Abstracts
Selective genotyping for a certain trait in individuals with extreme phenotypes contributes sufficient information to determine linkage between molecular markers and quantitative trait loci (QTL). In this experiment an F2 population, developed by crossing males from a broiler line with females from a layer line, was employed to detect QTL on chromosomes 3 and 5. Twenty-eight performance and carcass traits were measured in F2 offspring, and phenotypic correlations between traits were calculated. Body weight at 42 days (BW42) presented the greatest positive correlations with most other traits, with correlation between body weights at 35 and 41 days, weight gain between birth and 35, 41 and 42 days, as well as weights of carcass and some body parts superior to 0.8. One hundred-and-seventy F2 offspring, representing the top (4.5%) and the bottom (4.5%) of a normal distribution curve of BW42, were selected with equal proportions of males and females, and within dam family. Samples were genotyped for 19 informative markers on chromosome 3, and 11 markers on chromosome 5. Marker allelic frequencies of phenotypic groups with high and low BW42 were compared with a chi-square test. Four regions on chromosome 3 and three regions on chromosome 5 had markers that were suggestively associated with BW42 (P < 0.10), confirming and expanding previous studies.
QTL; broiler; selective genotyping; body weight
A genotipagem seletiva de indivíduos com fenótipos extremos para uma determinada característica contribui com informação suficiente para determinar a ligação entre marcadores moleculares e locos para características quantitativas (QTL). Neste estudo uma população F2, formada a partir do cruzamento de uma linha parental de aves para corte com uma linha de postura foi empregada para obtenção de medidas fenotípicas e genotipagem por marcadores microssatélites, posicionados nos cromossomos 3 e 5. Foram medidas 28 características de desempenho e carcaça e determinada a correlação fenotípica entre elas. A característica peso vivo aos 42 dias (BW42) apresentou maior correlação positiva com a maioria das características, com correlação entre pesos vivos aos 35, 41 dias, ganhos de peso do nascimento aos 35, 41 e 42 dias, e pesos de carcaça e partes superiores a 0,8. Cento e setenta aves F2, representando 4,5% das aves mais leves e 4,5% das mais pesadas para BW42 foram selecionadas dentro de famílias, na mesma proporção de machos e fêmeas e genotipadas para 19 marcadores informativos no cromossomo 3 e 11 no cromossomo 5. As freqüências alélicas dos marcadores nos grupos fenotípicos de alto e baixo BW42 foram comparadas empregando teste de qui-quadrado. Foram identificadas quatro regiões no cromossomo 3 e três regiões no cromossomo 5 sugestivamente ligadas a QTL para BW42 (P < 0,10), confirmando e expandindo estudos anteriores de mapeamento de QTL em aves.
QTL; aves; genotipagem seletiva; ganho de peso
ANIMAL SCIENCE AND PASTURES
Strategic marker selection to detect quantitative trait loci in chicken
Seleção estratégica de marcadores para detecção locos para características quantitativas em aves
Deborah Clea RuyI, II; Kátia NonesI; Erica Elias BaronI; Mônica Corrêa LedurIII; Cláudio Manoel Rodrigues de MeloI; Marcel AmboI; Raquel de Lello Rocha CamposI; Luiz Lehmann CoutinhoI, * * Corresponding author < llcoutin@esalq.usp.br>
IUSP/ESALQ - Depto. de Zootecnia, C.P. 9 - 13418-900 - Piracicaba, SP- Brasil
IIUnB - Faculdade de Agronomia e Medicina Veterinária, C.P. 4508 - 70910-970 - Brasília, DF-Brasil
IIIEmbrapa Suínos e Aves, C.P. 21 - 89000-700 - Concórdia, SC - Brasil
ABSTRACT
Selective genotyping for a certain trait in individuals with extreme phenotypes contributes sufficient information to determine linkage between molecular markers and quantitative trait loci (QTL). In this experiment an F2 population, developed by crossing males from a broiler line with females from a layer line, was employed to detect QTL on chromosomes 3 and 5. Twenty-eight performance and carcass traits were measured in F2 offspring, and phenotypic correlations between traits were calculated. Body weight at 42 days (BW42) presented the greatest positive correlations with most other traits, with correlation between body weights at 35 and 41 days, weight gain between birth and 35, 41 and 42 days, as well as weights of carcass and some body parts superior to 0.8. One hundred-and-seventy F2 offspring, representing the top (4.5%) and the bottom (4.5%) of a normal distribution curve of BW42, were selected with equal proportions of males and females, and within dam family. Samples were genotyped for 19 informative markers on chromosome 3, and 11 markers on chromosome 5. Marker allelic frequencies of phenotypic groups with high and low BW42 were compared with a chi-square test. Four regions on chromosome 3 and three regions on chromosome 5 had markers that were suggestively associated with BW42 (P < 0.10), confirming and expanding previous studies.
