Abstract
Purpose:
To compare the preventive effects of N-acetyl cysteine (NAC), ozone preconditioning and ozone treatment against contrast-induced nephropathy (CIN) in an experimental rat model.
Methods:
Thirty adult male Wistar rats were randomly distributed into five groups (n=6 for each group). Group I served as control and Group II had only contrast agent, while Group III received NAC and Group IV received intraperitoneal ozone 6 hours before and 6 hours after introduction of contrast agent. Ozone treatment was applied for 5 days after the contrast agent was introduced in Group V. After induction of CIN, groups were compared in terms of serum levels of urea, creatinine, neutrophil gelatinase associated lipocalin, protein carbonyl, total antioxidant capacity (TAC) as well as degree of renal injury at histopathologic level.
Results:
Groups II-V displayed more obvious histopathological alterations such as hemorrhage and renal tubular injury compared with Group I. TAC (p=0.043) and creatinine (p=0.046) levels increased significantly in Group II after the intervention. In Group III, protein carbonyl level diminished remarkably (p=0.046), while creatinine level was increased (p=0.046) following the intervention. TAC level was higher in Group IV (p=0.028) and Group V (p=0.026) following the procedure.
Conclusion:
The N-acetyl cysteine and ozone treatment may alleviate the biochemical and histopathological deleterious effects of contrast-induced nephropathy via enhancement of total antioxidant capacity and decreasing oxidative stress.
Key words:
Acetylcysteine; Ozone; Prevention; Rats
Introduction
Intravascular administration of radiographic contrast agents can be necessary for demonstration of vascular structures during diagnostic and therapeutic vascular interventions. Contrast-induced nephropathy (CIN) is an important adverse reaction following vascular procedures that involve the use of iodinated contrast agents. It is defined as an unexplained and acute impairment of renal functions reflected as a 25% or 0.5 mg/dL increase in serum creatinine levels within 48 to 72 hours following administration of contrast agent11 Morcos SK, Thomsen HS, Webb JA. Contrast-media-induced nephrotoxicity: a consensus report. Contrast Media Safety Committee, European Society of Urogenital Radiology (ESUR). Eur Radiol. 1999;9:1602-13. PMID: 10525875.. Contrast-induced nephropathy constitutes a frequent cause of acute kidney injury especially in hospitalized patients and in intensive care unit. It is associated with need for renal replacement therapy as well as prolonged hospitalization, and increased mortality22 Rihal CS, Textor SC, Grill DE, Berger PB, Ting HH, Best PJ, Singh M, Bell MR, Barsness GW, Mathew V, Garratt KN, Holmes DR. Incidence and prognostic importance of acute renal failure after percutaneous coronary intervention. Circulation. 2002;105:2259-64. PMID: 12010907..
The pathophysiological mechanisms responsible for CIN are still obscure. The possible pathophysiological mechanisms involve direct toxic effect of contrast agents on renal tubules and renal medullary hypoxia. There is evidence supporting that there are significant changes in renal microcirculation which lead to medullary hypoxia33 McCullough PA, Adam A, Becker CR, Davidson C, Lameire N, Stacul F, Tumlin J; CIN Consensus Working Panel. Epidemiology and prognostic implications of contrast-induced nephropathy. Am J Cardiol. 2006;98:5K-13K. PMID: 16949375.. Supression of renal cortical and medullary nitric oxide (NO) synthesis subsequent to the introduction of contrast agent may result in deterioration of renal microcirculation and lead to the formation of CIN44 Heyman SN, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy. Clin J Am Soc Nephrol. 2008;3:288-96. PMID: 18057308.. Even though various pharmacologic agents have been used for prophylaxis against CIN, there is no consensus for the protocol to be used for prevention55 Stacul F, Adam A, Becker CR, Davidson C, Lameire N, McCullough PA, Tumlin J; CIN Consensus Working Panel. Strategies to reduce the risk of contrast-induced nephropathy. Am J Cardiol. 2006;98:59K-77K. PMID: 16949381.
6 Walsh SR, Tang T, Gaunt ME, Boyle JR. Contrast-induced nephropathy. J Endovasc Ther. 2007;14:92-100. PMID: 17291156.-77 Pannu N, Wiebe N, Tonelli M; Alberta Kidney Disease Network. Prophylaxis strategies for contrast-induced nephropathy. J Am Med Assoc. 2006;295:2765-79. PMID: 16788132..
