ABSTRACT
Portable X-ray fluorescence spectrometer (pXRF) has been recently adopted by the Soil Science community for uses in both field and laboratory, obtaining the total content of several chemical elements in a few seconds. Sulfuric acid digestion is an expensive and time-consuming laboratory analysis that provides contents of Fe2O3, Al2O3, SiO2, TiO2 and P2O5, important for soil studies. Due to few pXRF studies in tropical soils, this work aimed to compare contents of Fe2O3, Al2O3, SiO2, TiO2 and P2O5 obtained by pXRF with sulfuric acid digestion results, and to evaluate the effects of varying forms of preparing soil samples and scanning with pXRF on the resulting values in Brazilian soils. Soils were scanned in five conditions in-field (in situ) and in laboratory, evaluating varying sample preparation methods, particle sizes and soil moisture. Four pXRF scanning operational modes were tested. Linear regressions were adjusted between results of pXRF and sulfuric acid digestion. Equations were validated with an independent set of samples. Statistical analyses compared the methods of preparing the samples. Adequate linear models reached R2 of 0.99 and 0.89 for Fe2O3 and TiO2, respectively. Validation promoted R2 greater than 0.97 and RMSE and ME close to zero for both oxides. Statistical differences of pXRF results were found among the methods of preparing samples. pXRF spectrometer has great potential to obtain Fe2O3 and TiO2 content rapidly and economically with high correspondence with laboratory results of sulfuric acid digestion analysis. Varying methods of preparing the samples promote differences in the results of pXRF.
Index terms:
Soil chemical properties; proximal sensor; sulfuric acid digestion analysis; soil properties prediction; oxides
RESUMO
O espectrômetro portátil de fluorescência de raios-X (pXRF) foi recentemente adotado pela Ciência do Solo, para uso em campo e laboratório, para obtenção do conteúdo total de vários elementos químicos em poucos segundos. A digestão com ácido sulfúrico é uma análise laboratorial cara e demorada que fornece teores de Fe2O3, Al2O3, SiO2, TiO2 e P2O5, importantes para estudos sobre solos. Devido aos poucos estudos sobre o pXRF em solos tropicais, este trabalho objetivou comparar os teores de Fe2O3, Al2O3, SiO2, TiO2 e P2O5 obtidos pelo pXRF com os resultados de digestão com ácido sulfúrico e avaliar os efeitos de diferentes formas de preparo de amostras de solo e leitura com o pXRF sobre seus resultados para solos brasileiros. Os solos foram submetidos a leituras com o pXRF em cinco condições, em campo (in situ) e em laboratório, avaliando variados métodos de preparo de amostras, tamanhos de partículas e umidade do solo. Quatro modos de operação do pXRF foram testados. Regressões lineares foram ajustadas entre os resultados do pXRF e digestão com ácido sulfúrico. As equações foram validadas com um conjunto independente de amostras. Análises estatísticas compararam os métodos de leitura de amostras. Modelos lineares adequados atingiram R2 de 0,99 e 0,89 para Fe2O3 e TiO2, respectivamente. A validação promoveu R2 maior que 0.97 e RMSE e ME próximos a zero para ambos os óxidos. Foram encontradas diferenças estatísticas dos resultados do pXRF entre os métodos de preparo de amostras. O pXRF possui um grande potencial para obter rápida e economicamente os teores de Fe2O3 e TiO2 com elevada correspondência com os resultados laboratoriais da análise da digestão com ácido sulfúrico. Métodos variáveis de preparo das amostras promovem diferenças nos resultados de pXRF.
Termos para indexação:
Atributos químicos do solo; sensor próximo; análise de digestão por ácido sulfúrico; predição de atributos do solo; óxidos
INTRODUCTION
X-ray fluorescence (XRF) technique has been widely used in studies of several branches of science, for identification and quantification of chemical elements in varying materials. It has advantages over other techniques for being rapid and without generation of chemical residues (Stockmann et al., 2016aSTOCKMANN, U. et al. Utilizing portable X-ray fluorescence spectrometry for in-field investigation of pedogenesis. Catena, 139:220-231, 2016a. ; Weindorf et al., 2014aWEINDORF, D. C. et al. Influence of ice on soil elemental characterization via portable X-ray fluorescence spectrometry. Pedosphere, 24(1):1-12, 2014a. ).
In recent years, portable X-ray fluorescence spectrometer (pXRF) was developed and it has been increasingly adopted by Soil Science community for allowing to access soil information in field or in the laboratory, in a few seconds, at low cost and without generating chemical residues. In this sense, pXRF has become an equipment of potential use by soil scientists, since it permits the evaluation of total content of elements from Mg to U of the Periodic Table, in soil under natural conditions, giving insight of some soil properties and contributing to the investigation of pedogenetic processes in the field (Weindorf et al., 2014aWEINDORF, D. C. et al. Influence of ice on soil elemental characterization via portable X-ray fluorescence spectrometry. Pedosphere, 24(1):1-12, 2014a. ).