Key words: QTL, broiler, selective genotyping, body weight
RESUMO
A genotipagem seletiva de indivíduos com fenótipos extremos para uma determinada característica contribui com informação suficiente para determinar a ligação entre marcadores moleculares e locos para características quantitativas (QTL). Neste estudo uma população F2, formada a partir do cruzamento de uma linha parental de aves para corte com uma linha de postura foi empregada para obtenção de medidas fenotípicas e genotipagem por marcadores microssatélites, posicionados nos cromossomos 3 e 5. Foram medidas 28 características de desempenho e carcaça e determinada a correlação fenotípica entre elas. A característica peso vivo aos 42 dias (BW42) apresentou maior correlação positiva com a maioria das características, com correlação entre pesos vivos aos 35, 41 dias, ganhos de peso do nascimento aos 35, 41 e 42 dias, e pesos de carcaça e partes superiores a 0,8. Cento e setenta aves F2, representando 4,5% das aves mais leves e 4,5% das mais pesadas para BW42 foram selecionadas dentro de famílias, na mesma proporção de machos e fêmeas e genotipadas para 19 marcadores informativos no cromossomo 3 e 11 no cromossomo 5. As freqüências alélicas dos marcadores nos grupos fenotípicos de alto e baixo BW42 foram comparadas empregando teste de qui-quadrado. Foram identificadas quatro regiões no cromossomo 3 e três regiões no cromossomo 5 sugestivamente ligadas a QTL para BW42 (P < 0,10), confirmando e expandindo estudos anteriores de mapeamento de QTL em aves.
Palavras-chave: QTL, aves, genotipagem seletiva, ganho de peso
INTRODUCTION
Quantitative trait loci (QTL) are genomic regions in which genes affecting quantitative characteristics are detected. The probability of detecting a QTL depends on linkage disequilibrium between a molecular marker and the QTL. The greater the difference among the lines used to form an experimental population the greater the linkage disequilibrium (Van Arendonk et al., 1994). The power to detect a QTL is directly related to the number of individuals genotyped; however, genotyping a large number of individuals for informative markers is very costly and may be viable for only a small population sample. Nevertheless, a random sample may result in a low power for QTL detection and thus only detect QTL that singularly explain more than 10% of the phenotypic variance (Muranty et al., 1997). Selective genotyping increases QTL detection power. This method entails genotyping only the individuals whose phenotypic values fall within the upper or lower tails of a normal distribution curve of an entire population sample, regardless of the number of animals phenotyped (Lander & Botstein, 1989; Darvasi & Soller, 1992; Muranty et al., 1997). Selective genotyping increases efficiency of QTL detection as much for characteristics for which the individuals are selected as for other correlated characteristics. On the other hand, detection accuracy of non-correlated characteristics is similar to random selection (Muranty & Goffinet, 1997). Mapping studies in poultry have identified QTL for performance, carcass and immune response characteristics (Vallejo et al., 1998, Van Kaam et al., 1998, 1999a, 1999b, Ikeobi et al., 2002 and Sewalem et al., 2002). The objective of the present study was to find linkage marker-QTL for body weight at 42 days of age on poultry chromosomes 3 and 5 using selective genotyping in a Brazilian F2 population.
