Abstract
The objective of the present study was to evaluate the quality of different glycerine sources produced in Brazil and to analyze their metabolizable energy levels and digestibility for broilers. Firstly, the composition of 10 samples of glycerine from different sources was analyzed. Secondly, glycerine from four different sources presenting contrasting compositions were selected to determine their metabolizable energy levels and digestibility in metabolism assays using 200 broilers between 21 and 29 days of age, applying the method of total excreta collection. The values of apparent metabolizable energy corrected for nitrogen balance of the four glycerine sources were 3145, 5026, 2828, and 2892 kcal/kg.
Alternative feedstuffs; broilers; metabolizable energy; glycerol
Metabolizable energy of different glycerine sources derived from national biodiesel production for broilers
Zavarize KCI; Menten JFMII; Pereira RII; Freitas LW; Romano GG; Bernardino M; Rosa AS
IUniversidade de São Paulo/ESALQ - Departamento de Zootecnia - Piracicaba/SP
Correspondence Correspondence : Kelen Cristiane Zavarize Departamento de Zootecnia - ESALQ/USP Avenida Pádua Dias, nº11 Agronomia - Piracicaba - SP CEP: 13418-900 E-mail: kelen_zavarize@yahoo.com.br
ABSTRACT
The objective of the present study was to evaluate the quality of different glycerine sources produced in Brazil and to analyze their metabolizable energy levels and digestibility for broilers. Firstly, the composition of 10 samples of glycerine from different sources was analyzed. Secondly, glycerine from four different sources presenting contrasting compositions were selected to determine their metabolizable energy levels and digestibility in metabolism assays using 200 broilers between 21 and 29 days of age, applying the method of total excreta collection. The values of apparent metabolizable energy corrected for nitrogen balance of the four glycerine sources were 3145, 5026, 2828, and 2892 kcal/kg.
Keywords: Alternative feedstuffs, broilers, metabolizable energy, glycerol
INTRODUCTION
Brazil is the third largest meat producer in the world. It produced 13 million tons of chicken meat in 2011, with approximately 3.5 million tons exported (AVISITE, 2011).
Corn is the main energy source used in poultry feeds. Approximately 80% of Brazilian corn production is used in the formulation of feeds. Corn accounts for 65-70% of poultry feed composition. It is a costly grain also used in human foods, and its replacement in feeds by other energy sources may reduce animal feeding costs.
The use of alternative feedstuffs, particularly in poultry production, may reduce production costs and increase the availability of traditional ingredients for human foods. According to Girotto et al. (2003), when considering the use of alternative feedstuffs, producers should take into account their availability in the market, quality, and price relative to the traditional ingredients in order to obtain price advantage, but not overlooking their quality.
Among alternative feedstuffs, glycerine is an energy-rich and low-cost ingredient. Crude glycerine obtained from biodiesel production contains 70-80% glycerol. It needs to be purified for plastic, resin, and adhesive industrial applications, and can be used in the cosmetic and pharmaceutical industries when presenting high purity.
The large volume of glycerine generated by the biodiesel industry may cause environmental problems if not properly disposed.
Glycerine is becoming an economically interesting energy feedstuff; however, its chemical composition can be widely variable, with consequent variable results in terms of poultry performance and health. Therefore, its composition and nutritional value should be known in order to determine its inclusion levels in feeds.
The objective of the present study was to evaluate the quality of different glycerine sources produced in Brazil and to analyze their metabolizable energy levels and digestibility for broilers.
MATERIALS AND METHODS
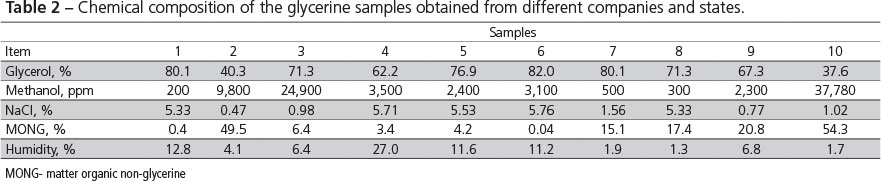
This study was performed at the Department of Animal Science of ESALQ/USP, Piracicaba, SP, Brazil. Glycerine samples were collected in 10 biodiesel companies in Brazil for chemical analyses. Companies were classified by region, size, and raw material (Table 1). A sample of 1000 mL of glycerine was collected per company and submitted to a specialized laboratory, blinded for company, region, and raw material for the determination of the percentages of glycerol, methanol, sodium chloride (NaCl), fatty acids, and moisture (Table 2).