Risk factors for development of CIN involve previous renal dysfunction, diabetic nephropathy, congestive heart failure, high-dose administration of contrast agents, and concomitant introduction of nephrotoxic drugs. The occurence of CIN is associated with increased morbidity, mortality, and the cost of medical care. The contrast agents may exert deleterious effects on renal function via a combination of renal vasoconstriction, hypoxia, and direct toxicity on tubular epithelial cells. Moreover, reactive oxygen species linked with the introduction of a contrast agent can contribute to the progression of CIN. Reactive oxygen species may have both direct and indirect roles on the cortical and medullary microcirculation88 Xu R, Tao A, Bai Y, Deng Y, Chen G. Effectiveness of n-acetylcysteine for the prevention of contrast-induced nephropathy: a systematic review and meta-analysis of randomized controlled trials. J Am Heart Assoc. 2016;5(9):e003968. PMID: 27663415.. CIN has become a more important problem since its incidence has increased attributed to the recent popularization of diagnostic and interventional radiologic procedures99 Benko A, Fraser-Hill M, Magner P, Capusten B, Barrett B, Myers A, Owen RJ; Canadian Association of Radiologists: consensus guidelines for the prevention of contrast-induced nephropathy. Can Assoc Radiol J. 2007 Apr;58(2):79-87. PMID: 17521052.. Its incidence varies between 2% and 12% after radiologic interventions such as angiography, computerized tomography (CT), and CT angiography1010 Krol AL, Dzialowski I, Roy J, Puetz V, Subramaniam S, Coutts SB, Demchuk AM. Incidence of radiocontrast nephropathy in patients undergoing acute stroke computed tomography angiography. Stroke. 2007 Aug;38(8):2364-6. PMID: 17600231.
11 Hipp A, Desai S, Lopez C, Sinert R. The incidence of contrast-induced nephropathy in trauma patients. Eur J Emerg Med. 2008 Jun;15(3):134-9. PMID: 18460952.-1212 Kim KS, Kim K, Hwang SS, Jo YH, Lee CC, Kim TY, Rhee JE, Suh GJ, Singer AJ, Kim HD. Risk stratification nomogram for nephropathy after abdominal contrast-enhanced computed tomography. Am J Emerg Med. 2011 May;29(4):412-7. PMID: 20825813..
Recently, neutrophil gelatinase associated lipocalin (NGAL) was identified as an early biomarker for acute renal injury after cardiac surgery and as one of the most strikingly induced proteins in kidney after ischemia1313 Ding H, He Y, Li K, Yang J, Li X, Lu R, Gao W. Urinary neutrophil gelatinase-associated lipocalin (NGAL) is an early biomarker for renal tubulointerstitial injury in IgA nephropathy. Clin Immunol. 2007 May;123(2):227-34. PMID: 17360238.. Protein carbonyl (PCO) content is an important parameter for evaluation of oxidative damage to proteins. PCO represents an irreversible protein modification, often leading to the inactivation of the proteins and it is a sensitive and an early marker of oxidative stress to tissues1414 Ben-Cherif W, Chahdoura H, Haouas Z, Ben-Attia M, Aouam K, Reinberg A, Boughattas NA. Dosing-time dependent oxidative effects of an immunosuppressive drug "Mycophenolate Mofetil" on rat kidneys. Biomed Pharmacother. 2017 Mar;87:509-18. PMID: 28076831..
N-acetylcysteine (NAC) is a cysteine derivative formed as an intermediate product during conversion of cysteine to glutathione1515 Demir M, Kutlucan A, Akin H, Aydin O, Sezer T. Comparison of different agents on radiographic contrast agent induced nephropathy. Eur J Gen Med. 2008;5(4):222-7. doi: 10.29333/ejgm/82611.
https://doi.org/10.29333/ejgm/82611...
. Sulfhydryl groups in NAC are capable of detoxifying free oxygen radicals and enhancing natural antioxidant capacity by increasing intracellular reduced glutathione concentration. Attributed to the effects of thiol group and nitric oxide, NAC may diminish the formation of nitrite and peroxynitrite1515 Demir M, Kutlucan A, Akin H, Aydin O, Sezer T. Comparison of different agents on radiographic contrast agent induced nephropathy. Eur J Gen Med. 2008;5(4):222-7. doi: 10.29333/ejgm/82611.
https://doi.org/10.29333/ejgm/82611...