Weindorf et al. (2012aWEINDORF, D. C. et al. Enhanced pedon horizonation using portable X-ray fluorescence spectrometry. Soil Science Society of America Journal , 76(2):522, 2012a. ) attempted to differentiate spodic and albic horizons using a pXRF and observed that this technique provides efficient and rapid analysis of soils originated from volcanic ash. Likewise, Stockmann et al. (2016aSTOCKMANN, U. et al. Utilizing portable X-ray fluorescence spectrometry for in-field investigation of pedogenesis. Catena, 139:220-231, 2016a. ), concluded that pXRF was adequate for determining soil parent material and pedogenetic processes, also noticing that pXRF results in the field are comparable to those from air-dried samples, since the geochemical indices showed similar trends in the soil profile, although samples were affected by some heterogeneity and moisture. Weindorf et al. (2012bWEINDORF, D. C. et al. Characterizing soils via portable X-ray fluorescence spectrometer: 2. Spodic and Albic horizons. Geoderma , 189-190:268-277, 2012b. ) also used pXRF to investigate soil profile development and found that elemental data aligned with the morphological descriptions of the horizons, concluding that pXRF could be used as an additional tool to distinguish soil horizons. In another work, Weindorf et al. (2015WEINDORF, D. C. et al. Lithologic discontinuity assessment in soils via portable X-ray fluorescence spectrometry and visible near-infrared diffuse reflectance spectroscopy. Soil Science Society of America Journal , 79(6):1704-1716, 2015. ), using pXRF for the determination of lithological discontinuities of soils from three different countries, observed an appropriate response of pXRF when compared with the laboratory data and the morphological descriptions of the profiles.
Although the aforementioned studies demonstrate the potential for using pXRF, due to the fact that pXRF is a recent tool, there is a lack of works worldwide associating pXRF with studies related to soil science, especially in developing countries, such as Brazil. In addition, the concentrations of the elements obtained by pXRF at varying methods of analysis, such as particle size of the samples, equipment operational mode, soil moisture, scanning place (field vs. laboratory) and comparisons of pXRF results with some most common laboratory tests, especially on tropical soils, are rare and still need to be evaluated. However, in soils of the United States, Sharma et al. (2014SHARMA, A. et al. Characterizing soils via portable X-ray fluorescence spectrometer: 3. Soil reaction (pH). Geoderma, 232-234:141-147, 2014. ) used pure pXRF elemental data and data from adjusted simple and multiple linear regressions to predict soil reaction (pH), obtaining better adjustments with multiple linear regressions.
In Brazil, sulfuric acid digestion analysis is performed for the determination of Fe2O3, Al2O3, SiO2, TiO2 and P2O5 contents (Empresa Brasileira de Pesquisa Agropecuária - Embrapa, 1997EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Manual de métodos de análises de solos, 2a. ed. Rio de Janeiro: Embrapa Solos, 1997. 212p. ). This method is well accepted by the scientific community for studies involving soil genesis, since its results enable the calculation of indexes that reflect the degree of soil weathering (Ki and Kr). In addition, according to the Brazilian Soil Classification System (Embrapa, 2013EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Sistema Brasileiro de Classificação de Solos. 3rd. ed. Brasília: Embrapa, 2013. 353p. ), these indexes and mainly Fe2O3 content are required for the classification of several soils at lower taxonomic levels. Although the sulfuric acid digestion analysis is widely used, it has as disadvantages the slow procedure of analysis, which is performed only by a few laboratories in Brazil, the generation of chemical residues, the high cost, besides the destruction of the sample. In view of the great need of sulfuric acid digestion analysis for the classification of soils in Brazil and its difficult and time-consuming analysis, pXRF potential for determination of those oxides should be evaluated, since it can obtain soil elemental contents faster and without production of chemical residues.
In this context, since there are very few studies evaluating the use of pXRF in tropical soils worldwide and due to its potential to aid obtaining information about soils at low cost and time, the objectives of this study were: a) to compare the pXRF contents of Fe2O3, Al2O3, SiO2, TiO2 and P2O5 with sulfuric acid digestion results and b) to evaluate the effects of varying forms of scanning soil samples with pXRF on the resulting values of those oxides, including different sample preparation methods and pXRF scanning modes in Brazilian soils.
MATERIAL AND METHODS
Study area, soil sampling and laboratory analyses
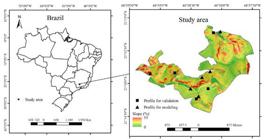
This study was carried out in the campus of the Federal University of Lavras, Minas Gerais State, Brazil, located at central latitude 21°34’46.04’’S and longitude 44°58’40.40’’W (Figure 1). Soil samples were collected in A and B horizons of six soil profiles, making up a total of 12 samples, encompassing six soil classes: Red Latosol (RL), Red-Yellow Latosol (RYL), Haplic Cambisol (HC), Yellow Argisol (YA), Red Argisol (RA), and Red Nitosol (RN), which are the most common soil classes in Brazil (Ker et al., 2012KER, J. C. et al. Pedologia - Fundamentos. 1. ed. Viçosa: SBCS, 2012. 343p. ) and are very common in the region of this study (Curi et al., 1990CURI, N. et al. Geomorfologia, física, química e mineralogia dos principais solos da região de Lavras (MG). Ciência e Prática, 14(3):297-307, 1990. ). RL and RA were developed from gabbro, RN was developed from gabbro with contributions of colluvium, while RYL, HC and YA are products of alteration of gneiss.
