Abstract
The objectives of the present study were to determine the parameters of Gompertz equations and to determine curves and growth rate, feed intake and body component deposition, as well as allometric coefficients of body water, protein, and fat relative to live weight of male and female broilers of intermediate performance (C44) and high performance (Cobb-500) genetic strains. In total, 384 one-d-old chicks were distributed into four treatments: male Cobb 500, male C44, female Cobb 500, and female C44, with six replicates of 16 birds, according to a completely randomized experimental design. Average body weight, weight gain, and feed intake were weekly determined, and six birds, representing the average weight of each treatment, were sacrificed to determine body composition. Growth curves were built applying Gompertz function, with excellent fit, and growth, feed intake, and tissue deposition rates were obtained by its derivatives. Superior growth rate was obtained for Cobb 500 male broilers. This genetic strain has higher feed intake capacity, which is achieved earlier than in the C44 strain. Protein and fat deposition maturity was reached earlier in males than in females in Cobb 500. The allometric coefficients showed earlier maturity for body water in C44 and females. In terms of body protein, male Cobb 500 broilers reached maturity earlier than females and C44. Body fat deposition maturity was reached earlier in Cobb 500 than in C44. The Gompertz equations obtained in the present study efficiently described body growth, feed intake, and deposition of body components, with a coefficient of determination higher than 0.99.
Body composition; Gompertz curve; growth rate; Cobb 500; C44
Growth and deposition of body components of intermediate and high performance broilers
Henn JDI,III; Bockor LI; Ribeiro AMLII; Coldebella AIII; Kessler A de MII
ILaboratório de Ensino Zootécnico (LEZO) - UFRGS. E-mail: joao.henn@embrapa.br; lubockor@hotmail.com
IIProfessor Associado do Departamento de Zootecnia, Programa de Pós-Graduação em Zootecnia da Faculdade de Agronomia (UFRGS).Avenida Bento Gonçalves, 7712. CEP: 91540-000, Porto Alegre, RS, Brasil, E-mail: aribeiro@ufrgs.br; akessler@ufrgs.br
IIIEmbrapa Suínos e Aves.BR 153, km 110, Distrito de Tamanduá, Caixa Postal 21, Concórdia, SC. Cep: 897000-000. E-mail: arlei.coldebella@embrapa.br
Correspondence Corresponding author e-mail address: Alexandre de Mello Kessler Av.Bento Gonçalves, 7712. 91.540-000. Porto Alegre, RS, Brazil Phone: (51) 3308 7424 E-mail: akessler@ufrgs.br
ABSTRACT
The objectives of the present study were to determine the parameters of Gompertz equations and to determine curves and growth rate, feed intake and body component deposition, as well as allometric coefficients of body water, protein, and fat relative to live weight of male and female broilers of intermediate performance (C44) and high performance (Cobb-500) genetic strains. In total, 384 one-d-old chicks were distributed into four treatments: male Cobb 500, male C44, female Cobb 500, and female C44, with six replicates of 16 birds, according to a completely randomized experimental design. Average body weight, weight gain, and feed intake were weekly determined, and six birds, representing the average weight of each treatment, were sacrificed to determine body composition. Growth curves were built applying Gompertz function, with excellent fit, and growth, feed intake, and tissue deposition rates were obtained by its derivatives. Superior growth rate was obtained for Cobb 500 male broilers. This genetic strain has higher feed intake capacity, which is achieved earlier than in the C44 strain. Protein and fat deposition maturity was reached earlier in males than in females in Cobb 500. The allometric coefficients showed earlier maturity for body water in C44 and females. In terms of body protein, male Cobb 500 broilers reached maturity earlier than females and C44. Body fat deposition maturity was reached earlier in Cobb 500 than in C44. The Gompertz equations obtained in the present study efficiently described body growth, feed intake, and deposition of body components, with a coefficient of determination higher than 0.99.
Keywords: Body composition, Gompertz curve, growth rate, Cobb 500, C44.