,1616 Diaz-Sandoval LJ, Kosowsky BD, Losordo DW. Acetylcysteine to prevent angiography-related renal tissue injury (the apart trial). Am J Cardiol. 2002 Feb 1;89(3):356-8. PMID: 11809444.. Thus, NAC may have a potential to be used as a prophylactic agent against CIN. Preventive effects of NAC against CIN may be related with its antioxidant and vasodilating features1616 Diaz-Sandoval LJ, Kosowsky BD, Losordo DW. Acetylcysteine to prevent angiography-related renal tissue injury (the apart trial). Am J Cardiol. 2002 Feb 1;89(3):356-8. PMID: 11809444..
Ozone is an oxidizing gas which may be synthesized by ultraviolet rays and it may eb used as a disinfectant in food and water industry. In spite of its toxic potential, ozone may also serve as a pro-drug at controlled non-toxic doses. It can aid in amelioration of ischemic injury in varios tissues1717 Ajamieh H, Merino N, Candelario-Jalil E, Menendez S, Martinez-Sanchez G, Re L, Giuliani A, Leon OS. Similar protective effect of ischaemic and ozone oxidative preconditionings in liver ischemia/reperfusion injury. Pharm Res. 2002 Apr;45(4):333-9. PMID: 12030798.. Ozone exerts its beneficial effects by decreasing the levels of free oxygen radicals, induction of the local migration of polymorphonuclear leukocytes and promotion of oxygen supply to tissues exposed to injury1818 Chen H, Xing B, Liu X, Zhan B, Zhou J, Zhu H, Chen Z. Ozone oxidative preconditioning inhibits inflammation and apoptosis in a rat model of renal ischemia/reperfusion injury. Eur J Pharmacol. 2008 Mar 10;581(3):306-14. PMID: 18093583..
This experimental study was implemented to investigate whether N-acetylcysteine and ozone display preventive effects against CIN and to determine the biochemical and histopathological impacts of these agents following CIN.
Methods
Experimental design
This study was carried out between June 2016 and August 2016 out in the experimental laboratory of our university after the approval of local animal experimentation ethics committee (KAÜ-HADYEK/2016-089). Adherence to the principles declared in the National Institute of Health’s Guide for the Care and Use of Laboratory Animals was provided.
Animals were fed on a standard rat chow diet and access to water and food was allowed ad libitum. Cages were kept at a temperature of 24 ± 2°C and humidity of 55 ± 5% at a 12 hour light & dark cycle. A total of 30 adult male Wistar rats (each weighing 250 to 300 g) were dehydrated by keeping away from water for 24 hours and blood samples (1 ml) were obtained from tail veins for determination of baseline serum levels of urea, creatinine, protein carbonyl and NGAL.
The rats were randomly distributed into one of the five following groups (n=6 for each group): Group I, which served as control, received no contrast agent, ozone and NAC. Group II had only contrast agent, sodium and meglumine ioxaglate (Hexabrix, Mallinckrodt, Inc., St. Louis, MO), at a dose of 10 ml/kg injected through the tail vein. In Group III, NAC (150 mg/kg/day) was administered intraperitoneally 6 hours before and 6 hours after introduction of contrast agent. Rats in Group IV received intraperitoneal ozone treatment (1 mg/kg, 5% O3-95% O2) 6 hours before and 6 hours after administration of the contrast agent. Ozone treatment (1 mg/kg, 5% O3-95% O2) was applied for 5 days after the contrast agent was introduced in Group V.
Induction of CIN
All groups of rats were left in normal routine nutrition environments after the procedure. Blood samples were again taken from rats under ether anesthesia at earliest in their 72th hours according to KM given times. Centrifugation was performed at 1500 g for 10 minutes to extract the serum and samples were maintained at -70°C until analysis. Renal functions as well as serum antioxidant status were studied from these samples. After the rats were sacrificed by exsanguination, kidneys were dissected and fixed at 10% formalin before histopathological examination (Figure 1).
