Abstracts
Due to the high importance of the venereal transmission of bovine leptospirosis, this study aimed to test the ability of PCR to detect Leptospira interrogans serovar hardjo DNA in experimentally contaminated bovine semen. Employing primers directed to the 16S rRNA gene, 10 bacteria/ml of semen could be detected by PCR. Results achieved in this work show that PCR can have a great potential to detect Leptospira spp. in insemination centers.
Leptospirosis; Leptospira; Semen; Polimerase Chain Reaction; Bovine
Considerando a importância do sêmen na transmissão da leptospira bovina, foi realizado o presente estudo que teve como objetivo aplicar a reação em cadeia pela polimerase (PCR) para a detecção de leptospiras em sêmen bovino experimentalmente contaminado. A reação de PCR foi capaz de amplificar um fragmento de DNA específico de 330 pares de bases a partir de cultivos puros de 26 sorovares de Leptospira spp. A contaminação experimental de sêmen com Leptospira interrogans serovar hardjo revelou que a técnica de PCR conseguiu detectar 10 bactérias/ml, concentração sensivelmente mais baixa que as 1.000 bactérias/ml detectadas através do cultivo microbiológico. Os resultados observados revelam o grande potencial da reação de PCR para a detecção de Leptospira spp. em sêmen bovino, notadamente em centrais de inseminação artificial.
Leptospirose; Leptospira; Sêmen; Reação em cadeia de polimerase; Bovinos
Detection of Leptospira spp. from pure cultures and from experimentally contaminated bovine semen by polymerase chain reaction
Detecção de Leptospira spp. através da reação em cadeia pela polimerase (PCR) em sêmen bovino experimentalmente contaminado
Marcos Bryan HEINEMANN1 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP ; José Fernando GARCIA2 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP ; Cáris Maroni NUNES2 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP ; Zenaide Maria Moraes HIGA1 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP ; Silvio Arruda VASCONCELLOS1 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP ; Leonardo José Richtzenhain1 1 Departamento de Medicina Veterinária Preventiva e Saúde Animal da Faculdade de Medicina Veterinária e Zootecnia da USP SP 2 Departamento de Apoio, Produção e Saúde Animal da Faculdade de Medicina Veterinária da UNESP, Araçatuba SP
Correspondence to:
Leonardo José Richtzenhain
Departamento de Medicina Preventiva e Saúde Animal
Faculdade de Medicina Veterinária e Zootecnia da USP
Cidade Universitária Armando de Salles Oliveira
Av. Orlando Marques de Paiva, 87
05508-900 São Paulo SP
e-mail: leonardo@usp.br
SUMMARY
Due to the high importance of the venereal transmission of bovine leptospirosis, this study aimed to test the ability of PCR to detect Leptospira interrogans serovar hardjo DNA in experimentally contaminated bovine semen. Employing primers directed to the 16S rRNA gene, 10 bacteria/ml of semen could be detected by PCR. Results achieved in this work show that PCR can have a great potential to detect Leptospira spp. in insemination centers.
UNITERMS: Leptospirosis; Leptospira; Semen; Polimerase Chain Reaction; Bovine.
INTRODUCTION
Artificial insemination is an important procedure for increasing animal production and millions of doses of frozen bovine semen are exchanged annually among different countries. To obtain high fertility and birth rates and to prevent infectious diseases, many tests are employed for the detection of infectious agents in bovine semen. Bovine leptospirosis is one of the important infectious diseases that can be transmitted by artificial insemination since Leptospira spp. can survive at the freezing temperatures needed to store semen1.
Direct diagnostic methods based on leptospira isolation from semen are not easily performed due to the rapid growth of other microorganisms8. In addition, the absence of detectable levels of serum antibodies in infected animals makes the indirect methods inefficient2.
Among the DNA-based techniques, the polymerase chain reaction (PCR) has been used for the diagnosis of slowly growing or fastidious microorganisms1. As to bovine leptospirosis, some investigators have used PCR to detect Leptospira spp. in bovine urine4,6,10, and recently in bovine semen5.
Employing primers directed to the Leptospira interrogans serovar canicola strain Moulton 16S rRNA gene6, this study aimed to test the ability of PCR for the detection of several Leptospira spp. from pure cultures and from bovine semen experimentally contaminated with Leptospira interrogans serovar hardjo.
MATERIAL AND METHOD
Leptospira serovars and growth conditions
The Leptospira serovars used in this study (Tab. 1) were cultivated at 28ºC in EMJH medium supplemented with inactivated rabbit serum as described by Turner9, and belong to the collection of the Laboratory of Bacterial Zoonoses of the Faculty of Veterinary Medicine and Zootechny, University of São Paulo - SP- Brazil.
