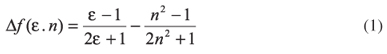Abstracts
This work studied the fluorescent and solvatochromic properties of coumarin and its derivatives aiming to contribute in the elucidation of the excited state behavior of the molecule in solvent mixtures. This approach has been developed through a set of measurements involving different solvent ratios, which present distinct dielectric constants. The excitation and fluorescence maxima were largely blue-shifted with increasing participation of the organic solvent and the Stokes shifts were unusually low in mixtures with lower solvent polarity parameter, Δf. The emission intensity of coumarin was extremely sensitive to the polarity and the different structural conformations of solvents. These results influenced the spectroscopic behavior and explain the deviation from the linearity of the Lippert-Mataga relationship. The use of fluorescence spectroscopy makes the understanding of the behavior of the systems studied when in the presence of different environments being extremely important for the identification and characterization of coumarin in different pharmaceutical formulations.
coumarin; fluorescence; solvatochromic properties; emission spectra; excitation spectra
Este trabalho estuda as propriedades fluorescentes e solvatocrômicas da cumarina e seus derivados visando contribuir na elucidação do comportamento do estado excitado dessas moléculas em misturas de solventes. Esta abordagem foi desenvolvida através de medidas que envolveram diferentes relações de solventes com constantes dielétricas distintas. Os máximos de excitação e emissão de fluorescência foram deslocados para o azul nas misturas com maior proporção do solvente orgânico e o deslocamento de Stokes foi menor em misturas com menor parâmetro de polaridade do solvente, Δf. A intensidade de emissão de cumarina foi extremamente sensível à polaridade e às diferentes conformações estruturais dos solventes. Estas conformações influenciaram diretamente no comportamento espectroscópico e explicam o desvio da linearidade da relação de Lippert-Mataga. A utilização de espectroscopia de fluorescência possibilita o conhecimento do comportamento destes derivados quando em diferentes ambientes, sendo de extrema importância para a identificação e caracterização desses compostos em formulações farmacêuticas.
ARTICLE
Photophysical properties of coumarin compounds in neat and binary solvent mixtures: evaluation and correlation between solvatochromism and solvent polarity parameters
Leonardo M. MoreiraI; Mirian M. de MeloII; Priscila A. MartinsII; Juliana P. LyonIII; Ana Paula RomaniIV; Lúcia CodognotoV, * * e-mail: lucia.codognoto@unifesp.br, hueder.paulo@ufabc.edu.br ; Sandra C. dos SantosVI; Hueder P. M. de OliveiraVII, * * e-mail: lucia.codognoto@unifesp.br, hueder.paulo@ufabc.edu.br
IDepartamento de Zootecnia (DEZOO), Universidade Federal de São João Del Rei (UFSJ), 36301-360 São João del Rei-MG, Brazil
IIInstituto de Pesquisa e Desenvolvimento, Universidade do Vale do Paraíba (UNIVAP), Av. Shishima Hifumi, 2911, Urbanova, 12244-000 São José dos Campos-SP, Brazil
IIIDepartamento de Ciências Naturais (DCNAT), Universidade Federal de São João Del Rei (UFSJ), 36301-160 São João del Rei-MG, Brazil
IVCentro de Engenharia, Modelagem e Ciências Sociais Aplicadas, Universidade Federal do ABC (UFABC), Avenida dos Estados, 5001, Bairro Bangu, 09210-580 Santo André-SP, Brazil
VDepartamento de Ciências Exatas e da Terra, Universidade Federal de São Paulo (Unifesp), Instituto de Ciências Ambientais, Químicas e Farmacêuticas, Rua Prof. Artur Riedel, 275, Bairro Eldorado, 09972-270 Diadema-SP, Brazil
VICentro de Ciências Químicas, Farmacêuticas e de Alimentos, Universidade Federal de Pelotas (UFPel), Campus Capão do Leão, 96160-000 Pelotas-RS, Brazil
VIICentro de Ciências Naturais e Humanas Universidade Federal do ABC (UFABC), Rua Santa Adélia, 166, Bairro Bangu, 09210-170 Santo André-SP, Brazil
ABSTRACT
This work studied the fluorescent and solvatochromic properties of coumarin and its derivatives aiming to contribute in the elucidation of the excited state behavior of the molecule in solvent mixtures. This approach has been developed through a set of measurements involving different solvent ratios, which present distinct dielectric constants. The excitation and fluorescence maxima were largely blue-shifted with increasing participation of the organic solvent and the Stokes shifts were unusually low in mixtures with lower solvent polarity parameter, Δf. The emission intensity of coumarin was extremely sensitive to the polarity and the different structural conformations of solvents. These results influenced the spectroscopic behavior and explain the deviation from the linearity of the Lippert-Mataga relationship. The use of fluorescence spectroscopy makes the understanding of the behavior of the systems studied when in the presence of different environments being extremely important for the identification and characterization of coumarin in different pharmaceutical formulations.