Ozone was generated by an ozone gas processor from medical grade oxygen via silent electric discharge (Turkozone Blue S; Ozon Health Services Ltd, Istanbul, Turkey). The delivery of ozone/oxygen gas mixture was performed by single-use silicon-treated polypropylene syringes.
Histopathological examination
A midline laparatomy was followed by dissection of both kidneys and the liver from the surrounding tissues. The kidneys were initially preserved in 10% phosphate buffered formalin before histopathological examination. Tissue samples were embedded in paraffin wax and sliced into sections of 4-mm thickness. All sections were stained with hematoxylin eosin and examined under light microscopy (Olympus BX50; Olympus Optical Co. Ltd., Tokyo, Japan) by a blinded pathologist.
Renal tissue samples were evaluated in terms of tubular injury, cast formation and hemorrhage. Histopathologic alterations were graded on a 3-point semiquantitative scale based on a previously described method as: 0 (no changes); 1 (focal, mild changes); 2 (multifocal, intermediate changes) and 3 (extensive, prominent changes)1919 Gulmen S, Kiris I, Narin C, Ceylan BG, Mermi B, Sutcu R, Meteoglu I. Tezosentan reduces the renal injury induced by abdominal aortic ischemia reperfusion in rats. J Surg Res. 2009 Nov;157(1):e7-e13. doi: 10.1016/j.jss.2008.08.011.
https://doi.org/10.1016/j.jss.2008.08.01...
.
Serum studies
Urea and creatinine levels
The concentration of urea in the serum was estimated by the urease-glutamate dehydrogenase enzymatic method, while serum creatinine level was measured by a modified Jaffe method2020 Talke H, Schubert GE. Enzymatic urea determination in the blood and serum in the Warburg optical test. Klin Wochenschr. 1965 Feb 1;43:174-5. PMID: 14258517.,2121 Fabiny DL, Ertinghausen G. Automated reaction-rate method for determination of serum creatinine with the CentrifiChem. Clin Chem. 1971 Aug;17(8):696-700. PMID: 5562281.. The analyses were performed using an automatic analyzer (Architect c800; Abbott Diagnostics, Lake Forest, IL, USA).
Total antioxidant capacity (TAC)
Commercially available kits based on colorimetric method and an autoanalyzer machine (Selectera XL, Holland) were used for assessment of TAC2222 Atashpour S, Jahromi HK, Jahromi ZK, Zarei S. Antioxidant effects of aqueous extract of Salep on Paraquat-induced rat liver injury. World J Hepatol. 2017 Feb 8;9(4):209-16. doi: 10.4254/wjh.v9.i4.209.
https://doi.org/10.4254/wjh.v9.i4.209...
.
Protein carbonyl content
Protein carbonyl content were measured spectrophotometrically by the Reznick and Packer method2323 Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol. 1994;233:357-63. PMID: 8015470.. The protein carbonyl content reacts with 2, 4-dinitrophenyl hydrazine (DNPH) to generate chromophoric dinitrophenylhydrazones. Accordingly, 0.5 ml of sample (1-2 mg), an equal volume of 10 mM DNPH in 2N HCl was added and incubated for 1 h. and shaken intermittently at room temperature. A corresponding blank was carried out by adding only 2 N HCl to the sample. Following incubation, the mixture was precipitated with 10% trichloroacetic acid and centrifuged. The precipitate was washed twice with ethanol: ethyl acetate (1:1) and finally dissolved in 1 ml of 6 M guanidine HCl, centrifuged at low speed and the supernatant was read at 366 nm. The difference in absorbance between the DNPH treated and HCl-treated sample was determined and expressed as nmoles of carbonyl groups per mg of protein, using the molar extinction coefficient of DNPH, e = 22.000 mM-1cm-1.
Neutrophil gelatinase associated lipocalin (NGAL) level
Level of NGAL was measured by quantitative sandwich ELISA in the serum samples which were maintained at -70°C until use. Rat monoclonal antibody (MAB1757, R&D Systems, USA) against NGAL were used as capturing and detecting antibodies. Recombinant NGAL (RC9209, Randox Laboratories, UK) was used as a standard (diluted from 1 to 1000 μg/L) to measure NGAL concentrations in 100 μL of serum samples in triplicate1313 Ding H, He Y, Li K, Yang J, Li X, Lu R, Gao W. Urinary neutrophil gelatinase-associated lipocalin (NGAL) is an early biomarker for renal tubulointerstitial injury in IgA nephropathy. Clin Immunol. 2007 May;123(2):227-34. PMID: 17360238..