MATERIAL AND METHODS
Experimental population
The present experiment utilized an experimental population in F2 design that was developed by Embrapa Swine and Poultry (Ledur et al., 2000). The first generation of this population was obtained by crossing seven males from a parent broiler line (TT) with each of seven females from a layer line (CC). Seven F1 males (TC) were selected and mated with each of three non-related F1 females (TC) for a total of 21 females. Approximately 100 offspring per female were generated after 17 incubations for a total of 2,063 F2 birds (Table 1) that were raised as broilers and slaughtered at 42 days.
Phenotypic Measures
Performance-related measures were taken or calculated after hatching at the following developmental stages in F2 animals: birth weight (BW), body weight at 35 days (BW35), 41 days (BW41) and 42 days (BW42) after transport to slaughterhouse; weight gain from birth to 35 days (WG35), birth to 41 days (WG41), birth to 42 days (WG42). Measurements from the 35th to the 41st days were taken for weight gain (WG35-41); feed intake (FI), feed conversion (FC) and feed efficiency (FE). After slaughter, measurements were taken for carcass weight including breast, back, wings, thighs and legs (CAR), individual weights for breast (BRT), back (BAK), both wings (WNG), both thighs and legs (THL), both feet (FET), head (HED), liver (LVR), gizzard (GZD), heart (HRT), lungs (LNG), abdominal fat (ABF); intestine length (INT) and percentages of carcass (PCAR), breast (PBRT) and abdominal fat (PABF). Hematocrit value (HEMAT) was also determined for each F2 bird.
DNA extraction, marker selection and genotyping
Blood was collected in tubes containing EDTA to prevent coagulation. Samples were then stored at -70ºC until use. Blood was extracted from the brachial vein in parents and through bleeding at slaughter in F1 and F2 animals. Genomic DNA was extracted using DNAzol® reagent (Invitrogen Life Technologies) following manufacturer's recommendations with modifications suggested by Campos et al. (2003). All samples were standardized for a final concentration of 20 ng µL-1 Afterwards, each DNA sample was submitted to electrophoresis in 1.0% agarose gel to verify DNA integrity.
Michigan State University, USA, provided primers that were part of the U.S. National Animal Genome Research Program (http//:genome.iastate.edu). DNA was amplified using PCR with reactions and temperature cycles according to the protocols described by Cheng et al. (1995). Annealing temperatures varied between 47ºC and 59ºC, depending on the primer-pair utilized. Amplifications were conducted in two thermal cyclers (MJ Research models PTC-100 and PTC-200).
Nineteen and eleven microssatelite markers, respectively distributed along chromosomes 3 and 5, were used. A MegaBace capillary sequencer (Amersham Biosciences) was used for genotyping. Up to three markers with different fragment size and fluorescence were combined. Samples were precipitated and resuspended with 4.75 µL loading solution (Amersham Biosciences) and 0.25 µL internal standard ET-ROX 400 (Amersham Biosciences). Samples were stored at -20ºC until shortly before use when they were denatured at 95ºC for one minute. Samples were run for a maximum of 100 minutes with pre-run injection conditions being 2 kV and run conditions of 9 kV. GENETIC PROFILER program (Amersham Biosciences) was employed to determine fragment size.
Statistical Analysis
An exploratory data analysis eliminated 108 animals due to unavailability of an F1 mother's blood. An additional eight measures with variations of more than four standard deviations for each characteristic were discarded. The latter were considered either true outliers or recording errors. Outliers were randomly distributed among the twenty-one families, indicating that no genetic component was involved. Statistical analysis considered phenotypic values of the remaining 1,859 birds (925 females and 934 males).
Phenotypic correlations among characteristics were calculated using the CORR procedure (SAS, 2001) to determine which characteristics were positively correlated with the greatest possible number of other characteristics. After choosing a characteristic for selective genotyping, values for this characteristic were analyzed separately in F2 males and females with GLM (SAS, 2001) to exclude the effect of sex on dependent variables. Phenotypic values were adjusted for incubation effect. After normal distribution curves were determined for each family, four-and-a-half percent of the top and bottom tails were selected from each family for genotyping. Number varied between six and ten birds per family depending on the number of offspring per F1 couple.
In the statistical analysis of selective genotyping, allele marker frequencies of extreme phenotypes (tails of a normal distribution curve) were compared with a chi-square test, using the following equation:
in which,
foi and fei are the observed and expected frequencies of allele i, in a total of k alleles. The null hypothesis (H0) assumes equal allele frequencies among groups. Analyses utilized PROC FREQ of SAS (2001). Markers with significance levels, P < 0.10, were considered to be suggestively linked to a QTL.