Study area and location of the places where the samples were collected for modeling and for validation of the models.
A pXRF (Bruker model S1 Titan LE) was used to obtain the contents of Fe2O3, TiO2, SiO2, Al2O3 and P2O5 through scanning samples. The pXRF contains a 50 kV and 100 µA X-ray tube, which allows for the detection of elements of the Periodic Table ranging from Mg to U in parts per million range (ppm). Since this equipment provides different scanning modes and because pXRF has been recently adopted in soil studies, we tested the different methods of scanning soil samples to assess their accuracy according to sulfuric acid digestion analysis.
Regarding the pXRF scanning modes, samples were scanned in triplicate for 30 and 60 seconds, in General (dual mining) and Trace (dual soil) modes. Also, five scanning conditions were performed regarding soil sample preparation methods: (a) Field - in the field directly at A and B horizons of the studied soil profiles, in triplicate; in the laboratory (b) After field - at the moment the samples arrived in the laboratory; (c) TF - after the samples being air-dried and passed through a 2 mm sieve; (d) M - after being air-dried and ground; and (e) MP - after being air-dried, ground and completely passed through a 150 µm sieve. The After field treatment was performed in order to evaluate the possible effects of alteration of soil structure in the pXRF results by scanning the samples with a soil moisture very similar to that found in the field (data presented later).
Prior to scanning the samples, three check samples STD-M2 (CS STD-M2), Montana soil I 2710a and Montana soil II 2711a whose content of the elements were known, being the first one certified by this pXRF manufacturer and the last two certified by NIST (National Institution of Standards and Technology) were scanned to assess the percentage of recovery of the equipment. The recovery results for the five oxides of study are presented in Table 1 for the different pXRF scanning modes.
Recovery values (%) obtained by different pXRF scanning modes compared with check samples with certified element contents.
For comparison purposes, soil moisture was obtained from the samples at the moment they were scanned in the soil profiles (Field), from the samples as they arrived in the laboratory (After field) and after being air-dried and sieved. For that, a subsample of each sample was weighted (wet weight - WW), left in an oven at 105 °C for 24 hours and then weighted again (dry weight - DW). Through the formula: [(WW-DW)/DW]x100 the soil moisture (in %) was obtained. Since soil moisture of the samples scanned in the soil profile (Field) and scanned as they arrived in the laboratory (After field) were similar (Table 2), but very different from the air-dried samples, it allowed us to compare the influence of soil moisture in the scanning. Thus, the effects of using pXRF in the field and in the laboratory under similar soil moisture (Field and After Field methods) might give an insight of soil structure disturbance on pXRF results by comparing Field and After field methods, besides allowing for the effects of soil particle sizes among these two and the other soil sample preparation methods.
Sulfuric acid digestion analysis was performed according to Embrapa (1997EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Manual de métodos de análises de solos, 2a. ed. Rio de Janeiro: Embrapa Solos, 1997. 212p. ), in which 1 g of TFSA is mixed to 500 mL of H2SO4 and 500 mL of water (1:1 diluted sulfuric acid). Then, it is heated and left boiling for 30 minutes. Afterwards, 50 mL of water is added to the mixture, which, in turn, is transferred to an erlenmeyer of 250 mL and, thus, filtered. In sequence the contents of Fe2O3, TiO2, SiO2, Al2O3 and P2O5 were obtained.
A linear model was adjusted between Fe2O3, TiO2, SiO2, Al2O3 and P2O5 contents obtained by sulfuric acid digestion analysis and by pXRF to assess the correspondence of the results. For the oxides that had an adequate adjustment, accuracy of the equations was assessed using an independent set of 8 samples of A and B horizons from 4 soil profiles (Red-Yellow Argisol, Red Argisol, Red-Yellow Latosol, Red Latosol) also collected in the study area (Figure 1). These samples were submitted to the different preparation methods, scanned using the 4 pXRF scanning modes and subjected to sulfuric acid digestion analysis. Root mean square error (RMSE) (Equation 1), mean error (ME) (Equation 2), R2 and adjusted R2, and a 1:1 graphic between values estimated by pXRF and results from sulfuric acid digestion were calculated. The best equation for each oxide was established and, hence, the best pXRF scanning mode (30s vs 60s; General mode vs. Trace mode) and sample preparation method were defined.
where n: number of observations, ei: sulfuric acid digestion values estimated by the equation, and mi: real values obtained from sulfuric acid digestion analysis in the laboratory.
Comparisons regarding sample preparation methods
In order to quantify the effects of sample preparation methods in the results of pXRF for Fe2O3, TiO2, SiO2, Al2O3 and P2O5 contents, after defining the best model and, hence the best method for scanning the samples with pXRF, statistical analysis through Scott-Knott test at 5% probability was performed per soil sample using SISVAR software (Ferreira, 2011FERREIRA, D. F. SISVAR: A Computer statistical analysis system. Ciência e Agrotecnologia, 35(6):1039-1042, 2011. ). These analyses compared the different manners of scanning the samples (Field, After field, TF, M, and MP) in order to give support on the influence of particle size as well as soil moisture on the results of pXRF. This procedure was carried out to help define the most precise and rapid manner of scanning soil samples with pXRF for this study conditions.
