with ischemic preconditioning (IPC) and pentoxifylline (PTX) in intestinal ischemia (I) and reperfusion (R) injury.
Methods:
Thirty male rats were assigned to 5 groups: (CG), no clamping of the superior mesenteric artery (90 minutes); (IR-SS) saline + ischemia (30 minutes) + reperfusion (60 minutes); (IR-PTX) PTX + ischemia (30 minutes) + reperfusion (60 minutes); (IPC-IR-SS) 5 minutes of ischemia + 5 minutes of reperfusion (IPC) + saline + I(30 minutes)+R(60 minutes); and (IPC-IR-PTX) IPC + PTX + I(30 minutes)+ R(60 minutes).
Results:
The application of IPC and PTX showed a significantly lower immunohistochemistry reaction for active caspase-3 (P<0.05) compared to IR+SS. The number of cells immunoreactive to BCL-2 was higher in the IR-PTX group (P>0.05). The NOS-2 expression (qRTPCR) in the IR-PTX group (P<0.05) was higher than the values for the IPC+IR-SS and IPC-IR-PTX groups. The NOS-3 expression was significantly upper in the IPC-IR-PTX group than in the CG (P<0.05), the IR-SS (P<0.05) and the IR-PTX (P<0.05) groups.
Conclusions:
The BCL-2 and active caspase-3 showed beneficial effects on PTX and IPC. The expression of NOS-2 and NOS-3 in the IPC and IPC-PTX groups showed no synergistic effect.
Key words:
Ischemic Preconditioning; Pentoxifylline; Ischemia; Reperfusion Injury; Nitric Oxide Synthase; Apoptosis; Rats.
Introduction
Intestinal ischemia-reperfusion injury (IR) is a frequent phenomenon with high morbidity and mortality. It is associated with a number of clinical conditions, including abdominal aortic aneurysm, surgery, cardiopulmonary bypass, strangulated hernias, intestinal obstruction, acute mesenteric ischemia, necrotizing colitis, collapse of systemic circulation (hypovolemic and septic shock), and intestinal transplantation11 Grootjans J, Lenaerts K, Buurman WA, Dejong CHC, Derikx JPM. Life and death at the mucosal-luminal interface: new perspectives on human intestinal ischemia-reperfusion. World J Gastroenterol. 2016;22(9):2760-70. doi: 10.3748/wjg.v22.i9.2760.
https://doi.org/10.3748/wjg.v22.i9.2760...
2 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
-33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
.
Interruption of blood supply to the intestine could result in tissue damage. However, the subsequent blood flow restoration may lead to additional injury, referred to as reperfusion injury11 Grootjans J, Lenaerts K, Buurman WA, Dejong CHC, Derikx JPM. Life and death at the mucosal-luminal interface: new perspectives on human intestinal ischemia-reperfusion. World J Gastroenterol. 2016;22(9):2760-70. doi: 10.3748/wjg.v22.i9.2760.
https://doi.org/10.3748/wjg.v22.i9.2760...
2 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
3 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
4 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
-55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
.
Although intestinal IR induced inflammation has been studied extensively in animal models and in humans, the induced inflammatory responses have yet to be characterized11 Grootjans J, Lenaerts K, Buurman WA, Dejong CHC, Derikx JPM. Life and death at the mucosal-luminal interface: new perspectives on human intestinal ischemia-reperfusion. World J Gastroenterol. 2016;22(9):2760-70. doi: 10.3748/wjg.v22.i9.2760.
https://doi.org/10.3748/wjg.v22.i9.2760...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
5 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
6 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
7 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
8 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
-99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
. There is abundant information on the compromising effects of IR on many processes in the intestine, and various protective treatment modalities have successfully been applied to mitigate reperfusion injury in animal models of reperfusion injury of the intestine, such as pharmacologic treatment, chemical agents, and ischemic preconditioning33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
7 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
8 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
9 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
10 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
11 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
-1212 Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
https://doi.org/10.1016/j.transproceed.2...
.
Among the available strategies is ischemic preconditioning; since its introduction more than thirty years ago in cardiac surgery99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1010 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
, ischemic preconditioning has demonstrated favorable effects in numerous other clinical situations or with experimental animals in different organs and tissues or in shock by retarding cell death33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
10 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
11 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
12 Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
https://doi.org/10.1016/j.transproceed.2...
13 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
14 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
15 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
-1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
. Ischemic preconditioning can be mechanical or pharmacological33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
. Direct mechanical preconditioning in which the target organ is exposed to brief ischemia prior to prolonged ischemia has the benefit of reducing ischemia-reperfusion injury33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
10 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
11 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
12 Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
https://doi.org/10.1016/j.transproceed.2...
13 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
14 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
15 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
-1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
.
Several reports have described the effects of IPC on the reduction of apoptosis of epithelial cells33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
, structural protection33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1212 Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
https://doi.org/10.1016/j.transproceed.2...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
, preservation of intestinal contractility1212 Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
https://doi.org/10.1016/j.transproceed.2...
, decreased expression of the transcription factor nuclear-kB (NF-kB), cell adhesion molecules and pro-inflammatory cytokines33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
, diminution of the inflammatory infiltrate33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
14 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
15 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
-1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
, and increased synthesis of nitric oxide (NO)1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
. NO synthesis derived from IPC has been associated with tissue protection66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
. However, the mechanisms by which IPC protects the intestine following IR are still not fully understood.
In addition to IPC, the use of drugs such as pentoxifylline (PTX), a methylxanthine derivative of theobromine, has been proposed to decrease the deleterious effects of IR33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
22 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
-2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.. Protective effects of PTX include the anti-inflammatory actions associated with inhibition of NF-kB activity1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
-2222 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
, down-regulation of interleukin (IL)-6, tumor necrosis factor (TNF)-alpha synthesis, and decreased apoptosis88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
22 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
-2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785. and inhibition of NO production by inflammatory signaling1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
22 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
-2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.. The activity of unspecific phosphodiesterase, an antioxidant, increases intracellular cyclic adenosine monophosphate (cAMP) concentrations, and it has been previously used to treat peripheral vascular disease and intermittent claudication1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
,1818 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
,2121 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
. However, the pharmacological properties of PTX are not fully understood.