RESULTS AND DISCUSSION
Among various correlated characteristics, BW42 was chosen as a variable in selective genotyping, since it presented the greatest general correlation with the other characteristics. In addition, BW42 is a primary consideration in broiler selection. Correlation values for BW42 with BW35, BW41, WG35, WG41, WG42, CAR, BRT, BAK, WNG, THL, HED, FET and LVR were greater than 0.80. The mean (standard deviation) for BW42 was 987.29 g (± 183.70) for all F2 birds.
A model based on father, mother, incubation and sex was chosen to test the existence of significant effect on phenotype, considering probability values of H0 given by type III sum of squares, indicated for analysis of unbalanced data. With proven effects of the mother, incubation and sex (P < 0.001), males and females were separated into distinct files to exclude the sex effect. Data was adjusted solely for incubation (BW42ajust); no corrections were made for mother effects, and selection was performed within families. Choice of birds to be selectively genotyped was made within families of F1 mothers, following recommendations of Hillel (1997) for crosses among commercial poultry lines, typically not endogamic. If families are not considered, individuals from different families may be over-represented in the extremes of a normal distribution (Muranty et al., 1997). Considering males and females together, birds chosen for selective genotyping from normal distribution curves for the variable BW42ajust presented means (standard deviation) of 801.5 g (± 93.8) and 1328.5 g (± 127.8) for birds considered lighter and heavier, respectively.
Informative markers were identified through evaluation of markers in F1 couples. Allelic evaluation of 17 markers amplified on chromosome 3 and ten markers amplified on chromosome 5 respectively, determined heterozygosity to be 0.57 and 0.72, polymorphism information content PIC to be 0.51 and 0.66, and the mean number of alleles per marker to be 4.71 and 6.89. Monomorphic, slightly polymorphic or difficult to amplify markers were discarded. Informative markers were genotyped in parents and in 170 F2 individuals from phenotypic extremes.
To reduce the distance between markers initially selected, additional markers were genotyped. For these new markers, selection was based solely on position on the chromosome. Selective genotyping on chromosome 3 utilized 19 informative markers with medium spacing (16.7 centiMorgans cM) in agreement with the estimated position on the consensus map (Groenen et al., 2000). The greatest interval was 36 cM and the least 3 cM, with estimated coverage of 341 cM, supposing that the markers positioned at the extremes would cover up to 20 cM for each side. Selective genotyping on chromosome 5 was completed with eleven markers with average spacing among them of 21.3 cM varying between 47 cM and 4 cM with estimated coverage of 222 cM. Coverage obtained includes the entire extension of the chromosomes with lengths given in the consensus map of 317 and 198 cM for chromosomes 3 and 5, respectively. The greatest distance present among the markers was located at the end of chromosome 5 between markers ADL 233 and ADL 298. The only marker available in this region, ADL166 would still maintain spacing greater than 30 cM (Groenen et al., 2000). Nevertheless, this marker was not polymorphic in our population.
The frequency of alleles observed for each marker within the light and heavy groups was compared with a chi-square test. Seeking to avoid false negatives, P < 0.10 was considered significant. This significance level is in agreement with levels employed in previous studies involving selective genotyping. Vallejo et al. (1998) investigated the presence of a QTL affecting susceptibility to Marek's disease by selective genotyping of 80 individuals from a total of 272 F2 progeny. Associations were made among phenotypes of these progenies for each marker locus tested utilizing analysis of variance. A P value < 0.20 was used to determine suggestive linkage with QTL. Yonash et al. (2001) studying immunological response to inoculation with Escherichia coli in broilers, genotyped 40% of the experimental population. Markers that did not indicate effect of the characteristics under study (P < 0.10) were discarded in later genotyping of the entire F2 and backcross populations. Studies to identify QTL in laboratory animals also utilize selective genotyping. Using mouse lines, Moody et al. (1999) sought to identify QTL related to energetic balance by selective genotyping animals from part of the population; markers with chi-square values > 2.71 (P < 0.10) were considered suggestively linked to QTL for the characteristic.