RESULTS AND DISCUSSION
Testing the equations
Taking into account Fe2O3, TiO2, SiO2, Al2O3 and P2O5 contents, the linear models between the oxide contents obtained by sulfuric acid digestion analyses and by pXRF for SiO2 and Al2O3 did not present high R2 values considering all the analyzed methods (mode and time of scanning, and methods of preparing soil samples). The oxides obtained by sulfuric acid digestion analysis are more correspondent to the oxides present in the clay fraction (Curi; Kämpf, 2012CURI, N.; KÄMPF, N. Caracterização do solo. In: KER, J. C. et al. (Eds.). Pedologia: Fundamentos. Viçosa: SBCS, 2012. p. 147-170. ), whereas results of pXRF includes elements present in clay and other particle size fractions of soil. Since quartz (SiO2) is commonly found in the sand fraction of Brazilian soils (Alves et al., 2013ALVES, M. J. F. et al. Reserva mineral de potássio em Latossolo cultivado com Pinus taeda L. Revista Brasileira de Ciência do Solo, 37(6):1599-1610, 2013. ; Araujo et al., 2014ARAUJO, M. A. et al. Paragênese mineral de solos desenvolvidos de diferentes litologias na região sul de Minas Gerais. Revista Brasileira de Ciência do Solo , 38(1):11-25, 2014. ; Lima et al., 2008LIMA, J. G. et al. Mineralogia de um Argissolo Vermelho-Amarelo da zona úmida costeira do estado de Pernambuco. Revista Brasileira de Ciência do Solo , 32(2):881-892, 2008. ; Ferreira; Fernandes; Curi, 1999FERREIRA, M. M.; FERNANDES, B.; CURI, N. Influência da mineralogia da fração argila nas propriedades fisicas de Latossolos da região sudeste do Brasil. Revista Brasileira de Ciência do Solo , 23(1):515-524, 1999. ) it may have contributed to the low R2 values of SiO2 contents from sulfuric acid digestion with those of pXRF.
Regarding P2O5, this oxide content was under the pXRF limit of detection (LOD) for all the studied soils. It might have been caused by the low natural P content in soils (Fernández et al., 2009FERNÁNDEZ R. I. E. et al. Reversibilidade do fósforo não-lábil em solos submetidos à redução microbiana e química. I - Alterações químicas e mineralógicas. Revista Brasileira de Ciência do Solo , 32(6):2307-2317, 2009. ; Ferreira; Fernandes; Curi, 1999FERREIRA, M. M.; FERNANDES, B.; CURI, N. Influência da mineralogia da fração argila nas propriedades fisicas de Latossolos da região sudeste do Brasil. Revista Brasileira de Ciência do Solo , 23(1):515-524, 1999. ; Towett et al., 2015TOWETT, E. K. et al. Total elemental composition of soils in Sub-Saharan Africa and relationship with soil forming factors. Geoderma Regional, 5:157-168, 2015. ). On the other hand, for Fe and Ti, all the linear models presented adequate adjustment among the values obtained by pXRF and sulfuric acid digestion. Table 3 shows the R2 of the linear models for all the oxides evaluated.
Analyzing Table 3, it is noticed the general adequate adjustment for Fe2O3 linear models independently of the soil preparation method and pXRF scanning mode. Comparing the pXRF scanning modes, independently of the sample preparation method, values of the Fe2O3 from the sulfuric acid digestion could be well adjusted to pXRF values, promoting adequate R2 results. However, scanning the samples directly in the soil profile in the field had the lowest R2 values when compared with the methods that scanned the samples in the laboratory, being the lowest value found for pXRF mode Trace 60s (R2 = 0.74). This fact may be attributed to the soil moisture in the soil profile being greater than 20% in most samples (Table 2), which is considered a cause of error in pXRF (Weindorf et al., 2014aWEINDORF, D. C. et al. Influence of ice on soil elemental characterization via portable X-ray fluorescence spectrometry. Pedosphere, 24(1):1-12, 2014a. ) or to soil structure, since better results were obtained for After Field preparation method, which contained soil moisture values similar to those in field condition. Stockmann et al. (2016bSTOCKMANN, U. et al. The effect of soil moisture and texture on Fe conentration using portable X-ray fluorescence spectrometers. In: HARTEMINK, A. E.; MINASNY, B. (Eds.). Digital soil morphometrics, Springer, 2016b. p. 63-72. ) found differences in Fe contents comparing results of pXRF used in laboratory on air-dried samples and in-field, and proposed an equation to correct this difference related to soil moisture.