Clinical biomarkers are used to diagnose the conditions of the development or inhibition of apoptosis and the deleterious effects of ischemia and reperfusion. BCL-2 (B-cell lymphoma 2), is a protein encoded in humans by the Bcl-2 gene and is specifically considered an important anti-apoptotic protein. Localized in the outer membrane of mitochondria, it plays a crucial role in promoting cellular survival and inhibiting the actions of pro-apoptotic proteins1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785..
On the other hand, caspase is a cysteine protease, and cleaved caspase-3, the active form of caspase-3, is well known as a marker for cells undergoing apoptosis; it is now widely accepted that cleaved caspase-3 staining is useful in scoring the apoptotic index1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.,2424 Gown AM, Willingham MC. Improved detection of apoptotic cells in archival paraffin sections: immunohistochemistry using antibodies to cleaved caspase 3. Histochem Cytochem. 2002;50:449-54. doi: 10.1177/002215540 20500 0401.
https://doi.org/10.1177/002215540...
. Studies in direct IPC have demonstrated the protective effect of NO on microcirculation22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1010 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
. NO is a free radical produced from L-arginine by the enzyme NO synthase, which has three forms: endogenous NOS (NOS-3), neuronal NOS (NOS-1), and inducible NOS (NOS-2)33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
. Of these, NOS-3 and NOS-1 are constitutively expressed, while NOS-2 is produced in response to cytokines and NF-kB. NOS-3 has a protective effect on the microcirculation and always produces NO in small amounts, which predominate in the circulation, producing a protective effect on the microcirculation that is of vital significance to the blood flow to the organs55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
10 Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
https://doi.org/10.1161/01.CIR.74.5.1124...
-1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
,1818 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
.
Histological evaluation in our previous publication showed a favorable effect in the IPC-IR-SS and IPC-IR-PTX groups, which is in accordance with the hypothesis of a beneficial effect of IPC alone or PTX; however, the simultaneous action of both did not provide better protection2626 Oliveira TRR, Oliveira GF, Simões RS, Tikazawa EH, Monteiro HP, Fagundes DJ, Taha MO. The role of ischemic preconditioning and pentoxifylline in intestinal ischemia/reperfusion injury of rats. Acta Cir Bras. 2017;32(7):559-67. doi: 10.1590/s0102-865020170070000007.
https://doi.org/10.1590/s0102-8650201700...
. Based on the common assumption that IR injury can go through distinct biomolecular pathways, we have proposed to study: (1) the immunochemistry expression of active caspase-3 and BCL-2 under the simultaneous action of IPC and PTX, which separately have a favorable effect on intestinal IR damages; and (2) the participation of NOS-2 and NOS-3 in the protective effects of IPC and PTX submitted to intestinal IR in rats, with the mRNA expression levels determined from epithelial cells.
Methods
All animal experiments were carried out in accordance with the recommendations of international legislation on animal protection. The Ethics Committee in Research of the Universidade Federal de São Paulo (UNIFESP) approved the study.
Thirty male Wistar-EPM-1 rats (250-300g) were purchased from the Center for the Development of Experimental Models for Medicine and Biology (CEDEME, Sao Paulo-SP, Brazil). The animals were housed under controlled temperature (23℃), 50% air humidity and light conditions with a 12-hour light/dark cycle. The rats had free access to water and standard pellet chow until 6 hours before the surgical procedures at the Experimental Surgery Department of UNIFESP. The individual cages had dimensions of 40 cm × 30 cm × 25 cm and, which were covered with previously autoclaved wood shavings.
All procedures involving anaesthesia and euthanasia were performed according to followed the guidelines of the National Council Animal Experimentation Control (SBCAL/COBEA - Brazil http://www.cobea.org.br/).
The surgical procedures were performed under intramuscular anesthesia with 80 mg/kg of ketamine (Dopalen® 80 mg/kg, Sespo Indústria Comércio, Paulínea-SP) and 10 mg/kg of xylazine (Dopaser® 5 mg/kg, Herpate Callieri, Juatuba-MG). The femoral vein was dissected and catheterized for drug infusion of 80 mg/kg pentoxifylline (Sigma Aldrich, São Paulo-SP) or 0.9% saline solution in the control group 5 minutes before ischemia. A median abdominal incision was made to identify the superior mesenteric vessels and clamping was done according to the group. The animals were randomly assigned into five groups:
-
CG - Control group: anesthesia + laparotomy + no clamping of the superior mesenteric artery (timing elapsed 90 minutes).
-
IR-SS group: saline + 30 minutes of ischemia + 60 minutes of reperfusion.
-
IR-PTX group: pentoxifylline + 30 minutes of ischemia + 60 minutes of reperfusion.
-
IPC-IR-SS group: five minutes of ischemia + five minutes of reperfusion (preconditioning) + saline + 30 minutes of ischemia + 60 minutes of reperfusion.
-
IPC-IR-PTX group, five minutes of ischemia + five minutes of reperfusion (preconditioning) + pentoxifylline + 30 minutes of ischemia + 60 minutes of reperfusion.
At the end of reperfusion, two samples of jejunal segments (3 cm long) were opened longitudinally, gently washed with saline solution, and one of them was wrapped in aluminum foil and frozen in liquid nitrogen. The other one was fixed in 10% formaldehyde phosphate buffered solution.
Immunohistochemistry (IHC) analysis
IHC is a commonly used technique to monitor protein expression and localization in the context of tissue morphology. Tissue samples were fixed in 10% neutral buffered formaldehyde, embedded in paraffin wax, cut into thin slices (3 µm) and mounted onto silanized slides. The sample was deparaffinized and hydrated. Assay procedures were performed according to the manufacturer’s instructions. The epitopes were blocked with peroxidase and incubated with a specific first antibody. The primary antibody to verify cell apoptosis was assessed using an anti-active caspase-3 antibody, diluted to 1:200 (rabbit heterologous polyclonal antibody ABCAM-AB2302, São Paulo-SP). The anti-apoptotic analysis was done using a primary antibody anti-BCL-2, diluted 1:100 (monoclonal mouse antibody, DAKO, M00884, São Paulo-SP). The sections were washed with PBS solution and incubated with biotinylated secondary antibody, avidin-chain enzyme, stained with DAB, and lastly with hematoxylin. The sections were dehydrated conventionally with ethanol, cleared with xylene, and mounted with synthetic resin. A scanner (3D HISTECH®, Biogen, São Paulo-SP) captured all images of the immunohistochemistry slides. The images were analyzed by specific software Case Viewer® (3D HISTECH®, Biogen, São Paulo-SP; magnification x20, x40, x50). A perimeter of 1.000 microns was determined on each slide. Within the perimeter, ten villi were randomly selected, and 100 epithelial cells were counted in 10 villi per slice for 5 treatment groups (each group, N=6), determining the score of immunohistochemically stained cells 60 villi or 10 villi per slice in 6 groups for each treatment. The data were expressed as mean ± standard error of the mean (x ± sem). The difference between the groups was processed by repeated measures analysis of variance and the Bonferroni post hoc comparison test. The results were processed by statistical software Stat Plus the Analyst Soft Inc® (version 9.4).