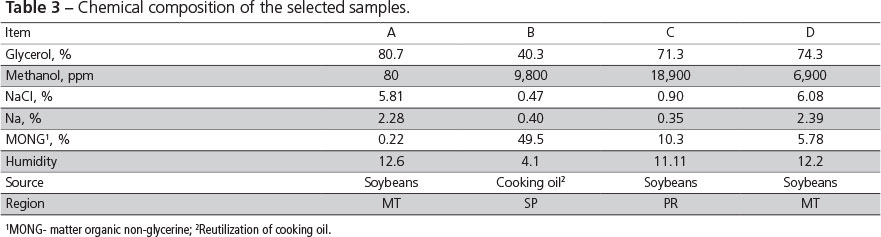
After chemical analyses, glycerine samples were grouped according to similarity of composition. Samples from four different companies, with variable glycerol and methanol composition, were selected. The selected companies then sent new glycerine samples that were again analyzed (Table 3), and used in the metabolism assay. Samples A, B, C, and D in Table 3 were obtained from glycerine from companies 1, 2, 3, and 4 in Table 2, respectively.
The metabolism assay with the glycerine samples selected in the previous phase was performed with broilers to estimate metabolizable energy values, using the traditional method of total excreta collection.
One-d-old Cobb 500 broilers were housed in an experimental broiler house and were offered a control diet and water ad libitum. On day 21, 200 birds were weighed and transferred to metabolic cages in a metabolism room.
A randomized block experimental design was applied, consisting of five treatments with eight replicates of five birds each. Treatments consisted of a reference diet and four test diets (four glycerine sources with different compositions - A, B, C, and D). The assay was carried out on days 21-29, with four days of adaptation to the cages and to the experimental diets, and four days of total excreta collection. Feed intake and excreta output were recorded during the collection period. Excreta were collected twice daily in order to prevent possible contamination and fermentation of the excreta. Plastic-lined trays were placed under each cage.
The reference diet (Table 1) was based on corn and soybean meal, and formulated according to the recommendations of Rostagno et al. (2005). The test diets consisted of 90% of the reference diet and 10% glycerine (sources A, B, C, or D). The beginning and end of the excreta collection period were determined by adding 1% ferric oxide to the diets and the output of pigmented excreta.
The collected excreta were placed in plastic bags and frozen at -18°C until the end of the collection period. At the end of the experimental period, feed intake was determined. Excreta were subsequently thawed, homogenized, and weighed, and a representative sample of each replicate was removed and submitted to analyses. These samples were pre-dried in a forced-ventilation oven at 65°C for 72 hours. Samples were then ground and stored in plastic recipients. Gross energy was determined in a bomb calorimeter (model Parr 1261). Nitrogen content in the diets, excreta, and glycerine samples was determined by combustion using automated equipment (LECO).
Chemical analysis results, feed intake, and excreta output were used to calculate apparent metabolizable energy (AME), apparent metabolizable energy corrected for nitrogen balance (AMEn), and gross energy metabolizability coefficient of the glycerine samples, according to the methodology of Sakomura & Rostagno (2007).
Results were submitted to analysis of variance using the PROC GLM (General Linear Model Procedures) of SAS® (Statistical Analysis System, 2006). Means were compared by the test of Tukey-Kramer, previously adjusted by the least-square mean method (LSMEANS).
RESULTS AND DISCUSSION
The chemical composition of the selected glycerine samples are presented in Table 3. The values show that the composition of glycerine produced in Brazil is widely variable and does not comply with the national standards issued by the Ministry of Agriculture (MAPA, 2010), which determines 150 ppm maximum methanol, 12% maximum humidity, and 80% minimum glycerol.
In the USA, glycerine obtained from different raw materials and companies also show wide variation in ash content and chemical composition due to the amount of catalyzers used by each company, as well as high methanol concentrations (GOTT, 2009). This is consistent with the findings of Kerr et al. (2009), who found 51-83% glycerol, 0.005-14% methanol, and 0.02-35% fatty acids in glycerine samples from different sources and companies in the USA.