Keywords: coumarin, fluorescence, solvatochromic properties, emission spectra, excitation spectra
RESUMO
Este trabalho estuda as propriedades fluorescentes e solvatocrômicas da cumarina e seus derivados visando contribuir na elucidação do comportamento do estado excitado dessas moléculas em misturas de solventes. Esta abordagem foi desenvolvida através de medidas que envolveram diferentes relações de solventes com constantes dielétricas distintas. Os máximos de excitação e emissão de fluorescência foram deslocados para o azul nas misturas com maior proporção do solvente orgânico e o deslocamento de Stokes foi menor em misturas com menor parâmetro de polaridade do solvente, Δf. A intensidade de emissão de cumarina foi extremamente sensível à polaridade e às diferentes conformações estruturais dos solventes. Estas conformações influenciaram diretamente no comportamento espectroscópico e explicam o desvio da linearidade da relação de Lippert-Mataga. A utilização de espectroscopia de fluorescência possibilita o conhecimento do comportamento destes derivados quando em diferentes ambientes, sendo de extrema importância para a identificação e caracterização desses compostos em formulações farmacêuticas.
Introduction
Coumarin compounds constitute a class of secondary metabolites widely found in plants.1,2 These compounds are recognized by a great variability of biological activities, especially chemotherapy agents, presenting antimicrobial, antiviral, antioxidant, antitumor, antispasmodic and anti-inflammatory activities.3-7 Coumarin compounds are usually employed as additive in dyes and sprays as well as in aromatization of foods.8 Thus, the wide utilization of these compounds in several areas has required the development of new methods aiming to improve the quality control of the pharmaceutical and cosmetic preparations.
Coumarin compounds belong to a large and important family of hydrophobic dyes due to their biological and physicochemical properties, which exhibit notable photophysical properties,9 being also used as dye laser in the blue-green region.10,11 Currently, there are several analytical methods for the determination of coumarin in commercial products, such as mass spectrometry (MS), nuclear magnetic resonance, capillary electrophoresis and chromatographic methods.12-14 In any case, in analyses focused on biological matrix, there is a higher prevalence of using high performance liquid chromatography (HPLC) with fluorimetric detection and MS.15,16 However, the technological advancement of instrumental devices associated with the spectroscopy of fluorescence has provoked a substantial increase of applications of this analytical methodology, mainly in studies developed directly upon the matrix of the medicine.17 This tool presents advantages regarding this kind of instrumental evaluation, such as high sensitivity when compared with the spectroscopy of electronic absorption in the ultraviolet-visible region (UV-Vis) and high selectivity to compounds that present fluorescent properties.
Considering that coumarin derivatives are fluorescent compounds, this type of spectroscopy analysis can be a very interesting alternative for several laboratories, since the use of this technique is significantly simple and technically accessible. Also, it presents low cost for a great number of biological matrix.3,18
These molecules usually show strong polarity-dependent Stokes shifts, large changes in dipole moments on excitation, and very high fluorescence quantum yields.19 Since these molecules have been shown to be excellent probes for solvation dynamics and local friction of different complex environments, the study of the interaction of these molecules with different solvents having different physicochemical properties forms an important subject that plays a significant role in the photophysics of the excited states.9,20
Thus, solvatochromic shifts can be used to obtain the ratio of the absorption/emission spectra and the polarity of the solvent as well as information on the electronic excited states of the molecule. Therefore, the objective of this work was to study the photophysical properties of coumarin to understand the behavior of the molecule in binary solvent mixtures of water and organic solvents such as ethanol (EtOH, polar and hydrogen-bond donor solvent), acetonitrile (ACN, polar and hydrogen-bond acceptor solvent) and dioxane (Diox, apolar and hydrogen-bond acceptor solvent). This work has been developed through a set of measurements involving different solvent ratios. Coumarin derivatives studied were C1 (7-diethylamino-4-methylcoumarin), C151 (7-amino-4-trifluoromethylcoumarin) and C500 (7-ethylamino-4-trifluoromethylcoumarin). The molecular structures of coumarin, 1,2-benzopyrone, and its derivatives are presented in Figure 1.