The analysis of mRNA NOS-2 and NOS-3 expression by qRT-PCR
After the storage period, all the tissue samples were prepared for qRT-PCR (LifeTechnologies™, Grand Island, NY, USA) for NOS-2(iNOS) and NOS-3(eNOS). Initially, for total RNA extraction, the tissue segment was lysed in Trizol (Trizol, Invitrogen™) and homogenized into the extractor (Polytron 3000). Purelink™ RNA Mini Kit and treatment Pure Link DNAse™ were used to achieve purification of the total RNA. The samples were subjected to electrophoresis on 1% agarose gel and reading by Spectrophotometer (NanodroP®) to assess the purity of the samples and their RNA concentrations (µg/mL), respectively. In the reverse transcription of total RNA, for the synthesis of the primary band of complementary DNA or cDNA, SuperScript™ Vilo™ Master Mix (Life Technologies™ - USA) and Reverse Transcriptase were used, and the sample was submitted to a GeneAmp 2700 thermocycler (Perkin-Elmer, Inc., Waltham, MA, USA) at 25°C temperature for 10 minutes, 42°C for 60 minutes and at 85°C for 5 minutes. The qRT-PCR reaction occurred from a sample-diluted cDNA and was amplified in 20 µL reaction volume distributed also in the wells. The final reaction mixture included the following: primers, 10 pmol; MgCl2, 2.5 mM; Deoxyribonucleosides triphosphate (dNTP) mixture (included in the TaqMan Gene Expression Master Mix kit, Life Technology™- São Paulo, SP, Brazil), 1 mM; 10 × RNA PCR buffer, 1x; 1x ExTaq™ DNA polymerase; and 0.5 U/ reaction of AmpliTaq Gold™ DNA Polymerase (included in the TaqMan Gene Expression Master Mix, Life Technology, São Paulo-SP). The PCR was performed with an initial denaturation at 95°C for 10 minutes, 40 cycles at 95°C for 15 seconds and 60°C for one minute. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH; TaqMan™ Endogenous Control, Invitrogen™ Rn01775763_g1) was used as a “housekeeping gene” (internal control gene) to indicate the amount of cDNA in the sample reference control and the experimental sample in PCR. The target genes were NOS-2 (iNOS) (TaqMan gene expression assay, Invitrogen/ Rn00561646_m1) and NOS-3 (eNOS) (TaqMan gene expression assay, Invitrogen/Rn02132634_s1). The samples were performed in triplicate. The products were amplified by Step One Plus vs 2.3 software (Applied Biosystems™) to obtain the reading of the amount of amplified cDNA during the reaction. To calculate the mean value of the cycle thresholds (Ct) relative on the reference gene, the value was subtracted from the mean value of the target gene Ct. The ∆∆Ct calculation was converted into a linear expression from the logarithmic exponential function (log 2)2727 Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-??ct method. Methods. 2001;25(4):402-8. doi: 10.1006/meth.2001.1262.
https://doi.org/10.1006/meth.2001.1262...
. Afterward, the results were expressed as the mean, standard error, 95% confidence interval, and an analysis of variance (ANOVA) and the Bonferroni post hoc test were performed to evaluate significant differences between the means of the experimental groups. The level of rejection of the null hypothesis was set at 5% (p ≤ 0.05). All calculations were performed using Stat software version 9.4®.
Results
Immunohistochemistry analysis with anti-active caspase-3 and anti BCL-2
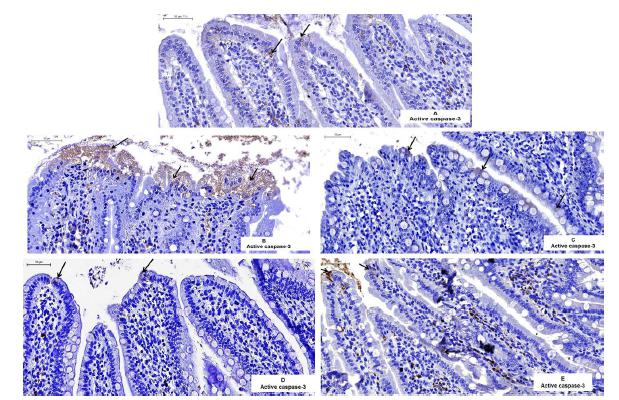
Analysis of the cell immunoreactivity (Figures 1 A-E) shows the photomicrographs of tissues incubated with activate caspase-3 antibody. The cells of the injured villi were measured, but the phagocytes were not measured. The average of the immunoreactive cells differed between the treatments (F= 47.2, p<0.05). The average scores in the immunoreactive cells in the IR-SS group (64.1 ± 3.02; 95% CI 58.1-70.1) were significantly higher than those in the CG (3.37 ± 0.403; 95% CI 2.56 - 4.17), IR-PTX (2.92 ± 0.488; 95% CI 1.94-3.89), IPC-IR-SS (0.72 ± 0.128; 95% CI 0.46 - 0.973), and IPC-IR-PTX (2.82 ± 0.373; 95% CI 2.07-3.56) groups. There was no significant difference among treatments for the other comparisons (Figure 2).
Photomicrography the histological sections of intestines of rats subjected to active caspase-3 immunostaining and counterstained with hematoxylin. Note the higher immunoreactivity (arrows) in the intestinal villi of the IR-SS group (B). Few active caspase-3-positive cells (arrows) can also be noticed in the CG (A), IR-PTX (C), IPC-IR-SS (D) and IPC-IR-IPTX (E) groups. Scale bare: 50 µm.