Experimental contamination of bovine semen
The semen for experimental contamination with leptospires was collected from a bull that presented no detectable antibodies against any of the bacteria listed in Tab. 1 as assessed by the microscopic agglutination test3, and was also free of leptospires, as determined by microbiological culture. The bovine semen described above was diluted with distilled water in order to obtain 50 x 106 spermatozoa/ml and contaminated with 101 to 106 Leptospira interrogans serovar hardjo/ml.
Microbiological culture
For microbiological culture 1 ml of the bovine semen contaminated with different bacterial concentrations was seeded in EMJH medium supplemented with inactivated rabbit serum, incubated at 28ºC and observed for 8 weeks.
PCR
Leptospires from experimentally contaminated bovine semen (serovar hardjo) or from pure cultures of all serovars listed in Tab. 1 were collected by centrifugation at 13,000 x g for 30 minutes at 4ºC and DNA was extracted as described by Sambrook et al.7.
The primers employed were those described by Mérien et al.6 corresponding to nucleotides 38 to 57 (5GGCGGCGCGTCTTAAACATG3) and 348 to 368 (5TCCCCCCATTGAGCAAGATT3) of the 16S rRNA gene of L. interrogans serovar canicola strain Moulton.
DNA amplification was performed in a total volume of 50 µl. The reaction mixture consisted of 200 mM Tris-HCl, pH 8.0, 500 mM KCl, 50 mM MgCl2, 1.25 mM each dATP, dTTP, dCTP, dGTP, 6.25 µM each oligonucleotide primer, 2 units of Taq DNA polymerase (Gibco BRL Gaythersburg, MD-USA) and the sample, comprised in 1/10 of the final volume. The reaction mixture was overlaid with 50 µl of mineral oil (Sigma). PCR was performed as described by Mérien et al.6 using a Perkin Elmer 480 thermal cycler.
The amplification products were separated by electrophoresis in a 2% agarose gel, stained with ethidium bromide and visualized under UV light.
The threshold of Leptospira detection by PCR and by microbiological culture
The threshold of Leptospira detection by PCR and by microbiological culture was evaluated in the experimentally contaminated bovine semen.
RESULTS AND DISCUSSION
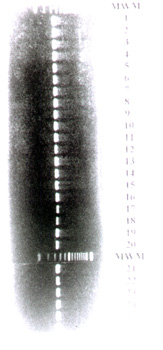
Fig. 1 shows that PCR amplified a 330 bp DNA fragment from the DNA of all Leptospira interrogans, Leptospira borgpetersenii, Leptospira noguchii, Leptospira santarosai and Leptospira biflexa serovars used in this study (Tab. 1).
The different papers that describes PCR assays for the detection of leptospires in bovine urine4,10 or semen5 were not able to identify specific serovars, since the primers amplified conserved regions of Leptospira spp. genome.
Recently Masri et al.5 described a PCR followed by a nested PCR assay to detect Leptospira spp. in bovine semen experimentally contaminated with the serovar hardjobovis. The primers employed by these authors were derived from a 3,800 bp EcoR1/BamH1 fragment of serovar hardjobovis genome and hybridized to 11 of 31 not specified serovars.
The broad spectrum of PCR in the detection of pathogenic and saprophytic leptospires, also observed in the present study, could be explained by the primers employed since the sequence of Leptospira 16S rRNA gene is highly conserved6.
In addition to the Leptospira serovars originally detected by PCR by Mérien et al.6, the following serovars were detected in the present study: andamana, brasiliensis, buenos aires, butembo, garcia, rufino and whitcombi.
Employing PCR and nested-PCR Masri et al.5 detected respectively 500 5,000 and 50 500 organisms/ml of semen. The PCR assay here described detected 10 Leptospira interrogans serovar hardjo/ml in bovine semen (Fig. 2), a threshold not reached by microbiological culture that needs a minimum of 1,000 bacteria/ml.
Among the different factors that could explain the lower threshold obtained in this study were: 1) The DNA extraction protocol that involved procedures used in the 3 different protocols evaluated by Masri et al.5; 2) The reagents, primers and DNA amplification conditions.
In addition to a lower detection limit, PCR has the advantage of being performed within a shorter period of time than Leptospira culture, which can take up to 2 months3.
Considering serology does not reflect the carrier or shedding status of cattle2 and the difficulties of Leptospira isolation from bovine semen, PCR may represent a useful tool for the rapid detection of Leptospira spp. in bovine semen at artificial insemination centers.