Introduction
Body growth is represented by the sum of protein, fat, water, and ash depositions. In broilers, the deposition rates and the ratio of these components characterize the physiological age of the birds and their maturity stage, which are influenced by genotype and environment (Vincek et al., 2011). During the last few decades, genetic improvement has developed broilers with high daily weight gain and high feed intake capacity (Sakomura et al., 2005; Sakomura et al., 2011). The selection for traits such as growth rate, body composition, and feed efficiency has allowed annual genetic gains of 2-3% in the efficiency of meat production. Improvements in other traits, such as robustness, specific and general disease resistance, and absence of metabolic defects, also contribute for this progress (McKay, 2008). When comparing a modern strain (Ross 708) with a strain that had not been selected since 1950, Schmidt et al. (2009) found, at 35 days of age, that the modern broilers presented higher growth capacity (1.8 vs. 1.0 kg live weight) and better feed efficiency. The modern birds also presented higher breast yield (18 vs. 9%), earlier liver development and function, and longer jejunum and ileum, suggesting better nutrient absorption and utilization.
Mathematical modeling have been increasingly applied in animal production to describe biological phenomena, allowing the comparison of growth rates and the description of weight and body tissue deposition evolution as a function of age (Gous et al., 1999; Freitas et al., 2005; Darmani Kuhi et al., 2010; Gous, 2014).
Several models are proposed in literature. However, the Gompertz function is preferred to describe growth and nutrient deposition in poultry, as only three parameters are required. These include, for instance, weight at maturity (asymptotic value), growth rate, and age when maximum weight gain is achieved. These parameters have biological meaning and allow better data fit than other more complex growth functions. Comparing several mathematical functions, Hruby et al. (1996) concluded that the Gompertz equation provided the most precise description of protein content at several ages. The desirable characteristics of the Gompertz equation were confirmed by Fialho (1999).
Different genetic strains and sexes of broilers may present different weight at maturity, body composition, and chemical component ratios that affect the characteristics of the growth curve. The simulation of growth and of body component deposition using mathematical models allows estimating live weight and body component growth as a function of age, and therefore, comparing relative body growth between sexes, as well as estimating feed conversion ratio, feed intake, and daily weight gain. The biological growth of broilers follow a sigmoidal pattern, with an initial slow growth rate, which increases as bird ages (acceleration) up to a maximum rate (inflection point), after which it gradually decreases (deceleration). After the inflection point, the slope of the curve becomes convex, rather than concave, corresponding to age at maximum growth. More important than the precise inflection point is the size of the section of the curve when growth rate is constant, when lean deposition is highest (Kessler et al., 2000). In general, the inflection point occurs at weights lower than half of mature weight (Darmani Kuhi et al., 2010), but this may be influenced by age sex, genetics, and bird type.
The objectives of the present study were to determine the parameters of Gompertz equations and to determine growth curves and rates, feed intake and body component deposition, as well as allometric coefficients of body water, protein, and fat relative to live weight of male and female broilers of intermediate performance (C44) and high performance (Cobb-500) genetic strains.
Material and methods
The procedures adopted in this experiment comply with the Brazil guidelines for the scientific use of animals (Federal Act n. 11794 as of October 8, 2008) and were approved by the Committee of Ethics of the Federal University of Rio Grande do Sul. The experiment was carried out at Laboratório de Ensino Zootécnico – LEZO, of Universidade Federal do Rio Grande do Sul, Porto Alegre, RS, Brazil (30°04'36"S.51°07'19"W). A number of 384 one-day-old chicks, with 192 from a high-performance strain (Cobb 500) and 192 from an intermediate-performance, free-range meat strain (C44). Birds were distributed into four treatments: Cobb 500 males (Cobb-M), C44 males (C44-M), Cobb 500 females (Cobb-F), and C44 females (C44-F), according to a completely randomized experimental design with six replicates of 16 birds per treatment. Birds were housed in an environmentally-controlled room, with central air conditioning and exhaustion fan, and were distributed into 24 pens (1 m2). Initially, there were 16 chicks per pen, and 10 during the last week due to the weekly slaughter. Room temperature and relative humidity were daily recorded during the entire experimental period (Table 1). New wood-shavings litter (predominantly consisting of Pinus elliottii) was used.