The expression of active caspase-3 was analyzed, and the difference was determined between groups for the model of repeated measures analysis of variance. (*) The difference was analyzed by pairs; statistically significant values are represented in the chart.
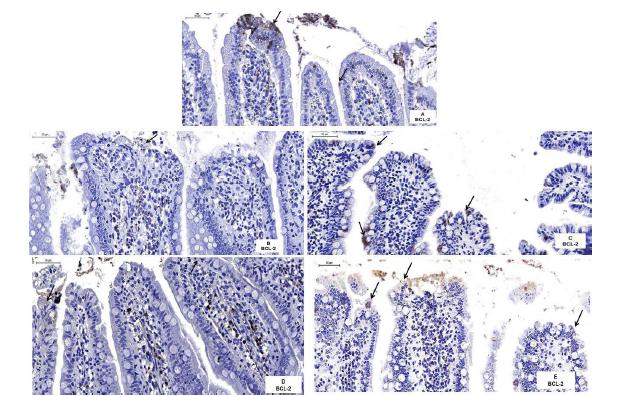
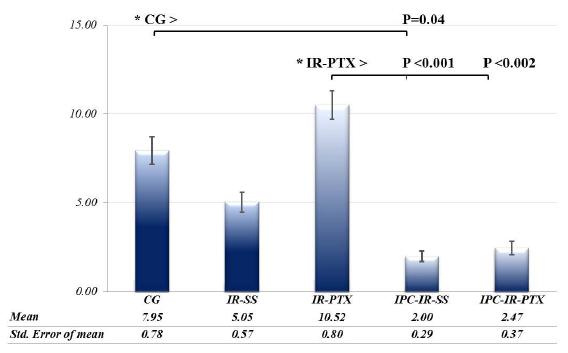
Figures 3A-E show the tissues of rats intestine incubated with BCL-2 antibody. The epithelial stain cells were measured, and the average analysis of the immunoreactive cells differed between the treatments (F = 4.96; P = 0.0015). The immunoreactive cells in the IR-PTX group (10.5 ± 0.795; 95% CI 0.832 - 1.1) were significantly higher than those in the IR-SS (5.05 ± 0.569; 95% CI 1.35 - 1.61), IPC-IR-SS (2.47 ± 0.372; 95% CI 0.828 - 1.04), and IPC-IR-PTX (2 ± 0.29; 95% CI 0.736-0.93) groups, whereas the averages in the IPC-IR-SS group were significantly lower than those in the CG group (7.95 ± 0.78; 95% CI 0.931-1.24). There was no significant difference between treatments for the other comparisons (Figure 4).
The photomicrographs of histological sections of rats intestines subjected to BCL-2 immunostaining and counterstained with hematoxylin. Note the higher presence of immunostained epithelial cells (arrows) in the intestinal villi of the CG (A) and IR-PTX (C) groups. Few immunostained epithelial cells (arrows) can also be observed in the IR-SS (B), IPC-IR-SS (D) and IPC-IR-IPTX (E) groups. Scale bare: 50 µm.
The expression of BCL-2 was analyzed, and the difference was determined between groups for the model of repeated measures analysis of variance. (*) Differences were analyzed by pairs. Statistically significant values are represented in the graphic.
Evaluated mRNA expression of NOS-2 and NOS-3 by qRT-PCR
The average analysis of expression of NOS-2 differed between the treatments (ANOVA, F= 8.11, P=0.0002). The IR-SS group exhibited a significant increase in NOS-2 expression (6.01± 1.34; 95% CI: 2.56 - 9.47). The NOS-2 level in the control group (1.38±0.52; 95% CI: 0.04 - 2.72) was lower than the level in the IR-SS group. The IR-PTX (2.27±1.28; 95% CI: -1.02 - 5.57) and IPC-IR-SS (0.23±0.10; 95% CI: 0.006 - 0.13) groups significantly reduced NOS-2 expression in comparison to the IR-SS group. The combination of procedures in the IPC-IR-PTX group (0.04±0.01; 95% CI: 0.006 - 0.08) drastically reduced NOS-2 expression (Figure 5).
Data from mRNA expression of NOS-2 by qRT-PCR. Differences among groups were analyzed by ANOVA followed by the Bonferroni test. (*) The differences were analyzed by pairs. Statistically significant values are represented in the graphic.
Our results showed that a significant difference between analysis of average of NOS-3 expressions in the treatments (ANOVA, F= 5.47, P=0.0027). The NOS-3 expression levels were also increased in the IPC-IR-SS group (1.61±0.11; 95% CI 1.32 - 1.89), IR-SS (1.24±0.16; 95% CI 0.83-1.65) and the IR-PTX group (1.29±0.08; 95% CI 1.08 - 1.49), but these levels were not significantly higher than the levels observed in the control group (1.10±0.20; 95% CI: 0.58 - 1.61). However, NOS-3 expression was significantly increased in the IPC-IR-PTX group (1.95±0.15; 95% CI: 1.55 - 2.35) compared with the groups CG, IR-SS, IR-PTX (Figure 6).
Data from mRNA expression of NOS-3 by qRT-PCR. Differences among groups were analyzed by ANOVA followed by the Bonferroni test. (*) The differences were analyzed by pairs. Statistically significant values are represented in the graphic.
Discussion
With the intent of studying the effects of intestinal ischemia, we have used an animal model in rats that have been subjected to 30 minutes of intestinal ischemia and 60 minutes of reperfusion (IR). We have investigated the effects of pentoxifylline (PTX - 80 mg/kg) applied (IV) before the onset of ischemia and/or ischemic preconditioning (IPC).
This study demonstrates that PTX (80 mg/kg) or IPC can protect against intestinal apoptosis in rats. The immunoreactivity is minimal to active caspase-3 and present to BCL-2 in the mechanic and pharmacology preconditioning. However, the combination of both did not show an adjuvant effect. In a study of pro-apoptotic genes, the caspase-3 expression was higher in tissues subjected to IR2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785. and lower in tissues subjected to IPC-IR1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
. It has been shown that the use of pretreatment with pentoxifylline attuned the expression of active caspase-32323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785. and decreased caspase-3/9 and the c-Jun N-terminal kinase (JNK) signaling pathway88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
.