Outcome parameters
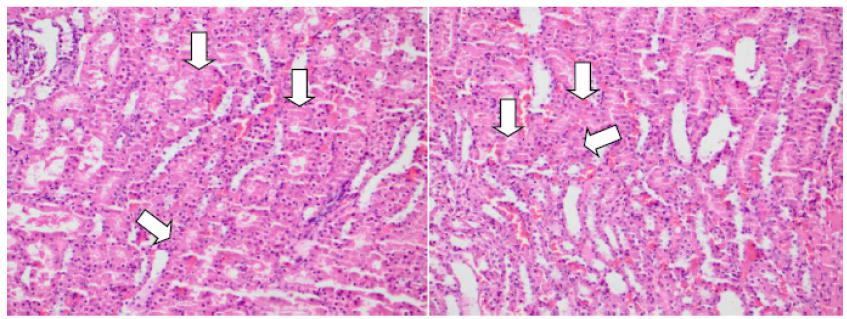
Biochemical analysis included comparison of serum levels of urea, creatinine, protein carbonyl, NGAL, and TAC within each group and between groups. Histopathologically, hemorrhage, renal tubular injury and cast formation were evaluated within each group and between groups. Intratubular hemorrhage and cast formation are evident in Figure 2 demonstrates, while renal tubular injury and regenerative changes are shown in Figures 3 and 4, respectively.
Statistical analysis
Our data was analyzed using IBM Statistical Package for Social Sciences Statistics 20 software (SPSS Inc., Chicago, IL, USA). Comparison of 5 groups in terms of quantitative variables was performed with Kruskal Wallis variance analysis. For posthoc comparisons between the control group and other groups, Mann Whitney U test was used based on a new p value 0.05/4=0.0125. Qualitative data was evaluated using Chi-square test. Comparison of variables within each group before and after the procedure was accomplished with Wilcoxon Signed Ranks test. For qualitative variables, level of significance was set at p<0.05.
Results
Table 1 demonstrates the levels of neutrophil gelatinase associated lipocalin (NGAL), total antioxidant capacity (TAC), protein carbonyl, urea and creatinine in 5 experimental groups before and after the procedures.
Levels of neutrophil gelatinase associated lipocalin (NGAL), total antioxidant capacity (TAC), protein carbonyl, urea and creatinine in 5 experimental groups before and after the procedures.
Levels of NGAL (p=0.002) and creatinine (p=0.004) in Group II were remarkably higher than Group I after the intervention. Similarly, NGAL (p=0.002) and creatinine (p=0.002) levels in Group III were remarkably higher than Group I after the intervention. NGAL level in Group IV was notably higher than that of Group I (p=0.009). TAC in Group V was remarkably higher compared to that in Group I (p=0.002) (Table 2).
For histopathologic parameters, cells in Table 3 with values below 5 comprise more than 20% of variables. Thus, validity of chi square results is under debate. However, conclusions can be drawn from tables made up of multiple cells. Accordingly, Groups II-V displayed more obvious histopathological alterations such as hemorrhage and renal tubular injury compared with Group I. No remarkable difference was detected between groups with respect to cast formation (Table 3).
Discussion
Contrast-induced nephropathy is a common and potentially serious complication following the administration of contrast media in patients at risk for acute renal injury. It is a complex disorder with vague pathophysiology. Patients who develop CIN have worse clinical outcomes, higher complication rates, longer hospital stay, and higher mortality. Mechanisms responsible for CIN are supposed to be increased vasoconstrictive forces, decreased local prostaglandin and NO-mediated vasodilatation, a direct toxic effect on renal tubular cells with damage caused by oxygen-free radicals, and increased oxygen consumption. These alterations lead to renal medullary ischemia that gives rise to CIN2424 Persson PB, Hansell P, Liss P. Pathophysiology of contrast medium-induced nephropathy. Kidney Int. 2005 Jul;68(1):14-22. PMID: 15954892..