Positions considered in the statistical analysis for selective genotyping were the ones presented in consensus map 2000 (Groenen et al., 2000) for markers LEI 43, MCW 169, LEI 118, ADL 127 and MCW 116 on chromosome 3 and MCW 193, MCW 90, LEI 145 and ADL 298 on chromosome 5. Remaining markers on the two chromosomes were positioned according to their maps of origin.
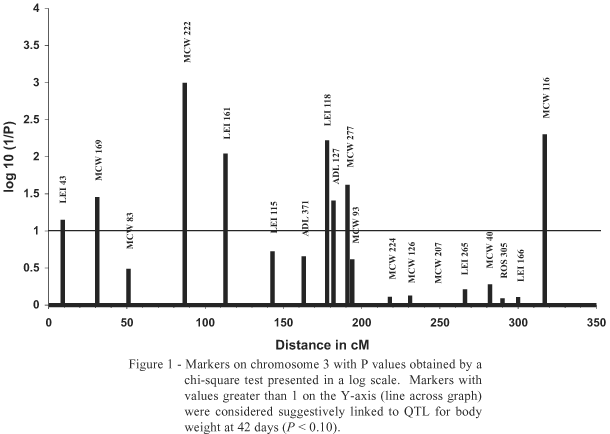
Four regions on chromosome 3 were observed to concentrate eight markers indicating suggestive marker linkage with QTL for BW42ajust (Figure 1). The first region is represented by LEI 43 (P = 0.071) and MCW 169 (P = 0.035) positioned at 9 and 31 cM, respectively (positions given on the consensus map). The region near marker MCW 169 coincides with the position described by McElroy et al. (2002), who identified QTL for body weight at six weeks after transport to slaughter.
The second region includes markers MCW 222 (P = 0.001) and LEI 161 (P = 0.009) positioned at 87 and 113 cM, respectively. No QTL was previously mapped in this region for performance and carcass characteristics that may be related to BW42.
The third region observed on chromosome 3 presented three close markers, LEI (P = 0.006), ADL 127 (P = 0.039) and MCW 277 (P = 0.024) located at 178, 182 and 191 cM, respectively. QTL for body weight had not been previously found in this region, nevertheless one QTL for conformation score was mapped by McElroy et al. (2002) at 188 cM.
In an experiment employing a population developed from layer lines, Tuiskula-Haavisto et al. (2002) found a QTL that was significant for age at first egg in the region between 153 and 201 cM. Although this characteristic was not evaluated in the present study, it is positively correlated with body weight in laying hens.
McElroy et al. (2002) detected QTL significant for body weight at slaughter and weights of half-carcass, white meat and fillet located at 154 cM on chromosome 3. In spite of two informative markers existing in this region, LEI 115 at 143 cM and ADL 371 at 163 cM, they were not significantly or suggestively linked to QTL and, at least in the present analysis of selective genotyping, these reported QTL were not confirmed.
The fourth region was related to marker MCW 116 (P = 0.005), located at 310 cM. In spite of not being previously described as a significant or suggestive QTL at this position, Van Kaam et al. (1999b) indicated the region positioned between markers LEI 166 and MCW 166 as potential for identifying QTL for BW48 and for carcass weight, since these authors found a significance level very near the suggestive level.
On chromosome 5, three regions were located where four markers with suggestive linkage with QTL for BW42ajust were found (Figure 2). The first region comprises markers MCW 193 (P = 0.067) and MCW 90 (P = 0.001) at 50 and 57 cM, respectively. McElroy et al. (2002) found linkage between MCW 193 and QTL, suggestive at 10%, for nine distinct characteristics including body weight at six week both pre- and post-transportation. However, these authors employed only this marker in all of chromosome 5, and detection of linkage between marker and QTL at significant levels depends, among other factors, on distance and degree of marker information. Using a similar population based on a broiler x layer line, Sewalen et al. (2002) mapped QTL for body weight at three weeks at 58 cM, coinciding with the region associated in this study to QTL for BW42.
