Abstract
Blood serum samples of HYBRO PG broilers were analyzed, with 30 samples collected from 21-day-old broilers (G1), 30 from 35-day-old birds (G2), and 30 from 42-day-old birds (G3), with the aim of establishing normal values of some blood serum parameters. The activities of the enzymes gamma-glutamyl-transferase (GGT), aspartate aminotransferase (AST), creatine kinase (CK), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH), serum levels of total calcium, calcium ion, phosphorus, sodium, potassium, magnesium, chlorides, creatinine, uric acid, triglycerides, cholesterol, total protein, albumin, total and indirect and direct bilirubin, and electrophoretic profile of serum proteins in acrylamide (SDS-PAGE) and agarose gel were determined. There was no influence of age on total bilirubin and albumin levels. All the other evaluated parameters presented differences in at least one age group. Protein electrophoretic profile also changed as a function of age. The obtained results can be considered as normal for the studied ages, and therefore be used as references for the interpretation of laboratory exams of broilers of this genetic line in the evaluated ages.
Broiler; serum biochemistry; serum protein test
Blood serum components and serum protein test of Hybro-PG broilers of different ages
Silva PRLI; Freitas Neto OCII; Laurentiz ACIII; Junqueira OMIII; Fagliari JJI
IDepartment of Veterinary Clinics and Surgery of the School of Agriculture and Veterinary Sciences of Jaboticabal (FCAV-Unesp)
IIDepartment of Animal Pathology of the School of Agriculture and Veterinary Sciences of Jaboticabal (FCAV-Unesp)
IIIDepartment de Animal Science of the School of Agriculture and Veterinary Sciences of Jaboticabal (FCAV-Unesp)
Mail Address Mail Address: Jose Jurandir Fagliari FCAV-Unesp Via de acesso Paulo Donato Castellane, s/n 14.884-900. Jaboticabal, SP, Brazil E-mail: fagliari@fcav.unesp.br
ABSTRACT
Blood serum samples of HYBRO PG broilers were analyzed, with 30 samples collected from 21-day-old broilers (G1), 30 from 35-day-old birds (G2), and 30 from 42-day-old birds (G3), with the aim of establishing normal values of some blood serum parameters. The activities of the enzymes gamma-glutamyl-transferase (GGT), aspartate aminotransferase (AST), creatine kinase (CK), alkaline phosphatase (ALP), and lactate dehydrogenase (LDH), serum levels of total calcium, calcium ion, phosphorus, sodium, potassium, magnesium, chlorides, creatinine, uric acid, triglycerides, cholesterol, total protein, albumin, total and indirect and direct bilirubin, and electrophoretic profile of serum proteins in acrylamide (SDS-PAGE) and agarose gel were determined. There was no influence of age on total bilirubin and albumin levels. All the other evaluated parameters presented differences in at least one age group. Protein electrophoretic profile also changed as a function of age. The obtained results can be considered as normal for the studied ages, and therefore be used as references for the interpretation of laboratory exams of broilers of this genetic line in the evaluated ages.
Keywords: Broiler, serum biochemistry, serum protein test.
INTRODUCTION
In the last few years, the poultry industry has shown extraordinary growth in Brazil, which is currently one of the main global producers and exporters of broilers.
When an animal population grows, there is often a proportional increase in disease incidence. Diseases present such a wide variety of symptoms that the physical exam is not sufficient to provide a diagnosis.
Blood components may be influenced by physiological factors, such as age and species, and by pathological factors (Szabo et al., 2005; Lloyd & Gibson, 2006). The loss of cell membrane integrity caused by hypoxia or traumatism causes enzymes to leak to the extracellular fluid, where they are measured, allowing to determine the degree of cell or tissue lesions. These analyses are discussed in several studies with domestic animals (Jain, 2000), but only a few specifically refer to broilers (Kaneko et al., 1997).
The evaluation of the levels of total protein and its fractions supply the information required to interpret the occurrence of dehydration, infections, immune diseases, and inflammatory responses.
The determination of blood component values using laboratory exams is an important procedure to aid the diagnosis of several diseases and dysfunctions, as they provide reliable results, and may also give inputs for research studies on nutrition, physiology, and pathology (Bounous et al., 2000).
This study aimed at determining the levels of blood serum components of broilers of different ages, including serum protein test in acrylamide gel (SDS-PAGE), in order to provide useful data for the diagnosis and prognosis of broiler diseases.
MATERIAL AND METHODS
Ninety blood serum samples were collected from HYBRO-PG broilers, being 30 samples collected at 21 days of age (G1), 30 colleted at 35 days of age (G2), and 30 collected at 42 days of age (G3). Broilers were fed a corn and soybean meal-based diet, which supplied the nutritional requirements recommended by Rostagno (2000). The ages chosen for blood collection coincided with diet change (21 and 35 days of age), and with market age (42 days of age.