All chicks were fed the same pre-starter diet during the first week. Thereafter, a starter and a grower diet (Tables 1 and 2) were supplied as mash ad libitum in tube feeders. Diets followed the specifications for genetic strain and sex and were formulated according to Rostagno et al. (2005). Clean and fresh water was supplied in nipple drinkers.

Click to enlarge
Birds and feeds were weekly weighed to determine average body weight, weight gain, and feed intake per bird in each replicate. Mortality was recorded to correct average feed intake and average feed conversion ratio at the end of each phase.
At the start of the experiment and at 7, 14, 21, 28, 35, 42, and 49 days of age, six birds representative of the average weight of each treatment (one per replicate) were sacrificed, plucked, and then their crop, gizzard, and intestinal contents removed and weighed. Feather percentage was estimated as the difference in body weight before and after plucking. Feather samples were collected for analyses. The remaining carcass with feet and head, offal, and blood was frozen at -20°C. , and then sawed and ground in an industrial meat grinder for three times to obtain good sample homogeneity. Subsamples weighing approximately 250 g were taken, dried in a forced-ventilation oven at 60°C for 72 hours, ground in a ball mill, and stored for subsequent analyses. Feedstuffs, experimental diets, and body samples were analyzed for dry matter, crude protein, and fat contents (AOAC, 1993).
In order to evaluated the growth of the two evaluate strains and sexes, the Gompertz equation (1825) was applied as suggested by Gous et al. (1999). Parameters a, b, and c are fit to the Gompertz model, modified according to Equation 1.
Equation 1:
Y=a*exp(-exp(-b*(Idade-c))), where
a = asymptotic value (for instance, weight at maturity or mature weight).
b = maturity rate, which is a function between maximum growth rate and mature weight. The higher this value, the earlier the bird will reach its mature weight.
c = age when maximum growth rate is achieved, and it is the inflection point of the sigmoidal curve.
Growth, feed intake and body component (water, protein, and fat, in g/day) deposition rates as a function of time were calculated using the derivative of the Gompertz equation (equation 2) presented by Fialho (1999). The allometric growth of body water, protein, and fat relative to live weight was calculated using exponential equations (Equation 3).
Equation 2:
Y=a*b*exp(-b*(age-c)-exp(-b*(age-c))).
Equation 3:
Y=aXb, where
Y = weight of each body component (total body water, protein, and fat)
X = body weight
a = intercept of the logarithm of the linear regression on Y.
b = relative growth coefficient or coefficient of allometry.
The allometric coefficients "b" and "a" value for body water, protein, and fat of each treatment were submitted to analysis of variance using the GLM procedure of SAS (SAS INSTITUTE INC, 2008). Finally, the t test was used to compare the means of every two treatments whenever the F test detected significant effect of treatments.
The NLIN procedure of SAS statistical package (SAS INSTITUTE INC, 2008) was used to fit the data and to estimate parameter values (a, b, c) of the Gompertz curve for the responses live weight, feed intake, and body water, protein, and fat deposition. The effect of treatments on the estimates of the curve parameters was tested by analysis of variance, using the GLM procedure. The t test was used to compare the means of every two treatments whenever the F test detected significant effect of treatments (p<0.05). Variables were also analyzed using the theory of mixed models for repeated measures and 16 types of variance and covariance matrix structures by PROC MIXED (Xavier, 2000). The choice of variance and covariance structure was based on the lowest value of the Akaike information criterion (AIC). Restricted maximum likelihood was used as estimation method.