Tuiskula-Haavisto et al. (2002), evaluating a layer population, found a QTL significant for egg specific gravity located between 32 and 112 cM on chromosome 5; however, it was not possible to establish a relationship between this characteristic and others evaluated in the present study.
The other two regions suggestively linked to QTL for BW42 on chromosome 5 were located at 151 cM and 198 cM, where markers ADL 233 (P = 0.071) and ADL 298 (P = 0.037) are respectively located. There is no previous mention of QTL for performance and carcass quality in these two regions; although marker ADL 298 has already been associated with immune response in an experiment conducted by Yonash et al. (2001).
CONCLUSIONS
Selective genotyping conducted in the present experiment allowed identification of four regions on chromosome 3 and three regions on chromosome 5 with suggestive marker-QTL linkage coinciding with QTL identified in other studies of different populations, thus indicating that this is a valuable strategy to conserve resources.
Some of the QTL regions identified in this work were associated in different studies with characteristics other than body weight, indicating that selective genotyping can possible identify QTL for characteristics that are correlated to the one use for selective genotyping.
Selective genotyping is an efficient tool for initially selecting markers, thus reducing cost of QTL experiments. Nevertheless, this process should be accompanied with later genotyping of the selected regions in more animals, so that an interval analysis may be conducted with various markers to more precisely define the location and the effect of the QTL.
ACKNOWLEDGEMENTS
Embrapa, PRODETAB and FUNCITEC for financial support of the project. CAPES for PICDT scholarship to the first author, and CNPq for a research productivity scholarship to L.L.C. and a post-doc scholarship to C.M.R.M. FAPESP for a doctorate scholarship to K.N. and E.E.B., and ESALQ for scholarships to M.A. and R.L.R.C., as well as Dr. Hans H. Cheng at Michigan State University for providing the primers used in this study.
Received April 08, 2004
Accepted December 23, 2004
- CAMPOS, R.L.R.; AMBO, M.; NONES, K.; RUY, D.C.; BARON, E.E.; LEDUR, M.C.; COUTINHO, L.L. Otimização e comparação de protocolos para extração de DNA de sangue de aves. In: CONGRESSO NACIONAL DE GENÉTICA, 49., Águas de Lindóia, 2003. Programa Águas de Lindóia: SBG, 2003. p.19.
- CHENG, H.H.; LEVIN, I.; VALLEJO, R.L.; KHATIB, H.; DODGSON, J.B.; CRITTENDEN, L.B.; HILLEL, J. Development of a genetic map of the chicken with markers of high utility. Poultry Science, v.74, p.1855-1874, 1995.
- DARVASI, A.; SOLLER, M. Selective genotyping for determination of linkage between a marker locus and a quantitative trait locus. Theoretical and Applied Genetics, v.85, p.353-359, 1992.
- GROENEN, M.A.M.; CHENG, H.H.; BUMSTEAD, N.; BENKEL, B.F.; BRILES, W.E.; BURKE, T.; BURT, D.W.; CRITTENDEN, L.B.; DODGSON, J.; HILLEL, J.; LAMONT, S.; PONCE DE LEON, A.; SOLLER, M.; TAKAHASHI, H.; VIGNAL, A. A consensus linkage map of the chicken genome. Genome Research, v.10, p.137-147, 2000.
- HILLEL, J. Map-based quantitative trait locus identification. Poultry Science, v.76, p.1115-1120, 1997.
- IKEOBI, C.O.N.; WOOLILIAMS, J.A.; MORRICE, D.R.; LAW, A.; WINDSOR, D.; BURT, D.W.; HOCKING, P.M. Quantitative trait loci affecting fatness in the chicken. Animal Genetics, v.33, p.428-435, 2002.
- LANDER, E.S.; BOTSTEIN, D. Mapping mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics, v.121, p.185-199, 1989.
- LEDUR, M.C.; ZANELLA, E.L.; SCHMIDT, G.S.; JAENISCH, F.R.F.; SAATKAMP, M.G.; BASSI, L.J.; COUTINHO, L.L. Peso e características de carcaça em linhagens utilizadas no desenvolvimento de populações referência para detecção de QTL em aves. Revista Brasileira de Ciência Avícola, Supl.2, p.73, 2000.