Experimental
Spectroscopic measurements were performed using a Jobin-Yvon Spex fluoromax-2 spectrofluorimeter with a scan between 200 and 800 nm, slits of 2 and 3 nm, respectively, and with quartz cuvette of 1 cm of optical path length at room temperature (25 ºC) and normal pressure (1 atm). Solvents used in the spectroscopic studies were deionized water (Milli-Q pattern), EtOH, Diox and ACN. Coumarin solutions were obtained from a 1.0 × 10-3 mol L-1 stock solution of ACN. Coumarin and its derivatives were purchased from Aldrich (99%). All reagents used in this work were of analytical grade and organic solvents were HPLC grade. Initially, we obtained the excitation spectra of coumarin and its derivatives by measuring the emission intensity at a fixed wavelength. In this case, the maximum wavelength was obtained in the emission spectra, while varying the excitation wavelength. Emission spectra were determined by measuring the change in emission intensity wavelength at a fixed excitation wavelength, where the maximum wavelength obtained in the excitation spectra was used.
The solvent polarity parameter, Δf, has been reported to correlate nicely with a variety of physicochemical properties of many probe molecules in different solvents and solvent mixtures. The Δf of the solvents can be calculated as defined by Lippert and Mataga:21,22
where the dielectric permittivity (ε) and refractive index (n) of the pure solvents and mixed solvents were taken from literature.
Results and Discussion
Excitation and fluorescence spectra of coumarin and its derivatives were recorded in different solvents and solvent mixtures. The emission spectra of coumarin and its derivatives in EtOH at the concentration of 4.0 × 10-5 mol L-1 are given in Figure S1 in the Supplementary Information (SI) section. The maximum emission wavelength could be observed at 380, 444, 475 and 488 nm for coumarin, C1, C151 and C500, respectively, denoting that the wavelengths of each respective compound are significantly different. The longer wavelengths found for C151 and C500 compared to C1 can be explained by the replacement of a CH3 group by a CF3 group.4 The energy gap between the highest occupied and the lowest unoccupied orbitals of an amino substituted molecule is considerably lower than the difference between the highest occupied and the lowest unoccupied orbitals of unsubstituted molecules. This effect is responsible for the greatest length of emission found for the CF3 substituted molecule. In the case of fluorine being an electron-withdrawing group, the excited state of the molecule is more polar than the ground state due to intermolecular charge transfer.23
The excitation and emission spectra of coumarin-derivatives dissolved in water, EtOH, ACN and Diox, using a concentration of 4.0 × 10-5 mol L-1 are given in Figure S2. Both the excitation and fluorescence spectra are strongly dependent on the polarities of the solvents used. In all spectra, it is remarkable that, depending on the solvent, we observe different emission wavelengths (between 407-507 nm). We observed a shift to longer wavelengths in solvents with higher polarity, in this case, water. The increasing solvent polarity is in accordance with the expected stabilization of the intramolecular charge transfer (ICT) character of the excited singlet state in polar solvents.24 The intensity of fluorescence was higher in lower polarity solvents. This is reported to show low fluorescence quantum yields in solvents of higher polarities.10
Figure 2 shows the emission spectra of C1 (a), C151 (b) and C500 (c) as function of the solvent composition, organic solvent-water. In the case of solvent mixtures, the increase of the proportion of organic solvent reduces the value of the dielectric constant and the polarity of the system, thus generating blue shifted spectral bands compared to the data obtained in water (higher polarity). The shift in the fluorescence band position observed towards longer wavelengths could be due to the different excited state charge distribution of the solute than that in the ground state, which would result in a stronger interaction with polar solvents in the excited state.25
In the case of coumarin, the maximum quantum yield was obtained in water, while with EtOH the quantum yield was very low. Furthermore, results have shown that the solvent mixtures containing 80% and 60% of water present a significant intensity of emission. A similar spectroscopic behavior was observed with the Diox-water mixture. Interestingly, the system containing 20% of ACN presented the lowest fluorescence intensity, which is an experimental result quite different from that obtained with the EtOH-water system. This result demonstrated that the association between ACN and water molecules is very peculiar, presenting physicochemical properties different from those obtained for pure ACN and water, respectively. The reason for this non-ideality is due to micro-heterogeneity of the mixtures, as shown by Catalán and Seoud.26,27 Since ACN molecules are receptors of hydrogen bonds, they interact in an additional form with the water clusters and are not part of the network of hydrogen bonds formed in the cluster of water. Thus, they interact only between the spaces between clusters of water in an additional form.28 Thus, this water-ACN solvent system is less effective to form hydrogen bonds with coumarin and coumarin-derivative molecules when compared with pure water and pure ACN. Therefore, it seems that the interaction between ACN and water is more effective than the interaction of the ACN-water system with the solute.