Results and Discussion
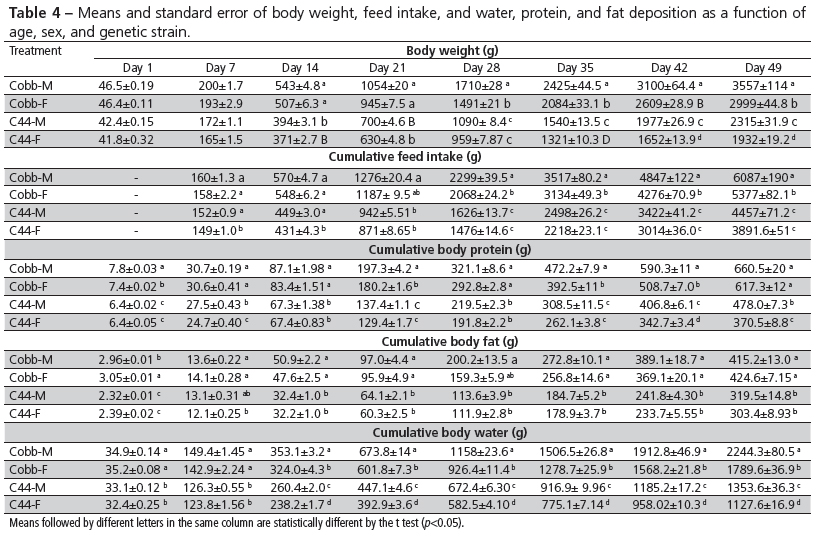
Significant differences (p<0.05) between genetic strains and between sexes within each strain were detected, with higher averages obtained in Cobb 500 broilers and male broilers (Table 4).
For most Gompertz parameters (Table 5), Cobb broilers presented higher asymptotic values than C44 broilers, and within strain, males presented higher asymptotic values than females. Considering growth curves, males and females of the same strain presented different (p<0.05) weight at maturity (a) and similar age at maximum growth rate (c), indicating that the curves have a similar shape (Figure 1), but higher daily weight gain for males due to their heavier weight at maturity (a). Maximum growth rate (b) in grams was higher in Cobb 500 and male broilers of both evaluated strains. Maturity rate value was higher in Cobb 500 and equal for sexes of both strains, showing that C44 broilers grow slower as a function of their slower maturity rate and older age at maximum growth rate.
There is a large variation when the values obtained in the present study are compared with the findings of other studies, mostly due to genetic strain and age. Marcato et al. (2008) found that Cobb 500 males were heavier at maturity (6812 g), but females were not different (4282 g) compared to the present study (5178 e 4256 g, for males and females, respectively). Also, age at maximum growth was nine days later for males (39.41 vs. 29.6) and three days later for females (32.07 vs. 28.66), indicating that this strain currently has higher growth rate and matures earlier. When compared with the results of Sakomura et al. (2005), age at maximum growth was achieved nine days earlier in males and females, whereas in comparison with the findings of Gous et al. (1999), age at maximum growth was achieved 10 days earlier in males and 13 days earlier in females in the present study.
Evaluating slower growing strains, Dourado et al. (2009) obtained 4301 to 2009 g of weight at maturity for male and female Sasso and ISA Label broilers, and observed higher values for Sasso broilers of both sexes. On the other hand, Santos et al. (2005) obtained higher values for the strain Paraíso Pedrês (4764 and 4222 g at the ages of 44 and 48 days for males and females, respectively) and for ISA Label broilers (4230 and 3136 g at the ages of 52 and 53 days for males and females, respectively). Using the Gompertz equation, Hancock et al. (1995) obtained mature weights of 5171-6145 for males and 4279-4705 for females of six different strains, with maximum growth rates of 0.0355-0.0371 for males and 0.0363-0.0382 for females, with age at the inflection point between 39.2 and 41.8 for females and 41.9 and 44.2 for males.
Feed intake curves were characterized by lower asymptotic value for C44 females, and similar values for the other treatments. The b value was higher in Cobb 500 and similar for males and females, independently of strain. Cobb 500 broilers were younger at maximum feed intake than C44. Males presented higher maximum feed intake rates than females in both strains.