Kerr (2007) analyzed two crude glycerine samples from a same biodiesel company collected 90 days apart and obtained different chemical compositions, particularly relative to glycerol and methanol levels. This difference in chemical composition was also detected in the samples collected 60 days apart from companies B, C, and D in the present study (Tables 2 and 3). These differences in the chemical composition of glycerine may be associated with the raw material and the lack of process standardization by the companies. The levels of methanol, sodium chloride, and potassium chloride found in glycerine depend on from the biodiesel extraction and glycerine purification methods.
The efficiency of the transesterification process determines the energy concentration of the glycerine produced by each company. Lower gross energy levels indicate better utilization of fatty acids during processing, i.e., no triglycerides are left intact. On the other hand, high gross energy levels may signal the inefficiency of the process, which results in a higher concentration of residual products (Penz Jr. & Gianfelice, 2008).
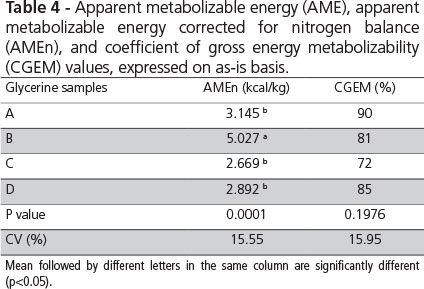
Apparent metabolizable energy (AME) and apparent metabolizable energy corrected for nitrogen balance (AMEn) contents, and coefficients of gross energy metabolizability (CGEM), expressed on as-is basis, are shown in Table 4. AMEn and CGEM varied according to glycerine composition (p<0.05), particularly to glycerol and fatty acid levels. Lammers et al. (2008) observed that metabolizable energy may be directly related to glycerol content in glycerine samples. In addition, fatty acid content also directly influenced glycerine gross energy levels.
The AMEn value of glycerine A was 3.145 kcal/kg and its CGEM was 90%, which are close to those obtained by Dozier et al. (2008), who evaluated a glycerine sample containing 86.9T glycerol, 5.33% humidity, 1.26% sodium, and 3.625 kcal/kg GE in 38- to 45-d-old broilers, and obtained 3.349 kcal AMEn/kg on as-is basis and 92% CGEM. Lammers et al. (2008b), working with 40-wk-old layers, obtained 3.800 kcal AMEn/kg. These differences in metabolizable energy values are probably due to the age of the chickens used in the trials and/or glycerine composition. Energy digestibility values tend to increase with bird age due to the development of the digestive tract, which presents higher nutrient and energy utilization capacity as birds age. Another aspect to be considered is that glycerine A used in the present study contained low glycerol levels, which may have reduced AMEn.
There is a close correlation between gross energy and metabolizable energy values in glycerine, indicating its high digestibility (Penz Jr. & Gianfelice, 2008). In the present study, glycerine A had a CGEM of 90%, which demonstrates its high utilization by the birds. Glycerine B presented 5.027 kcal AMEn/kg and 81% CGEM (Table 4). This high energy value is explained by the high levels of fatty acids, but the lower CGEM may be related to the concentration of free fatty acids. Higher free fatty acid levels reduce fat digestibility (Gaiotto et al., 2000).
AMEn values of 3,276 kcal/kg (Gianfelice, 2009) and of 4,556 kcal/kg (Carvalho, 2010) were obtained with semi-purified vegetable glycerine fed to broilers and pigs, respectively. It should be considered that glycerine AMEn changes according to its inclusion levels in the diet. Gianfelice (2009) included 5 and 15% glycerine in the diet and obtained energy values of 1,527 and 4,890 kcal/kg, respectively, suggesting that average values not important.
In pigs, the dietary inclusion of glycerine obtained from different biodiesel companies resulted in metabolizable energy levels ranging between 2,535 and 5,206 kcal/kg. Kerr et al. (2009) concluded that glycerol, methanol and fatty acid contents directly affect ME of glycerine, but CGEM was not different among the tested sources (85%). The obtained CGEM value range of 72-90% indicates that energy utilization may vary according to glycerine source, despite the lack of statistical differences.