Blood samples were collected by puncture of the jugular vein in 21-day-old broilers, and axillary vein in 31- and 42-day-old broilers, using 5-ml plastic syringes, and 25 X 7 mm needles.
The activities of the enzymes gamma-glutamyl-transferase (modified Szasz' method), aspartate aminotransferase (Reitman-Frankel' method), creatine kinase (IFCC method), alkaline phosphatase (Bowers and McComb's method), lactate dehyidrogenase (piruvate-lactate method), serum levels of total calcium (Labtest method), calcium ion (ion-selective method), phosphorus (Basques-Lustosa's method), magnesium (Tonks' method), sodium (ion-selective method), potassium (ion-selective method), chlorine (Labtest method), creatinine (Basques-Lustosa's method), uric acid (Trinder's enzymic method), triglycerides (Trinder's enzymic method), cholesterol (Trinder's enzymic method), albumin (bromocresol green method), total protein (biuret method), total and direct bilirubin (Sims-Horn's method) were determined using of commercial kits (LABTEST). Samples reading were performed using spectrophotometry (LABQUEST semi-automatic spectrophotometer) with light wave length adequate for each test. Indirect bilirubin content was calculated by subtracting direct bilirubin level from total bilirubin level. Protein fractions were determined by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), as described by Laemmli (1970).
Serum components were expressed as means and standard deviation, which were submitted to statistical analysis. Means were compared by the test of Tukey after sample homogeneity was verified (Zar, 1999). Significance was considered at 5% probability level. Serum protein test results were expressed as lower and upper limits.
RESULTS AND DISCUSSION
Table 1 shows the means and standard deviations of the serum activities of the enzymes AST, ALP, GGT, LDH, and CK of healthy broilers of different ages: 21 days of age (G1), 35 days of age (G2), and 42 days of age (G3). Serum activity of all enzymes changed as a function of bird age. AST serum activity progressively increased with age, and LDH and CK activities were higher at older ages, which is consistent with the observations of other studies (Sandhu et al., 1998; Agawane & Lonkar, 2004). This is possibly due to the increase of liver metabolism and to the significant muscle development that usually happens during this period, as observed by Szabo et al. (2005) in turkeys of commercial strains. ALP serum activity, as opposed to AST, LDH, and CK, was higher at 21 days of age due to a higher bone development, as compared to older ages. The activity of GGT was not different between extreme ages (G1 and G3), but it was higher at 35 days of age, a phase when there is rapid body development due to higher feed intake and feed gain (Almeida et al., 2006), with a concurrent significant increase of liver metabolism. The differences detected among groups relative to serum enzymes are probably due to physiological changes that are normal in these different ages. Nevertheless, birds were not tested for mycotoxins, which may also influence these parameters and it is frequently present in farms.
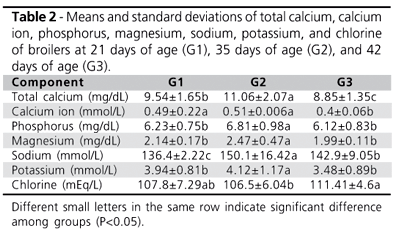
Table 2 shows the means and standard deviations of mineral serum levels. There were changes in the levels of Ca, P, Mg, Na, and K, with the highest values obtained at 35 days of age. Chlorine serum level was the lowest in G2. Calcium and phosphorus level at 21 days of age were similar to those observed in the control group in the study of Oguz et al. (2000), whereas sodium and potassium levels at 42 days of age were different from those analyzed in healthy broilers by Oguz et al. (2002). Harr (2002) stressed that blood mineral results levels may be different as a function of the methodology applied, and breed and genetic line may also influence these parameters (Gyenis et al., 2006). The higher mineral levels observed in the sera of 35-day-old birds are probably due to the higher nutrient supply (Rajman et al., 2006), and to the more intense metabolism that commonly occurs at this age (Almeida et al., 2006). Therefore, the obtained results suggest that the different blood mineral components usually vary with age due to physiological changes that happen at each development phase.
Serum levels of creatinine, uric acid, triglycerides, cholesterol, total protein, albumin, as well as total, direct and indirect bilirubin levels are presented in Table 3. It is observed that age did not influence total bilirubin and albumin levels, but all other components changed as a function of age. Creatinine level at 35 days of age was higher than in the other age groups, and no difference was detected between G1 and G3. The level of this catabolite directly related to muscle volume and activity, and therefore its lower blood levels in old and young chickens (Rajman et al., 2006; Agawane & Lonkar 2004; Sandhu et al., 1998). Uric acid level was higher in 21-day-old birds, and there was no difference between G2 and G3. the highest dietary protein level is supplied in the starter phase of production, and a study (Szabo et al., 2005) found a direct relation between the amount of ingested protein and uric acid serum level. Rajman et al. (2006) found higher uric acid levels in broiler breeders during the rearing phase, at 30 and 44 days of age, as compared to older breeders.