The Diox-water system presented a spectroscopic behavior completely different from that of the ACN-water system. In spite of the higher intensity of emission that was obtained with 100% of water, the solvent systems with 20% of Diox also presented significant emission intensity. Probably, the presence of Diox in a low concentration does not affect the ability of the water to form hydrogen bonds, even in a system containing 80% of water. This may occur due to a weak interaction between the molecules of Diox and water, considering that Diox does not present high polarity.29
Regarding coumarin-derivatives, the enhancement of the organic solvent probably increases the fluorescence quantum yield. However, some authors have shown that C151 presents unusual low fluorescence quantum yields and lifetimes in non-polar solvents.30,31 This can be explained by a possible enhancement in the intersystem crossing process in non-polar solvents or by different conformations in non-polar and other solvents.30,31 The excitation spectra of C1 in Diox-water system are also very peculiar, since the highest intensity was obtained with 80% of Diox and the lowest with a system containing 100% of water.
The maximum emission regarding C151 was obtained with ACN. The solvent mixture containing 80% ACN presented the lowest quantum yield for the C151 derivative. These results corroborated the previous spectroscopic results that suggested a very characteristic photophysical behavior for the ACN-water systems, denoting that this combination is quite different of each respective solvent when analyzed alone.27
C151 presented a blue-shifted band in the presence of 100% Diox when compared to the solvent system with 100% water. It is important to note that the emission band of C151 in 100% Diox is more intensely blue-shifted when compared with the excitation band of C151 in the same solvent system. Moreover, the emission band of C151 in 100% water is red-shifted when compared to other solvent ratios, which does not occur with the respective excitation band in 100% of water. The results for C500 in Diox-water mixture are presented in the SI section. The highest quantum yield was obtained in the presence of 100% Diox. The lowest emission was obtained in the presence of 100% of water, which was accompanied by the excitation spectra of this solvation condition; the minimum quantum yield was found to be similar to that obtained in 100% water. All the coumarin-derivatives presented higher intensity of fluorescence emission when compared with the precursor coumarin. Furthermore, the highest quantum yield was obtained in the presence of 100% Diox, whereas in the presence of 100% water (opposite condition), it represents in several cases a condition of low quantum yield.