Cerrate et al. (2006) verified that glycerine ME was 95% of its GE value in diet formulation. However, glycerine metabolism may be limited by its dietary inclusion level. Glycerine AME is reduced due to the lack of renal reabsorption of glycerol, which excess is excreted in the urine (Bartlet & Schnieder, 2002) and to metabolization losses (Mendoza et al., 2010).
Differences in the chemical of glycerine may affect its metabolizable energy content. A study comparing the inclusion of mixed crude glycerine (55% glycerol, 50,500 ppm methanol; 1.99% Na; 21.5% fatty acids; 5,242 kcal GE/kg) with vegetable crude glycerine (55% glycerol; 10,000 ppm methanol; 1.62% Na; 23% fatty acids; 5,247 kcal GE/kg) showed very close ME (4,488 and 4,556 kcal/kg) and CGEM (85 and 87%) values (Carvalho, 2010).
In Japanese quails, AMEn values also vary according to glycerine source. Values of 4,839 and 2,476 kcal/kg (Pasquetti, 2011) and of 4,564 and 3,069 kcal/kg (Batista, 2010), expressed on as-fed basis, were determined for crude and semi-purified glycerine, respectively. Both studies obtained high levels of fatty acids in crude glycerine, and therefore, high energy values. In the study of Pasqueti (2011), crude glycerine CGEM was higher than that of semi-purified glycerine (90 vs. 77%). In the present study, semi-purified glycerine presented better CGEM (90%), which is consistent with the findings of Lammers et al. (2008).
Methanol levels were higher than those established by the Brazilian Ministry of Agriculture (150 ppm). Methanol affects the central nervous system causing intoxication symptoms 12-24 hours after ingestion in humans, and its minimum lethal dose ranges between 0.3 and 1.0 g/kg (International Programme on Chemical Safety, 1997). No studies were found in literature relative to methanol poisoning, but in the present study, no visual effects of poisoning were observed in none of the methanol concentrations determined (80-18,900 ppm).
Glycerine C presented 2,669 kcal/kg AMEn and 72% CGEM, which were lower than in the other tested glycerine samples (Table 4), but are consistent with the results of Guerra (2010). Working with 18- to 28-d-old broilers and a crude glycerine composition of 87.5% glycerol, 19% humidity, 2.3% Na, and 3,458 kcal GE/kg, that author obtained 2,485 kcal AME/kg as fed and a coefficient of metabolization of 71.85%. Guerra (2010) suggested that this low coefficient is due to glycerol hygroscopicity and the 2.3% Na level, because Na increases water intake and excretion, thereby reducing glycerine absorption, explaining the lower CGEM obtained.
Although glycerine C presented AMEn and CGEM similar to those found by Guerra (2010), its sodium concentration was low. In addition, glycerine sources A and D contained 2.3 and 2.3% Na, respectively, but their CGEM were high (90 and 85%, respectively). Abd-Elsamee et al. (2010), working with 28-wk-old roosters and glycerine with 3.41% Na, obtained 96% CGEM. Therefore, the low CGEM cannot be explained by high sodium levels in glycerine.
When glycerine complies with the standards established by the Brazilian Ministry of Agriculture (Brasil, 2010), its metabolizable energy values are very close to the apparent metabolizable energy values obtained in pigs (3,340 kcal/kg) and poultry (3,381 kcal/kg) (Rostagno et al., 2011). This demonstrates the potential of glycerine inclusion as an energy feedstuff in pig and poultry diets. However, when formulating pig and poultry diets, the fact that glycerine metabolizable energy value is proportional to its glycerol and fatty acid levels.
Pure glycerol ME values for broilers, layers and pig may vary according to its dietary inclusion levels (Bartlet & Schnieder, 2002). AME may be reduced to the lack of renal reabsorption of glycerol, which excess is excreted in the urine. Gianfelice (2010) also suggests that there is a threshold of glycerol metabolization, after which increasing dietary inclusion levels increase glycerol blood levels, and consequent excretion in the urine.
It should be noted, therefore, that the different energy values of glycerine reported in literature are due to the different glycerol, water, and fatty acid contents of this by-product.
CONCLUSIONS
Glycerine composition directly affects its metabolizable energy content and coefficient of metabolizability; when it contains high levels of glycerol, it may be considered for the inclusion in broiler feeds due to high metabolizable energy content.