- McELROY, J.P.; HARRY, D.E.; DEKKERS, J.C.M.; LAMONT, S.J. Molecular markers associated with growth and carcass traits in meat-type chickens. In: WORLD CONGRESS ON GENETICS APPLIED TO LIVESTOCK PRODUCTION, 7., Montpellier, 2002. Papers Montpellier, 2002. (Session, 4: Poultry and rabbit breeding). 1 CD-ROM.
- MOODY, D.E.; POMP, D.; NIELSEN, M.K.; VAN VLECK, L.D. Identification of quantitative trait loci influencing traits related to energy balance in selection and inbred lines of mice. Genetics, v.152, p.699-711, 1999.
- MURANTY, H.; GOFFINET, B. Selective genotyping for location and estimulation of the effect of a quantitative trait locus. Biometrics, v.53, p.629-643, 1997.
- MURANTY, H.; GOFFINET, B.; SANTI, F. Multitrait and multipopulation QTL search using selective genotyping, Genetic Research of Cambridge, v.70, p.259-265, 1997.
- SAS INSTITUTE. SAS/STAT 2001: user's guide: statistics version 8.2. Cary, 2001. 1 CD-ROM.
- SEWALEM, A.; MORRICE, D.M.; LAW, A.; WINDSOR, D.; Haley, C.S.; IKEOBI, C.O.N.; BURT, D.W.; HOCKING, P.M. Mapping of quantitative trait loci for body weight at three, six, and nine weeks of age in a broiler layer cross. Poultry Science, v.81, p.1775-1781, 2002.
- TUISKULA-HAAVISTO, M.; HONKATUKIA, M.; KONING, D.J.; SCHULMAN, N.F.; MÄKI-TANILA, A. Mapping of quantitative trait loci affecting quality and production traits in egg layers. Poultry science, v.81, p.919-927, 2002.
- VALLEJO, R.L.; BACON, L.D.; LIU, H.C.; WITTER, R.L.; GROENEN, M.A.M.; HILLEL, J.; CHENG, H.H. Genetic mapping of quantitative trait loci affecting susceptibility to marek's disease virus induced tumors in F2 intercross chickens. Genetics, v.148, p.349-360, 1998.
- VAN ARENDONK, J.A.M.; BOVENHUIS, H.; VAN DER BEEK, S.; GROEN, A.F. Detection and exploitation of markers linked to quantitative traits in farm animals. In: WORLD CONGRESS OF GENETICS AND LIVESTOCK PRODUCTION, 5., Guelph, 1994. Proocedings Guelph: University of Guelph, 1994. p.193-200.
- VAN KAAM, J.B.C.H.M.; VAN ARENDONK, J.A.M.; GROENEN, M.A.M.; BOVENHUIS, H.; VEREIJKEN, A.L.J.; CROOIJMANS, R.P.M.A.; VAN DER POEL, J.J.; VEENENDAAL, A. Whole genome scan for quantitative trait loci affecting body weight in chickens using a three generation design. Livestock Production Science, v.54, p.133-150, 1998.
- VAN KAAM, J.B.C.H.M.; GROENEN, M.A.M.; BOVENHUIS, H.; VEENENDAAL, A.; VEREIJKEN, A.L.J.; VAN ARENDONK, J.A.M. Whole genome scan in chickens for quantitative trait loci affecting growth and feed efficiency. Poultry Science, v.78, p.15-23, 1999a.
- VAN KAAM, J.B.C.H.M.; GROENEN, M.A.M.; BOVENHUIS, H.; VEENENDAAL, A.; VEREIJKEN, A.L.J.; VAN ARENDONK, J.A.M. Whole genome scan in chickens for quantitative trait loci affecting carcass traits. Poultry Science, v.78, p.1091-1099, 1999b.
- YONASH, N.; CHENG, H.H.; HILLEL, J.; HELLER, D.E.; CAHANER, A. DNA microsatellites linked to quantitative trait loci affecting antibody response and survival rate in meat-type chickens. Poultry Science, v.80, p.22-28, 2001.
Publication Dates
-
Publication in this collection
08 Apr 2005 -
Date of issue
Apr 2005
History
-
Accepted
23 Dec 2004 -
Received
08 Apr 2004