Figure 3 shows the relative intensity and the maximum emission wavelength of C1 (a), C151 (b) and C500 (c) as function of the solvent mixture ratio. Coumarin-derivatives presented a decrease of the relative intensity as the proportion of water was increased. This shows a decrease in the fluorescence yield with the increase of the solvent polarity, which may be due to the associated interaction with the organic solvent.21
The molar electronic transition energy, ET (kcal mol-1), gives the direct measure of stabilization energy on account of solvation, whose relationship with absorption maximum (λmax) is given as32-37
To show the stability of different compounds in the corresponding binary mixtures, the inset of Figure 3 shows the molar transition energy of C1 (a), C151 (b) and C500 (c) as function of the solvent mixture ratio. In terms of molar transition energy, ET, the variation is shown as a function of the water percentage in the binary mixture. These variations are not linear, due to the sites of specific/nonspecific interactions between the dye molecule and one of the components of the mixture, characterizing the phenomenon that is known as preferential solvation. Furthermore, the deviation from the ideal behavior suggests that the binary solvent mixture is micro-heterogeneous.34
The spectral shifts (measured in fluorescence and absorption experiments) caused by the solvent effect can be used to estimate the ground- and excited-state dipole moments.20,38 There is a linear correlation between the Stokes shifts, Δν, and a solvent polarity function which involves both dielectric permittivity (ε) and refractive index (n) of the medium.20 The solvent sensitivity of the Stokes shift is commonly explained by the Lippert-Mataga equation (equation 3) which is based on Onsager's reaction field theory:39,40
where νaand νfare the wavenumbers of absorption and fluorescence transition, respectively, h is the Planck constant, c is the speed of light, ε0 is the electric constant, µeand µgare the excited state and ground state dipole moments of a solute molecule, and a0is the cavity. The values of the solute cavity radii (ao) were calculated from the molecular volume of molecules according to Suppan's equation:41
where δ is the density of the solute molecule, M is the molecular weight of solute and N is Avogadro's number. The ao calculated for the solutes are 5.14, 4.63 and 4.96 Å, respectively, for C1, C151 and C500.
In the case of binary mixtures, the value of Δf(ε,n) could be calculated from experimental values using equation 1. To understand the dependence of the spectral shifts on solvent polarity, a method that uses the solvent polarity parameter, Δf(ε,n), was developed. Table 1 shows the solvent polarity parameter, Δf(ε,n), which was estimated using equation 1, the physicochemical parameters of solvents and their mixtures (dielectric constant (ε) and refractive index (n)), the wavelengths of the maximum excitation (λexc), emission (λF), and Stokes shift (Δν) obtained for each system. Table 1 shows that the increasing participation of the organic compound results in an enhancement of the intensity together with a slight blue shift in the emission maximum. This maximum and the Stokes shift decrease as the organic portion is increased, i.e., in solvents with low polarity, smaller values of Δν were obtained. This indicates greater changes between interactions with more polar solvents relative to those less polar.20
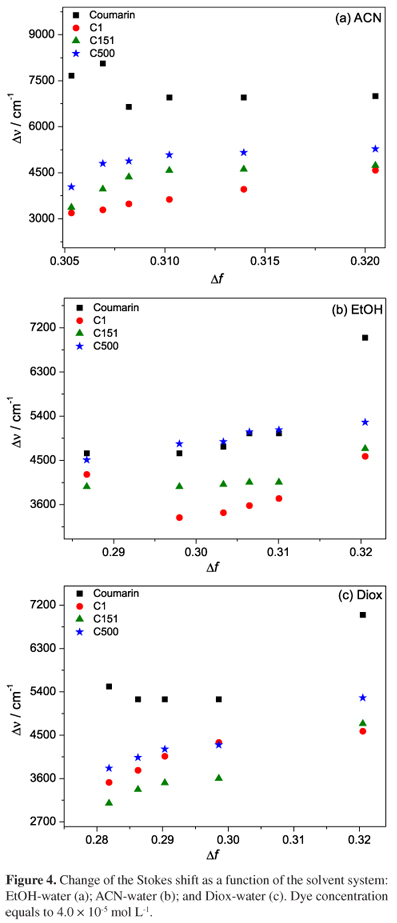
Figure 4 shows the change of Stokes shift (Δν) with the solvent polarity function Δf(ε,n). The decrease of the Stokes shift as a function of Δf(ε,n) indicates a lower solvation of the excited electronic state of the coumarin. High shift values suggest a reorganization of the orientation of polar groups found in the molecular structure of coumarin. We observed an increase of fluorescence intensity in some coumarin-derivatives when the solvent system was changed, which can be assigned to the polarity of the solvents. This effect increases the activation energy associated with the conversion of the planar excited electronic state to an intramolecular charge transfer, favoring the quantum yield of fluorescence.
The dipole moment of the excited state can increase and/or decrease as compared to ground state due to redistribution of charges and electron densities that occur naturally with conformational changes in the excited state.48 The increase in the Stokes shift with increasing solvent polarity indicates that there is an increase in the dipole moment on excitation.20 The results of the statistical treatment of the Lippert-Mataga correlations, namely the slopes and number of data, are shown in Table 2. The variation of dipole moments (µeµg) of the ground state (µg) and excited state (µe) are determined from the slopes of the Lippert-Mataga correlations. This evaluation indicates that the dipole moment of coumarin in the excited electronic state is higher than in the fundamental electronic state. There was an exception in the case of coumarin that showed a lower dipole moment in the presence of ACN-water and Diox-water mixtures.