Submitted: October/2013
Approved: June/2014
- Abd-Elsamee MO, Abdo SMA, El-Manylawi MAF. Use of crude glycerol in broilers diets. Egypty Poultry Science 2010;30:281-295.
- AviSite. Estatísticas. 2011 [cited 2012 Jul 20]. Available from: htpp://www.avisite.com.br
- Barlet J, Schneider D. Investigation on the energy value of glycerol in the feeding of poultry and pig. Union for the promotion of oilseeds Schriften Heft 2002;17:15-36.
- Carvalho PLO, Moreira I, Piano ML, Toledo JB, Gallego, AG, Crus TMP. Valor nutriocional da glicerina bruta e semi-purificada na alimentação de suínos na fase de crescimento [CD-ROM]. Anais da 47ª Reunião Anual Da Sociedade Brasileira de Zootecnia; 2010; Salvador. Bahia, Salvador. Brasil: SBZ, 2010.
- Dozier WA, Kerr BJ, Corzo A, Kidd MT, Weber TE, Bregendahl K. Apparent metabolizable energy of glycerin for broiler chickens. Poultry Science 2008;87:317-322.
- Gaiotto JB, Menten JFM, Racanicci AMC, Iafigliola M.C. Óleo de soja, óleo ácido de soja e sebo bovino como fontes de gordura em rações de frangos de corte. Revista Brasileira de Ciências Avícolas 2000;2(3):219-227.
- Gianfelice MF. Uso do glicerol como fonte alternativa para frangos de corte [dissertação]. Porto Alegre (RS): Universidade Federal do Rio Grande do Sul; 2009.
- Gott P. Variation in the chemical composition of crude glycerin [cited 2011 Jul 8]. The Ohio State University; 2009. Available from:http://hdl.handle.net/1811/37082
- Kerr BJ. Feeding bioenergy coproducts to swine [cited 2011 Abr 25]. Sheffield: Iowa State University, University Extension; 2007. Available from: http://www.ipic.iastate.edu/publications/IPIC11b.pdf
- Kerr BJ, Weber TE, Dozier WA, Kidd MT. Digestible and metabolizable energy content of crude glycerin originating from different sources in nursery pigs. Journal of Animal Science 2009;87:4042-4049.
- Guerra LRH. Glicerina bruta na alimentação de frangos de corte [dissertação]. Maringá (PR): Universidade Estadual de Maringá; 2010. 67p.
- International Programme on Chemical Safety. Envirommental health criteria 196-methanol. Geneva: WHO; 1997. 345p.
- Lammers P, Kerr BJ, Honeyman M, Stalder K, Dozier III WA, Weber TE, Kidd T, Bregendahl K. Nitrogen-corrected apparent metabolizable energy value of crude glycerol for laying hens. Journal of Animal Science 2008b;87(1):104-107.
- Ministério da Agricultura, Pecuária e Abastecimento. Brasília, DF: Departamento de Fiscalização dos Insumos Pecuários (DFIP-MAPA); 2010
- Penz Junior AM, Gianfelice M. O que fazer para substituir os insumos que podem migrar para a produção de biocombustível. Acta Scientiae Veterinariae 2008;36 Supl 1 :107-117
- Rostagno HS. Tabelas brasileiras para aves e suínos: composição de alimentos e exigências nutricionais. 3ªed. Viçosa: Universidade Federal de Zootecnia; 2011. 244p.
- Rostagno HS, Albino LFT, Donzele JL, Gomes PC, Oliveria RF, Lopes DC, Ferreira AS, Barr SLT. Tabelas brasileiras para aves e suínos: composição de alimentos e exigências nutricionais. Viçosa: Universidade Federal de Viçosa; 2005. 186p.
- Sakamura NK, Rostagno HS. Métodos de pesquisa em nutrição de monogástricos. Jaboticabal: FUNEP; 2007. 283 p.
- Sas Institute. Statistical analysis system: user's guide. Version 9.1 ed. Cary; 2006.
Correspondence
Publication Dates
-
Publication in this collection
06 Jan 2015 -
Date of issue
Dec 2014
History
-
Received
Oct 2013 -
Accepted
June 2014