Despite of the field scanning, all the other sample preparation methods presented high R2, being them always equals to or greater than 0.98, and with 12 out of the 16 remaining combinations (scanning modes and soil sample preparation methods) reaching R2 = 0.99. These results suggest that using pXRF to assess Fe2O3 content in soil using an adjusted equation should be adequate, since it has high correspondence with the results of sulfuric acid digestion analysis. Additionally, since Fe2O3 from the sulfuric acid digestion analysis is important for soil classification purposes according to Brazilian Soil Classification System (Embrapa, 2013EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Sistema Brasileiro de Classificação de Solos. 3rd. ed. Brasília: Embrapa, 2013. 353p. ), pXRF can accelerate the process of classifying soils at lower taxonomical levels (with more details), besides avoiding both the costs of laboratory analyses and the production of chemical residues. For illustration purposes, Figure 2a shows the adjustment of an equation that presented adequate result to estimate Fe2O3 of sulfuric acid digestion from Fe2O3 values obtained by pXRF scanning mode General 30s and M sample preparation method.
Regarding TiO2, the linear models had more variable R2 and they were always lower than those for Fe2O3. Similarly to the findings for Fe2O3, when scanning was performed on the soil profile in the field, the R2 results were lower than those for scanning in the laboratory, being the lowest found for scanning mode General 30s (R2 = 0.40). Also, the R2 among the preparation methods was more variable within the same scanning mode, demonstrating the effect of sample preparation method on TiO2 estimates by pXRF. For instance, for scanning mode General 30s, R2 varied from 0.69 to 0.88 for After Field and MP sample preparation methods, respectively. In the same way, within the same sample preparation method, R2 also varied according to the pXRF scanning mode, such as for M preparation method that had R2 = 0.89 in Trace 30s against R2 =0.71 in General 60s. For illustration purposes, Figure 2b shows the adjustment of an equation for estimating TiO2 by sulfuric acid digestion generated from TiO2 values obtained by scanning mode Trace 30s and M sample preparation method.
Linear regression for Fe2O3 (a) and TiO2 (b) obtained by results of a portable X-ray fluorescence spectrometer (pXRF) and sulfuric acid digestion (SAD).
Ti importance in soil studies is related to soil genesis, since this element is considered less mobile in soils and, thus, represents a correspondence with Ti contents in the soil parent material. In this context, soils developed from different parent materials tend to present varying Ti contents (Curi; Franzmeier, 1987CURI, N.; FRANZMEIER, D. P. Effect of parent rocks on chemical and mineralogical properties of some Oxisols in Brazil. Soil Science Society of America Journal, 51:153-158, 1987. ; Moreira; Oliveira, 2008MOREIRA, H. L.; OLIVEIRA, V. Á. Evolução e gênese de um Plintossolo Pétrico Concrecionário Êutrico Argissólico no município de Ouro Verde de Goiás. Revista Brasileira de Ciência do Solo , 32(4):1683-1690, 2008. ).
Table 4 presents the equations for obtaining Fe2O3 and TiO2 of sulfuric acid digestion from these element contents resulted from pXRF analyses.
Equations for estimating Fe2O3 and TiO2 of sulfuric acid digestion analyses (in %) from these element contents resulted from portable X-ray fluorescence (pXRF) spectrometer (in %). For Trace mode, Ti e Fe contents obtained by pXRF must be converted into TiO2 and Fe2O3 to be used in the equations.
Validation of the equations for predicting Fe 2 O 3 and TiO 2 contents from sulfuric acid digestion analysis
Table 5 shows the parameters for validation of Fe2O3 and TiO2 equations for the four scanning modes and the best three sample preparation methods (TF, M, and MP), since equations for SiO2 and Al2O3 did not have adequate adjustment. It is noticed that most of the methods presented adequate results, which confirms the trend of using pXRF to estimate Fe2O3 and TiO2 contents equivalent to those obtained from sulfuric acid digestion.
Considering the sample preparation methods in Table 3, the validation results for TF presented the highest R2 and R2 adj in all the pXRF scanning modes, independently of sample preparation method for both Fe2O3 and TiO2. In addition, RMSE and ME were the lowest for TF in 5 out of the 8 possible conditions (4 scanning modes and 2 oxides). It indicates that scanning the soil samples prepared as TF promoted better results than M and MP methods, which is interesting since preparation of samples in TF particle size is more rapid and easier than preparation in M and MP size fractions. However, in most cases, the statistical parameters used for validating the equations had good results. Furthermore, among the preparation methods, MP had lower R2 and R2 adj, although they were all greater than 0.93, and higher RMSE and ME than the other methods, indicating a slightly worse performance. These findings may contribute to future works, aiding researchers to define a proper way of preparing a soil sample intended to be scanned using pXRF.
Analyzing the performance of the scanning modes, all of them had adequate results in general, being the R2 and R2 adj always greater than 0.94 and 0.93, respectively, and some of them reaching 0.99. However, when observing these results in more detail, a slight difference among the statistical parameters was found between the modes General and Trace. The Trace mode, which is recommended for scanning soils, showed better results than those for General, with higher R2 and R2 adj and lower RMSE and ME in most cases. The necessity of testing both methods showed up from the observation in the literature of works that used the two methods in the soil according to the element they want to obtain the contents.