In intestinal IR, we observed a significant difference in the BCL-2 expression with treatment of IR-PTX compared with the CG, IR and IPC groups. Furthermore, if pentoxifylline could suppress caspase-3/9 and JNK88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
, this could be interpreted as a significant difference between the anti-apoptotic agents (BCL-2) in this group. In addition, in IPC in the experimental study in the intestine, it could be suggested that the lower stimulation of the apoptotic protein caspase-366 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
would be able maintain the necessary level of BLC-233 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
. It provide protections: blocking oxidative damage to cell components and influencing cell membrane transport, altering the distribution of calcium, inhibiting release of cytochrome C from the mitochondria to the cytoplasm and protecting DNA fragmentation1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
.
The cell death pathways, such as necrosis, autophagy, the death receptor apoptotic pathway, post-ischemic inflammation and necroptosis, are operating simultaneously22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
. All these pathways could get activated at different times and in certain time gaps. The therapy for prevention of IR, for example, could depend on the mitochondrial apoptotic pathway alone or assessment at a particular time point, or the therapy could release other endogenous substances at the plasma membrane or the cytoplasmic pathways, for instance, anion channel, BCL-2 family proteins and JNK, among others molecular pathways22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
.
After IR in the current molecular study, a significant increase in the expression of NOS-2 was observed. The expression of nitric oxide synthase (NOS-2 and NOS-3) was evaluated, and the results showed that the mean NOS-2 expression in the ischemia and reperfusion group was higher than that for the control group; we also found that the PTX produced a significant reduction of NOS-2. On the other hand, the reduction promoted by IPC, alone or in association with PTX, was intense and almost neutralized the expression of the NOS-2. The NOS-2 enzyme is regulated at the transcriptional level, and its activation is independent of the intracellular calcium concentration22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
. Experimental research related the anti-inflammatory effect and involvement in the control of NOS-288 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
and NOS-32828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
for the action of PTX1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
22 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
-2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785. and IPC 33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
15 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
-1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,3030 Bjornsson B, Winbladh A, Bojmar L, Sundqvist T, Gullstrand P, Sandstrom P. Conventional, but not remote ischemic preconditioning, reduces iNOS transcription in liver ischemia/reperfusion. World J Gastroenterol. 2014;20:9506-12. doi: 10.3748/wjg.v20.i28.9506.
https://doi.org/10.3748/wjg.v20.i28.9506...
. High NO levels associated with inflammatory conditions and activation of NOS-2 have been related to tissue injury33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
.
The synthesis of NO by NOS-2 and NOS-3 depends on the induction mechanism, half-life, and the period of stimulation2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. NO in such conditions may promote tissue damage or beneficial effects22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. Analyses of the expression levels of 84 genes encoding proteins that are associated with oxidative stress during 120 minutes of IR in mouse intestine indicated 47 genes, including NOS-2, showed increased expression2929 Bertoletto PR, Ikejiri AT, Somaio Neto F, Chaves JC, Teruya R, Bertoletto ER, Taha MO, Fagundes DJ. Oxidative stress gene expression profile in inbred mouse after ischemia/reperfusion small bowel injury. Acta Cir Bras. 2012;27:773-82. doi: 10.1590/S0102-86502012001100006.
https://doi.org/10.1590/S0102-8650201200...
. Previously, mRNA expression levels of NOS-2 and NOS-3 were evaluated during IR of the superior mesenteric artery (40 minutes of ischemia and 300 minutes of reperfusion) in the rat intestine. The observed effects included increased NO synthesis, decreased mesenteric vascular response, and increased production of the toxic oxidizing species peroxynitrite (ONOO)2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
. In IR, ROS such as O2- are produced concomitantly with NOS-2-derived NO. Both reactive species efficiently react, generating ONOO-, which initiates lipid peroxidation22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
3 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
4 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
5 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
-66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
,1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,2929 Bertoletto PR, Ikejiri AT, Somaio Neto F, Chaves JC, Teruya R, Bertoletto ER, Taha MO, Fagundes DJ. Oxidative stress gene expression profile in inbred mouse after ischemia/reperfusion small bowel injury. Acta Cir Bras. 2012;27:773-82. doi: 10.1590/S0102-86502012001100006.
https://doi.org/10.1590/S0102-8650201200...
. In the other direction, NO binds thiol groups of cysteine of proteins resulting in S- nitrosylation, an important cell signaling pathways99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
.
NO expression is also affected by ischemic preconditioning1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
, by reduction in the generation of pro-inflammatory cytokines and the preservation of blood flow, oxygenation, and mitochondrial function22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
3 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
4 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
-55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
. Furthermore, in the intestine, IPC protection has been associated with inhibition of NOS-2 expression levels44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,3030 Bjornsson B, Winbladh A, Bojmar L, Sundqvist T, Gullstrand P, Sandstrom P. Conventional, but not remote ischemic preconditioning, reduces iNOS transcription in liver ischemia/reperfusion. World J Gastroenterol. 2014;20:9506-12. doi: 10.3748/wjg.v20.i28.9506.
https://doi.org/10.3748/wjg.v20.i28.9506...
. A study in rat intestines indicated that IPC reduced the expression of leukocyte adhesion proteins, endothelial cell adhesion proteins, and the NF-κB-dependent expression of TNF-α1515 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
,1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
. Data from animal model have shown that IPC reduced NO levels in serum and lung after intestinal IR66 Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
https://doi.org/10.1007/s10753-014-0047-...
. An investigation on the transcriptional activation of NOS-2 during hepatic hypoxia via intestinal IR found that NFκB activates the transcription of multiple inflammatory genes, including TNFα and IL1β. The research suggested that IPC may inhibit NF-κB, p65 activation and decrease liver injury and the infiltration of inflammatory cells1313 Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
https://doi.org/10.3892/mmr.2016.4817...
. Several studies described the inhibition of NOS-2 expression levels by IPC in heart tissue submitted to IR, and in these conditions, IPC has been associated with cardio protection99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
. These findings, as well as those of the present study, suggest that NOS2 levels decreased in IPC.
NOS-2 mRNA expression levels by the PTX group downregulated TNF-alpha88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
, derived inhibitory effects on NF-kB activity88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,1818 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
-2020 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
,2222 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
,2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.. PTX is described to diminished interleukin-1 and interleukin-61717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
,2020 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
,2121 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
,2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785., increase intracellular levels of cAMP, and exerts its effects through both protein kinase A (PKA) mechanisms1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
-1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
. Moreover, cAMP and PKA activation have been shown to inhibit signaling via the NFk-B pathway and decrease TNF1818 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
,1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
and pro-inflammatory cytokines2121 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
.Thus, our hypothesis confirms the beneficial effects listed in these previous findings.