The current trial was carried out to assess the biochemical and histopathological impacts of NAC and ozone treatment on CIN in an experimental setting. Our results indicated that ozone treatment may enhance and improve TAC against CIN, while NAC was more likely to diminish oxidative stress as reflected in protein carbonyl levels. Determination of therapeutic efficacy of NAC and ozone treatment and establishment of preventive protocols against CIN warrants design of multi-centric clinical trials. Multiple doses of ozone treatment seemed to offer no additional benefit over single dose against development of CIN.
In the present study, the cascade leading to CIN was reflected as elevation of serum creatinine levels. NAC led to diminution of protein carbonyl levels which was consistent with decreased oxidative stress. Prolonged administration of ozone treatment after introduction of contrast agent seemed to ameliorate the unfavourable histopathological results linked with CIN. With respect to renal tubular necrosis and hemorrhage, preconditioning with ozone and NAC displayed similar outcomes. Interestingly, creatinine levels were not increased in rats preconditioning with ozone. Therefore, we speculate that preconditioning with ozone may have a beneficial impact against acute mechanisms initiating injury associated with contrast agent. This finding is consistent with the data supporting that ozone pretreatment activates renal antioxidant mechanisms potently2525 Kurtoglu T, Durmaz S, Akgullu C, Gungor H, Eryilmaz U, Meteoglu I, Karul A, Boga A. Ozone preconditioning attenuates contrast-induced nephropathy in rats. J Surg Res. 2015 May 15;195(2):604-11. doi: 10.1016/j.jss.2015.01.041.
https://doi.org/10.1016/j.jss.2015.01.04...
. On the other hand, prolonged course of ozone treatment after contrast agent is given may alleviate the histopathologic injury. A complex and multidisciplinary approach is necessary for management of biochemical and histopathological results of CIN.
Exposure to contrast agents may adversely affect renal oxygen supply and induce renal medullary hypoxia leading to CIN44 Heyman SN, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy. Clin J Am Soc Nephrol. 2008;3:288-96. PMID: 18057308.. 4 In conjunction with other experimental studies, we observed that use of contrast agents may be associated with hazardous effects of oxidative stress and histopathologic damage2626 Devrim E, Cetin M, Namuslu M, Erguder IB, Cetin R, Durak I. Oxidant stress due to non ionic low osmolar contrast medium in rat kidney. Indian J Med Res. 2009;130:433-6. PMID: 19942798..
We suggest that preconditioning with ozone, NAC and ozone treatment after administration of contrast agent may affect different steps of the pathophysiological reactions involved in CIN. Therefore, combination of NAC and ozone treatment may offer more promising protective effects against CIN. Timing of ozone application may have important clinical implications on its utility against CIN.
Kurtoglu et al.2525 Kurtoglu T, Durmaz S, Akgullu C, Gungor H, Eryilmaz U, Meteoglu I, Karul A, Boga A. Ozone preconditioning attenuates contrast-induced nephropathy in rats. J Surg Res. 2015 May 15;195(2):604-11. doi: 10.1016/j.jss.2015.01.041.
https://doi.org/10.1016/j.jss.2015.01.04...
suggested that reduction of the oxidative stress in the animals exposed to contrast agents was linked with enhancement of the renal antioxidant systems and increased endogenous capacity for ROS scavenging. In the literature, there is contradictory data on the beneficial effect of NAC for prevention of CIN. A meta-analysis revaled that data on intravenous NAC treatment against CIN was inconsistent to make a conclusion on its efficacy. There has been a substantial lack of pharmacokinetic or pharmacodynamic data and the exact mechanism by which NAC acts is still obscure2727 Sun Z, Fu Q, Cao L, Jin W, Cheng L, Li Z. Intravenous N-Acetylcysteine for prevention of contrast-induced nephropathy: a meta-analysis of randomized, controlled trials. PLoS One. 2013;8(1):e55124. PMID: 23383076..
To the best of our knowledge, this is the first study comparing the effectivenesses of NAC, ozone preconditioning and ozone treatment after administration of contrast agent. The seek for determination of ideal preventive regimen against CIN require further investigation and efforts must be spent to explore the efficacy, safety and optimization of combination treatment protocols. Hopefully, our findings may provide novel insights about the preventive and therapeutic mechanisms against CIN.