The plots in Figure 4 are clearly nonlinear. This suggests that the dipole-dipole interaction between the solute and solvent is responsible for the large solvent-dependent fluorescence shift.49 Thus, a shift in the Lippert relationship in binary mixtures was observed in these results. Regarding pure solvents, the deviation was more pronounced. This is explained because of the different physicochemical and structural properties of compounds that end up directly influencing their spectroscopic properties.27 When considering the interactions between probe-solvent, deviations from linearity may be called "preferential solvation" of the probe for one of the components of the mixture.27,50,51 The solvation parameters as well as the local mole fraction (X1L and X2L) and preferential solvation constant (K12) are used to study the preferential solvation. The X1L and X2L parameters are the mole fraction of solvent 1 and solvent 2, respectively, in the cybotactic region of the dye and were calculated from:51
where ν1 and ν2 are the emission maximum expressed in wavenumber of solvent 1 and solvent 2 and ν2 is the emission maximum of binary mixtures in cm-1.
The preferential solvation constant (K12) was calculated according to the equation 6:
where K12 > 1 indicates a preference for solvent 2 over solvent 1 and if K12 < 1 indicates a preference for solvent 1 over solvent 2.51,52
The preferential solvation may include contributions from specific interactions between probe-solvent (for example, hydrogen bonds and hydrophobic interactions) or may be due to different interactions in the existing microdomains dependent on the mole fraction of the solvent due to the micro-heterogeneity of the mixtures.27 The preferential solvation data for coumarin, C1, C151 and C500 in the binary mixtures are given in Table S1 in the SI section. In the case of water-ACN mixture, despite the high solubility of ACN in water, this mixture has a micro-heterogeneous characteristic showing different regions dependent on the physicochemical properties of each component.26 In ACN-water mixture, K12 values are greater than 1, indicating that the solute is preferentially solvated by acetonitrile than water. The existence of clusters in the mixtures is essential for these results. Micro-heterogeneous structure (clusters) formed between the solvents in the mixtures at the molecular level can explain the behavior of specific thermodynamic and physicochemical properties of the mixture and consequently have a large influence on the photophysical data obtained.53 Regarding the mixture of EtOH and water, the results obtained may be dependent on a strong interaction between the solvents. For C151, in all mixture proportions, and for C1 and C500 in higher concentrations of water, the K12 values are smaller than 1, indicating a preference for water. These interactions occur through the formation of hydrogen bonds.54 In the presence of Diox-water mixtures, for all compounds studied, the K12 values are greater than 1, which indicates a preference for Diox. In the presence of higher concentration of water, in the Diox-water mixtures, C151 and C500 showed a preferential solvation for water.
The formation of clusters changes the type of coumarin-water-organic solvent interactions, modifying the kinetics of deactivation of the excited state of the probe. These factors associated with the different abilities of hydrogen bond formation and medium polarity would generate the very peculiar solvatochromic effect observed in the measurements involving these coumarin-derivatives.
Conclusions
Results showed the complexity of the solvatochromic behavior of coumarin and the relevance of the studies focused on the mixture of solvents in order to understand the physicochemical properties of this organic compound. Coumarin-derivatives with higher non-polar character present higher tendency to generate high quantum yield in the presence of non-polar solvents, while coumarin-derivatives with higher polar character would present higher quantum yield with more polar solvents. In any case, some solvent mixtures, such as ACN-water, present very peculiar properties, such as the predominance of the solvent-solvent interaction compared to the solute-solvent mixture interaction. This competition is associated with more differentiated spectroscopic behaviors observed in some solvent associations such as the ACN-water system, which seems to be the most complex solvent system, probably due to the characteristic interaction involving the molecules of ACN and water. The shifts in the emission bands with solvent polarity changes are more pronounced than those of absorption bands, which is an indication of higher dipole moments in the excited state than in the ground state. Thus, from the data presented concerning the properties of the binary mixtures studied in this work, we can say that the dependence of these properties on the composition of each solvent mixture is not linear, being dependent on the type of interaction between the solvents. This behavior can be explained based on microscopic structures of clusters. In fact, a series of micro-heterogeneous systems occurred in the different solvent mixtures evaluated. The understanding of these results, which involve the learning of weak interactions, as well as the nature of intra-and intermolecular physicochemical studies, is highly important for further research.