Regarding the time of scanning within the same mode (General or Trace), the results were very similar for both Fe2O3 and TiO2. Thus, since good results were obtained independently of the time of scanning, we considered that using 30s as the time for scanning should promote more rapid analyses, which is advantageous when the number of samples to be scanned is large. However, Hou, He and Jones (2004)HOU, X.; HE, Y.; JONES, B. T. Recent advances in portable X-ray fluorescence spectrometry. Applied Spectroscopy Reviews, 39:1-25, 2004. have suggested that longer scanning times should provide better precision on elemental content estimates. However, for the samples of this present study, it was defined that the best method to scan was Trace 30s in TF sample preparation method. Figures 3a and 3b, respectively, show the 1:1 ratio graphics comparing the Fe2O3 and TiO2 contents of sulfuric acid digestion estimated by using pXRF to obtain Fe2O3 and TiO2 contents to be inserted into the formerly adjusted equations for TF in Trace 30s scanning mode (estimated values) versus Fe2O3 and TiO2 contents obtained by sulfuric acid digestion analysis (real values).
Graphics of sulfuric acid digestion values of Fe2O3 (a) and TiO2 (b) versus these same values resulted from the insertion of values from portable X-ray fluorescence spectrometer (pXRF) in the previously adjusted equations for TF sample preparation method scanned in mode Trace 30s.
Statistical differences of Fe 2 O 3 and TiO 2 contents among the sample preparation methods
In order to better quantify the differences promoted by scanning samples with different preparation methods, statistical test was performed and the results are expressed in Table 6. Analyzing Table 6, it can be noticed that significant statistical differences, according to Scott-Knott test at 5% probability, occurred among the preparation methods in all soil samples.
In general, scannings performed on the soil profile in the field promoted the lowest contents of Fe2O3 and TiO2, being statistically different from those in the laboratory for 79% of the soils, according to Scott-Knott test at 5% probability. However, this fact does not impede the use of pXRF on such conditions, since equations can be created to correct the contents obtained with pXRF in the field, as Stockmann et al. (2016bSTOCKMANN, U. et al. The effect of soil moisture and texture on Fe conentration using portable X-ray fluorescence spectrometers. In: HARTEMINK, A. E.; MINASNY, B. (Eds.). Digital soil morphometrics, Springer, 2016b. p. 63-72. ) demonstrated for Fe. Stockmann et al. (2016aSTOCKMANN, U. et al. Utilizing portable X-ray fluorescence spectrometry for in-field investigation of pedogenesis. Catena, 139:220-231, 2016a. ) used pXRF to quantify the several element contents in three soil profiles and compared these results with air-dried and ground samples, finding that, for two soils, the Fe contents were greater when samples were scanned in the laboratory in comparison with field scanning. For Ti, these same authors found contrasting results according to the soil class: one had greater Ti content for the lower part of E horizon and the whole B horizon, another had similar contents in all depths for both scanning conditions, and the third had greater Ti content in air-dried and ground soil than in the field for the most part of the soil profile.
Since the samples of the After Field preparation methods contained similar moisture to those in the field, and promoted different results in comparison with the field scanning, it demonstrates the effect of the scanning conditions and, hence, soil structure disturbance, on the results, drawing attention to great differences among the contents of Fe2O3 and TiO2 for those scanning conditions. Although Sahraoui and Hachicha (2017SAHRAOUI, H.; HACHICHA, M. Effect of soil moisture on trace elements concentrations. Journal of Fundamental and Applied Sciences, 9(1):468-484, 2017. ), studying the effects of soil moisture on element contents obtained by pXRF, noticed that greater amounts for Fe were found as the samples got drier, here we found that greater contents were found when modifying the natural soil condition (in field), as obtained by scanning samples in the laboratory with similar soil moisture to the field condition. Weindorf et al. (2014b), studying soils under influence of ice, noticed underestimation of values obtained by pXRF when the soil was wet in comparison with dry soil, suggesting a correction of soil moisture for better correlations with dry soil.
Comparing the sample conditions that were scanned in the laboratory, the results were variable according to the soil classes, which depends on the soil properties variability inherent of the soil forming factors (Jenny, 1941JENNY, H. Factors of soil formation: A system of quantitative Pedology. New York: McGraw-Hill Book Co., Inc., 1941. 281p. ) and processes that led to soils development. For Fe2O3, in 11 out of the 12 soils analyzed, the highest contents were obtained by scanning the samples in M condition, whereas this same fact occurred in 9 soils for TiO2. This result shows that grinding the samples to fine sizes, such as MP (< 150 µm), smaller than M, does not always promote higher content estimates for such oxides. In this sense, it is suggested other works in order to compare the contents of elements obtained by pXRF and by other chemical methods to certify which of these sample preparation methods would be more efficient in predicting the real content of elements in tropical soils, which was not the objective of this study.
It is important to highlight the coefficient of variation (CV) of Fe2O3 and TiO2 values of the replicates of the samples scanned in the laboratory were mostly below 5% (data not presented), whereas the CV for the samples scanned in the field were close to 20%. For example, CV in average was 1.78% for Fe2O3 and 3.72% for TiO2 in TF sample preparation method and Trace 30s scanning mode, whereas for the same scanning mode performed in the field, these values were, respectively, 18.83% and 19.73%. These findings suggest that larger number of replicates should be made in the field.