In this study, the NOS-3 expression was significantly higher in the IPC-IR-PTX group than in the IR-PTX, IR-SS and CG groups. In a study of intestines submitted to IR, an increase in NO and a reduction in intestinal smooth muscle contraction and reactivity to vasoconstrictors was detected2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
. As stated previously, IPC reduced the generation of pro-inflammatory cytokines, preserving blood flow, oxygenation, and mitochondrial function22 Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
https://doi.org/10.1038/nm.2507...
3 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
4 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
-55 Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
https://doi.org/10.1016/j.redox.2015.08....
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
, and it has been associated with inhibition of NOS-2 expression levels44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,3030 Bjornsson B, Winbladh A, Bojmar L, Sundqvist T, Gullstrand P, Sandstrom P. Conventional, but not remote ischemic preconditioning, reduces iNOS transcription in liver ischemia/reperfusion. World J Gastroenterol. 2014;20:9506-12. doi: 10.3748/wjg.v20.i28.9506.
https://doi.org/10.3748/wjg.v20.i28.9506...
. Preconditioning is triggered by an initial increase in nitric oxide synthesis1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
.
The NOS-3 exerts a function as a vasodilator and an anti-inflammatory mediator2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. The NOS-3 phosphorylation is regulated up and down and depends on the phosphorylation or dephosphorylation domains at each of the respective sites, the protein-protein interactions, and the bioavailability of cofactors; these can occur simultaneously, with all or combinations of these proceeding to activation. For instance: three domains that contain phosphorylation sites could be inhibiting or stimulating NOS-3; oxygenase, Calmodulin (CaM) and reductase domains. These domains modulate NOS-3 activity and specific stimuli influence multiple pathways such as: AMPK phosphorylates the vasodilator bradykinin and the shear stress, resulting in decreasing NO or increasing NO production, according to the specific activated domain2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
.
Another pathway involving caveolins is associated with the PI3K/Akt signaling pathway2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. The activity of NOS-3 is regulated by plasma membrane invaginations known as caveolae, which contain G protein-coupled receptors, heterotrimeric G proteins, tyrosine kinase type receptors, mitogen-activated protein (MAP) kinase pathway proteins, Src tyrosine kinases, and NOS399 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. The activation of NOS3 also depends on the formation of the Ca++/calmodulin complex using calcium from the endoplasmic reticulum or the extracellular milieu2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. Nitric oxide elevation could be from diffusion into the adjacent cell on the smooth muscle of the intestinal vessels and exerting control over the vascular tone2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. NO inhibits the adhesion of platelets and leukocytes to endothelial cells and decreases platelet aggregation; it also participates in the control of the release of a plasminogen-activating factor and its inhibitor, PAI-1, thereby improving hemodynamics1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
.
The importance of higher concentrations of pentoxifylline has been highlighted. The PTX, as a non-selective phosphodiesterase inhibitor, was associated with increases in cAMP levels1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
-1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
and an amplified signaling pathway1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
-1919 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
. One of the PI3K/Akt signaling pathways that actives the cyclic guanosine monophosphate (cGMP), when in smooth muscle cells constituent of endothelium vessels, raises resistance vessels, which include NOS-3 expression activated via sensitivity to shear stress or other stimuli2828 Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
https://doi.org/10.1007/s00018-015-2021-...
. The effects that increase vasodilation, as well a consequence of decreases in leukocyte adhesion, increase erythrocyte flexibility17,28 and act in the signaling pathways involved in the synthesis of NF-κB expression88 Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
https://doi.org/10.3748/wjg.v19.i30.4925...
,1818 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
-2020 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
,2222 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
,2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785..
The lack of consensus on research protocols related to the best IR duration limits the comparison of our results with those of other studies.
Future studies of IPC and pentoxifylline may focus on other pathways besides the apoptosis inhibition pathways, such as molecular pathways associated with cGMP and cAMP in IR with the ambivalent effect of NO on these pathways. Besides that NF-κB modulation was including the phosphatidylinositol 3 kinase (PI3K/Akt) pathway.
The results of previous studies and this study indicate that pentoxifylline1717 McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
https://doi.org/10.1136/openhrt-2015-000...
18 Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
https://doi.org/10.1097/SHK.0b013e3181cd...
19 Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
https://doi.org/10.1097/TA.0b013e318191b...
20 Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
https://doi.org/10.1016/j.ijsu.2014.04.0...
21 Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
https://doi.org/10.6002/ect.2012.0222...
22 Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
https://doi.org/10.1590/S0102-8650201400...
-2323 Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.,2626 Oliveira TRR, Oliveira GF, Simões RS, Tikazawa EH, Monteiro HP, Fagundes DJ, Taha MO. The role of ischemic preconditioning and pentoxifylline in intestinal ischemia/reperfusion injury of rats. Acta Cir Bras. 2017;32(7):559-67. doi: 10.1590/s0102-865020170070000007.
https://doi.org/10.1590/s0102-8650201700...
and IPC33 Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
https://doi.org/10.1023/B:DDAS.000004223...
,44 Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
https://doi.org/10.3748/wjg.v20.i13.3572...
,77 Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
https://doi.org/10.1016/j.transproceed.2...
,99 Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
https://doi.org/10.1161/CIRCRESAHA.116.3...
,1111 Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
https://doi.org/10.1006/bbrc.1996.0692...
,1414 Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
https://doi.org/10.1097/00000658-2001070...
15 Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
https://doi.org/10.1016/j.transproceed...
-1616 Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
https://doi.org/10.3748/wjg.v21.i26.8081...
,2525 Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
https://doi.org/10.1016/j.transproceed.2...
,2626 Oliveira TRR, Oliveira GF, Simões RS, Tikazawa EH, Monteiro HP, Fagundes DJ, Taha MO. The role of ischemic preconditioning and pentoxifylline in intestinal ischemia/reperfusion injury of rats. Acta Cir Bras. 2017;32(7):559-67. doi: 10.1590/s0102-865020170070000007.
https://doi.org/10.1590/s0102-8650201700...