Relative to body water deposition, Cobb 500 males presented higher asymptotic values than females, and Cobb 500 of both sexes presented higher values than C44, which did not present any differences between sexes. Maximum water deposition rate was higher in Cobb 500 and male broilers of both evaluated strains.
Relative to body protein deposition, the asymptotic values obtained for Cobb 500 males and females were similar and higher than C44 broilers, which males presented higher values. Protein deposition maturity rate was higher in Cobb 500 males than in females, and higher in Cobb 500 broilers and males in general.
The asymptotic values of body fat deposition were not different (p<0.05) among treatments due to the high variability in the obtained data. This variability may have been due to the earlier slaughter (49 days) compared with other studies (Gous et al., 1999; Sakomura et al., 2005), where birds were slaughtered at 120 days of age. Slaughter at older ages to determine Gompertz parameters seems to be more important for slow-growing strains, such as C44, and for traits that are manifested later, such as fat deposition. Cobb 500 males presented higher growth rate (b), indicating they deposit fat earlier compared with the other treatments, which presented similar values. Maximum fat deposition rate was obtained earlier in Cobb 500 compared with C44, independently of sex. C44 females presented the lowest maximum body fat deposition rate, whereas it was not different in the other treatments. According to Kessler & Snizek (2001), protein deposition is controlled by genetics, and therefore, there is a limit for its daily deposition, independently of nutrient intake. However, it may be influenced by feed intake, diet, and environmental conditions. On the other hand, fat deposition in any rearing phase is directly related to the amount of nutrients (proteins, carbohydrates, fats) available for its synthesis, and it is particularly influenced by energy intake.
The higher growth and feed intake rates (p<0.05) observed in males (Table 5) are explained by their higher potential of gain relative to females within each strain (Figure 1).
Growth rate increases with age until a maximum rate is achieved, and then gradually decreases. This is called the inflection point of the curve, which shape changes from concave to convex, and corresponds to age at maximum growth (Kessler et al, 2000). The differences observed in the parameter "b" of the equation indicate differences in maximum growth and tissue deposition rates. According to Fialho (1999), higher rates indicate that birds are younger (c) when the maximum rate is achieved. Longo et al. (2000) observed that males have higher growth potential, and consequently higher capacity of nutrient deposition than females, except for fat deposition.
Although males presented higher body growth rates than females, the behavior of both growth curves was similar: growth rates rapidly increased up to 30 days of age, and then decelerated. On the other hand, protein deposition rates presented different behavior between sexes, and were higher in males.
In slow-growing strains, Figueiredo et al. (2003) observed that EMBRAPA 041 broilers presented higher growth rate (43.3 g/d at 56 days of age) compared with Label Rouge broilers, which growth rate was slower (38.5 g/d at 56 days of age). The authors also obtained 45.41 g/d at 49 days of age in males and 35.46 g/d in females of the Sasso strains, whereas for the ISA Label strains, 40.33 g/d and 31.77 g/d were calculated for 49-d-old males and 42-d-old females, respectively. In the present study, daily weight gain of C44 males was 62.1 g/d at 32 days of age and 50.0 g/d for C44 females at 30 days of age (Table 4), which values are higher than those found in the above-mentioned studies.
Feed intake rates were similar for C44 males and females, with maximum intake at 44 days of age, with 142 g/d for males and 120 g/d for females. The feed intake capacity of Cobb 500 broilers was much higher: 188 g/d for males and 163 g/d for females, both at 38 days of age.
Figure 2 shows body water, protein, and fat deposition curves and rates (g/d) based on the values on Table 5. Male Cobb 500 broilers presented higher and earlier maximum protein deposition rate than females. However, after 40 days of age, the situation was reversed: females presented higher protein deposition rate because it decreased in males. Cobb 500 broilers presented higher rate than C44 broilers. C44 males also presented higher maximum protein deposition rate than females, but at an older age than females. Fat deposition occurred later in C44 than in Cobb 500. Cobb 500 males reached maximum rate earlier than females (days 29 and 37, respectively), but equivalent maximum rate values. Maximum water deposition rate was higher in Cobb 500 and in males of both strains, but at different ages.