Some weaknesses of our study must be remembered during interpretation of our results. Experimental design, the complexity of counteracting oxidants and antioxidants in biological systems, relatively subjective methodology and limitations of statistical analysis particularly for histopathologic data as well as lack of tissue studies for oxidative stress markers should be taken into account before extrapolation of our data to clinical setting. Strengths of the present study involve assessment of both biochemical and histopathologic aspects of CIN and inclusion of multiple biomarkers of renal injury such as NGAL, protein carbonyl, urea, creatinine and TAC.
Conclusion
The N-acetyl cysteine and ozone treatment may alleviate the biochemical and histopathological deleterious effects of contrast-induced nephropathy via enhancement of total antioxidant capacity and decreasing oxidative stress.
References
-
1Morcos SK, Thomsen HS, Webb JA. Contrast-media-induced nephrotoxicity: a consensus report. Contrast Media Safety Committee, European Society of Urogenital Radiology (ESUR). Eur Radiol. 1999;9:1602-13. PMID: 10525875.
-
2Rihal CS, Textor SC, Grill DE, Berger PB, Ting HH, Best PJ, Singh M, Bell MR, Barsness GW, Mathew V, Garratt KN, Holmes DR. Incidence and prognostic importance of acute renal failure after percutaneous coronary intervention. Circulation. 2002;105:2259-64. PMID: 12010907.
-
3McCullough PA, Adam A, Becker CR, Davidson C, Lameire N, Stacul F, Tumlin J; CIN Consensus Working Panel. Epidemiology and prognostic implications of contrast-induced nephropathy. Am J Cardiol. 2006;98:5K-13K. PMID: 16949375.
-
4Heyman SN, Rosen S, Rosenberger C. Renal parenchymal hypoxia, hypoxia adaptation, and the pathogenesis of radiocontrast nephropathy. Clin J Am Soc Nephrol. 2008;3:288-96. PMID: 18057308.
-
5Stacul F, Adam A, Becker CR, Davidson C, Lameire N, McCullough PA, Tumlin J; CIN Consensus Working Panel. Strategies to reduce the risk of contrast-induced nephropathy. Am J Cardiol. 2006;98:59K-77K. PMID: 16949381.
-
6Walsh SR, Tang T, Gaunt ME, Boyle JR. Contrast-induced nephropathy. J Endovasc Ther. 2007;14:92-100. PMID: 17291156.
-
7Pannu N, Wiebe N, Tonelli M; Alberta Kidney Disease Network. Prophylaxis strategies for contrast-induced nephropathy. J Am Med Assoc. 2006;295:2765-79. PMID: 16788132.
-
8Xu R, Tao A, Bai Y, Deng Y, Chen G. Effectiveness of n-acetylcysteine for the prevention of contrast-induced nephropathy: a systematic review and meta-analysis of randomized controlled trials. J Am Heart Assoc. 2016;5(9):e003968. PMID: 27663415.
-
9Benko A, Fraser-Hill M, Magner P, Capusten B, Barrett B, Myers A, Owen RJ; Canadian Association of Radiologists: consensus guidelines for the prevention of contrast-induced nephropathy. Can Assoc Radiol J. 2007 Apr;58(2):79-87. PMID: 17521052.
-
10Krol AL, Dzialowski I, Roy J, Puetz V, Subramaniam S, Coutts SB, Demchuk AM. Incidence of radiocontrast nephropathy in patients undergoing acute stroke computed tomography angiography. Stroke. 2007 Aug;38(8):2364-6. PMID: 17600231.
-
11Hipp A, Desai S, Lopez C, Sinert R. The incidence of contrast-induced nephropathy in trauma patients. Eur J Emerg Med. 2008 Jun;15(3):134-9. PMID: 18460952.
-
12Kim KS, Kim K, Hwang SS, Jo YH, Lee CC, Kim TY, Rhee JE, Suh GJ, Singer AJ, Kim HD. Risk stratification nomogram for nephropathy after abdominal contrast-enhanced computed tomography. Am J Emerg Med. 2011 May;29(4):412-7. PMID: 20825813.
-
13Ding H, He Y, Li K, Yang J, Li X, Lu R, Gao W. Urinary neutrophil gelatinase-associated lipocalin (NGAL) is an early biomarker for renal tubulointerstitial injury in IgA nephropathy. Clin Immunol. 2007 May;123(2):227-34. PMID: 17360238.