,3030 Bjornsson B, Winbladh A, Bojmar L, Sundqvist T, Gullstrand P, Sandstrom P. Conventional, but not remote ischemic preconditioning, reduces iNOS transcription in liver ischemia/reperfusion. World J Gastroenterol. 2014;20:9506-12. doi: 10.3748/wjg.v20.i28.9506.
https://doi.org/10.3748/wjg.v20.i28.9506...
have robust protective effects in the intestine of rats subjected to IR. For this reason, pentoxifylline has anti-inflammatory activity, increases vascular resistance and improves erythrocyte flexibility; this drug can be used in some clinical conditions, including during an acute deficiency in the blood supply and for the attenuation of the adverse effects of organ and tissue transplantation.
The data on the integrity of the rat jejunum mucosa2626 Oliveira TRR, Oliveira GF, Simões RS, Tikazawa EH, Monteiro HP, Fagundes DJ, Taha MO. The role of ischemic preconditioning and pentoxifylline in intestinal ischemia/reperfusion injury of rats. Acta Cir Bras. 2017;32(7):559-67. doi: 10.1590/s0102-865020170070000007.
https://doi.org/10.1590/s0102-8650201700...
, control the apoptosis and modulation of NOS are relevant to future study of the signaling pathway control of nitric oxide and nitrosylation.
Conclusions
The IPC and pentoxifylline alone: decrease NOS-2 expression, a compensatory up-regulation of NOS-3 expression and control the apoptosis after intestinal IR injury. When viewed the IPC-PTX and IPC groups, no synergism effects was observed.
Acknowledgement
To Prof. Manuel de Jesus Simões, Chairman of Division of Histology and Structural Biology, Department of Morphology and Genetic, UNIFESP.
References
-
1Grootjans J, Lenaerts K, Buurman WA, Dejong CHC, Derikx JPM. Life and death at the mucosal-luminal interface: new perspectives on human intestinal ischemia-reperfusion. World J Gastroenterol. 2016;22(9):2760-70. doi: 10.3748/wjg.v22.i9.2760.
» https://doi.org/10.3748/wjg.v22.i9.2760 -
2Eltzschig HK, Eckle T. Ischemia and reperfusion-from mechanism to translation. Nat Med. 2011;17(11):1391-401. doi: 10.1038/nm.2507.
» https://doi.org/10.1038/nm.2507 -
3Mallick IH, Yang W, Winslet MC, Seifalian AM. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49(9):1359-77. doi: 10.1023/B:DDAS.0000042232.98927.91.
» https://doi.org/10.1023/B:DDAS.0000042232.98927.91 -
4Camara-Lemarroy CR. Remote ischemic preconditioning as treatment for non-ischemic gastrointestinal disorders: Beyond ischemia-reperfusion injury. World J Gastroenterol. 2014;20:3572-81. doi: 10.3748/wjg.v20.i13.3572.
» https://doi.org/10.3748/wjg.v20.i13.3572 -
5Granger DN, Kvietys PR. Reperfusion injury and reactive oxygen species: the evolution of a concept. Redox Biol. 2015;6:524-51. doi: 10.1016/j.redox.2015.08.020.
» https://doi.org/10.1016/j.redox.2015.08.020 -
6Wang Z, Ji Y, Wang S, Wang R, Li Z, Kang A, Xu H, Shi M, Zhao M. Protective effect of intestinal ischemic preconditioning on ischemia reperfusion-caused lung injury in rats. Inflammation. 2015;38(1):424-32. doi: 10.1007/s10753-014-0047-3.
» https://doi.org/10.1007/s10753-014-0047-3 -
7Camprodon RA, Bowles MJ, Pockley AG, de Oca J. Anti-inflammatory effects of ischemic preconditioning on rat small bowel allografts. Transplant Proc. 2014;46(6):2146-9. doi: 10.1016/j.transproceed.2014.06.038.
» https://doi.org/10.1016/j.transproceed.2014.06.038 -
8Yang Q, Zheng FP, Zhan YS, Tao J, Tan SW, Liu HL, Wu B. Tumor necrosis factor-alpha mediates JNK activation response to intestinal ischemia-reperfusion injury. World J Gastroenterol. 2013;19:4925-34. doi: 10.3748/wjg.v19.i30.4925.
» https://doi.org/10.3748/wjg.v19.i30.4925 -
9Heusch G. Molecular basis of cardioprotection signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res. 2015;116:674-99. doi: 10.1161/CIRCRESAHA.116.305348.
» https://doi.org/10.1161/CIRCRESAHA.116.305348 -
10Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124-36. doi: 10.1161/01.CIR.74.5.1124.
» https://doi.org/10.1161/01.CIR.74.5.1124 -
11Hotter G, Closa D, Prados M, Fernandez-Cruz L, Prats N, Gelpi E, Rosello-Catafau J. Intestinal preconditioning is mediated by a transient increase in nitric oxide. Biochem Biophys Res Commun. 1996;222 (1):27-32. doi: 10.1006/bbrc.1996.0692.
» https://doi.org/10.1006/bbrc.1996.0692 -
12Taha MO, Miranda-Ferreira R, Chang AC, Rodrigues AM, Fonseca IS, Toral LB, Cardoso MR, Simões MJ, Oliveira-Junior IS, Monteiro HP, Fagundes DJ, Taha NS, Caricati-Neto A. Effect of ischemic preconditioning on injuries caused by ischemia and reperfusion in rat intestine. Transplant Proc. 2012;44:2304-08. doi: 10.1016/j.transproceed.2012.07.056.
» https://doi.org/10.1016/j.transproceed.2012.07.056 -
13Xue TM, Tao LD, Zhang J, Xue TM., Tao, LD., Zhang, J, Zhang, PJ, Liu, X., Chen, GF, ZHU, Y.J. Intestinal ischemic preconditioning reduces liver ischemia reperfusion injury in rats. Mol Med Rep. 2016;13(3):2511-7. doi: 10.3892/ mmr.2016.4817.
» https://doi.org/10.3892/mmr.2016.4817 -
14Sola A, De Oca J, González R, Prats N, Roselló-Catafau J, Gelpí E, Jaurrieta E, Hotter G. Protective effect of ischemic preconditioning on cold preservation and reperfusion injury associated with rat intestinal transplantation. Ann Surg. 2001;234:98-106. doi: 10.1097/00000658-200107000-00015.