Supplementary Information
Supplementary data are available free of charge at http://jbcs.sbq.org.br as PDF file.
Acknowledgements
The authors wish to thank CNPq (479655/2008-1 and 474019/2012-8), FAPESP (08/50588-6 and 06/56701-3), and L'Oréal/Academia Brasileira de Ciências/Unesco (Grant for Women in Science 2007) for the financial support.
Submitted: November 11, 2013
Published online: March 14, 2014
FAPESP has sponsored the publication of this article.
Supplementary Data
The supplementary data is available in pdf: [Supplementary data]
- 1. Reddy, T. S.; Reddy, A. R.; Dyes Pigm. 2013, 96, 525.
- 2. Hoult, J. R; Payá, M.; Gen. Pharmacol 1996, 27, 713.
- 3. Lake, B. G.; Food Chem. Toxicol. 1999, 37, 423.
- 4. Sharma, V. K.; Saharo, P. D.; Sharma, N.; Rastogi, R. C.; Ghoshal, S. K.; Mohan, D.; Spectrochim. Acta, Part A 2003, 59, 1161.
- 5. Rosskoph, F.; Kraus, J.; Franz, G.; Pharmazie 1992, 47, 139.
- 6. Kirkiacharian, S.; Thuy, D. T.; Sicsic, S.; Bakhchinian, R.; Kurkjian, R.; Tonnaire, T.; Farmaco 2002, 57, 703.
- 7. Brar, S. S.; Kennedy, T. P.; Whorton, A. R.; Am. J. Physiol. 2001, 280, 659.
- 8. Bhattacharya, S.; Banerji, J.; Ghosh, P. C.; Indian J. Chem. 1977, 15B, 214.
- 9. Hierrezuelo, J. M.; Ruiz, C. C.; J. Phys. Chem. A 2012, 116, 12476.
- 10. Nad, S.; Pal, H.; J. Phys. Chem. A 2003, 107, 501.
- 11. Gustavsson, T.; Cassara, L.; Gulbinas, V.; Gurzadyan, G.; Mialocq, J. C.; Pommeret, S.; Sorgius, M.; Van der Meulen, P.; J. Phys. Chem. A 1998, 102, 4229.
- 12. Rychlik, M.; J. Agric. Food Chem. 2008, 56, 796.
- 13. Wang, W.; Tang, J.; Wang, S.; Zhou, L.; Hu, Z.; J. Chromatogr. A 2007, 1148, 108.
- 14. Wang, L. H.; Lien, C. L.; J. Liq. Chromatogr. Relat. Technol. 2004, 27, 3077.
- 15. Jager, L. S.; Perfetti, G. A.; Diachenko, G. W.; J. Chromatogr. A 2007, 1145, 83.
- 16. Kollroser, M.; Schober, C.; Clin. Chem. 2002, 48, 84.
- 17. Marcolan, M.; Martins, P. A.; Pedrosa, V. A.; Magini, M. R. R.; Oliveira, H. P. M.; Codognoto, L.; J. Fluoresc. 2011, 21, 733.
- 18. Sotomayor, M. D. P.; Dias, I. L. T.; Lanza, M. R. V.; Moreira, A. B.; Kubota, L. T.; Quim. Nova 2008, 31, 1755.
- 19. Krystkowiak, E.; Dobek, K.; Burdziński, G.; Maciejewski, A.; Photochem. Photobiol. Sci. 2012, 11, 1322.
- 20. Homocianu, M.; Airinei, A.; Dorohoi, D. O.; J. Adv. Res. Phys. 2011, 2, 1.
- 21. Lakowicz, J. R.; Principles of Fluorescence Spectroscopy; Plenum Press: New York, 1983.
- 22. Dahiya, P.; Kumbhakar, M.; Mukherjee, T.; Pal, H.; Chem. Phys. Lett. 2005, 414, 148.
- 23. Mannekutla, J. R.; Mulimani, B. G.; Inamdar, S. R.; Spectrochim. Acta, Part A 2008, 69, 419.