Boekholt et al. (1994) observed lower lipid deposition and higher protein deposition at the same final weight in fast-growing broilers compared with those of a slow-growing strain. The studies of Gous et al. (1999) and Longo (2000) obtained higher fat deposition rate at maturity in males than in females, as observed in Cobb 500 broilers in the present study. According to Kessler et al. (2000), higher and longer plateau of protein deposition indicate that broilers present better meat production efficiency and better carcass composition.
All the evaluated responses presented excellent fit to the applied non-linear model, with a R2>0.99. Although feed intake responses do not have asymptotic behavior, we chose to maintain the same model because it had excellent fit and because the estimates of this study were limited to slaughter age, and not to mature age.
Theoretically, when b=1, allometric growth is called isogonic, indicating that development rates from "X" to "Y" are similar within the growth interval considered (Table 6). When b≠1, growth is called heterogonic, and it is positive (b>1) when growth is slow or negative (b<1) when growth is fast. Water deposition occurred earlier than protein, followed by fat deposition (p<0.05). C44 broilers deposited water earlier than Cobb 500, as well as females compared with males (p<0.05). In general, body water deposition occurred earlier than weight gain. All allometric coefficients were significant (p<0.05) for all parameters and treatments.
The allometric coefficients of protein were lower for Cobb 500 than for C44, and for Cobb 500 male than Cobb 500 females. No differences were detected between C44 males and females. Protein deposition was similar to weight gain, and it occurred slightly late. This may be explained by the proportional increase in protein deposited in feathers as birds age, as feathers have high protein and low water content. Fat was the tissue with the latest deposition. It occurred earlier in Cobb 500 than in C44 and in males compared with females, independently of genetic strain.
CONCLUSIONS
The Gompertz equations obtained in the present study efficiently described body growth, feed intake, and deposition of body components, with a coefficient of determination higher than 0.99.
Submitted: December/2013
Approved: May/2014
- Association of Official Agricultural Chemists (AOAC). Official Methods of Analysis. 16 ed. Washington, 1993.
- Boekholt HA, Van der Grinten PH, Schrours VVAM, Los MJN, Lefferinge CP. Effects of divergent selection for body weight on three skeletal muscles characteristics in the chicken. British Poultry Science, v.35, p.603-614, 1994.
- Darmani HK, Porter T, LÓPEZ S, Kebreab E, Strathe AB, Dumas A, Dijkstra J, France J. A review of mathematical functions for the analysis of growth in poultry. World's Poultry Science Journal, 66, p. 227-240, 2010.
- Dourado LRB, Sakomura NK, Nascimento DCN, Dorigam JC, Marcato S M, Fermandes JBK. Crescimento e desempenho de linhagens de aves pescoço pelado criadas em sistema semi-confinado. Ciência e Agrotecnologia, Lavras, v. 33, n.3, p. 875-881, maio/junho de 2009.
- Fialho FB. Interpretação da curva de crescimento de Gompertz. Concórdia. Embrapa- CNPSA, p.1-4. (Comunicado Técnico 237), 1999.
- Figueiredo EAP, Avila VS, Schmidt GS, Baroni Júnior W, Coldebella A, Piccinin I. Curvas de crescimento de linhagens criadas em sistema alternativo. Revista Brasileira de Ciências Avícolas, Campinas, v.5, n.5, p. 111, 2003. Suplemento.
- Freitas AR. Curvas de crescimento na produção animal. Revista Brasileira de Zootecnia, v.34, n.3, p. 786-795, 2005.
- Gompertz B. On the nature of the function expressive of the law of human mortality and on a new method of determining the value of life contingencies. Philosophical Transactions of the Royal Society, v.115, p. 513-585, 1925.