» https://doi.org/10.1097/00000658-200107000-00015 -
15Takeshita M, Tani T, Harada S, Hayashi H, Itoh H, Tajima H, Ohnishi I, Takamura H, Fushida S, Kayahara M. Role of transcription factors in small intestinal ischemia-reperfusion injury and tolerance induced by ischemic preconditioning. Transplant Proc. 2010;42:3406-13. doi: 10.1016/j.transproceed. 2010.06.038.
» https://doi.org/10.1016/j.transproceed -
16Ji YY, Wang ZD, Wang SF, Wang BT, Yang ZA, Zhou XR, Lei NN, Yue WN. Ischemic preconditioning ameliorates intestinal injury induced by ischemia-reperfusion in rats. World J Gastroenterol. 2015;21:8081-8. doi: 10.3748/wjg.v21.i26.8081.
» https://doi.org/10.3748/wjg.v21.i26.8081 -
17McCarty MF, O'Keefe JH, DiNicolantonio JJ. Pentoxifylline for vascular health: a brief review of the literature. Open Heart. 2016;3:e000365. doi: 10.1136/openhrt-2015-000365.
» https://doi.org/10.1136/openhrt-2015-000365 -
18Kreth S, Ledderose C, Luchting B, Weis F, Thiel M. Immunomodulatory properties of pentoxifylline are mediated via adenosine-dependent pathways. Shock. 2010;34(1):10-6. doi: 10.1097/SHK.0b013e3181cdc3e2.
» https://doi.org/10.1097/SHK.0b013e3181cdc3e2 -
19Costantini TW, Loomis WH, Putnam JG, Kroll L, Eliceiri BP, Baird A, Bansal V, Coimbra R. Pentoxifylline modulates intestinal tight junction signaling after burn injury: Effects on myosin light chain kinase. J Trauma. 2009;66:17-24. doi: 10.1097/TA.0b013e318191bb1f.
» https://doi.org/10.1097/TA.0b013e318191bb1f -
20Rasslan R, Utiyama EM, Marques GM, Ferreira TC, da Costa VA, de Victo NC, Rasslan S, Montero EF. Inflammatory activity modulation by hypertonic saline and pentoxifylline in a rat model of strangulated closed loop small bowel obstruction. Int J Surg. 2014;12:594-600. doi: 10.1016/j.ijsu.2014.04.007.
» https://doi.org/10.1016/j.ijsu.2014.04.007 -
21Carsi JML, Lapena DC, Toledo AH, Fernandez CZ, Pereyra LHT. Pentoxifylline protects the small intestine after severe ischemia and reperfusion. Exp Clin Transplant. 2013;11:250-8. doi: 10.6002/ect.2012.0222.
» https://doi.org/10.6002/ect.2012.0222 -
22Marques GMN, Rasslan R, Belon AR, Carvalho JG, Felice Neto R, Rasslan S, Utiyama EM, Montero EFS. Pentoxifylline associated to hypertonic saline solution attenuates inflammatory process and apoptosis after intestinal ischemia/reperfusion in rats. Acta Cir Bras. 2014;29:735-41. doi: 10.1590/S0102-86502014001800007.
» https://doi.org/10.1590/S0102-86502014001800007 -
23Ji Q, Zhang L, Jia H, Xu J. Pentoxifylline inhibits endotoxin-induced NF-kappa B activation and associated production of proinflammatory cytokines. Ann Clin Lab Sci. 2004;34:427-36. PMID: 15648785.
-
24Gown AM, Willingham MC. Improved detection of apoptotic cells in archival paraffin sections: immunohistochemistry using antibodies to cleaved caspase 3. Histochem Cytochem. 2002;50:449-54. doi: 10.1177/002215540 20500 0401.
» https://doi.org/10.1177/002215540 -
25Chen CF, Leu FJ, Chen HI, Wang D, Chou SJ. Ischemia/reperfusion-induced low reactivity of the rat superior mesenteric vascular bed is associated with expression of nitric oxide synthases. Transplant Proc. 2006;38:2216-20. doi: 10.1016/j.transproceed.2006.07.022.
» https://doi.org/10.1016/j.transproceed.2006.07.022 -
26Oliveira TRR, Oliveira GF, Simões RS, Tikazawa EH, Monteiro HP, Fagundes DJ, Taha MO. The role of ischemic preconditioning and pentoxifylline in intestinal ischemia/reperfusion injury of rats. Acta Cir Bras. 2017;32(7):559-67. doi: 10.1590/s0102-865020170070000007.
» https://doi.org/10.1590/s0102-865020170070000007 -
27Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2-??ct method. Methods. 2001;25(4):402-8. doi: 10.1006/meth.2001.1262.
» https://doi.org/10.1006/meth.2001.1262 -
28Shu X, Keller TCt, Begandt D, Butcher JT, Biwer L, Keller AS, Columbus L, Isakson BE. Endothelial nitric oxide synthase in the microcirculation. Cell Mol Life Sci. 2015;72:4561-75. doi: 10.1007/s00018-015-2021-0.
» https://doi.org/10.1007/s00018-015-2021-0 -
29Bertoletto PR, Ikejiri AT, Somaio Neto F, Chaves JC, Teruya R, Bertoletto ER, Taha MO, Fagundes DJ. Oxidative stress gene expression profile in inbred mouse after ischemia/reperfusion small bowel injury. Acta Cir Bras. 2012;27:773-82. doi: 10.1590/S0102-86502012001100006.
» https://doi.org/10.1590/S0102-86502012001100006 -
30Bjornsson B, Winbladh A, Bojmar L, Sundqvist T, Gullstrand P, Sandstrom P. Conventional, but not remote ischemic preconditioning, reduces iNOS transcription in liver ischemia/reperfusion. World J Gastroenterol. 2014;20:9506-12. doi: 10.3748/wjg.v20.i28.9506.
» https://doi.org/10.3748/wjg.v20.i28.9506
-
Financial source:
Ministério da Educação do Brasil, UFGD
-
1
Research performed at Division of Surgical Techniques and Experimental Surgery, Department of Surgery and Therapy Center of Cellular and Molecular (CTCMol), Universidade Federal de São Paulo (UNIFESP), Brazil.
Publication Dates
-
Publication in this collection
Nov 2017
History
-
Received
02 July 2017 -
Reviewed
05 Sept 2017 -
Accepted
08 Oct 2017