Triglycerides and total cholesterol present a similar pattern of variation as a function of age, with higher values in G1 and G3 birds, as observed in other studies (Sandhu et al., 1998; Oguz et al., 2002). The higher values of these components in G1 correspond to their low mobilization by tissues and to their intense synthesis by the liver (Szabo et al., 2005). At 42 days of age, the increase of these values is related to the higher dietary energy supply (Rajman et al., 2006). The lower triglycerides and cholesterol levels at 35 days of age are due to the bird's high energy requirement at this stage caused by high body development (Almeida et al., 2006).
The evaluation of total protein serum levels did not reveal any differences between G2 and G3; however, at 21 days of age, its level was lower, due to the high requirement for growth. These data are compatible with those presented above for uric acid level, main catabolite of protein metabolism, which presented the highest serum levels at 21 days of age. Direct and indirect bilirubin levels were different in the three studied ages, and their variation is probably linked to variation in liver metabolism as a function of age.
Table 4 shows the results of the serum protein test obtained in acrylamide gel SDS-PAGE). Electrophoretic fractioning performed by this technique allowed the identification of 24 proteins, out of which 11 were identified by their name, including acute-phase proteins and immunoglobulins, whereas the remaining 13 were identified on molecular weight basis.
Acknowledgements
The authors thank FAPESP for providing financial support, and Renata Lemos Nagib Jorge and Paulo César Silva for their technical cooperation.
Arrived: June / 2007
Approved: January / 2008
- Agawane SB, Lonkar PS. Effect of prbiotic containing Saccharomyces boulardii on experimental on experimental ochratoxicosis in broilers: hematobiochemical studies. Journal of Veterinary Science. 2004; 5(4):359-367.
- Almeida JG, Vieira SL, Gallo BB, Conde ORA, Olmos AR. Period of incubation and posthatching holding time influence on broiler performance. Brazilian Journal of Poultry Science 2006; 8 (3):153-158.
- Bounous ID, Wyatt RD, Gibbs PS, Kilburn JV, Quist CF. Hematologic and serum biochemical reference intervals for juvenile wild turkeys. Journal of Wildlife Diseases 2000; 36(2):393-396.
- Gyenis J, Suto Z, Romvari R, Horn P. Tracking the development of serum biochemical parameters in two laying hen strains: a comparative study. Archieve. Tierzlander 2006; 49(6):593-606.
- Jain NC. Schalm's veterinary hematology. 5th ed. Baltimore: Lippincott; 2000.
- Kaneko JJ, Harvey JW, Bruss ML. Clinical biochemestry of domestic animals. 5th ed. San Diego: Academic Press; 1997.
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophague T4. Nature 1970; 227:680-685.
- Lloyd S, Gibson JS. Haematology and biochemistry in healthy young pheasants and red-legged partridges and effects of spironucleosis on these parameters. Avian Patology 2006; 35(4):335-340.
- Oguz H, Kecect T, Birdane YO, Onder F, Kurtoglu V. Effect of clinoptilolite on serum biochemical and haematological characters of broiler chickens during the aflatoxicosis. Research in Veterinary Science 2000; 73:101-103.
- Oguz H, Kurtoglu F, Kurtoglu V, Birdane YO. Evaluation of biochemical characters of broiler chickens during dietary aflatoxin (50 and 100 ppb) and clinoptilolite exposure. Research in Veterinary Science 2002; 73:101-103.
- Rajman M, Jurani M, Lamosova D, Macajova M, Sedlackova M, Kostal L, Jesova D, Vyboh P. The effects of feeed restriction on plasma biochemistry in growing meat type chickens (Gallus gallus). Comparative Biochemistry and Phisiology 2006; 145:363-371.
- Rostagno HS, Albino LFT, Donzele JL. Composição de alimentos e exigências nutricionais de aves [tese]. Viçosa: Universidade Federal de Viçosa; 2000.
- Sandhu BS, Singh B, Brar RS. Haematological and biochemical studies in broiler chickens fed ochratoxin and inoculated with inclusion body hepatitis virus, singly and in concurrence. Veterinary Research Communications 1998; 22:335 346.
- Szabo A, Mezes M, Horn P, Suto Z, Bazar G, Romvari R. Developmental dynamics of some blood biochemical parameters in the growing turkey (Meleagris Gallopavo). Acta Veterinary Hungary 2005; 53 (4):397-409.
- Zar JH. Bioestatistical analysis. 4th ed. New Jersey: Prentice Hall; 1999.
Mail Address:
Publication Dates
-
Publication in this collection
12 Mar 2008 -
Date of issue
Dec 2007
History
-
Accepted
Jan 2007 -
Received
June 2007