-
14Ben-Cherif W, Chahdoura H, Haouas Z, Ben-Attia M, Aouam K, Reinberg A, Boughattas NA. Dosing-time dependent oxidative effects of an immunosuppressive drug "Mycophenolate Mofetil" on rat kidneys. Biomed Pharmacother. 2017 Mar;87:509-18. PMID: 28076831.
-
15Demir M, Kutlucan A, Akin H, Aydin O, Sezer T. Comparison of different agents on radiographic contrast agent induced nephropathy. Eur J Gen Med. 2008;5(4):222-7. doi: 10.29333/ejgm/82611.
» https://doi.org/10.29333/ejgm/82611 -
16Diaz-Sandoval LJ, Kosowsky BD, Losordo DW. Acetylcysteine to prevent angiography-related renal tissue injury (the apart trial). Am J Cardiol. 2002 Feb 1;89(3):356-8. PMID: 11809444.
-
17Ajamieh H, Merino N, Candelario-Jalil E, Menendez S, Martinez-Sanchez G, Re L, Giuliani A, Leon OS. Similar protective effect of ischaemic and ozone oxidative preconditionings in liver ischemia/reperfusion injury. Pharm Res. 2002 Apr;45(4):333-9. PMID: 12030798.
-
18Chen H, Xing B, Liu X, Zhan B, Zhou J, Zhu H, Chen Z. Ozone oxidative preconditioning inhibits inflammation and apoptosis in a rat model of renal ischemia/reperfusion injury. Eur J Pharmacol. 2008 Mar 10;581(3):306-14. PMID: 18093583.
-
19Gulmen S, Kiris I, Narin C, Ceylan BG, Mermi B, Sutcu R, Meteoglu I. Tezosentan reduces the renal injury induced by abdominal aortic ischemia reperfusion in rats. J Surg Res. 2009 Nov;157(1):e7-e13. doi: 10.1016/j.jss.2008.08.011.
» https://doi.org/10.1016/j.jss.2008.08.011 -
20Talke H, Schubert GE. Enzymatic urea determination in the blood and serum in the Warburg optical test. Klin Wochenschr. 1965 Feb 1;43:174-5. PMID: 14258517.
-
21Fabiny DL, Ertinghausen G. Automated reaction-rate method for determination of serum creatinine with the CentrifiChem. Clin Chem. 1971 Aug;17(8):696-700. PMID: 5562281.
-
22Atashpour S, Jahromi HK, Jahromi ZK, Zarei S. Antioxidant effects of aqueous extract of Salep on Paraquat-induced rat liver injury. World J Hepatol. 2017 Feb 8;9(4):209-16. doi: 10.4254/wjh.v9.i4.209.
» https://doi.org/10.4254/wjh.v9.i4.209 -
23Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol. 1994;233:357-63. PMID: 8015470.
-
24Persson PB, Hansell P, Liss P. Pathophysiology of contrast medium-induced nephropathy. Kidney Int. 2005 Jul;68(1):14-22. PMID: 15954892.
-
25Kurtoglu T, Durmaz S, Akgullu C, Gungor H, Eryilmaz U, Meteoglu I, Karul A, Boga A. Ozone preconditioning attenuates contrast-induced nephropathy in rats. J Surg Res. 2015 May 15;195(2):604-11. doi: 10.1016/j.jss.2015.01.041.
» https://doi.org/10.1016/j.jss.2015.01.041 -
26Devrim E, Cetin M, Namuslu M, Erguder IB, Cetin R, Durak I. Oxidant stress due to non ionic low osmolar contrast medium in rat kidney. Indian J Med Res. 2009;130:433-6. PMID: 19942798.
-
27Sun Z, Fu Q, Cao L, Jin W, Cheng L, Li Z. Intravenous N-Acetylcysteine for prevention of contrast-induced nephropathy: a meta-analysis of randomized, controlled trials. PLoS One. 2013;8(1):e55124. PMID: 23383076.
-
Financial source:
none
-
1
Research performed at Animal Study Laboratory, Kafkas University, Kars, Turkey.
Publication Dates
-
Publication in this collection
June 2018
History
-
Received
08 Feb 2018 -
Reviewed
09 Apr 2018 -
Accepted
11 May 2018