The fact that sulfuric acid digestion results can be adequately obtained after adjusting an equation with these values and those obtained by pXRF scanning for Fe2O3 and TiO2 can aid in reducing the amount of residues produced from sulfuric acid digestion analysis. Although an adequate adjustment of equations could not be made for SiO2, Al2O3 and P2O5 contents, soil classification can be aided with the use of pXRF especially for countries whose classification system requires the Fe2O3 by sulfuric acid digestion at lower taxonomical levels, such as the Brazilian Soil Classification System (Embrapa, 2013EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Sistema Brasileiro de Classificação de Solos. 3rd. ed. Brasília: Embrapa, 2013. 353p. ). Also, TiO2, which is one of the oxides commonly used for studies of soil genesis, can be easily estimated with pXRF with high correspondence to sulfuric acid digestion analysis, giving an insight of differences in soil parent material and soil forming processes. Finally, the fact that pXRF promotes a rapid access of several element contents in soil (although it still requires further tests in tropical soils), both in the laboratory and in the field, at low cost and no production of chemical residues, it shows pXRF potential to be used in studies of varying natures, such as geology (Milić, 2014MILIĆ, M. PXRF characterisation of obsidian from central Anatolia, the Aegean and central Europe. Journal of Archaeological Science, 41:285-296, 2014. ) and archeology (Mehta et al., 2017MEHTA, J. M. et al. Geochemical source evaluation of archaeological chert from the Carson mounds site in northwestern Mississippi using portable X-ray fluorescence (pXRF). Journal of Archaeological Science: Reports. 11:381-389, 2017. ), as well as in soil surveys to help separating soil classes. Silva et al. (2016SILVA, S. H. G. et al. Proximal sensing and digital terrain models applied to digital soil mapping and modeling of Brazilian Latosols (Oxisols). Remote Sensing, 8:614-635, 2016. ) in a work that aimed to map Latosols (Oxisols) developed from varying parent materials at lower taxonomical levels in Brazil, used several terrain attribute maps, such as slope gradient and topographic wetness index (Beven; Kirkby, 1979BEVEN, K. J.; KIRKBY, M. J. A physically based, variable contributing area model of basin hydrology / Un modèle à base physique de zone d’appel variable de l’hydrologie du bassin versant. Hydrological Sciences Bulletin, 24(1):43-69, 1979. ) which distinguishes places more likely to accumulate water, in addition to soil magnetic susceptibility and elements estimated by pXRF, e.g. Fe, Ti, Si, and other as variables. They found that magnetic susceptibility and some element contents obtained by pXRF helped improving the soil mapping processes, demonstrating that pXRF can provide more variables to improve soil surveys and mapping.
CONCLUSIONS
Results of sulfuric acid digestion analysis for Fe2O3 and TiO2 have adequate correlation with these oxide contents obtained by pXRF for different soils, whereas for SiO2 and Al2O3 adequate correlations were not obtained. Sample preparation methods with varying particle sizes and the place where the samples were scanned by pXRF (in the field or laboratory) presented statistical differences for Fe2O3 and TiO2 contents, whereas all the pXRF scanning modes provided adequate results for varying soils. The use of pXRF can help in obtaining Fe2O3 and TiO2 content data correspondent to sulfuric acid digestion analysis in a rapid and economical way, both in the field and in laboratory, reducing the amount of residues originated from chemical analysis, confirming the potential use of pXRF for several studies regarding soils.
ACKNOWLEDGEMENTS
The authors thank the following funding agencies: National Council for Scientific and Technological Development (CNPq), Coordination for the Improvement of Higher Education Personnel (CAPES) and Minas Gerais Foundation for Research Support (FAPEMIG) for the financial support that enabled us to develop this work.
REFERENCES
- ALVES, M. J. F. et al. Reserva mineral de potássio em Latossolo cultivado com Pinus taeda L. Revista Brasileira de Ciência do Solo, 37(6):1599-1610, 2013.
- ARAUJO, M. A. et al. Paragênese mineral de solos desenvolvidos de diferentes litologias na região sul de Minas Gerais. Revista Brasileira de Ciência do Solo , 38(1):11-25, 2014.
- BEVEN, K. J.; KIRKBY, M. J. A physically based, variable contributing area model of basin hydrology / Un modèle à base physique de zone d’appel variable de l’hydrologie du bassin versant. Hydrological Sciences Bulletin, 24(1):43-69, 1979.
- CURI, N. et al. Geomorfologia, física, química e mineralogia dos principais solos da região de Lavras (MG). Ciência e Prática, 14(3):297-307, 1990.
- CURI, N.; FRANZMEIER, D. P. Effect of parent rocks on chemical and mineralogical properties of some Oxisols in Brazil. Soil Science Society of America Journal, 51:153-158, 1987.
- CURI, N.; KÄMPF, N. Caracterização do solo. In: KER, J. C. et al. (Eds.). Pedologia: Fundamentos. Viçosa: SBCS, 2012. p. 147-170.
- EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Manual de métodos de análises de solos, 2a. ed. Rio de Janeiro: Embrapa Solos, 1997. 212p.
- EMPRESA BRASILEIRA DE PESQUISA AGROPECUÁRIA - EMBRAPA. Sistema Brasileiro de Classificação de Solos. 3rd. ed. Brasília: Embrapa, 2013. 353p.
- FERNÁNDEZ R. I. E. et al. Reversibilidade do fósforo não-lábil em solos submetidos à redução microbiana e química. I - Alterações químicas e mineralógicas. Revista Brasileira de Ciência do Solo , 32(6):2307-2317, 2009.
- FERREIRA, D. F. SISVAR: A Computer statistical analysis system. Ciência e Agrotecnologia, 35(6):1039-1042, 2011.
- FERREIRA, M. M.; FERNANDES, B.; CURI, N. Influência da mineralogia da fração argila nas propriedades fisicas de Latossolos da região sudeste do Brasil. Revista Brasileira de Ciência do Solo , 23(1):515-524, 1999.
- HOU, X.; HE, Y.; JONES, B. T. Recent advances in portable X-ray fluorescence spectrometry. Applied Spectroscopy Reviews, 39:1-25, 2004.
- JENNY, H. Factors of soil formation: A system of quantitative Pedology. New York: McGraw-Hill Book Co., Inc., 1941. 281p.
- KALNICKY, D. J.; SINGHVI, R. Field portable XRF analysis of environmental samples. Journal of Hazardous Materials, 83(1-2):93-122, 2001.
- KER, J. C. et al. Pedologia - Fundamentos. 1. ed. Viçosa: SBCS, 2012. 343p.
- LIMA, J. G. et al. Mineralogia de um Argissolo Vermelho-Amarelo da zona úmida costeira do estado de Pernambuco. Revista Brasileira de Ciência do Solo , 32(2):881-892, 2008.
- MEHTA, J. M. et al. Geochemical source evaluation of archaeological chert from the Carson mounds site in northwestern Mississippi using portable X-ray fluorescence (pXRF). Journal of Archaeological Science: Reports. 11:381-389, 2017.
- MILIĆ, M. PXRF characterisation of obsidian from central Anatolia, the Aegean and central Europe. Journal of Archaeological Science, 41:285-296, 2014.
- MOREIRA, H. L.; OLIVEIRA, V. Á. Evolução e gênese de um Plintossolo Pétrico Concrecionário Êutrico Argissólico no município de Ouro Verde de Goiás. Revista Brasileira de Ciência do Solo , 32(4):1683-1690, 2008.
- SAHRAOUI, H.; HACHICHA, M. Effect of soil moisture on trace elements concentrations. Journal of Fundamental and Applied Sciences, 9(1):468-484, 2017.
- SHARMA, A. et al. Characterizing soils via portable X-ray fluorescence spectrometer: 3. Soil reaction (pH). Geoderma, 232-234:141-147, 2014.
- SILVA, S. H. G. et al. Proximal sensing and digital terrain models applied to digital soil mapping and modeling of Brazilian Latosols (Oxisols). Remote Sensing, 8:614-635, 2016.
- STOCKMANN, U. et al. Utilizing portable X-ray fluorescence spectrometry for in-field investigation of pedogenesis. Catena, 139:220-231, 2016a.
- STOCKMANN, U. et al. The effect of soil moisture and texture on Fe conentration using portable X-ray fluorescence spectrometers. In: HARTEMINK, A. E.; MINASNY, B. (Eds.). Digital soil morphometrics, Springer, 2016b. p. 63-72.
- TERRA, J. et al. Análise multielementar de solos: Uma proposta envolvendo equipamento portátil de fluorescência de raios X. Semina: Ciências Exatas e Tecnológicas, 35(2):207-214, 2014.
- TOWETT, E. K. et al. Total elemental composition of soils in Sub-Saharan Africa and relationship with soil forming factors. Geoderma Regional, 5:157-168, 2015.
- WEINDORF, D. C. et al. Enhanced pedon horizonation using portable X-ray fluorescence spectrometry. Soil Science Society of America Journal , 76(2):522, 2012a.
- WEINDORF, D. C. et al. Characterizing soils via portable X-ray fluorescence spectrometer: 2. Spodic and Albic horizons. Geoderma , 189-190:268-277, 2012b.
- WEINDORF, D. C. et al. Influence of ice on soil elemental characterization via portable X-ray fluorescence spectrometry. Pedosphere, 24(1):1-12, 2014a.
- WEINDORF, D. C. et al. Lithologic discontinuity assessment in soils via portable X-ray fluorescence spectrometry and visible near-infrared diffuse reflectance spectroscopy. Soil Science Society of America Journal , 79(6):1704-1716, 2015.
- WEINDORF, D. C.; BAKR, N.; ZHU, Y. Advances in portable X-ray fluorescence (pXRF) for environmental, pedological, and agronomic applications. Advances in Agronomy, 128:1-45, 2014
Publication Dates
-
Publication in this collection
Jan-Feb 2018
History
-
Received
31 Mar 2017 -
Accepted
16 May 2017