Gel electrophoresis performed on 2% agarose of PCR-amplified products from 26 Leptospira serovars. Lanes 1= australis; 2= autumnalis; 3= butembo; 4= castellonis; 5= bataviae; 6= brasiliensis; 7= canicola; 8= whitcombi; 9= cynopteri; 10= hebdomadis; 11= copenhageni; 12= icterohaemorrhagiae; 13= grippotyphosa; 14= javanica; 15= panama; 16= pomona; 17= pyrogenes; 18= hardjo; 19= wolffi; 20= shermani; 21= tarassovi; 22= andamana; 23= buenos aires; 24= garcia; 25= rufino and 26= patoc. A 330 bp amplified DNA fragment can be observed in all serovars. MWM= molecular weight markers ranging from 100 to 1,500 bp at 100 bp increments (Gibco-BRL Gaythersburg, MD-USA). The bands were visualized by ethidium bromide staining under ultraviolet light.
RESUMO
Considerando a importância do sêmen na transmissão da leptospira bovina, foi realizado o presente estudo que teve como objetivo aplicar a reação em cadeia pela polimerase (PCR) para a detecção de leptospiras em sêmen bovino experimentalmente contaminado. A reação de PCR foi capaz de amplificar um fragmento de DNA específico de 330 pares de bases a partir de cultivos puros de 26 sorovares de Leptospira spp. A contaminação experimental de sêmen com Leptospira interrogans serovar hardjo revelou que a técnica de PCR conseguiu detectar 10 bactérias/ml, concentração sensivelmente mais baixa que as 1.000 bactérias/ml detectadas através do cultivo microbiológico. Os resultados observados revelam o grande potencial da reação de PCR para a detecção de Leptospira spp. em sêmen bovino, notadamente em centrais de inseminação artificial.
UNITERMOS: Leptospirose; Leptospira; Sêmen; Reação em cadeia de polimerase; Bovinos.
Received:15/10/97
Accepted:17/06/98
-
1- EAGLESOME, M.D.; GARCIA, M.M. Microbial agents associated with bovine genital tract infections and semen. Part 1. Brucella abortus, Leptospira, Campylobacter fetus and Tritrichomonas foetus Veterinary Bulletin, v.62, n.8, p.743-75, 1992.
-
2- ELLIS, W.A.; CASSELS, J.A.; DOYLE, J. Genital leptospirosis in bull. Veterinary Record, v.118, n.3, p.333, 1986.
-
3- FAINE, S. Guidelines for leptospirosis control. World Health Organization, Geneva : WHO, 1982. 171p.
-
4- GERRITSEN, M.J.; OLYHOEK, T.; SMITS, M.A.; BOKHOUT, B.A. Sample preparation method for polymerase chain reaction-based semiquantitative detection of Leptospira interrogans serovar hardjo subtype hardjobovis in bovine urine. Journal of Clinical Microbiology, v.29, n.12, p.2805-8, 1991.
-
5- MASRI, S.A.; NGUYEN, P.T.; GALE, S.P.; HOWARD, C.J.; JUNG, S.C. A polymerase chain reaction for the detection of Leptospira spp. in bovine semen. Canadian Journal of Veterinary Research, v.61, n.1, p.15-20, 1997.
-
6- MÉRIEN, F.; AMOURIAUX, P.; PEROLAT, P.; BARANTON, G.; SAINT GIRON, I. Polymerase chain reaction for detection of Leptospira spp. in clinical samples. Journal of Clinical Microbiology, v.30, n.9, p.2219-24, 1992.
-
7- SAMBROOK, J.; FRITSCH; E.F.; MANIATIS, T. Molecular cloning: a laboratory manual. Cold Spring : Cold Spring Harbor, 1989. 956p.
-
8- SCHÖNBERG, A. Studies on the effect of antibiotic substances on leptospires and their cultivation from material with a high bacterial count. Zentralblatt für Bakteriologie, Parasitenkunde Infektionkrankheiten und Hygiene, v.249, n.5, p.400-6, 1981.
-
9- TURNER, L.H. Leptospirosis III. Maintenance, isolation and demonstration of leptospiras. Transactions of the Royal Society of Tropical Medicine and Hygiene, v.64, n.4, p.623-46, 1970.
-
10- VAN EYS, G.J.; GRAVEKAMP, C.; GERRITSEN, M.J.; QUINT, W.; CORNELISSEN, M.P.E.; SCHEGGET, J.T.; TERPSTRA, W.J. Detection of leptospires in urine by polymerase chain reaction. Journal of Clinical Microbiology, v.27, n.10, p.2258-62, 1989.
Publication Dates
-
Publication in this collection
20 Sept 2000 -
Date of issue
1999
History
-
Accepted
17 June 1998 -
Received
15 Oct 1997