- Gous RM, Moran Jr ET, Stilborn HR, Bradford GD, Emmans GC. Evaluation of the parameters needed to describe the overall growth, the chemical growth, and the growth of feathers and breast muscles of broilers. Poultry Science, v.78, n.6, p.812-821, 1999.
- Gous RM. Modeling as a research tool in poultry science. Poultry Science, 93: 1-7, 2014.
- Hancock CE, Bradford GD, Emmans GC, Gous RM. The evaluation of the growth parameters of six strains of comercial broiler chickens. British Poultry Science, Basingstoke, v.36, p.247-264, 1995.
- Hruby M, Haure KL, Coon CN. Non-linear and linear functions in body protein growth. Journal of Applied Poultry Research, v.5, p. 109-115, 1996.
- Kessler AM, Snizek PN, Brugalli I. Manipulação da quantidade de gordura na carcaça de frangos. Conferência Apinco de Ciência e Tecnologia Avícolas. FACTA, Campinas, SP, Brazil. p.117-133, 2000.
- Kessler AM, Snizek PN. Considerações sobre a quantidade de gordura na carcaça do frango. In: REUNIÃO ANUAL DA SOCIEDADE BRASILEIRA DE ZOOTECNIA, 2001, Piracicaba. Anais... Piracicaba: Sociedade Brasileira de Zootecnia, 2001. p.111-159.
- Longo F. Estudo do metabolismo energético e do crescimento de frangos de corte. (Dissertação). Jaboticabal (SP): Faculdade de Ciências Agrárias e Veterinárias – Universidade Estadual Paulista; 2000.
- Marcatto SM, Sakomura NK, Munari DP, Fernandes JBK, Kawauchi IM, Bonato MA. Growth and body nutrient deposition of two broiler commercial genetic lines. Brazilian Journal of Poultry Science, v. 10, n.2, p. 117-123, 2008.
- McKay JC. The genetics of modern commercial poultry. In: Proceedings of the XXIII World's Poultry Congress, Brisbane, Australia. 2008.
- Rostagno HS, Albino LFT, Donzelle JL. Tabelas brasileiras para aves e suínos: Composição de alimentos e exigências nutricionais. 2 ed. Viçosa: UFV. Departamento de Zootecnia, 186p., 2005.
- Sakomura NK, Longo FA, Oviedo-Rondon EO, Boa-Viagem C, Ferraudo A. Modeling energy utilization and growth parameter description for broiler chickens. Poultry Science, 84: 1363-1369, 2005.
- Sakomura NK, Gous R M, Marcato SM, Fernandes JBK. A description of the growth of the major body components of 2 broiler chicken strains. Poultry Science, 90: 2888-2896, 2011.
- Santos AL, Sakomura NK, Freitas ER, Fortes CMLS, Carrilho ENVM, Fernandes JBK. Estudo do crescimento, desempenho, rendimento de carcaça e qualidade de carne de três linhagens de frango de corte. Revista Brasileira de Zootecnia, v.34, n.5, p.1589-1598, 2005.
- Schmidt CJ, Persia ME, Feierstein B, Kingham B, Saylor WW. Comparison of a modern broiler line and a heritage line unselected since the 1950s. Poultry Science, 84: 2610 - 2619, 2009.
- SAS INSTITUTE INC. SAS/STAT software: changes and enhancement through release 9.2. Cary: SAS Institute, 2008.
- Vincek D, Kralik G, Kusec G, Sabo K, Scitovski R. Aplication of growth functions in the prediction of live weight of domestic animals. Central European Journal of Operations Research, p.1-15, 2011.
- Xavier LH. Modelos univariado e multivariado para análise de medidas repetidas e verificação da acurácia do modelo univariado por meio de simulação. Piracicaba, 2000. 91 p. Dissertação (mestrado) – Escola Superior de Agricultura "Luiz de Queiroz", Universidade de São Paulo, 2000.
Publication Dates
-
Publication in this collection
01 Oct 2014 -
Date of issue
Sept 2014
History
-
Accepted
May 2014 -
Received
Dec 2013