- 24. Cigán, M.; Donovalová, J.; Szöcs, V.; Gaspar, J.; Jakusová, K.; Gáplovský, A.; J. Phys. Chem. A 2013, 117, 4870.
- 25. Giri, R.; Curr. Sci. 1992, 62, 522.
- 26. Catalán, J.; Díaz, C.; Blanco, F. G.; Org. Biomol. Chem. 2003, 1, 575.
- 27. Seoud, O. A. E.; Pure Appl. Chem. 2009, 81, 697.
- 28. Wakisaka, A.; Abdoul-Carime, H.; Yamamoto, Y.; Kiyozumi, Y.; J. Chem. Soc., Faraday Trans. 1998, 94, 369.
- 29. Hovorka, F.; Schaefer, R. A.; Dreisbach, D.; J. Am. Chem. Soc. 1936, 58, 2264.
- 30. Satpati, A. K.; Kumbhakar, M.; Maity, D. K.; Pal, H.; Chem. Phys. Lett. 2005, 407, 114.
- 31. Pal, H.; Nad, S.; Kumbhakar, M.; J. Chem. Phys. 2003, 119, 443.
- 32. Reichardt, C.; Chem. Rev. 1994, 94, 2319.
- 33. Tada, E. B.; Silva, P. L.; Tavares, C.; Seoud, O. A. E; J. Phys. Org. Chem. 2005, 18, 398.
- 34. Gupta, S.; Rafiq, S.; Kund, M.; Sem, P.; J. Phys. Chem. B 2012, 116, 1345.
- 35. Ortega, J.; Ràfols, C.; Bosch, E.; Rosés, M.; J. Chem. Soc., Perkin Trans. 2 1996, 2, 1497.
- 36. Reichardt, C.; Pure Appl. Chem. 2004, 76, 1903.
- 37. Catalán, J.; Paz, J. L. G.; J. Phys. Chem. A 2010, 114, 6226.
- 38. Sun, M.; Song, P. S.; Photochem. Photobiol 1977, 25, 3.
- 39. Nad, S.; Pal, H.; J. Phys. Chem. A 2001, 105, 1097.
- 40. Mataga, N.; Kaifu, Y.; Koizumi, M.; Bull. Chem. Soc. Jpn. 1956, 29, 465.
- 41. Suppan, P.; Chem. Phys. Lett. 1983, 94, 272.
- 42. Moreau, C.; Douhéret, G.; J. Chem. Thermodyn 1976, 8, 403.
- 43. Smallwood, I. M.; Handbook of Organic Solvent Properties; John Wiley & Sons: New York, 1996.
- 44. Faraji, M.; Farajtabar, A.; Gharib, F.; J. Appl. Chem. Res. 2009, 9, 7.
- 45. Akerlof, G.; Short, O. A.; J. Am. Chem. Soc. 1936, 58, 1241.
- 46. Aminabhav, T. M.; Gopalakrishna, B.; J. Chem. Eng. Data 1995, 40, 856.
- 47. Arce, A.; Blanco, A.; Soto, A.; Souza, P.; Vidal, I.; Fluid Phase Equilib. 1993, 87, 347.
- 48. Sidir, Y. S.; Sidir, I.; Spectrochim. Acta, Part A 2013, 102, 286.
- 49. Singh, T. S.; Mitra, S.; J. Lumin. 2007, 127, 508.
- 50. Das, P. K.; Pramanik, R.; Bagchi, S.; Spectrochim. Acta, Part A 2003, 59, 1681.
- 51. Suganthi, G.; Sivakolunthu, S.; Ramakrishnan, V.; J. Fluoresc. 2010, 20, 1181.
- 52. Sasirekha, V.; Vanelle, P.; Terme, T.; Ramakrishman, V.; J. Fluoresc. 2008, 9, 428.
- 53. Wakisaka, A.; Matsuura, K.; J. Mol. Liq. 2006, 129, 25.
- 54. Petong, P.; Pottel, R.; Kaatze, U.; J. Phys. Chem. A 2000, 104, 7420.
Publication Dates
-
Publication in this collection
30 May 2014 -
Date of issue
May 2014
History
-
Accepted
14 Mar 2014 -
Received
11 Nov 2013